The alkali metals consist of the
chemical element
A chemical element is a species of atoms that have a given number of protons in their nuclei, including the pure substance consisting only of that species. Unlike chemical compounds, chemical elements cannot be broken down into simpler sub ...
s
lithium
Lithium (from el, λίθος, lithos, lit=stone) is a chemical element with the symbol Li and atomic number 3. It is a soft, silvery-white alkali metal. Under standard conditions, it is the least dense metal and the least dense solid el ...
(Li),
sodium
Sodium is a chemical element with the symbol Na (from Latin ''natrium'') and atomic number 11. It is a soft, silvery-white, highly reactive metal. Sodium is an alkali metal, being in group 1 of the periodic table. Its only stable iso ...
(Na),
potassium
Potassium is the chemical element with the symbol K (from Neo-Latin ''kalium'') and atomic number19. Potassium is a silvery-white metal that is soft enough to be cut with a knife with little force. Potassium metal reacts rapidly with atmosphe ...
(K),
[The symbols Na and K for sodium and potassium are derived from their Latin names, ''natrium'' and ''kalium''; these are still the origins of the names for the elements in some languages, such as German and Russian.] rubidium
Rubidium is the chemical element with the symbol Rb and atomic number 37. It is a very soft, whitish-grey solid in the alkali metal group, similar to potassium and caesium. Rubidium is the first alkali metal in the group to have a density higher ...
(Rb),
caesium
Caesium (IUPAC spelling) (or cesium in American English) is a chemical element with the symbol Cs and atomic number 55. It is a soft, silvery-golden alkali metal with a melting point of , which makes it one of only five elemental metals that a ...
(Cs), and
francium
Francium is a chemical element with the symbol Fr and atomic number 87. It is extremely radioactive; its most stable isotope, francium-223 (originally called actinium K after the natural decay chain it appears in), has a half-life of only 22&nb ...
(Fr). Together with
hydrogen
Hydrogen is the chemical element with the symbol H and atomic number 1. Hydrogen is the lightest element. At standard conditions hydrogen is a gas of diatomic molecules having the formula . It is colorless, odorless, tasteless, non-toxic, an ...
they constitute
group 1 Group 1 may refer to:
* Alkali metal, a chemical element classification for Alkali metal
* Group 1 (racing), a historic (until 1981) classification for Touring car racing, applied to standard touring cars. Comparable to modern FIA Group N
* Group On ...
, which lies in the
s-block
A block of the periodic table is a set of elements unified by the atomic orbitals their valence electrons or vacancies lie in. The term appears to have been first used by Charles Janet. Each block is named after its characteristic orbital: s-blo ...
of the
periodic table
The periodic table, also known as the periodic table of the (chemical) elements, is a rows and columns arrangement of the chemical elements. It is widely used in chemistry, physics, and other sciences, and is generally seen as an icon of ch ...
. All alkali metals have their outermost electron in an
s-orbital
In atomic theory and quantum mechanics, an atomic orbital is a function describing the location and wave-like behavior of an electron in an atom. This function can be used to calculate the probability of finding any electron of an atom in any sp ...
: this shared electron configuration results in their having very similar characteristic properties. Indeed, the alkali metals provide the best example of
group trends in properties in the periodic table, with elements exhibiting well-characterised
homologous behaviour.
This family of elements is also known as the lithium family after its leading element.
The alkali metals are all shiny,
soft, highly
reactive
Reactive may refer to:
*Generally, capable of having a reaction (disambiguation)
*An adjective abbreviation denoting a bowling ball coverstock made of reactive resin
*Reactivity (chemistry)
*Reactive mind
*Reactive programming
See also
*Reactanc ...
metals
A metal (from Greek μέταλλον ''métallon'', "mine, quarry, metal") is a material that, when freshly prepared, polished, or fractured, shows a lustrous appearance, and conducts electricity and heat relatively well. Metals are typicall ...
at
standard temperature and pressure
Standard temperature and pressure (STP) are standard sets of conditions for experimental measurements to be established to allow comparisons to be made between different sets of data. The most used standards are those of the International Union o ...
and readily lose their
outermost electron to form
cations
An ion () is an atom or molecule with a net electrical charge.
The charge of an electron is considered to be negative by convention and this charge is equal and opposite to the charge of a proton, which is considered to be positive by convent ...
with
charge
Charge or charged may refer to:
Arts, entertainment, and media Films
* '' Charge, Zero Emissions/Maximum Speed'', a 2011 documentary
Music
* ''Charge'' (David Ford album)
* ''Charge'' (Machel Montano album)
* ''Charge!!'', an album by The Aqu ...
+1. They can all be cut easily with a knife due to their softness, exposing a shiny surface that tarnishes rapidly in air due to
oxidation
Redox (reduction–oxidation, , ) is a type of chemical reaction in which the oxidation states of substrate change. Oxidation is the loss of electrons or an increase in the oxidation state, while reduction is the gain of electrons or a d ...
by atmospheric moisture and
oxygen
Oxygen is the chemical element with the symbol O and atomic number 8. It is a member of the chalcogen group in the periodic table, a highly reactive nonmetal, and an oxidizing agent that readily forms oxides with most elements as wel ...
(and in the case of lithium,
nitrogen
Nitrogen is the chemical element with the symbol N and atomic number 7. Nitrogen is a nonmetal and the lightest member of group 15 of the periodic table, often called the pnictogens. It is a common element in the universe, estimated at se ...
). Because of their high reactivity, they must be stored under oil to prevent reaction with air, and are found naturally only in
salts
In chemistry, a salt is a chemical compound consisting of an ionic assembly of positively charged cations and negatively charged anions, which results in a compound with no net electric charge. A common example is table salt, with positively cha ...
and never as the free elements. Caesium, the fifth alkali metal, is the most reactive of all the metals. All the alkali metals react with water, with the heavier alkali metals reacting more vigorously than the lighter ones.
All of the discovered alkali metals occur in nature as their compounds: in order of
abundance
Abundance may refer to:
In science and technology
* Abundance (economics), the opposite of scarcities
* Abundance (ecology), the relative representation of a species in a community
* Abundance (programming language), a Forth-like computer prog ...
, sodium is the most abundant, followed by potassium, lithium, rubidium, caesium, and finally francium, which is very rare due to its extremely high
radioactivity; francium occurs only in minute
traces
Traces may refer to:
Literature
* ''Traces'' (book), a 1998 short-story collection by Stephen Baxter
* ''Traces'' series, a series of novels by Malcolm Rose
Music Albums
* ''Traces'' (Classics IV album) or the title song (see below), 1969
* ''Tra ...
in nature as an intermediate step in some obscure side branches of the natural
decay chain
In nuclear science, the decay chain refers to a series of radioactive decays of different radioactive decay products as a sequential series of transformations. It is also known as a "radioactive cascade". Most radioisotopes do not decay direct ...
s. Experiments have been conducted to attempt the synthesis of
ununennium
Ununennium, also known as eka-francium or element 119, is the hypothetical chemical element with symbol Uue and atomic number 119. ''Ununennium'' and ''Uue'' are the temporary systematic IUPAC name and symbol respectively, which are used until th ...
(Uue), which is likely to be the next member of the group; none were successful. However, ununennium may not be an alkali metal due to
relativistic effects
Relativistic quantum chemistry combines relativistic mechanics with quantum chemistry to calculate elemental properties and structure, especially for the heavier elements of the periodic table. A prominent example is an explanation for the color of ...
, which are predicted to have a large influence on the chemical properties of
superheavy element
Superheavy elements, also known as transactinide elements, transactinides, or super-heavy elements, are the chemical elements with atomic number greater than 103. The superheavy elements are those beyond the actinides in the periodic table; the l ...
s; even if it does turn out to be an alkali metal, it is predicted to have some differences in physical and chemical properties from its lighter homologues.
Most alkali metals have many different applications. One of the best-known applications of the pure elements is the use of rubidium and caesium in
atomic clock
An atomic clock is a clock that measures time by monitoring the resonant frequency of atoms. It is based on atoms having different energy levels. Electron states in an atom are associated with different energy levels, and in transitions betwee ...
s, of which caesium atomic clocks form the basis of the
second
The second (symbol: s) is the unit of time in the International System of Units (SI), historically defined as of a day – this factor derived from the division of the day first into 24 hours, then to 60 minutes and finally to 60 seconds ...
. A common application of the compounds of sodium is the
sodium-vapour lamp
A sodium-vapor lamp is a gas-discharge lamp that uses sodium in an excited state to produce light at a characteristic wavelength near 589 nm.
Two varieties of such lamps exist: low pressure and high pressure. Low-pressure sodium lamps are ...
, which emits light very efficiently.
Table salt
Salt is a mineral composed primarily of sodium chloride (NaCl), a chemical compound belonging to the larger class of salts; salt in the form of a natural crystalline mineral is known as rock salt or halite. Salt is present in vast quantitie ...
, or sodium chloride, has been used since antiquity.
Lithium
Lithium (from el, λίθος, lithos, lit=stone) is a chemical element with the symbol Li and atomic number 3. It is a soft, silvery-white alkali metal. Under standard conditions, it is the least dense metal and the least dense solid el ...
finds use as a psychiatric medication and as an
anode
An anode is an electrode of a polarized electrical device through which conventional current enters the device. This contrasts with a cathode, an electrode of the device through which conventional current leaves the device. A common mnemonic is ...
in
lithium batteries
Lithium battery may refer to:
* Lithium metal battery, a non-rechargeable battery with lithium as an anode
** Rechargeable lithium metal battery, a rechargeable counterpart to the lithium metal battery
* Lithium-ion battery, a rechargeable batte ...
. Sodium, potassium and lithium are
essential element
In the context of nutrition, a mineral is a chemical element required as an essential nutrient by organisms to perform functions necessary for life. However, the four major structural elements in the human body by weight (oxygen, hydrogen, carbon ...
s, having major biological roles as
electrolytes
An electrolyte is a medium containing ions that is electrically conducting through the movement of those ions, but not conducting electrons. This includes most soluble salts, acids, and bases dissolved in a polar solvent, such as water. Upon di ...
, and although the other alkali metals are not essential, they also have various effects on the body, both beneficial and harmful.
__TOC__
History

Sodium compounds have been known since ancient times; salt (
sodium chloride
Sodium chloride , commonly known as salt (although sea salt also contains other chemical salts), is an ionic compound with the chemical formula NaCl, representing a 1:1 ratio of sodium and chloride ions. With molar masses of 22.99 and 35.45 g ...
) has been an important commodity in human activities, as testified by the English word ''salary'', referring to ''salarium'', money paid to Roman soldiers for the purchase of salt. While
potash
Potash () includes various mined and manufactured salts that contain potassium in water-soluble form. has been used since ancient times, it was not understood for most of its history to be a fundamentally different substance from sodium mineral salts.
Georg Ernst Stahl
Georg Ernst Stahl (22 October 1659 – 24 May 1734) was a German chemist, physician and philosopher. He was a supporter of vitalism, and until the late 18th century his works on phlogiston were accepted as an explanation for chemical processes.Ku ...
obtained experimental evidence which led him to suggest the fundamental difference of sodium and potassium salts in 1702,
and
Henri-Louis Duhamel du Monceau
Henri-Louis Duhamel du Monceau (20 July 1700, Paris13 August 1782, Paris), was a French physician, naval engineer and botanist.
Biography
Henri-Louis Duhamel du Monceau was born in Paris in 1700, the son of Alexandre Duhamel, lord of Denai ...
was able to prove this difference in 1736. The exact chemical composition of potassium and sodium compounds, and the status as chemical element of potassium and sodium, was not known then, and thus
Antoine Lavoisier
Antoine-Laurent de Lavoisier ( , ; ; 26 August 17438 May 1794), When reduced without charcoal, it gave off an air which supported respiration and combustion in an enhanced way. He concluded that this was just a pure form of common air and th ...
did not include either alkali in his list of chemical elements in 1789.
Pure potassium was first isolated in 1807 in England by
Humphry Davy
Sir Humphry Davy, 1st Baronet, (17 December 177829 May 1829) was a British chemist and inventor who invented the Davy lamp and a very early form of arc lamp. He is also remembered for isolating, by using electricity, several elements for t ...
, who derived it from
caustic potash
Potassium hydroxide is an inorganic compound with the formula K OH, and is commonly called caustic potash.
Along with sodium hydroxide (NaOH), KOH is a prototypical strong base. It has many industrial and niche applications, most of which explo ...
(KOH, potassium hydroxide) by the use of electrolysis of the molten salt with the newly invented
voltaic pile
upright=1.2, Schematic diagram of a copper–zinc voltaic pile. The copper and zinc discs were separated by cardboard or felt spacers soaked in salt water (the electrolyte). Volta's original piles contained an additional zinc disk at the bottom, ...
. Previous attempts at electrolysis of the aqueous salt were unsuccessful due to potassium's extreme reactivity.
Potassium was the first metal that was isolated by electrolysis.
Later that same year, Davy reported extraction of sodium from the similar substance
caustic soda
Sodium hydroxide, also known as lye and caustic soda, is an inorganic compound with the formula NaOH. It is a white solid ionic compound consisting of sodium cations and hydroxide anions .
Sodium hydroxide is a highly caustic base and alka ...
(NaOH, lye) by a similar technique, demonstrating the elements, and thus the salts, to be different.

Petalite
Petalite, also known as castorite, is a lithium aluminum phyllosilicate mineral Li Al Si4 O10, crystallizing in the monoclinic system. Petalite occurs as colorless, pink, grey, yellow, yellow grey, to white tabular crystals and columnar masses. ...
(
Li Al Si4 O10) was discovered in 1800 by the
Brazil
Brazil ( pt, Brasil; ), officially the Federative Republic of Brazil (Portuguese: ), is the largest country in both South America and Latin America. At and with over 217 million people, Brazil is the world's fifth-largest country by area ...
ian chemist
José Bonifácio de Andrada
José is a predominantly Spanish and Portuguese form of the given name Joseph. While spelled alike, this name is pronounced differently in each language: Spanish ; Portuguese (or ).
In French, the name ''José'', pronounced , is an old vernacul ...
in a mine on the island of
Utö, Sweden
Utö is a small island in the East of Stockholm archipelago, known for its nature. Utö means "outer island" in Swedish. It is a part of Haninge Municipality.
Geology
Utö and the surrounding islands are unique from a geological point of view ...
.
However, it was not until 1817 that
Johan August Arfwedson
Johan August Arfwedson (12 January 1792 – 28 October 1841) was a Swedish chemist who discovered the chemical element lithium in 1817 by isolating it as a salt.
Life and work
Arfwedson belonged to a wealthy bourgeois family, the son of t ...
, then working in the laboratory of the chemist
Jöns Jacob Berzelius
Baron Jöns Jacob Berzelius (; by himself and his contemporaries named only Jacob Berzelius, 20 August 1779 – 7 August 1848) was a Swedish chemist. Berzelius is considered, along with Robert Boyle, John Dalton, and Antoine Lavoisier, to be on ...
,
detected the presence of a new element while analysing petalite
ore
Ore is natural rock or sediment that contains one or more valuable minerals, typically containing metals, that can be mined, treated and sold at a profit.Encyclopædia Britannica. "Ore". Encyclopædia Britannica Online. Retrieved 7 Apr ...
.
This new element was noted by him to form compounds similar to those of sodium and potassium, though its
carbonate
A carbonate is a salt of carbonic acid (H2CO3), characterized by the presence of the carbonate ion, a polyatomic ion with the formula . The word ''carbonate'' may also refer to a carbonate ester, an organic compound containing the carbonate g ...
and
hydroxide
Hydroxide is a diatomic anion with chemical formula OH−. It consists of an oxygen and hydrogen atom held together by a single covalent bond, and carries a negative electric charge. It is an important but usually minor constituent of water. I ...
were less
soluble in water and more
alkaline
In chemistry, an alkali (; from ar, القلوي, al-qaly, lit=ashes of the saltwort) is a base (chemistry), basic, ionic compound, ionic salt (chemistry), salt of an alkali metal or an alkaline earth metal. An alkali can also be defined as ...
than the other alkali metals.
Berzelius gave the unknown material the name "''lithion''/''lithina''", from the
Greek
Greek may refer to:
Greece
Anything of, from, or related to Greece, a country in Southern Europe:
*Greeks, an ethnic group.
*Greek language, a branch of the Indo-European language family.
**Proto-Greek language, the assumed last common ancestor ...
word ''λιθoς'' (transliterated as ''lithos'', meaning "stone"), to reflect its discovery in a solid mineral, as opposed to potassium, which had been discovered in plant ashes, and sodium, which was known partly for its high abundance in animal blood. He named the metal inside the material "''lithium''".
Lithium, sodium, and potassium were part of the discovery of
periodicity
Periodicity or periodic may refer to:
Mathematics
* Bott periodicity theorem, addresses Bott periodicity: a modulo-8 recurrence relation in the homotopy groups of classical groups
* Periodic function, a function whose output contains values tha ...
, as they are among a series of triads of elements in the same
group
A group is a number of persons or things that are located, gathered, or classed together.
Groups of people
* Cultural group, a group whose members share the same cultural identity
* Ethnic group, a group whose members share the same ethnic ide ...
that were noted by
Johann Wolfgang Döbereiner
Johann Wolfgang Döbereiner (13 December 1780 – 24 March 1849) was a German chemist who is best known for work that foreshadowed the periodic law for the chemical elements, and for inventing the first lighter, which was known as the Döberei ...
in 1850 as having similar properties.

Rubidium and caesium were the first elements to be discovered using the
spectroscope
An optical spectrometer (spectrophotometer, spectrograph or spectroscope) is an instrument used to measure properties of light over a specific portion of the electromagnetic spectrum, typically used in spectroscopic analysis to identify mate ...
, invented in 1859 by
Robert Bunsen
Robert Wilhelm Eberhard Bunsen (;
30 March 1811
– 16 August 1899) was a German chemist. He investigated emission spectra of heated elements, and discovered caesium (in 1860) and rubidium (in 1861) with the physicist Gustav Kirchhoff. The Bu ...
and
Gustav Kirchhoff
Gustav Robert Kirchhoff (; 12 March 1824 – 17 October 1887) was a German physicist who contributed to the fundamental understanding of electrical circuits, spectroscopy, and the emission of black-body radiation by heated objects.
He coine ...
.
The next year, they discovered caesium in the
mineral water
Mineral water is water from a mineral spring that contains various minerals, such as salts and sulfur compounds. Mineral water may usually be still or sparkling (carbonated/effervescent) according to the presence or absence of added gases.
Tra ...
from
Bad Dürkheim
Bad Dürkheim () is a spa town in the Rhine-Neckar urban agglomeration, and is the seat of the Bad Dürkheim district in Rhineland-Palatinate, Germany.
Geography
Location
Bad Dürkheim lies at the edge of Palatinate Forest on the German Wine ...
, Germany. Their discovery of rubidium came the following year in
Heidelberg
Heidelberg (; Palatine German language, Palatine German: ''Heidlberg'') is a city in the States of Germany, German state of Baden-Württemberg, situated on the river Neckar in south-west Germany. As of the 2016 census, its population was 159,914 ...
, Germany, finding it in the mineral
lepidolite
Lepidolite is a lilac-gray or rose-colored member of the mica group of minerals with chemical formula . It is the most abundant lithium-bearing mineral and is a secondary source of this metal. It is the major source of the alkali metal rubidium.
...
.
The names of rubidium and caesium come from the most prominent lines in their
emission spectra
The emission spectrum of a chemical element or chemical compound is the spectrum of frequencies of electromagnetic radiation emitted due to an electron making a atomic electron transition, transition from a high energy state to a lower energy st ...
: a bright red line for rubidium (from the
Latin
Latin (, or , ) is a classical language belonging to the Italic branch of the Indo-European languages. Latin was originally a dialect spoken in the lower Tiber area (then known as Latium) around present-day Rome, but through the power of the ...
word ''rubidus'', meaning dark red or bright red), and a sky-blue line for caesium (derived from the Latin word ''caesius'', meaning sky-blue).
Around 1865
John Newlands produced a series of papers where he listed the elements in order of increasing atomic weight and similar physical and chemical properties that recurred at intervals of eight; he likened such periodicity to the
octave
In music, an octave ( la, octavus: eighth) or perfect octave (sometimes called the diapason) is the interval between one musical pitch and another with double its frequency. The octave relationship is a natural phenomenon that has been refer ...
s of music, where notes an octave apart have similar musical functions. His version put all the alkali metals then known (lithium to caesium), as well as
copper
Copper is a chemical element with the symbol Cu (from la, cuprum) and atomic number 29. It is a soft, malleable, and ductile metal with very high thermal and electrical conductivity. A freshly exposed surface of pure copper has a pinkis ...
,
silver
Silver is a chemical element with the Symbol (chemistry), symbol Ag (from the Latin ', derived from the Proto-Indo-European wikt:Reconstruction:Proto-Indo-European/h₂erǵ-, ''h₂erǵ'': "shiny" or "white") and atomic number 47. A soft, whi ...
, and
thallium
Thallium is a chemical element with the Symbol (chemistry), symbol Tl and atomic number 81. It is a gray post-transition metal that is not found free in nature. When isolated, thallium resembles tin, but discolors when exposed to air. Chemists W ...
(which show the +1 oxidation state characteristic of the alkali metals), together into a group. His table placed hydrogen with the
halogen
The halogens () are a group in the periodic table consisting of five or six chemically related elements: fluorine (F), chlorine (Cl), bromine (Br), iodine (I), astatine (At), and tennessine (Ts). In the modern IUPAC nomenclature, this group is ...
s.
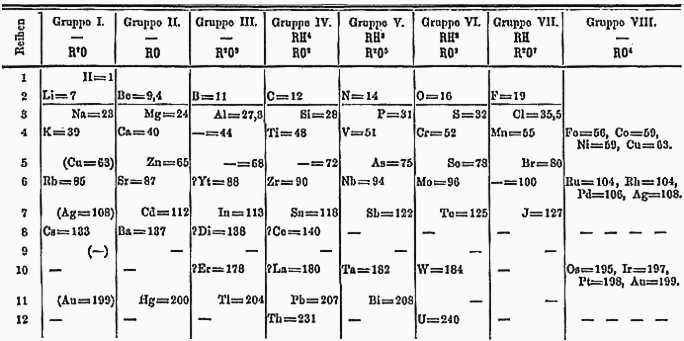
After 1869,
Dmitri Mendeleev
Dmitri Ivanovich Mendeleev (sometimes transliterated as Mendeleyev or Mendeleef) ( ; russian: links=no, Дмитрий Иванович Менделеев, tr. , ; 8 February Old_Style_and_New_Style_dates">O.S._27_January.html" ;"title="O ...
proposed his periodic table placing lithium at the top of a group with sodium, potassium, rubidium, caesium, and thallium. Two years later, Mendeleev revised his table, placing hydrogen in group 1 above lithium, and also moving thallium to the
boron group
The boron group are the chemical elements in group 13 of the periodic table, comprising boron (B), aluminium (Al), gallium (Ga), indium (In), thallium (Tl), and nihonium (Nh). The elements in the boron group are characterized by having three vale ...
. In this 1871 version, copper, silver, and
gold
Gold is a chemical element with the symbol Au (from la, aurum) and atomic number 79. This makes it one of the higher atomic number elements that occur naturally. It is a bright, slightly orange-yellow, dense, soft, malleable, and ductile met ...
were placed twice, once as part of
group IB, and once as part of a "group VIII" encompassing today's groups
8 to 11.
[In the 1869 version of Mendeleev's periodic table, copper and silver were placed in their own group, aligned with hydrogen and ]mercury
Mercury commonly refers to:
* Mercury (planet), the nearest planet to the Sun
* Mercury (element), a metallic chemical element with the symbol Hg
* Mercury (mythology), a Roman god
Mercury or The Mercury may also refer to:
Companies
* Merc ...
, while gold was tentatively placed under uranium
Uranium is a chemical element with the symbol U and atomic number 92. It is a silvery-grey metal in the actinide series of the periodic table. A uranium atom has 92 protons and 92 electrons, of which 6 are valence electrons. Uranium is weak ...
and the undiscovered eka-aluminium in the boron group
The boron group are the chemical elements in group 13 of the periodic table, comprising boron (B), aluminium (Al), gallium (Ga), indium (In), thallium (Tl), and nihonium (Nh). The elements in the boron group are characterized by having three vale ...
. After the introduction of the 18-column table, the group IB elements were moved to their current position in the
d-block
A block of the periodic table is a set of elements unified by the atomic orbitals their valence electrons or vacancies lie in. The term appears to have been first used by Charles Janet. Each block is named after its characteristic orbital: s-bloc ...
, while alkali metals were left in ''group IA''. Later the group's name was changed to ''group 1'' in 1988.
[ The ]trivial name
In chemistry, a trivial name is a nonsystematic name for a chemical substance. That is, the name is not recognized according to the rules of any formal system of chemical nomenclature such as IUPAC nomenclature of inorganic chemistry, IUPAC inor ...
"alkali metals" comes from the fact that the hydroxides of the group 1 elements are all strong alkali
In chemistry, an alkali (; from ar, القلوي, al-qaly, lit=ashes of the saltwort) is a basic, ionic salt of an alkali metal or an alkaline earth metal. An alkali can also be defined as a base that dissolves in water. A solution of a ...
s when dissolved in water.Marguerite Perey
Marguerite Catherine Perey (19 October 1909 – 13 May 1975) was a French physicist and a student of Marie Curie. In 1939, Perey discovered the element francium by purifying samples of lanthanum that contained actinium. In 1962, she was the firs ...
of the Curie Institute in Paris, France discovered francium in 1939 by purifying a sample of actinium-227
Actinium (89Ac) has no stable isotopes and no characteristic terrestrial isotopic composition, thus a standard atomic weight cannot be given. There are 33 known isotopes, from 204Ac to 236Ac, and 7 isomers. Three isotopes are found in nature, 225 ...
, which had been reported to have a decay energy of 220 keV Kev can refer to:
Given name
* Kev Adams, French comedian, actor, screenwriter and film producer born Kevin Smadja in 1991
* Kevin Kev Carmody (born 1946), Indigenous Australian singer-songwriter
* Kev Coghlan (born 1988), Scottish Grand Prix motor ...
. However, Perey noticed decay particles with an energy level below 80 keV. Perey thought this decay activity might have been caused by a previously unidentified decay product, one that was separated during purification, but emerged again out of the pure actinium
Actinium is a chemical element with the symbol Ac and atomic number 89. It was first isolated by Friedrich Oskar Giesel in 1902, who gave it the name ''emanium''; the element got its name by being wrongly identified with a substance And ...
-227. Various tests eliminated the possibility of the unknown element being thorium
Thorium is a weakly radioactive metallic chemical element with the symbol Th and atomic number 90. Thorium is silvery and tarnishes black when it is exposed to air, forming thorium dioxide; it is moderately soft and malleable and has a high me ...
, radium
Radium is a chemical element with the symbol Ra and atomic number 88. It is the sixth element in group 2 of the periodic table, also known as the alkaline earth metals. Pure radium is silvery-white, but it readily reacts with nitrogen (rather t ...
, lead
Lead is a chemical element with the symbol Pb (from the Latin ) and atomic number 82. It is a heavy metal that is denser than most common materials. Lead is soft and malleable, and also has a relatively low melting point. When freshly cu ...
, bismuth
Bismuth is a chemical element with the Symbol (chemistry), symbol Bi and atomic number 83. It is a post-transition metal and one of the pnictogens, with chemical properties resembling its lighter group 15 siblings arsenic and antimony. Elemental ...
, or thallium
Thallium is a chemical element with the Symbol (chemistry), symbol Tl and atomic number 81. It is a gray post-transition metal that is not found free in nature. When isolated, thallium resembles tin, but discolors when exposed to air. Chemists W ...
. The new product exhibited chemical properties of an alkali metal (such as coprecipitating with caesium salts), which led Perey to believe that it was element 87, caused by the alpha decay
Alpha decay or α-decay is a type of radioactive decay in which an atomic nucleus emits an alpha particle (helium nucleus) and thereby transforms or 'decays' into a different atomic nucleus, with a mass number that is reduced by four and an atom ...
of actinium-227.[Adloff, Jean-Pierre; Kaufman, George B. (25 September 2005)]
Francium (Atomic Number 87), the Last Discovered Natural Element
. ''The Chemical Educator'' 10 (5). Retrieved 26 March 2007. Perey then attempted to determine the proportion of beta decay
In nuclear physics, beta decay (β-decay) is a type of radioactive decay in which a beta particle (fast energetic electron or positron) is emitted from an atomic nucleus, transforming the original nuclide to an isobar of that nuclide. For ...
to alpha decay in actinium-227. Her first test put the alpha branching at 0.6%, a figure that she later revised to 1%.ununennium
Ununennium, also known as eka-francium or element 119, is the hypothetical chemical element with symbol Uue and atomic number 119. ''Ununennium'' and ''Uue'' are the temporary systematic IUPAC name and symbol respectively, which are used until th ...
(Uue), element 119.einsteinium
Einsteinium is a synthetic element with the symbol Es and atomic number 99. Einsteinium is a member of the actinide series and it is the seventh transuranium element. It was named in honor of Albert Einstein.
Einsteinium was discovered as a compo ...
-254 with calcium
Calcium is a chemical element with the symbol Ca and atomic number 20. As an alkaline earth metal, calcium is a reactive metal that forms a dark oxide-nitride layer when exposed to air. Its physical and chemical properties are most similar to ...
-48 ions at the superHILAC accelerator at Berkeley, California. No atoms were identified, leading to a limiting yield of 300 nb.[The ]asterisk
The asterisk ( ), from Late Latin , from Ancient Greek , ''asteriskos'', "little star", is a typographical symbol. It is so called because it resembles a conventional image of a heraldic star.
Computer scientists and mathematicians often voc ...
denotes an excited state
In quantum mechanics, an excited state of a system (such as an atom, molecule or nucleus) is any quantum state of the system that has a higher energy than the ground state (that is, more energy than the absolute minimum). Excitation refers to a ...
.
It is highly unlikelyextended periodic table
An extended periodic table theorises about chemical elements beyond those currently known in the periodic table and proven. , the element with the highest atomic number known is oganesson (''Z'' = 118), which completes the seventh period (row) ...
, it may well be discovered in the near future through other reactions, and indeed an attempt to synthesise it is currently ongoing in Japan.
Occurrence
In the Solar System
 The
The Oddo–Harkins rule The Oddo–Harkins rule holds that an element with an even atomic number (such as carbon, element 6) is more abundant than both elements with the adjacently larger and smaller odd atomic numbers (such as boron, element 5, and nitrogen, element 7, ...
holds that elements with even atomic numbers are more common that those with odd atomic numbers, with the exception of hydrogen. This rule argues that elements with odd atomic numbers have one unpaired proton and are more likely to capture another, thus increasing their atomic number. In elements with even atomic numbers, protons are paired, with each member of the pair offsetting the spin of the other, enhancing stability.noble gas
The noble gases (historically also the inert gases; sometimes referred to as aerogens) make up a class of chemical elements with similar properties; under standard conditions, they are all odorless, colorless, monatomic gases with very low chemi ...
es and the alkaline earth metal
The alkaline earth metals are six chemical elements in group 2 of the periodic table. They are beryllium (Be), magnesium (Mg), calcium (Ca), strontium (Sr), barium (Ba), and radium (Ra).. The elements have very similar properties: they are al ...
s) in the Solar System. The heavier alkali metals are also less abundant than the lighter ones as the alkali metals from rubidium onward can only be synthesised in supernova
A supernova is a powerful and luminous explosion of a star. It has the plural form supernovae or supernovas, and is abbreviated SN or SNe. This transient astronomical event occurs during the last evolutionary stages of a massive star or when ...
e and not in stellar nucleosynthesis
Stellar nucleosynthesis is the creation (nucleosynthesis) of chemical elements by nuclear fusion reactions within stars. Stellar nucleosynthesis has occurred since the original creation of hydrogen, helium and lithium during the Big Bang. As a ...
. Lithium is also much less abundant than sodium and potassium as it is poorly synthesised in both Big Bang nucleosynthesis
In physical cosmology, Big Bang nucleosynthesis (abbreviated BBN, also known as primordial nucleosynthesis) is the production of nuclei other than those of the lightest isotope of hydrogen (hydrogen-1, 1H, having a single proton as a nucleus) du ...
and in stars: the Big Bang could only produce trace quantities of lithium, beryllium
Beryllium is a chemical element with the symbol Be and atomic number 4. It is a steel-gray, strong, lightweight and brittle alkaline earth metal. It is a divalent element that occurs naturally only in combination with other elements to form mi ...
and boron
Boron is a chemical element with the symbol B and atomic number 5. In its crystalline form it is a brittle, dark, lustrous metalloid; in its amorphous form it is a brown powder. As the lightest element of the ''boron group'' it has th ...
due to the absence of a stable nucleus with 5 or 8 nucleon
In physics and chemistry, a nucleon is either a proton or a neutron, considered in its role as a component of an atomic nucleus. The number of nucleons in a nucleus defines the atom's mass number (nucleon number).
Until the 1960s, nucleons were ...
s, and stellar nucleosynthesis could only pass this bottleneck by the triple-alpha process
The triple-alpha process is a set of nuclear fusion reactions by which three helium-4 nuclei (alpha particles) are transformed into carbon.
Triple-alpha process in stars
Helium accumulates in the cores of stars as a result of the proton–pro ...
, fusing three helium nuclei to form carbon
Carbon () is a chemical element with the symbol C and atomic number 6. It is nonmetallic and tetravalent
In chemistry, the valence (US spelling) or valency (British spelling) of an element is the measure of its combining capacity with o ...
, and skipping over those three elements.
On Earth
 The
The Earth
Earth is the third planet from the Sun and the only astronomical object known to harbor life. While large volumes of water can be found throughout the Solar System, only Earth sustains liquid surface water. About 71% of Earth's surfa ...
formed from the same cloud of matter that formed the Sun, but the planets acquired different compositions during the formation and evolution of the solar system
The formation of the Solar System began about 4.6 billion years ago with the gravitational collapse of a small part of a giant molecular cloud. Most of the collapsing mass collected in the center, forming the Sun, while the rest flattened into a ...
. In turn, the natural history of the Earth caused parts of this planet to have differing concentrations of the elements. The mass of the Earth is approximately 5.98 kg. It is composed mostly of iron
Iron () is a chemical element with symbol Fe (from la, ferrum) and atomic number 26. It is a metal that belongs to the first transition series and group 8 of the periodic table. It is, by mass, the most common element on Earth, right in f ...
(32.1%), oxygen
Oxygen is the chemical element with the symbol O and atomic number 8. It is a member of the chalcogen group in the periodic table, a highly reactive nonmetal, and an oxidizing agent that readily forms oxides with most elements as wel ...
(30.1%), silicon
Silicon is a chemical element with the symbol Si and atomic number 14. It is a hard, brittle crystalline solid with a blue-grey metallic luster, and is a tetravalent metalloid and semiconductor. It is a member of group 14 in the periodic tab ...
(15.1%), magnesium
Magnesium is a chemical element with the symbol Mg and atomic number 12. It is a shiny gray metal having a low density, low melting point and high chemical reactivity. Like the other alkaline earth metals (group 2 of the periodic ta ...
(13.9%), sulfur
Sulfur (or sulphur in British English) is a chemical element with the symbol S and atomic number 16. It is abundant, multivalent and nonmetallic. Under normal conditions, sulfur atoms form cyclic octatomic molecules with a chemical formula ...
(2.9%), nickel
Nickel is a chemical element with symbol Ni and atomic number 28. It is a silvery-white lustrous metal with a slight golden tinge. Nickel is a hard and ductile transition metal. Pure nickel is chemically reactive but large pieces are slow to ...
(1.8%), calcium
Calcium is a chemical element with the symbol Ca and atomic number 20. As an alkaline earth metal, calcium is a reactive metal that forms a dark oxide-nitride layer when exposed to air. Its physical and chemical properties are most similar to ...
(1.5%), and aluminium
Aluminium (aluminum in American and Canadian English) is a chemical element with the symbol Al and atomic number 13. Aluminium has a density lower than those of other common metals, at approximately one third that of steel. I ...
(1.4%); with the remaining 1.2% consisting of trace amounts of other elements. Due to planetary differentiation
In planetary science, planetary differentiation is the process by which the chemical elements of a planetary body accumulate in different areas of that body, due to their physical or chemical behavior (e.g. density and chemical affinities). The ...
, the core region is believed to be primarily composed of iron (88.8%), with smaller amounts of nickel (5.8%), sulfur (4.5%), and less than 1% trace elements.lithophiles
Lithophiles are micro-organisms that can live within the pore interstices of sedimentary and even fractured igneous rocks to depths of several kilometers.
Some are known to live on surface rocks, and make use of photosynthesis for energy.
Thos ...
and therefore remain close to the Earth's surface because they combine readily with oxygen
Oxygen is the chemical element with the symbol O and atomic number 8. It is a member of the chalcogen group in the periodic table, a highly reactive nonmetal, and an oxidizing agent that readily forms oxides with most elements as wel ...
and so associate strongly with silica
Silicon dioxide, also known as silica, is an oxide of silicon with the chemical formula , most commonly found in nature as quartz and in various living organisms. In many parts of the world, silica is the major constituent of sand. Silica is one ...
, forming relatively low-density minerals that do not sink down into the Earth's core. Potassium, rubidium and caesium are also incompatible element In petrology and geochemistry, an incompatible element is one that is unsuitable in size and/or charge to the cation sites of the minerals of which it is included. It is defined by the partition coefficient between rock-forming minerals and melt b ...
s due to their large ionic radii
Ionic radius, ''r''ion, is the radius of a monatomic ion in an ionic crystal structure. Although neither atoms nor ions have sharp boundaries, they are treated as if they were hard spheres with radii such that the sum of ionic radii of the cation ...
.Earth
Earth is the third planet from the Sun and the only astronomical object known to harbor life. While large volumes of water can be found throughout the Solar System, only Earth sustains liquid surface water. About 71% of Earth's surfa ...
's crust measured by weight, making it the sixth most abundant element overallhalite
Halite (), commonly known as rock salt, is a type of salt, the mineral (natural) form of sodium chloride ( Na Cl). Halite forms isometric crystals. The mineral is typically colorless or white, but may also be light blue, dark blue, purple, p ...
, amphibole
Amphibole () is a group of inosilicate minerals, forming prism or needlelike crystals, composed of double chain tetrahedra, linked at the vertices and generally containing ions of iron and/or magnesium in their structures. Its IMA symbol is A ...
, cryolite
Cryolite ( Na3 Al F6, sodium hexafluoroaluminate) is an uncommon mineral identified with the once-large deposit at Ivittuut on the west coast of Greenland, mined commercially until 1987.
History
Cryolite was first described in 1798 by Danish vete ...
, nitratine
Nitratine or nitratite, also known as cubic niter (UK: nitre), soda niter or Chile saltpeter (UK: Chile saltpetre), is a mineral, the naturally occurring form of sodium nitrate, NaNO3. Chemically it is the sodium analogue of saltpeter. Nitratine ...
, and zeolite
Zeolites are microporous, crystalline aluminosilicate materials commonly used as commercial adsorbents and catalysts. They mainly consist of silicon, aluminium, oxygen, and have the general formula ・y where is either a metal ion or H+. These pos ...
.Utah
Utah ( , ) is a state in the Mountain West subregion of the Western United States. Utah is a landlocked U.S. state bordered to its east by Colorado, to its northeast by Wyoming, to its north by Idaho, to its south by Arizona, and to it ...
's Great Salt Lake
The Great Salt Lake is the largest saltwater lake in the Western Hemisphere and the eighth-largest terminal lake in the world. It lies in the northern part of the U.S. state of Utah and has a substantial impact upon the local climate, particula ...
and the Dead Sea
The Dead Sea ( he, יַם הַמֶּלַח, ''Yam hamMelaḥ''; ar, اَلْبَحْرُ الْمَيْتُ, ''Āl-Baḥrū l-Maytū''), also known by other names, is a salt lake bordered by Jordan to the east and Israel and the West Bank ...
.micromolar
Molar concentration (also called molarity, amount concentration or substance concentration) is a measure of the concentration of a chemical species, in particular of a solute in a solution, in terms of amount of substance per unit volume of sol ...
. Its diagonal relationship with magnesium often allows it to replace magnesium in ferromagnesium minerals, where its crustal concentration is about 18 ppm, comparable to that of gallium
Gallium is a chemical element with the symbol Ga and atomic number 31. Discovered by French chemist Paul-Émile Lecoq de Boisbaudran in 1875, Gallium is in group 13 of the periodic table and is similar to the other metals of the group (aluminiu ...
and niobium
Niobium is a chemical element with chemical symbol Nb (formerly columbium, Cb) and atomic number 41. It is a light grey, crystalline, and ductile transition metal. Pure niobium has a Mohs hardness rating similar to pure titanium, and it has sim ...
. Commercially, the most important lithium mineral is spodumene
Spodumene is a pyroxene mineral consisting of lithium aluminium inosilicate, Li Al( Si O3)2, and is a source of lithium. It occurs as colorless to yellowish, purplish, or lilac kunzite (see below), yellowish-green or emerald-green hiddenite, pri ...
, which occurs in large deposits worldwide.zinc
Zinc is a chemical element with the symbol Zn and atomic number 30. Zinc is a slightly brittle metal at room temperature and has a shiny-greyish appearance when oxidation is removed. It is the first element in group 12 (IIB) of the periodi ...
and more abundant than copper. It occurs naturally in the minerals leucite
Leucite is a rock-forming mineral of the feldspathoid group, silica-undersaturated and composed of potassium and aluminium tectosilicate KAlSi2O6. Crystals have the form of cubic icositetrahedra but, as first observed by Sir David Brewster in 18 ...
, pollucite
Pollucite is a zeolite mineral with the formula with iron, calcium, rubidium and potassium as common substituting elements. It is important as a significant ore of caesium and sometimes rubidium. It forms a solid solution series with analcime. I ...
, carnallite
Carnallite (also carnalite) is an evaporite mineral, a hydrated potassium magnesium chloride with formula KMgCl3·6(H2O). It is variably colored yellow to white, reddish, and sometimes colorless or blue. It is usually massive to fibrous with rare ...
, zinnwaldite
Zinnwaldite, K Li Fe Al(Al Si3) O10(O H, F)2, potassium lithium iron aluminium silicate hydroxide fluoride is a silicate mineral in the mica group. The IMA status is as a series between siderophyllite (KFe2Al(Al2Si2)O10(F,OH)2) and polylithion ...
, and lepidolite
Lepidolite is a lilac-gray or rose-colored member of the mica group of minerals with chemical formula . It is the most abundant lithium-bearing mineral and is a secondary source of this metal. It is the major source of the alkali metal rubidium.
...
, although none of these contain only rubidium and no other alkali metals.antimony
Antimony is a chemical element with the symbol Sb (from la, stibium) and atomic number 51. A lustrous gray metalloid, it is found in nature mainly as the sulfide mineral stibnite (Sb2S3). Antimony compounds have been known since ancient time ...
, cadmium
Cadmium is a chemical element with the symbol Cd and atomic number 48. This soft, silvery-white metal is chemically similar to the two other stable metals in group 12, zinc and mercury. Like zinc, it demonstrates oxidation state +2 in most of ...
, tin
Tin is a chemical element with the symbol Sn (from la, stannum) and atomic number 50. Tin is a silvery-coloured metal.
Tin is soft enough to be cut with little force and a bar of tin can be bent by hand with little effort. When bent, t ...
, and tungsten
Tungsten, or wolfram, is a chemical element with the symbol W and atomic number 74. Tungsten is a rare metal found naturally on Earth almost exclusively as compounds with other elements. It was identified as a new element in 1781 and first isolat ...
, but is much less abundant than rubidium.Francium-223
Francium (87Fr) has no stable isotopes. A standard atomic weight cannot be given. Its most stable isotope is 223Fr with a half-life of 22 minutes, occurring in trace quantities as an intermediate decay product of 235U.
Of elements whose most sta ...
, the only naturally occurring isotope of francium,product
Product may refer to:
Business
* Product (business), an item that serves as a solution to a specific consumer problem.
* Product (project management), a deliverable or set of deliverables that contribute to a business solution
Mathematics
* Produ ...
of the alpha decay
Alpha decay or α-decay is a type of radioactive decay in which an atomic nucleus emits an alpha particle (helium nucleus) and thereby transforms or 'decays' into a different atomic nucleus, with a mass number that is reduced by four and an atom ...
of actinium-227 and can be found in trace amounts in uranium
Uranium is a chemical element with the symbol U and atomic number 92. It is a silvery-grey metal in the actinide series of the periodic table. A uranium atom has 92 protons and 92 electrons, of which 6 are valence electrons. Uranium is weak ...
minerals.earth's crust
Earth's crust is Earth's thin outer shell of rock, referring to less than 1% of Earth's radius and volume. It is the top component of the lithosphere, a division of Earth's layers that includes the crust and the upper part of the mantle. The ...
at any time, due to its extremely short half-life
Half-life (symbol ) is the time required for a quantity (of substance) to reduce to half of its initial value. The term is commonly used in nuclear physics to describe how quickly unstable atoms undergo radioactive decay or how long stable ato ...
of 22 minutes.
Properties
Physical and chemical
The physical and chemical properties of the alkali metals can be readily explained by their having an ns1 valence electron configuration
In atomic physics and quantum chemistry, the electron configuration is the distribution of electrons of an atom or molecule (or other physical structure) in atomic or molecular orbitals. For example, the electron configuration of the neon atom ...
, which results in weak metallic bonding
Metallic bonding is a type of chemical bonding that arises from the electrostatic attractive force between conduction electrons (in the form of an electron cloud of delocalized electrons) and positively charged metal ions. It may be descri ...
. Hence, all the alkali metals are soft and have low densities
Density (volumetric mass density or specific mass) is the substance's mass per unit of volume. The symbol most often used for density is ''ρ'' (the lower case Greek language, Greek letter Rho (letter), rho), although the Latin letter ''D'' ca ...
,melting
Melting, or fusion, is a physical process that results in the phase transition of a substance from a solid to a liquid. This occurs when the internal energy of the solid increases, typically by the application of heat or pressure, which incre ...
boiling point
The boiling point of a substance is the temperature at which the vapor pressure of a liquid equals the pressure surrounding the liquid and the liquid changes into a vapor.
The boiling point of a liquid varies depending upon the surrounding envir ...
s,vaporisation
Vaporization (or vaporisation) of an element or compound is a phase transition from the liquid phase to vapor. There are two types of vaporization: evaporation and boiling. Evaporation is a surface phenomenon, whereas boiling is a bulk phenomenon ...
, and dissociation
Dissociation, in the wide sense of the word, is an act of disuniting or separating a complex object into parts. Dissociation may also refer to:
* Dissociation (chemistry), general process in which molecules or ionic compounds (complexes, or salts) ...
.body-centered cubic
In crystallography, the cubic (or isometric) crystal system is a crystal system where the unit cell is in the shape of a cube. This is one of the most common and simplest shapes found in crystals and minerals.
There are three main varieties of ...
crystal structure,ionic radii
Ionic radius, ''r''ion, is the radius of a monatomic ion in an ionic crystal structure. Although neither atoms nor ions have sharp boundaries, they are treated as if they were hard spheres with radii such that the sum of ionic radii of the cation ...
, as well as very high thermal
A thermal column (or thermal) is a rising mass of buoyant air, a convective current in the atmosphere, that transfers heat energy vertically. Thermals are created by the uneven heating of Earth's surface from solar radiation, and are an example ...
and electrical conductivity
Electrical resistivity (also called specific electrical resistance or volume resistivity) is a fundamental property of a material that measures how strongly it resists electric current. A low resistivity indicates a material that readily allow ...
.group
A group is a number of persons or things that are located, gathered, or classed together.
Groups of people
* Cultural group, a group whose members share the same cultural identity
* Ethnic group, a group whose members share the same ethnic ide ...
are to each other.ionic radii
Ionic radius, ''r''ion, is the radius of a monatomic ion in an ionic crystal structure. Although neither atoms nor ions have sharp boundaries, they are treated as if they were hard spheres with radii such that the sum of ionic radii of the cation ...
; lithium and sodium are more distinct. For instance, when moving down the table, all known alkali metals show increasing atomic radius
The atomic radius of a chemical element is a measure of the size of its atom, usually the mean or typical distance from the center of the nucleus to the outermost isolated electron. Since the boundary is not a well-defined physical entity, there ...
,electronegativity
Electronegativity, symbolized as , is the tendency for an atom of a given chemical element to attract shared electrons (or electron density) when forming a chemical bond. An atom's electronegativity is affected by both its atomic number and the d ...
,densities
Density (volumetric mass density or specific mass) is the substance's mass per unit of volume. The symbol most often used for density is ''ρ'' (the lower case Greek language, Greek letter Rho (letter), rho), although the Latin letter ''D'' ca ...
increase when moving down the table, with the exception that potassium is less dense than sodium.reduction potential
Redox potential (also known as oxidation / reduction potential, ''ORP'', ''pe'', ''E_'', or E_) is a measure of the tendency of a chemical species to acquire electrons from or lose electrons to an electrode and thereby be reduced or oxidised respe ...
s: lithium's value is anomalous, being more negative than the others.hydration energy
In chemistry, hydration energy (also hydration enthalpy) is the amount of energy released when one mole of ions undergoes hydration. Hydration energy is one component in the quantitative analysis of solvation. It is a particular special case of ...
in the gas phase: though the lithium ion disrupts the structure of water significantly, causing a higher change in entropy, this high hydration energy is enough to make the reduction potentials indicate it as being the most electropositive alkali metal, despite the difficulty of ionising it in the gas phase.alkaline earth metal
The alkaline earth metals are six chemical elements in group 2 of the periodic table. They are beryllium (Be), magnesium (Mg), calcium (Ca), strontium (Sr), barium (Ba), and radium (Ra).. The elements have very similar properties: they are al ...
s calcium
Calcium is a chemical element with the symbol Ca and atomic number 20. As an alkaline earth metal, calcium is a reactive metal that forms a dark oxide-nitride layer when exposed to air. Its physical and chemical properties are most similar to ...
, strontium
Strontium is the chemical element with the symbol Sr and atomic number 38. An alkaline earth metal, strontium is a soft silver-white yellowish metallic element that is highly chemically reactive. The metal forms a dark oxide layer when it is ex ...
, and barium
Barium is a chemical element with the symbol Ba and atomic number 56. It is the fifth element in group 2 and is a soft, silvery alkaline earth metal. Because of its high chemical reactivity, barium is never found in nature as a free element.
Th ...
, as well as the divalent lanthanide
The lanthanide () or lanthanoid () series of chemical elements comprises the 15 metallic chemical elements with atomic numbers 57–71, from lanthanum through lutetium. These elements, along with the chemically similar elements scandium and yttr ...
s europium
Europium is a chemical element with the symbol Eu and atomic number 63. Europium is the most reactive lanthanide by far, having to be stored under an inert fluid to protect it from atmospheric oxygen or moisture. Europium is also the softest lanth ...
and ytterbium
Ytterbium is a chemical element with the symbol Yb and atomic number 70. It is a metal, the fourteenth and penultimate element in the lanthanide series, which is the basis of the relative stability of its +2 oxidation state. However, like the othe ...
, are pale yellow, though the colour is much less prominent than it is for caesium.body-centered cubic
In crystallography, the cubic (or isometric) crystal system is a crystal system where the unit cell is in the shape of a cube. This is one of the most common and simplest shapes found in crystals and minerals.
There are three main varieties of ...
crystal structure,mineral oil
Mineral oil is any of various colorless, odorless, light mixtures of higher alkanes from a mineral source, particularly a distillate of petroleum, as distinct from usually edible vegetable oils.
The name 'mineral oil' by itself is imprecise, ...
or kerosene
Kerosene, paraffin, or lamp oil is a combustible hydrocarbon liquid which is derived from petroleum. It is widely used as a fuel in aviation as well as households. Its name derives from el, κηρός (''keros'') meaning "wax", and was regi ...
(paraffin oil).halogen
The halogens () are a group in the periodic table consisting of five or six chemically related elements: fluorine (F), chlorine (Cl), bromine (Br), iodine (I), astatine (At), and tennessine (Ts). In the modern IUPAC nomenclature, this group is ...
s to form the alkali metal halide
In chemistry, alkali metal halides, or alkali halides, are the family of inorganic compounds with the chemical formula MX, where M is an alkali metal and X is a halogen. These compounds are the often commercially significant sources of these m ...
s, which are white ionic crystal
In chemistry, an ionic crystal is a crystalline form of an ionic compound. They are solids consisting of ions bound together by their electrostatic attraction into a regular Crystal structure, lattice. Examples of such crystals are the alkali hal ...
line compounds that are all soluble
In chemistry, solubility is the ability of a substance, the solute, to form a solution with another substance, the solvent. Insolubility is the opposite property, the inability of the solute to form such a solution.
The extent of the solubil ...
in water except lithium fluoride
Lithium fluoride is an inorganic compound with the chemical formula LiF. It is a colorless solid, that transitions to white with decreasing crystal size. Although odorless, lithium fluoride has a bitter-saline taste. Its structure is analogous to ...
( Li F).alkali
In chemistry, an alkali (; from ar, القلوي, al-qaly, lit=ashes of the saltwort) is a basic, ionic salt of an alkali metal or an alkaline earth metal. An alkali can also be defined as a base that dissolves in water. A solution of a ...
ne hydroxide
Hydroxide is a diatomic anion with chemical formula OH−. It consists of an oxygen and hydrogen atom held together by a single covalent bond, and carries a negative electric charge. It is an important but usually minor constituent of water. I ...
s and thus should be handled with great care. The heavier alkali metals react more vigorously than the lighter ones; for example, when dropped into water, caesium produces a larger explosion than potassium if the same number of moles of each metal is used.periodic table
The periodic table, also known as the periodic table of the (chemical) elements, is a rows and columns arrangement of the chemical elements. It is widely used in chemistry, physics, and other sciences, and is generally seen as an icon of ch ...
effective nuclear charge In atomic physics, the effective nuclear charge is the actual amount of positive (nuclear) charge experienced by an electron in a multi-electron atom. The term "effective" is used because the shielding effect of negatively charged electrons prevent ...
noble gas
The noble gases (historically also the inert gases; sometimes referred to as aerogens) make up a class of chemical elements with similar properties; under standard conditions, they are all odorless, colorless, monatomic gases with very low chemi ...
configuration by losing just one electron
The electron ( or ) is a subatomic particle with a negative one elementary electric charge. Electrons belong to the first generation of the lepton particle family,
and are generally thought to be elementary particles because they have no kn ...
.alcohol
Alcohol most commonly refers to:
* Alcohol (chemistry), an organic compound in which a hydroxyl group is bound to a carbon atom
* Alcohol (drug), an intoxicant found in alcoholic drinks
Alcohol may also refer to:
Chemicals
* Ethanol, one of sev ...
s and phenols
In organic chemistry, phenols, sometimes called phenolics, are a class of chemical compounds consisting of one or more hydroxyl groups (— O H) bonded directly to an aromatic hydrocarbon group. The simplest is phenol, . Phenolic compounds are c ...
, gaseous ammonia
Ammonia is an inorganic compound of nitrogen and hydrogen with the formula . A stable binary hydride, and the simplest pnictogen hydride, ammonia is a colourless gas with a distinct pungent smell. Biologically, it is a common nitrogenous was ...
, and alkyne
\ce
\ce
Acetylene
\ce
\ce
\ce
Propyne
\ce
\ce
\ce
\ce
1-Butyne
In organic chemistry, an alkyne is an unsaturated hydrocarbon containing at least one carbon—carbon triple bond. The simplest acyclic alkynes with only one triple bond and n ...
s, the last demonstrating the phenomenal degree of their reactivity. Their great power as reducing agents makes them very useful in liberating other metals from their oxides or halides.alkalide An alkalide is a chemical compound in which alkali metal atoms are anions (negative ions) with a charge or oxidation state of −1. Until the first discovery of alkalides in the 1970s, alkali metals were known to appear in salts only as cations ...
s are an exception: they are unstable compounds which contain alkali metals in a −1 oxidation state, which is very unusual as before the discovery of the alkalides, the alkali metals were not expected to be able to form anion
An ion () is an atom or molecule with a net electrical charge.
The charge of an electron is considered to be negative by convention and this charge is equal and opposite to the charge of a proton, which is considered to be positive by convent ...
s and were thought to be able to appear in salts
In chemistry, a salt is a chemical compound consisting of an ionic assembly of positively charged cations and negatively charged anions, which results in a compound with no net electric charge. A common example is table salt, with positively cha ...
only as cations. The alkalide anions have filled s-subshells, which gives them enough stability to exist. All the stable alkali metals except lithium are known to be able to form alkalides, and the alkalides have much theoretical interest due to their unusual stoichiometry
Stoichiometry refers to the relationship between the quantities of reactants and products before, during, and following chemical reactions.
Stoichiometry is founded on the law of conservation of mass where the total mass of the reactants equal ...
and low ionisation potentials. Alkalides are chemically similar to the electride
An electride is an ionic compound in which an electron is the anion. Solutions of alkali metals in ammonia are electride salts. In the case of sodium, these blue solutions consist of a(NH3)6sup>+ and solvated electrons:
:Na + 6 NH3 → ...
s, which are salts with trapped electron
The electron ( or ) is a subatomic particle with a negative one elementary electric charge. Electrons belong to the first generation of the lepton particle family,
and are generally thought to be elementary particles because they have no kn ...
s acting as anions.sodium hydride
Sodium hydride is the chemical compound with the empirical formula Na H. This alkali metal hydride is primarily used as a strong yet combustible base in organic synthesis. NaH is a saline (salt-like) hydride, composed of Na+ and H− ions, in co ...
", H+Na− (both ions being complexed), as opposed to the usual sodium hydride, Na+H−:metastable
In chemistry and physics, metastability denotes an intermediate Energy level, energetic state within a dynamical system other than the system's ground state, state of least energy.
A ball resting in a hollow on a slope is a simple example of me ...
or stable.coordination number
In chemistry, crystallography, and materials science, the coordination number, also called ligancy, of a central atom in a molecule or crystal is the number of atoms, molecules or ions bonded to it. The ion/molecule/atom surrounding the central i ...
s and shapes agree well with those expected from their ionic radii. In aqueous solution the water molecules directly attached to the metal ion are said to belong to the first coordination sphere
In coordination chemistry, the first coordination sphere refers to the array of molecules and ions (the ligands) directly attached to the central metal atom. The second coordination sphere consists of molecules and ions that attached in various ...
, also known as the first, or primary, solvation shell. The bond between a water molecule and the metal ion is a dative covalent bond
In coordination chemistry, a coordinate covalent bond, also known as a dative bond, dipolar bond, or coordinate bond is a kind of two-center, two-electron covalent bond in which the two electrons derive from the same atom. The bonding of metal io ...
, with the oxygen atom donating both electrons to the bond. Each coordinated water molecule may be attached by hydrogen bond
In chemistry, a hydrogen bond (or H-bond) is a primarily electrostatic force of attraction between a hydrogen (H) atom which is covalently bound to a more electronegative "donor" atom or group (Dn), and another electronegative atom bearing a ...
s to other water molecules. The latter are said to reside in the second coordination sphere. However, for the alkali metal cations, the second coordination sphere is not well-defined as the +1 charge on the cation is not high enough to polarise the water molecules in the primary solvation shell enough for them to form strong hydrogen bonds with those in the second coordination sphere, producing a more stable entity.tetrahedral
In geometry, a tetrahedron (plural: tetrahedra or tetrahedrons), also known as a triangular pyramid, is a polyhedron composed of four triangular faces, six straight edges, and four vertex corners. The tetrahedron is the simplest of all the o ...
i(H2O)4sup>+: while solvation numbers of 3 to 6 have been found for lithium aqua ions, solvation numbers less than 4 may be the result of the formation of contact ion pair
In chemistry, ion association is a chemical reaction whereby ions of opposite electric charge come together in solution to form a distinct chemical entity. Ion associates are classified, according to the number of ions that associate with each o ...
s, and the higher solvation numbers may be interpreted in terms of water molecules that approach i(H2O)4sup>+ through a face of the tetrahedron, though molecular dynamic simulations may indicate the existence of an octahedral
In geometry, an octahedron (plural: octahedra, octahedrons) is a polyhedron with eight faces. The term is most commonly used to refer to the regular octahedron, a Platonic solid composed of eight equilateral triangles, four of which meet at ea ...
hexaaqua ion. There are also probably six water molecules in the primary solvation sphere of the sodium ion, forming the octahedral a(H2O)6sup>+ ion.square antiprism
In geometry, the square antiprism is the second in an infinite family of antiprisms formed by an even-numbered sequence of triangle sides closed by two polygon caps. It is also known as an ''anticube''.
If all its faces are regular, it is a sem ...
atic structure, and that caesium forms the 12-coordinate s(H2O)12sup>+ ion.
Lithium
The chemistry of lithium shows several differences from that of the rest of the group as the small Li+ cation polarises anion
An ion () is an atom or molecule with a net electrical charge.
The charge of an electron is considered to be negative by convention and this charge is equal and opposite to the charge of a proton, which is considered to be positive by convent ...
s and gives its compounds a more covalent
A covalent bond is a chemical bond that involves the sharing of electrons to form electron pairs between atoms. These electron pairs are known as shared pairs or bonding pairs. The stable balance of attractive and repulsive forces between atoms ...
character.magnesium
Magnesium is a chemical element with the symbol Mg and atomic number 12. It is a shiny gray metal having a low density, low melting point and high chemical reactivity. Like the other alkaline earth metals (group 2 of the periodic ta ...
have a diagonal relationship
A diagonal relationship is said to exist between certain pairs of diagonally adjacent elements in the second and third periods (first 20 elements) of the periodic table. These pairs (lithium (Li) and magnesium (Mg), beryllium (Be) and aluminium ...
due to their similar atomic radii,nitride
In chemistry, a nitride is an inorganic compound of nitrogen. The "nitride" anion, N3- ion, is very elusive but compounds of nitride are numerous, although rarely naturally occuring. Some nitrides have a find applications, such as wear-resistant ...
, a property common among all the alkaline earth metal
The alkaline earth metals are six chemical elements in group 2 of the periodic table. They are beryllium (Be), magnesium (Mg), calcium (Ca), strontium (Sr), barium (Ba), and radium (Ra).. The elements have very similar properties: they are al ...
s (magnesium's group) but unique among the alkali metals.organometallic compound
Organometallic chemistry is the study of organometallic compounds, chemical compounds containing at least one chemical bond between a carbon atom of an organic molecule and a metal, including alkali, alkaline earth, and transition metals, and so ...
s with significant covalent character (e.g. Li Me and MgMe2).lithium hydroxide
Lithium hydroxide is an inorganic compound with the formula LiOH. It can exist as anhydrous or hydrated, and both forms are white hygroscopic solids. They are soluble in water and slightly soluble in ethanol. Both are available commercially. While ...
is the only alkali metal hydroxide that is not deliquescent
Hygroscopy is the phenomenon of attracting and holding water molecules via either absorption or adsorption from the surrounding environment, which is usually at normal or room temperature. If water molecules become suspended among the substance ...
.lithium perchlorate
Lithium perchlorate is the inorganic compound with the formula LiClO4. This white or colourless crystalline salt is noteworthy for its high solubility in many solvents. It exists both in anhydrous form and as a trihydrate.
Applications Inorga ...
and other lithium salts with large anions that cannot be polarised are much more stable than the analogous compounds of the other alkali metals, probably because Li+ has a high solvation energy
Solvation (or dissolution) describes the interaction of a solvent with dissolved molecules. Both ionized and uncharged molecules interact strongly with a solvent, and the strength and nature of this interaction influence many properties of the ...
.hygroscopic
Hygroscopy is the phenomenon of attracting and holding water molecules via either absorption or adsorption from the surrounding environment, which is usually at normal or room temperature. If water molecules become suspended among the substance ...
: this allows salts like lithium chloride
Lithium chloride is a chemical compound with the formula Li Cl. The salt is a typical ionic compound (with certain covalent characteristics), although the small size of the Li+ ion gives rise to properties not seen for other alkali metal chlor ...
and lithium bromide
Lithium bromide (LiBr) is a chemical compound of lithium and bromine. Its extreme hygroscopic character makes LiBr useful as a desiccant in certain air conditioning systems.Wietelmann, Ulrich and Bauer, Richard J. (2005) "Lithium and Lithium Compo ...
to be used in dehumidifier
A dehumidifier is an air conditioning device which reduces and maintains the level of humidity in the air. This is done usually for health or thermal comfort reasons, or to eliminate musty odor and to prevent the growth of mildew by extracting w ...
s and air-conditioner
Air conditioning, often abbreviated as A/C or AC, is the process of removing heat from an enclosed space to achieve a more comfortable interior environment (sometimes referred to as 'comfort cooling') and in some cases also strictly controlling ...
s.
Francium
Francium is also predicted to show some differences due to its high atomic weight
Relative atomic mass (symbol: ''A''; sometimes abbreviated RAM or r.a.m.), also known by the deprecated synonym atomic weight, is a dimensionless physical quantity defined as the ratio of the average mass of atoms of a chemical element in a giv ...
, causing its electrons to travel at considerable fractions of the speed of light and thus making relativistic effects
Relativistic quantum chemistry combines relativistic mechanics with quantum chemistry to calculate elemental properties and structure, especially for the heavier elements of the periodic table. A prominent example is an explanation for the color of ...
more prominent. In contrast to the trend of decreasing electronegativities
Electronegativity, symbolized as , is the tendency for an atom of a given chemical element to attract shared electrons (or electron density) when forming a chemical bond. An atom's electronegativity is affected by both its atomic number and the d ...
and ionisation energies of the alkali metals, francium's electronegativity and ionisation energy are predicted to be higher than caesium's due to the relativistic stabilisation of the 7s electrons; also, its atomic radius
The atomic radius of a chemical element is a measure of the size of its atom, usually the mean or typical distance from the center of the nucleus to the outermost isolated electron. Since the boundary is not a well-defined physical entity, there ...
is expected to be abnormally low. Thus, contrary to expectation, caesium is the most reactive of the alkali metals, not francium.
Nuclear
All the alkali metals have odd atomic numbers; hence, their isotopes must be either odd–odd (both proton and neutron number
The neutron number, symbol ''N'', is the number of neutrons in a nuclide.
Atomic number (proton number) plus neutron number equals mass number: . The difference between the neutron number and the atomic number is known as the neutron excess: . ...
are odd) or odd–even (proton number
The atomic number or nuclear charge number (symbol ''Z'') of a chemical element is the charge number of an atomic nucleus. For ordinary nuclei, this is equal to the proton number (''n''p) or the number of protons found in the nucleus of every ...
is odd, but neutron number is even). Odd–odd nuclei have even mass number
The mass number (symbol ''A'', from the German word ''Atomgewicht'' tomic weight, also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. It is approxima ...
s, whereas odd–even nuclei have odd mass numbers. Odd–odd primordial nuclide
In geochemistry, geophysics and nuclear physics, primordial nuclides, also known as primordial isotopes, are nuclides found on Earth that have existed in their current form since before Earth was formed. Primordial nuclides were present in the ...
s are rare because most odd–odd nuclei are highly unstable with respect to beta decay
In nuclear physics, beta decay (β-decay) is a type of radioactive decay in which a beta particle (fast energetic electron or positron) is emitted from an atomic nucleus, transforming the original nuclide to an isobar of that nuclide. For ...
, because the decay products are even–even, and are therefore more strongly bound, due to nuclear pairing effects.radioisotope
A radionuclide (radioactive nuclide, radioisotope or radioactive isotope) is a nuclide that has excess nuclear energy, making it unstable. This excess energy can be used in one of three ways: emitted from the nucleus as gamma radiation; transferr ...
potassium-40). For a given odd mass number, there can be only a single beta-stable nuclide, since there is not a difference in binding energy between even–odd and odd–even comparable to that between even–even and odd–odd, leaving other nuclides of the same mass number ( isobars) free to beta decay
In nuclear physics, beta decay (β-decay) is a type of radioactive decay in which a beta particle (fast energetic electron or positron) is emitted from an atomic nucleus, transforming the original nuclide to an isobar of that nuclide. For ...
toward the lowest-mass nuclide. An effect of the instability of an odd number of either type of nucleons is that odd-numbered elements, such as the alkali metals, tend to have fewer stable isotopes than even-numbered elements. Of the 26 monoisotopic element
A monoisotopic element is an element which has only a single stable isotope (nuclide). There are only 26 elements that have this property. A list is given in a following section.
Stability is experimentally defined for chemical elements, as ther ...
s that have only a single stable isotope, all but one have an odd atomic number and all but one also have an even number of neutrons. Beryllium
Beryllium is a chemical element with the symbol Be and atomic number 4. It is a steel-gray, strong, lightweight and brittle alkaline earth metal. It is a divalent element that occurs naturally only in combination with other elements to form mi ...
is the single exception to both rules, due to its low atomic number.radioisotope
A radionuclide (radioactive nuclide, radioisotope or radioactive isotope) is a nuclide that has excess nuclear energy, making it unstable. This excess energy can be used in one of three ways: emitted from the nucleus as gamma radiation; transferr ...
: sodium-22
There are 22 isotopes of sodium (11Na), ranging from to , and two isomers ( and ). is the only stable (and the only primordial) isotope. It is considered a monoisotopic element and it has a standard atomic weight of . Sodium has two radioact ...
and sodium-24
There are 22 isotopes of sodium (11Na), ranging from to , and two isomers ( and ). is the only stable (and the only primordial) isotope. It is considered a monoisotopic element and it has a standard atomic weight of . Sodium has two radioactive ...
are trace radioisotope
A trace radioisotope is a radioisotope that occurs naturally in trace amounts (i.e. extremely small). Generally speaking, trace radioisotopes have half-lives that are short in comparison with the age of the Earth, since primordial nuclides tend to ...
s produced cosmogenic
Cosmogenic nuclides (or cosmogenic isotopes) are rare nuclides (isotopes) created when a high-energy cosmic ray interacts with the nucleus of an ''in situ'' Solar System atom, causing nucleons (protons and neutrons) to be expelled from the atom ...
ally, potassium-40 and rubidium-87
Rubidium (37Rb) has 36 isotopes, with naturally occurring rubidium being composed of just two isotopes; 85Rb (72.2%) and the radioactive 87Rb (27.8%). Normal mixes of rubidium are radioactive enough to fog photographic film in approximately 30 ...
have very long half-lives
Half-life (symbol ) is the time required for a quantity (of substance) to reduce to half of its initial value. The term is commonly used in nuclear physics to describe how quickly unstable atoms undergo radioactive decay or how long stable at ...
and thus occur naturally,isotopes of francium
Francium (87Fr) has no stable isotopes. A standard atomic weight cannot be given. Its most stable isotope is 223Fr with a half-life of 22 minutes, occurring in trace quantities as an intermediate decay product of 235U.
Of elements whose most sta ...
are radioactive
Radioactive decay (also known as nuclear decay, radioactivity, radioactive disintegration, or nuclear disintegration) is the process by which an unstable atomic nucleus loses energy by radiation. A material containing unstable nuclei is consid ...
.Caesium-137
Caesium-137 (), cesium-137 (US), or radiocaesium, is a radioactive isotope of caesium that is formed as one of the more common fission products by the nuclear fission of uranium-235 and other fissionable isotopes in nuclear reactors and nucl ...
, with a half-life of 30.17 years, is one of the two principal medium-lived fission product
Long-lived fission products (LLFPs) are radioactive materials with a long half-life (more than 200,000 years) produced by nuclear fission of uranium and plutonium. Because of their persistent radiotoxicity it is necessary to isolate them from man ...
s, along with strontium-90
Strontium-90 () is a radioactive isotope of strontium produced by nuclear fission, with a half-life of 28.8 years. It undergoes β− decay into yttrium-90, with a decay energy of 0.546 MeV. Strontium-90 has applications in medicine and i ...
, which are responsible for most of the radioactivity of spent nuclear fuel
Spent nuclear fuel, occasionally called used nuclear fuel, is nuclear fuel that has been irradiated in a nuclear reactor (usually at a nuclear power plant). It is no longer useful in sustaining a nuclear reaction in an ordinary thermal reactor an ...
after several years of cooling, up to several hundred years after use. It constitutes most of the radioactivity still left from the Chernobyl accident
The Chernobyl disaster was a nuclear accident that occurred on 26 April 1986 at the No. 4 reactor in the Chernobyl Nuclear Power Plant, near the city of Pripyat in the north of the Ukrainian SSR in the Soviet Union. It is one of only two nu ...
. Caesium-137 undergoes high-energy beta decay and eventually becomes stable barium-137
Naturally occurring barium (56Ba) is a mix of six stable isotopes and one very long-lived radioactive primordial isotope, barium-130, identified as being unstable by geochemical means (from analysis of the presence of its daughter xenon-130 in roc ...
. It is a strong emitter of gamma radiation. Caesium-137 has a very low rate of neutron capture and cannot be feasibly disposed of in this way, but must be allowed to decay.tritium
Tritium ( or , ) or hydrogen-3 (symbol T or H) is a rare and radioactive isotope of hydrogen with half-life about 12 years. The nucleus of tritium (t, sometimes called a ''triton'') contains one proton and two neutrons, whereas the nucleus o ...
. Small amounts of caesium-134
Caesium (55Cs) has 40 known isotopes, making it, along with barium and mercury, one of the elements with the most isotopes. The atomic masses of these isotopes range from 112 to 151. Only one isotope, 133Cs, is stable. The longest-lived radioisot ...
and caesium-137 were released into the environment during nearly all nuclear weapon test
Nuclear weapons tests are experiments carried out to determine nuclear weapons' effectiveness, yield, and explosive capability. Testing nuclear weapons offers practical information about how the weapons function, how detonations are affected by ...
s and some nuclear accident
A nuclear and radiation accident is defined by the International Atomic Energy Agency (IAEA) as "an event that has led to significant consequences to people, the environment or the facility. Examples include lethal effects to individuals, lar ...
s, most notably the Goiânia accident
The Goiânia accident was a radioactive contamination accident that occurred on September 13, 1987, in Goiânia, Goiás, Brazil, after a forgotten radiotherapy source was stolen from an abandoned hospital site in the city. It was subsequent ...
and the Chernobyl disaster
The Chernobyl disaster was a nuclear accident that occurred on 26 April 1986 at the No. 4 reactor in the Chernobyl Nuclear Power Plant, near the city of Pripyat in the north of the Ukrainian SSR in the Soviet Union. It is one of only two nuc ...
. As of 2005, caesium-137 is the principal source of radiation in the zone of alienation
The Chernobyl Nuclear Power Plant Zone of Alienation, Belarusian: Хона адчужэння Чарнобыльскай АЭС, ''Zona adčužennia Čarnobyĺskaj AES'', russian: Зона отчуждения Чернобыльской АЭС ...
around the Chernobyl nuclear power plant
The Chernobyl Nuclear Power Plant (ChNPP; ; ), is a nuclear power plant undergoing decommissioning. ChNPP is located near the abandoned city of Pripyat in northern Ukraine northwest of the city of Chernobyl, from the Belarus–Ukraine border, ...
.
Periodic trends
The alkali metals are more similar to each other than the elements in any other group
A group is a number of persons or things that are located, gathered, or classed together.
Groups of people
* Cultural group, a group whose members share the same cultural identity
* Ethnic group, a group whose members share the same ethnic ide ...
are to each other.atomic radius
The atomic radius of a chemical element is a measure of the size of its atom, usually the mean or typical distance from the center of the nucleus to the outermost isolated electron. Since the boundary is not a well-defined physical entity, there ...
,electronegativity
Electronegativity, symbolized as , is the tendency for an atom of a given chemical element to attract shared electrons (or electron density) when forming a chemical bond. An atom's electronegativity is affected by both its atomic number and the d ...
,densities
Density (volumetric mass density or specific mass) is the substance's mass per unit of volume. The symbol most often used for density is ''ρ'' (the lower case Greek language, Greek letter Rho (letter), rho), although the Latin letter ''D'' ca ...
increase when moving down the table, with the exception that potassium is less dense than sodium.
Atomic and ionic radii
 The atomic radii of the alkali metals increase going down the group.
The atomic radii of the alkali metals increase going down the group.shielding effect
In chemistry, the shielding effect sometimes referred to as atomic shielding or electron shielding describes the attraction between an electron and the nucleus in any atom with more than one electron. The shielding effect can be defined as a re ...
, when an atom has more than one electron shell
In chemistry and atomic physics, an electron shell may be thought of as an orbit followed by electrons around an atom's nucleus. The closest shell to the nucleus is called the "1 shell" (also called the "K shell"), followed by the "2 shell" (o ...
, each electron feels electric repulsion from the other electrons as well as electric attraction from the nucleus.nuclear charge In atomic physics, the effective nuclear charge is the actual amount of positive (nuclear) charge experienced by an electron in a multi-electron atom. The term "effective" is used because the shielding effect of negatively charged electrons prevent ...
(which is equal to the atomic number
The atomic number or nuclear charge number (symbol ''Z'') of a chemical element is the charge number of an atomic nucleus. For ordinary nuclei, this is equal to the proton number (''n''p) or the number of protons found in the nucleus of every ...
) is cancelled by the inner electrons; the number of inner electrons of an alkali metal is always one less than the nuclear charge. Therefore, the only factor which affects the atomic radius of the alkali metals is the number of electron shells. Since this number increases down the group, the atomic radius must also increase down the group.ionic radii
Ionic radius, ''r''ion, is the radius of a monatomic ion in an ionic crystal structure. Although neither atoms nor ions have sharp boundaries, they are treated as if they were hard spheres with radii such that the sum of ionic radii of the cation ...
of the alkali metals are much smaller than their atomic radii. This is because the outermost electron of the alkali metals is in a different electron shell
In chemistry and atomic physics, an electron shell may be thought of as an orbit followed by electrons around an atom's nucleus. The closest shell to the nucleus is called the "1 shell" (also called the "K shell"), followed by the "2 shell" (o ...
than the inner electrons, and thus when it is removed the resulting atom has one fewer electron shell and is smaller. Additionally, the effective nuclear charge In atomic physics, the effective nuclear charge is the actual amount of positive (nuclear) charge experienced by an electron in a multi-electron atom. The term "effective" is used because the shielding effect of negatively charged electrons prevent ...
has increased, and thus the electrons are attracted more strongly towards the nucleus and the ionic radius decreases.
First ionisation energy
 The first ionisation energy of an element or
The first ionisation energy of an element or molecule
A molecule is a group of two or more atoms held together by attractive forces known as chemical bonds; depending on context, the term may or may not include ions which satisfy this criterion. In quantum physics, organic chemistry, and bioch ...
is the energy required to move the most loosely held electron from one mole
Mole (or Molé) may refer to:
Animals
* Mole (animal) or "true mole", mammals in the family Talpidae, found in Eurasia and North America
* Golden moles, southern African mammals in the family Chrysochloridae, similar to but unrelated to Talpida ...
of gaseous atoms of the element or molecules to form one mole of gaseous ions with electric charge
Electric charge is the physical property of matter that causes charged matter to experience a force when placed in an electromagnetic field. Electric charge can be ''positive'' or ''negative'' (commonly carried by protons and electrons respe ...
+1. The factors affecting the first ionisation energy are the nuclear charge In atomic physics, the effective nuclear charge is the actual amount of positive (nuclear) charge experienced by an electron in a multi-electron atom. The term "effective" is used because the shielding effect of negatively charged electrons prevent ...
, the amount of shielding by the inner electrons and the distance from the most loosely held electron from the nucleus, which is always an outer electron in main group elements. The first two factors change the effective nuclear charge the most loosely held electron feels. Since the outermost electron of alkali metals always feels the same effective nuclear charge (+1), the only factor which affects the first ionisation energy is the distance from the outermost electron to the nucleus. Since this distance increases down the group, the outermost electron feels less attraction from the nucleus and thus the first ionisation energy decreases.electron shell
In chemistry and atomic physics, an electron shell may be thought of as an orbit followed by electrons around an atom's nucleus. The closest shell to the nucleus is called the "1 shell" (also called the "K shell"), followed by the "2 shell" (o ...
and is thus difficult to remove.
Reactivity
The reactivities of the alkali metals increase going down the group. This is the result of a combination of two factors: the first ionisation energies and atomisation energies of the alkali metals. Because the first ionisation energy of the alkali metals decreases down the group, it is easier for the outermost electron to be removed from the atom and participate in chemical reaction
A chemical reaction is a process that leads to the IUPAC nomenclature for organic transformations, chemical transformation of one set of chemical substances to another. Classically, chemical reactions encompass changes that only involve the pos ...
s, thus increasing reactivity down the group. The atomisation energy measures the strength of the metallic bond
Metallic bonding is a type of chemical bonding that arises from the electrostatic attractive force between conduction electrons (in the form of an electron cloud of delocalized electrons) and positively charged metal ions. It may be descri ...
of an element, which falls down the group as the atoms increase in radius
In classical geometry, a radius ( : radii) of a circle or sphere is any of the line segments from its center to its perimeter, and in more modern usage, it is also their length. The name comes from the latin ''radius'', meaning ray but also the ...
and thus the metallic bond must increase in length, making the delocalised electrons further away from the attraction of the nuclei of the heavier alkali metals. Adding the atomisation and first ionisation energies gives a quantity closely related to (but not equal to) the activation energy
In chemistry and physics, activation energy is the minimum amount of energy that must be provided for compounds to result in a chemical reaction. The activation energy (''E''a) of a reaction is measured in joules per mole (J/mol), kilojoules pe ...
of the reaction of an alkali metal with another substance. This quantity decreases going down the group, and so does the activation energy; thus, chemical reactions can occur faster and the reactivity increases down the group.
Electronegativity
 Electronegativity is a
Electronegativity is a chemical property
A chemical property is any of a material's properties that becomes evident during, or after, a chemical reaction; that is, any quality that can be established only by changing a substance's chemical identity.William L. Masterton, Cecile N. Hurley ...
that describes the tendency of an atom
Every atom is composed of a nucleus and one or more electrons bound to the nucleus. The nucleus is made of one or more protons and a number of neutrons. Only the most common variety of hydrogen has no neutrons.
Every solid, liquid, gas, and ...
or a functional group
In organic chemistry, a functional group is a substituent or moiety in a molecule that causes the molecule's characteristic chemical reactions. The same functional group will undergo the same or similar chemical reactions regardless of the rest ...
to attract electron
The electron ( or ) is a subatomic particle with a negative one elementary electric charge. Electrons belong to the first generation of the lepton particle family,
and are generally thought to be elementary particles because they have no kn ...
s (or electron density
In quantum chemistry, electron density or electronic density is the measure of the probability of an electron being present at an infinitesimal element of space surrounding any given point. It is a scalar quantity depending upon three spatial va ...
) towards itself.sodium
Sodium is a chemical element with the symbol Na (from Latin ''natrium'') and atomic number 11. It is a soft, silvery-white, highly reactive metal. Sodium is an alkali metal, being in group 1 of the periodic table. Its only stable iso ...
and chlorine
Chlorine is a chemical element with the Symbol (chemistry), symbol Cl and atomic number 17. The second-lightest of the halogens, it appears between fluorine and bromine in the periodic table and its properties are mostly intermediate betwee ...
in sodium chloride
Sodium chloride , commonly known as salt (although sea salt also contains other chemical salts), is an ionic compound with the chemical formula NaCl, representing a 1:1 ratio of sodium and chloride ions. With molar masses of 22.99 and 35.45 g ...
were covalent
A covalent bond is a chemical bond that involves the sharing of electrons to form electron pairs between atoms. These electron pairs are known as shared pairs or bonding pairs. The stable balance of attractive and repulsive forces between atoms ...
, the pair of shared electrons would be attracted to the chlorine because the effective nuclear charge on the outer electrons is +7 in chlorine but is only +1 in sodium. The electron pair is attracted so close to the chlorine atom that they are practically transferred to the chlorine atom (an ionic bond
Ionic bonding is a type of chemical bonding that involves the electrostatic attraction between oppositely charged ions, or between two atoms with sharply different electronegativities, and is the primary interaction occurring in ionic compounds. ...
). However, if the sodium atom was replaced by a lithium atom, the electrons will not be attracted as close to the chlorine atom as before because the lithium atom is smaller, making the electron pair more strongly attracted to the closer effective nuclear charge from lithium. Hence, the larger alkali metal atoms (further down the group) will be less electronegative as the bonding pair is less strongly attracted towards them. As mentioned previously, francium is expected to be an exception.lithium iodide
Lithium iodide, or LiI, is a compound of lithium and iodine. When exposed to air, it becomes yellow in color, due to the oxidation of iodide to iodine. It crystallizes in the NaCl motif. It can participate in various hydrates.Wietelmann, Ulrich an ...
( Li I) will dissolve in organic solvent
A solvent (s) (from the Latin '' solvō'', "loosen, untie, solve") is a substance that dissolves a solute, resulting in a solution. A solvent is usually a liquid but can also be a solid, a gas, or a supercritical fluid. Water is a solvent for p ...
s, a property of most covalent compounds.Lithium fluoride
Lithium fluoride is an inorganic compound with the chemical formula LiF. It is a colorless solid, that transitions to white with decreasing crystal size. Although odorless, lithium fluoride has a bitter-saline taste. Its structure is analogous to ...
(Li F) is the only alkali halide
In chemistry, alkali metal halides, or alkali halides, are the family of inorganic compounds with the chemical formula MX, where M is an alkali metal and X is a halogen. These compounds are the often commercially significant sources of these metal ...
that is not soluble in water,lithium hydroxide
Lithium hydroxide is an inorganic compound with the formula LiOH. It can exist as anhydrous or hydrated, and both forms are white hygroscopic solids. They are soluble in water and slightly soluble in ethanol. Both are available commercially. While ...
(Li OH) is the only alkali metal hydroxide
The alkali hydroxides are a class of chemical compounds which are composed of an alkali metal cation and the hydroxide anion (OH−). The alkali hydroxides are:
*Lithium hydroxide (LiOH)
* Sodium hydroxide (NaOH)
*Potassium hydroxide (KOH)
*Rubi ...
that is not deliquescent
Hygroscopy is the phenomenon of attracting and holding water molecules via either absorption or adsorption from the surrounding environment, which is usually at normal or room temperature. If water molecules become suspended among the substance ...
.
Melting and boiling points
The melting point of a substance is the point where it changes state
State may refer to:
Arts, entertainment, and media Literature
* ''State Magazine'', a monthly magazine published by the U.S. Department of State
* ''The State'' (newspaper), a daily newspaper in Columbia, South Carolina, United States
* ''Our S ...
from solid
Solid is one of the State of matter#Four fundamental states, four fundamental states of matter (the others being liquid, gas, and Plasma (physics), plasma). The molecules in a solid are closely packed together and contain the least amount o ...
to liquid
A liquid is a nearly incompressible fluid that conforms to the shape of its container but retains a (nearly) constant volume independent of pressure. As such, it is one of the four fundamental states of matter (the others being solid, gas, a ...
while the boiling point of a substance (in liquid state) is the point where the vapour pressure
Vapor pressure (or vapour pressure in English-speaking countries other than the US; see spelling differences) or equilibrium vapor pressure is defined as the pressure exerted by a vapor in thermodynamic equilibrium with its condensed phases ...
of the liquid equals the environmental pressure surrounding the liquid and all the liquid changes state to gas
Gas is one of the four fundamental states of matter (the others being solid, liquid, and plasma).
A pure gas may be made up of individual atoms (e.g. a noble gas like neon), elemental molecules made from one type of atom (e.g. oxygen), or ...
. As a metal is heated to its melting point, the metallic bond
Metallic bonding is a type of chemical bonding that arises from the electrostatic attractive force between conduction electrons (in the form of an electron cloud of delocalized electrons) and positively charged metal ions. It may be descri ...
s keeping the atoms in place weaken so that the atoms can move around, and the metallic bonds eventually break completely at the metal's boiling point.
Density
The alkali metals all have the same crystal structure
In crystallography, crystal structure is a description of the ordered arrangement of atoms, ions or molecules in a crystal, crystalline material. Ordered structures occur from the intrinsic nature of the constituent particles to form symmetric pat ...
(body-centred cubic
In crystallography, the cubic (or isometric) crystal system is a crystal system where the unit cell is in the shape of a cube. This is one of the most common and simplest shapes found in crystals and minerals.
There are three main varieties of ...
)room temperature
Colloquially, "room temperature" is a range of air temperatures that most people prefer for indoor settings. It feels comfortable to a person when they are wearing typical indoor clothing. Human comfort can extend beyond this range depending on ...
.
Compounds
The alkali metals form complete series of compounds with all usually encountered anions, which well illustrate group trends. These compounds can be described as involving the alkali metals losing electrons to acceptor species and forming monopositive ions.ionic bond
Ionic bonding is a type of chemical bonding that involves the electrostatic attraction between oppositely charged ions, or between two atoms with sharply different electronegativities, and is the primary interaction occurring in ionic compounds. ...
ing gives way to metallic bond
Metallic bonding is a type of chemical bonding that arises from the electrostatic attractive force between conduction electrons (in the form of an electron cloud of delocalized electrons) and positively charged metal ions. It may be descri ...
ing along the series NaCl, Na2O, Na2S, Na3P, Na3As, Na3Sb, Na3Bi, Na.
Hydroxides
 All the alkali metals react vigorously or explosively with cold water, producing an
All the alkali metals react vigorously or explosively with cold water, producing an aqueous solution
An aqueous solution is a solution in which the solvent is water. It is mostly shown in chemical equations by appending (aq) to the relevant chemical formula. For example, a solution of table salt, or sodium chloride (NaCl), in water would be re ...
of a strongly basic
BASIC (Beginners' All-purpose Symbolic Instruction Code) is a family of general-purpose, high-level programming languages designed for ease of use. The original version was created by John G. Kemeny and Thomas E. Kurtz at Dartmouth College ...
alkali metal hydroxide
Hydroxide is a diatomic anion with chemical formula OH−. It consists of an oxygen and hydrogen atom held together by a single covalent bond, and carries a negative electric charge. It is an important but usually minor constituent of water. I ...
and releasing hydrogen gas.effervescence
Effervescence is the escape of gas from an aqueous solution and the foaming or fizzing that results from that release. The word effervescence is derived from the Latin verb ''fervere'' (to boil), preceded by the adverb ''ex''. It has the same lin ...
, but sodium and potassium can ignite, and rubidium and caesium sink in water and generate hydrogen gas so rapidly that shock waves form in the water that may shatter glass containers.hydrogen
Hydrogen is the chemical element with the symbol H and atomic number 1. Hydrogen is the lightest element. At standard conditions hydrogen is a gas of diatomic molecules having the formula . It is colorless, odorless, tasteless, non-toxic, an ...
gas; this takes place faster for the more reactive heavier alkali metals. Second, the heat generated by the first part of the reaction often ignites the hydrogen gas, causing it to burn explosively into the surrounding air. This secondary hydrogen gas explosion produces the visible flame above the bowl of water, lake or other body of water, not the initial reaction of the metal with water (which tends to happen mostly under water).Coulomb explosion
In condensed-matter physics, Coulombic explosions are a mechanism for transforming energy in intense electromagnetic fields into atomic motion and are thus useful for controlled destruction of relatively robust molecules. The explosions are a pr ...
rather than solely by rapid generation of hydrogen itself.oligomer
In chemistry and biochemistry, an oligomer () is a molecule that consists of a few repeating units which could be derived, actually or conceptually, from smaller molecules, monomers.Quote: ''Oligomer molecule: A molecule of intermediate relativ ...
ic alkoxide
In chemistry, an alkoxide is the conjugate base of an alcohol and therefore consists of an organic group bonded to a negatively charged oxygen atom. They are written as , where R is the organic substituent. Alkoxides are strong bases and, whe ...
s. They easily react with carbon dioxide
Carbon dioxide (chemical formula ) is a chemical compound made up of molecules that each have one carbon atom covalently double bonded to two oxygen atoms. It is found in the gas state at room temperature. In the air, carbon dioxide is transpar ...
to form carbonate
A carbonate is a salt of carbonic acid (H2CO3), characterized by the presence of the carbonate ion, a polyatomic ion with the formula . The word ''carbonate'' may also refer to a carbonate ester, an organic compound containing the carbonate g ...
s or bicarbonate
In inorganic chemistry, bicarbonate (IUPAC-recommended nomenclature: hydrogencarbonate) is an intermediate form in the deprotonation of carbonic acid. It is a polyatomic anion with the chemical formula .
Bicarbonate serves a crucial biochemic ...
s, or with hydrogen sulfide
Hydrogen sulfide is a chemical compound with the formula . It is a colorless chalcogen-hydride gas, and is poisonous, corrosive, and flammable, with trace amounts in ambient atmosphere having a characteristic foul odor of rotten eggs. The unde ...
to form sulfide
Sulfide (British English also sulphide) is an inorganic anion of sulfur with the chemical formula S2− or a compound containing one or more S2− ions. Solutions of sulfide salts are corrosive. ''Sulfide'' also refers to chemical compounds lar ...
s or bisulfide
Bisulfide (or bisulphide in British English) is an inorganic anion with the chemical formula HS− (also written as SH−). It contributes no color to bisulfide salts, and its salts may have a distinctive putrid smell. It is a strong base. Bisul ...
s, and may be used to separate thiol
In organic chemistry, a thiol (; ), or thiol derivative, is any organosulfur compound of the form , where R represents an alkyl or other organic substituent. The functional group itself is referred to as either a thiol group or a sulfhydryl gro ...
s from petroleum. They react with amphoteric oxides: for example, the oxides of aluminium
Aluminium (aluminum in American and Canadian English) is a chemical element with the symbol Al and atomic number 13. Aluminium has a density lower than those of other common metals, at approximately one third that of steel. I ...
, zinc
Zinc is a chemical element with the symbol Zn and atomic number 30. Zinc is a slightly brittle metal at room temperature and has a shiny-greyish appearance when oxidation is removed. It is the first element in group 12 (IIB) of the periodi ...
, tin
Tin is a chemical element with the symbol Sn (from la, stannum) and atomic number 50. Tin is a silvery-coloured metal.
Tin is soft enough to be cut with little force and a bar of tin can be bent by hand with little effort. When bent, t ...
, and lead
Lead is a chemical element with the symbol Pb (from the Latin ) and atomic number 82. It is a heavy metal that is denser than most common materials. Lead is soft and malleable, and also has a relatively low melting point. When freshly cu ...
react with the alkali metal hydroxides to give aluminates, zincates, stannates, and plumbates. Silicon dioxide
Silicon dioxide, also known as silica, is an oxide of silicon with the chemical formula , most commonly found in nature as quartz and in various living organisms. In many parts of the world, silica is the major constituent of sand. Silica is one ...
is acidic, and thus the alkali metal hydroxides can also attack silicate glass
Glass is a non-crystalline, often transparent, amorphous solid that has widespread practical, technological, and decorative use in, for example, window panes, tableware, and optics. Glass is most often formed by rapid cooling (quenching) of ...
.
Intermetallic compounds
 The alkali metals form many
The alkali metals form many intermetallic compound
An intermetallic (also called an intermetallic compound, intermetallic alloy, ordered intermetallic alloy, and a long-range-ordered alloy) is a type of metallic bonding, metallic alloy that forms an ordered solid-state Chemical compound, compoun ...
s with each other and the elements from groups 2 to 13 in the periodic table of varying stoichiometries,sodium amalgam
Sodium amalgam, commonly denoted Na(Hg), is an alloy of mercury and sodium. The term amalgam is used for alloys, intermetallic compounds, and solutions (both solid solutions and liquid solutions) involving mercury as a major component. Sodium amal ...
s with mercury
Mercury commonly refers to:
* Mercury (planet), the nearest planet to the Sun
* Mercury (element), a metallic chemical element with the symbol Hg
* Mercury (mythology), a Roman god
Mercury or The Mercury may also refer to:
Companies
* Merc ...
, including Na5Hg8 and Na3Hg. Some of these have ionic characteristics: taking the alloys with gold
Gold is a chemical element with the symbol Au (from la, aurum) and atomic number 79. This makes it one of the higher atomic number elements that occur naturally. It is a bright, slightly orange-yellow, dense, soft, malleable, and ductile met ...
, the most electronegative of metals, as an example, NaAu and KAu are metallic, but RbAu and CsAu are semiconductors.NaK
In data networking, telecommunications, and computer buses, an acknowledgment (ACK) is a signal that is passed between communicating processes, computers, or devices to signify acknowledgment, or receipt of message, as part of a communicatio ...
is an alloy of sodium and potassium that is very useful because it is liquid at room temperature, although precautions must be taken due to its extreme reactivity towards water and air. The eutectic mixture
A eutectic system or eutectic mixture ( ) is a homogeneous mixture that has a melting point lower than those of the constituents. The lowest possible melting point over all of the mixing ratios of the constituents is called the ''eutectic tempe ...
melts at −12.6 °C.
Compounds with the group 13 elements
The intermetallic compounds of the alkali metals with the heavier group 13 elements (aluminium
Aluminium (aluminum in American and Canadian English) is a chemical element with the symbol Al and atomic number 13. Aluminium has a density lower than those of other common metals, at approximately one third that of steel. I ...
, gallium
Gallium is a chemical element with the symbol Ga and atomic number 31. Discovered by French chemist Paul-Émile Lecoq de Boisbaudran in 1875, Gallium is in group 13 of the periodic table and is similar to the other metals of the group (aluminiu ...
, indium
Indium is a chemical element with the symbol In and atomic number 49. Indium is the softest metal that is not an alkali metal. It is a silvery-white metal that resembles tin in appearance. It is a post-transition metal that makes up 0.21 parts p ...
, and thallium
Thallium is a chemical element with the Symbol (chemistry), symbol Tl and atomic number 81. It is a gray post-transition metal that is not found free in nature. When isolated, thallium resembles tin, but discolors when exposed to air. Chemists W ...
), such as NaTl, are poor conductors or semiconductor
A semiconductor is a material which has an electrical resistivity and conductivity, electrical conductivity value falling between that of a electrical conductor, conductor, such as copper, and an insulator (electricity), insulator, such as glas ...
s, unlike the normal alloys with the preceding elements, implying that the alkali metal involved has lost an electron to the Zintl anions involved.[Sevov, S.C]
"Zintl Phases"
pp. 113–132 in ''Intermetallic Compounds, Principles and Practice: Progress'', Vol. 3. Westbrook, J.H.; Freisher, R.L.: Eds.; John Wiley & Sons. Ltd., Chichester, England Nevertheless, while the elements in group 14 and beyond tend to form discrete anionic clusters, group 13 elements tend to form polymeric ions with the alkali metal cations located between the giant ionic lattice. For example, NaTl consists of a polymeric anion (—Tl−—)n with a covalent diamond cubic
The diamond cubic crystal structure is a repeating pattern of 8 atoms that certain materials may adopt as they solidify. While the first known example was diamond, other elements in group 14 also adopt this structure, including α-tin, the sem ...
structure with Na+ ions located between the anionic lattice. The larger alkali metals cannot fit similarly into an anionic lattice and tend to force the heavier group 13 elements to form anionic clusters.[S.M. Kauzlarich, Encyclopedia of Inorganic chemistry, 1994, John Wiley & Sons, ]
Boron
Boron is a chemical element with the symbol B and atomic number 5. In its crystalline form it is a brittle, dark, lustrous metalloid; in its amorphous form it is a brown powder. As the lightest element of the ''boron group'' it has th ...
is a special case, being the only nonmetal in group 13. The alkali metal boride A boride is a compound between boron and a less electronegative element, for example silicon boride (SiB3 and SiB6). The borides are a very large group of compounds that are generally high melting and are covalent more than ionic in nature. Some bo ...
s tend to be boron-rich, involving appreciable boron–boron bonding involving deltahedral structures,vapour pressure
Vapor pressure (or vapour pressure in English-speaking countries other than the US; see spelling differences) or equilibrium vapor pressure is defined as the pressure exerted by a vapor in thermodynamic equilibrium with its condensed phases ...
at elevated temperatures. This makes direct synthesis problematic because the alkali metals do not react with boron below 700 °C, and thus this must be accomplished in sealed containers with the alkali metal in excess. Furthermore, exceptionally in this group, reactivity with boron decreases down the group: lithium reacts completely at 700 °C, but sodium at 900 °C and potassium not until 1200 °C, and the reaction is instantaneous for lithium but takes hours for potassium. Rubidium and caesium borides have not even been characterised. Various phases are known, such as LiB10, NaB6, NaB15, and KB6. Under high pressure the boron–boron bonding in the lithium borides changes from following Wade's rules to forming Zintl anions like the rest of group 13.
Compounds with the group 14 elements
Lithium and sodium react with carbon
Carbon () is a chemical element with the symbol C and atomic number 6. It is nonmetallic and tetravalent
In chemistry, the valence (US spelling) or valency (British spelling) of an element is the measure of its combining capacity with o ...
to form acetylide
In organometallic chemistry, acetylide refers to chemical compounds with the chemical formulas and , where M is a metal. The term is used loosely and can refer to substituted acetylides having the general structure (where R is an organic side c ...
s, Li2C2 and Na2C2, which can also be obtained by reaction of the metal with acetylene
Acetylene (systematic name: ethyne) is the chemical compound with the formula and structure . It is a hydrocarbon and the simplest alkyne. This colorless gas is widely used as a fuel and a chemical building block. It is unstable in its pure ...
. Potassium, rubidium, and caesium react with graphite
Graphite () is a crystalline form of the element carbon. It consists of stacked layers of graphene. Graphite occurs naturally and is the most stable form of carbon under standard conditions. Synthetic and natural graphite are consumed on large ...
; their atoms are intercalated between the hexagonal graphite layers, forming graphite intercalation compound
Graphite intercalation compounds are complex materials having a formula where the ion or is inserted ( intercalated) between the oppositely charged carbon layers. Typically ''m'' is much less than 1. These materials are deeply colored solids t ...
s of formulae MC60 (dark grey, almost black), MC48 (dark grey, almost black), MC36 (blue), MC24 (steel blue), and MC8 (bronze) (M = K, Rb, or Cs). These compounds are over 200 times more electrically conductive than pure graphite, suggesting that the valence electron of the alkali metal is transferred to the graphite layers (e.g. ).reducing agent
In chemistry, a reducing agent (also known as a reductant, reducer, or electron donor) is a chemical species that "donates" an electron to an (called the , , , or ).
Examples of substances that are commonly reducing agents include the Earth meta ...
and is pyrophoric and explodes on contact with water.buckminsterfullerene
Buckminsterfullerene is a type of fullerene with the formula C60. It has a cage-like fused-ring structure (truncated icosahedron) made of twenty hexagons and twelve pentagons, and resembles a soccer ball. Each of its 60 carbon atoms is bonded ...
to produce solid fullerides M''n''C60; sodium, potassium, rubidium, and caesium can form fullerides where ''n'' = 2, 3, 4, or 6, and rubidium and caesium additionally can achieve ''n'' = 1.carbon group
The carbon group is a periodic table group consisting of carbon (C), silicon (Si), germanium (Ge), tin (Sn), lead (Pb), and flerovium (Fl). It lies within the p-block.
In modern IUPAC notation, it is called group 14. In the field of semicond ...
(silicon
Silicon is a chemical element with the symbol Si and atomic number 14. It is a hard, brittle crystalline solid with a blue-grey metallic luster, and is a tetravalent metalloid and semiconductor. It is a member of group 14 in the periodic tab ...
, germanium
Germanium is a chemical element with the symbol Ge and atomic number 32. It is lustrous, hard-brittle, grayish-white and similar in appearance to silicon. It is a metalloid in the carbon group that is chemically similar to its group neighbors s ...
, tin
Tin is a chemical element with the symbol Sn (from la, stannum) and atomic number 50. Tin is a silvery-coloured metal.
Tin is soft enough to be cut with little force and a bar of tin can be bent by hand with little effort. When bent, t ...
, and lead
Lead is a chemical element with the symbol Pb (from the Latin ) and atomic number 82. It is a heavy metal that is denser than most common materials. Lead is soft and malleable, and also has a relatively low melting point. When freshly cu ...
), ionic substances with cage-like structures are formed, such as the silicide
A silicide is a type of chemical compound that combines silicon and a (usually) more electropositive element.
Silicon is more electropositive than carbon. Silicides are structurally closer to borides than to carbides.
Similar to borides and carb ...
s M4 Si4 (M = K, Rb, or Cs), which contains M+ and tetrahedral ions.germanide A germanide is any binary compound of germanium and a more electropositive element. The composition of most germanides is analogous to that of the corresponding silicides and does not follow formal valence rules. The germanides of alkali and alkali ...
s, involving the germanide ion Ge4− and other cluster ( Zintl) ions such as , , , and Ge9)2sup>6−, is largely analogous to that of the corresponding silicides.plumbide
A plumbide can refer to one of two things: an intermetallic compound that contains lead, or a Zintl phase compound with lead as the anion.
Zintl phase
Plumbides can be formed when lead forms a Zintl phase compound with a more metallic element. O ...
ion ( Pb4−) is unknown, and indeed its formation is predicted to be energetically unfavourable; alkali metal plumbides have complex Zintl ions, such as . These alkali metal germanides, stannides, and plumbides may be produced by reducing germanium, tin, and lead with sodium metal in liquid ammonia.
Nitrides and pnictides
 Lithium, the lightest of the alkali metals, is the only alkali metal which reacts with
Lithium, the lightest of the alkali metals, is the only alkali metal which reacts with nitrogen
Nitrogen is the chemical element with the symbol N and atomic number 7. Nitrogen is a nonmetal and the lightest member of group 15 of the periodic table, often called the pnictogens. It is a common element in the universe, estimated at se ...
at standard conditions
Standard temperature and pressure (STP) are standard sets of conditions for experimental measurements to be established to allow comparisons to be made between different sets of data. The most used standards are those of the International Union ...
, and its nitride
In chemistry, a nitride is an inorganic compound of nitrogen. The "nitride" anion, N3- ion, is very elusive but compounds of nitride are numerous, although rarely naturally occuring. Some nitrides have a find applications, such as wear-resistant ...
is the only stable alkali metal nitride. Nitrogen is an unreactive gas because breaking the strong triple bond
A triple bond in chemistry is a chemical bond between two atoms involving six bonding electrons instead of the usual two in a covalent single bond. Triple bonds are stronger than the equivalent single bonds or double bonds, with a bond order o ...
in the dinitrogen molecule (N2) requires a lot of energy. The formation of an alkali metal nitride would consume the ionisation energy of the alkali metal (forming M+ ions), the energy required to break the triple bond in N2 and the formation of N3− ions, and all the energy released from the formation of an alkali metal nitride is from the lattice energy
In chemistry, the lattice energy is the energy change upon formation of one mole of a crystalline ionic compound from its constituent ions, which are assumed to initially be in the gaseous state. It is a measure of the cohesive forces that bind ...
of the alkali metal nitride. The lattice energy is maximised with small, highly charged ions; the alkali metals do not form highly charged ions, only forming ions with a charge of +1, so only lithium, the smallest alkali metal, can release enough lattice energy to make the reaction with nitrogen exothermic
In thermodynamics, an exothermic process () is a thermodynamic process or reaction that releases energy from the system to its surroundings, usually in the form of heat, but also in a form of light (e.g. a spark, flame, or flash), electricity (e ...
, forming lithium nitride
Lithium nitride is a compound with the formula Li3N. It is the only stable alkali metal nitride. The solid has a reddish-pink color and high melting point.
Preparation and handling
Lithium nitride is prepared by direct combination of elemental ...
. The reactions of the other alkali metals with nitrogen would not release enough lattice energy and would thus be endothermic
In thermochemistry, an endothermic process () is any thermodynamic process with an increase in the enthalpy (or internal energy ) of the system.Oxtoby, D. W; Gillis, H.P., Butler, L. J. (2015).''Principle of Modern Chemistry'', Brooks Cole. p. ...
, so they do not form nitrides at standard conditions.Sodium nitride
Sodium nitride is the inorganic compound with the chemical formula Na3N. In contrast to lithium nitride and some other nitrides, sodium nitride is an extremely unstable alkali metal nitride. It can be generated by combining atomic beams of sodium ...
(Na3N) and potassium nitride
Potassium nitride is an unstable chemical compound. Several syntheses were erroneously claimed in the 19th century, and by 1894 it was assumed that it did not exist.
However, a synthesis of this compound was claimed in 2004. It is observed to have ...
(K3N), while existing, are extremely unstable, being prone to decomposing back into their constituent elements, and cannot be produced by reacting the elements with each other at standard conditions.
'Elusive Binary Compound Prepared'
''Chemical & Engineering News'' 80 No. 20 (20 May 2002)azide
In chemistry, azide is a linear, polyatomic anion with the formula and structure . It is the conjugate base of hydrazoic acid . Organic azides are organic compounds with the formula , containing the azide functional group. The dominant applic ...
salts involving the linear anion; due to the large size of the alkali metal cations, they are thermally stable enough to be able to melt before decomposing.phosphorus
Phosphorus is a chemical element with the symbol P and atomic number 15. Elemental phosphorus exists in two major forms, white phosphorus and red phosphorus, but because it is highly reactive, phosphorus is never found as a free element on Ear ...
and arsenic
Arsenic is a chemical element with the symbol As and atomic number 33. Arsenic occurs in many minerals, usually in combination with sulfur and metals, but also as a pure elemental crystal. Arsenic is a metalloid. It has various allotropes, but ...
to form phosphides and arsenides with the formula M3Pn (where M represents an alkali metal and Pn represents a pnictogen
A pnictogen ( or ; from grc, πνῑ́γω "to choke" and -gen, "generator") is any of the chemical elements in group 15 of the periodic table. Group 15 is also known as the nitrogen group or nitrogen family. Group 15 consists of the ele ...
– phosphorus, arsenic, antimony
Antimony is a chemical element with the symbol Sb (from la, stibium) and atomic number 51. A lustrous gray metalloid, it is found in nature mainly as the sulfide mineral stibnite (Sb2S3). Antimony compounds have been known since ancient time ...
, or bismuth
Bismuth is a chemical element with the Symbol (chemistry), symbol Bi and atomic number 83. It is a post-transition metal and one of the pnictogens, with chemical properties resembling its lighter group 15 siblings arsenic and antimony. Elemental ...
). This is due to the greater size of the P3− and As3− ions, so that less lattice energy needs to be released for the salts to form.[H.G. Von Schnering, W. Hönle ''Phosphides – Solid-state Chemistry'' Encyclopedia of Inorganic Chemistry Ed. R. Bruce King (1994) John Wiley & Sons ] While most metals form arsenides, only the alkali and alkaline earth metals form mostly ionic arsenides. The structure of Na3As is complex with unusually short Na–Na distances of 328–330 pm which are shorter than in sodium metal, and this indicates that even with these electropositive metals the bonding cannot be straightforwardly ionic.metallic bond
Metallic bonding is a type of chemical bonding that arises from the electrostatic attractive force between conduction electrons (in the form of an electron cloud of delocalized electrons) and positively charged metal ions. It may be descri ...
ing.antimonide Antimonides (sometimes called stibnides) are compounds of antimony with more electropositive elements. The antimonide ion is Sb3−.
Reduction of antimony by alkali metals or by other methods leads to alkali metal antimonides of various types. K ...
s are unstable and reactive as the Sb3− ion is a strong reducing agent; reaction of them with acids form the toxic and unstable gas stibine (SbH3). Indeed, they have some metallic properties, and the alkali metal antimonides of stoichiometry MSb involve antimony atoms bonded in a spiral Zintl structure.Bismuth
Bismuth is a chemical element with the Symbol (chemistry), symbol Bi and atomic number 83. It is a post-transition metal and one of the pnictogens, with chemical properties resembling its lighter group 15 siblings arsenic and antimony. Elemental ...
ides are not even wholly ionic; they are intermetallic compound
An intermetallic (also called an intermetallic compound, intermetallic alloy, ordered intermetallic alloy, and a long-range-ordered alloy) is a type of metallic bonding, metallic alloy that forms an ordered solid-state Chemical compound, compoun ...
s containing partially metallic and partially ionic bonds.
Oxides and chalcogenides
All the alkali metals react vigorously with oxygen
Oxygen is the chemical element with the symbol O and atomic number 8. It is a member of the chalcogen group in the periodic table, a highly reactive nonmetal, and an oxidizing agent that readily forms oxides with most elements as wel ...
at standard conditions. They form various types of oxides, such as simple oxide
An oxide () is a chemical compound that contains at least one oxygen atom and one other element in its chemical formula. "Oxide" itself is the dianion of oxygen, an O2– (molecular) ion. with oxygen in the oxidation state of −2. Most of the E ...
s (containing the O2− ion), peroxide
In chemistry, peroxides are a group of compounds with the structure , where R = any element. The group in a peroxide is called the peroxide group or peroxo group. The nomenclature is somewhat variable.
The most common peroxide is hydrogen p ...
s (containing the ion, where there is a single bond
In chemistry, a single bond is a chemical bond between two atoms involving two valence electrons. That is, the atoms share one pair of electrons where the bond forms. Therefore, a single bond is a type of covalent bond. When shared, each of th ...
between the two oxygen atoms), superoxide
In chemistry, a superoxide is a compound that contains the superoxide ion, which has the chemical formula . The systematic name of the anion is dioxide(1−). The reactive oxygen ion superoxide is particularly important as the product of the ...
s (containing the ion), and many others. Lithium burns in air to form lithium oxide
Lithium oxide ( O) or lithia is an inorganic chemical compound. It is a white solid. Although not specifically important, many materials are assessed on the basis of their Li2O content. For example, the Li2O content of the principal lithium miner ...
, but sodium reacts with oxygen to form a mixture of sodium oxide
Sodium oxide is a chemical compound with the formula Na2 O. It is used in ceramics and glasses. It is a white solid but the compound is rarely encountered. Instead "sodium oxide" is used to describe components of various materials such as glass ...
and sodium peroxide
Sodium peroxide is an inorganic compound with the formula Na2O2. This yellowish solid is the product of sodium ignited in excess oxygen. It is a strong base. This metal peroxide exists in several hydrates and peroxyhydrates including Na2O2·2H2O ...
. Potassium forms a mixture of potassium peroxide
Potassium peroxide is an inorganic compound with the molecular formula K2O2. It is formed as potassium reacts with oxygen in the air, along with potassium oxide (K2O) and potassium superoxide (KO2).
Potassium peroxide reacts with water to form p ...
and potassium superoxide
Potassium superoxide is an inorganic compound with the formula KO2. It is a yellow paramagnetic solid that decomposes in moist air. It is a rare example of a stable salt of the superoxide anion. It is used as a scrubber, dehumidifier, and gen ...
, while rubidium and caesium form the superoxide exclusively. Their reactivity increases going down the group: while lithium, sodium and potassium merely burn in air, rubidium and caesium are pyrophoric
A substance is pyrophoric (from grc-gre, πυροφόρος, , 'fire-bearing') if it ignites spontaneously in air at or below (for gases) or within 5 minutes after coming into contact with air (for liquids and solids). Examples are organolith ...
(spontaneously catch fire in air).oxidising agent
An oxidizing agent (also known as an oxidant, oxidizer, electron recipient, or electron acceptor) is a substance in a redox chemical reaction that gains or " accepts"/"receives" an electron from a (called the , , or ). In other words, an oxi ...
s. Sodium peroxide
Sodium peroxide is an inorganic compound with the formula Na2O2. This yellowish solid is the product of sodium ignited in excess oxygen. It is a strong base. This metal peroxide exists in several hydrates and peroxyhydrates including Na2O2·2H2O ...
and potassium superoxide
Potassium superoxide is an inorganic compound with the formula KO2. It is a yellow paramagnetic solid that decomposes in moist air. It is a rare example of a stable salt of the superoxide anion. It is used as a scrubber, dehumidifier, and gen ...
react with carbon dioxide
Carbon dioxide (chemical formula ) is a chemical compound made up of molecules that each have one carbon atom covalently double bonded to two oxygen atoms. It is found in the gas state at room temperature. In the air, carbon dioxide is transpar ...
to form the alkali metal carbonate and oxygen gas, which allows them to be used in submarine
A submarine (or sub) is a watercraft capable of independent operation underwater. It differs from a submersible, which has more limited underwater capability. The term is also sometimes used historically or colloquially to refer to remotely op ...
air purifiers; the presence of water vapour
(99.9839 °C)
, -
, Boiling point
,
, -
, specific gas constant
, 461.5 J/( kg·K)
, -
, Heat of vaporization
, 2.27 MJ/kg
, -
, Heat capacity
, 1.864 kJ/(kg·K)
Water vapor, water vapour or aqueous vapor is the gaseous pha ...
, naturally present in breath, makes the removal of carbon dioxide by potassium superoxide even more efficient.ozonide
Ozonide is the polyatomic anion . Cyclic organic compounds formed by the addition of ozone () to an alkene are also called ozonides.
Ionic ozonides
Inorganic ozonides are dark red salts. The anion has the bent shape of the ozone molecule.
Inor ...
s (MO3) through low-temperature reaction of the powdered anhydrous hydroxide with ozone
Ozone (), or trioxygen, is an inorganic molecule with the chemical formula . It is a pale blue gas with a distinctively pungent smell. It is an allotrope of oxygen that is much less stable than the diatomic allotrope , breaking down in the lo ...
: the ozonides may be then extracted using liquid ammonia
Ammonia is an inorganic compound of nitrogen and hydrogen with the formula . A stable binary hydride, and the simplest pnictogen hydride, ammonia is a colourless gas with a distinct pungent smell. Biologically, it is a common nitrogenous was ...
. They slowly decompose at standard conditions to the superoxides and oxygen, and hydrolyse immediately to the hydroxides when in contact with water.suboxide
Suboxides are a class of oxides wherein the electropositive element is in excess relative to the “normal” oxides. When the electropositive element is a metal, the compounds are sometimes referred to as “metal-rich”. Thus the normal oxide ...
s,sulfur
Sulfur (or sulphur in British English) is a chemical element with the symbol S and atomic number 16. It is abundant, multivalent and nonmetallic. Under normal conditions, sulfur atoms form cyclic octatomic molecules with a chemical formula ...
, selenium
Selenium is a chemical element with the symbol Se and atomic number 34. It is a nonmetal (more rarely considered a metalloid) with properties that are intermediate between the elements above and below in the periodic table, sulfur and tellurium, ...
, tellurium
Tellurium is a chemical element with the symbol Te and atomic number 52. It is a brittle, mildly toxic, rare, silver-white metalloid. Tellurium is chemically related to selenium and sulfur, all three of which are chalcogens. It is occasionally fou ...
, and polonium
Polonium is a chemical element with the symbol Po and atomic number 84. Polonium is a chalcogen. A rare and highly radioactive metal with no stable isotopes, polonium is chemically similar to selenium and tellurium, though its metallic character ...
), and all the alkali metal chalcogenides are known (with the exception of francium's). Reaction with an excess of the chalcogen can similarly result in lower chalcogenides, with chalcogen ions containing chains of the chalcogen atoms in question. For example, sodium can react with sulfur to form the sulfide
Sulfide (British English also sulphide) is an inorganic anion of sulfur with the chemical formula S2− or a compound containing one or more S2− ions. Solutions of sulfide salts are corrosive. ''Sulfide'' also refers to chemical compounds lar ...
( Na2S) and various polysulfide
Polysulfides are a class of chemical compounds containing chains of sulfur atoms. There are two main classes of polysulfides: inorganic and organic. Among the inorganic polysulfides, there are ones which contain anions, which have the general formu ...
s with the formula Na2S''x'' (''x'' from 2 to 6), containing the ions.selenide A selenide is a chemical compound containing a selenium anion with oxidation number of −2 (Se2−), much as sulfur does in a sulfide. The chemistry of the selenides and sulfides is similar. Similar to sulfide, in aqueous solution, the selenide ion ...
s and tellurides
The telluride ion is the anion Te2− and its derivatives. It is analogous to the other chalcogenide anions, the lighter O2−, S2−, and Se2−, and the heavier Po2−.
In principle, Te2− is formed by the two-e− reduction of telluri ...
are alkaline in solution; when reacted directly with selenium and tellurium, alkali metal polyselenides and polytellurides are formed along with the selenides and tellurides with the and ions.polonide
A polonide is a chemical compound of the radioactive element polonium with any element less electronegative than polonium. Polonides are usually prepared by a direct reaction between the elements at temperatures of around 300–400 °C... They a ...
s are all ionic compounds containing the Po2− ion; they are very chemically stable and can be produced by direct reaction of the elements at around 300–400 °C.
Halides, hydrides, and pseudohalides
The alkali metals are among the most electropositive
Electronegativity, symbolized as , is the tendency for an atom of a given chemical element to attract shared electrons (or electron density) when forming a chemical bond. An atom's electronegativity is affected by both its atomic number and the d ...
elements on the periodic table and thus tend to bond ionically to the most electronegative
Electronegativity, symbolized as , is the tendency for an atom of a given chemical element to attract shared electrons (or electron density) when forming a chemical bond. An atom's electronegativity is affected by both its atomic number and the d ...
elements on the periodic table, the halogen
The halogens () are a group in the periodic table consisting of five or six chemically related elements: fluorine (F), chlorine (Cl), bromine (Br), iodine (I), astatine (At), and tennessine (Ts). In the modern IUPAC nomenclature, this group is ...
s (fluorine
Fluorine is a chemical element with the symbol F and atomic number 9. It is the lightest halogen and exists at standard conditions as a highly toxic, pale yellow diatomic gas. As the most electronegative reactive element, it is extremely reacti ...
, chlorine
Chlorine is a chemical element with the Symbol (chemistry), symbol Cl and atomic number 17. The second-lightest of the halogens, it appears between fluorine and bromine in the periodic table and its properties are mostly intermediate betwee ...
, bromine
Bromine is a chemical element with the symbol Br and atomic number 35. It is the third-lightest element in group 17 of the periodic table (halogens) and is a volatile red-brown liquid at room temperature that evaporates readily to form a simila ...
, iodine
Iodine is a chemical element with the symbol I and atomic number 53. The heaviest of the stable halogens, it exists as a semi-lustrous, non-metallic solid at standard conditions that melts to form a deep violet liquid at , and boils to a vi ...
, and astatine
Astatine is a chemical element with the symbol At and atomic number 85. It is the rarest naturally occurring element in the Earth's crust, occurring only as the decay product of various heavier elements. All of astatine's isotopes are short-lived; ...
), forming salts
In chemistry, a salt is a chemical compound consisting of an ionic assembly of positively charged cations and negatively charged anions, which results in a compound with no net electric charge. A common example is table salt, with positively cha ...
known as the alkali metal halides. The reaction is very vigorous and can sometimes result in explosions.sodium chloride
Sodium chloride , commonly known as salt (although sea salt also contains other chemical salts), is an ionic compound with the chemical formula NaCl, representing a 1:1 ratio of sodium and chloride ions. With molar masses of 22.99 and 35.45 g ...
, otherwise known as common salt. All of the stable alkali metal halides have the formula MX where M is an alkali metal and X is a halogen. They are all white ionic crystalline solids that have high melting points.soluble
In chemistry, solubility is the ability of a substance, the solute, to form a solution with another substance, the solvent. Insolubility is the opposite property, the inability of the solute to form such a solution.
The extent of the solubil ...
in water except for lithium fluoride
Lithium fluoride is an inorganic compound with the chemical formula LiF. It is a colorless solid, that transitions to white with decreasing crystal size. Although odorless, lithium fluoride has a bitter-saline taste. Its structure is analogous to ...
(LiF), which is insoluble in water due to its very high lattice enthalpy
In chemistry, the lattice energy is the energy change upon formation of one mole of a crystalline ionic compound from its constituent ions, which are assumed to initially be in the gaseous state. It is a measure of the cohesive forces that bind ...
. The high lattice enthalpy of lithium fluoride is due to the small sizes of the Li+ and F− ions, causing the electrostatic interaction
Electrostatics is a branch of physics that studies electric charges at rest (static electricity).
Since classical times, it has been known that some materials, such as amber, attract lightweight particles after triboelectric effect, rubbing. ...
s between them to be strong:magnesium fluoride
Magnesium fluoride is an inorganic compound with the formula MgF2. The compound is a white crystalline salt and is transparent over a wide range of wavelengths, with commercial uses in optics that are also used in space telescopes. It occurs natur ...
, consistent with the diagonal relationship between lithium and magnesium.hydride
In chemistry, a hydride is formally the anion of hydrogen( H−). The term is applied loosely. At one extreme, all compounds containing covalently bound H atoms are called hydrides: water (H2O) is a hydride of oxygen, ammonia is a hydride of ...
anion acts as a pseudohalide
Pseudohalogens are polyatomic analogues of halogens, whose chemistry, resembling that of the true halogens, allows them to substitute for halogens in several classes of chemical compounds. Pseudohalogens occur in pseudohalogen molecules, inorganic ...
: these are often used as reducing agents, producing hydrides, complex metal hydrides, or hydrogen gas.cyanide
Cyanide is a naturally occurring, rapidly acting, toxic chemical that can exist in many different forms.
In chemistry, a cyanide () is a chemical compound that contains a functional group. This group, known as the cyano group, consists of ...
s. These are isostructural to the respective halides except for lithium cyanide
Lithium cyanide is an inorganic compound with the chemical formula LiCN. It is a toxic, white colored, hygroscopic, water-soluble salt that finds only niche uses.
Preparation
LiCN arises from the interaction of lithium hydroxide and hydrogen cyan ...
, indicating that the cyanide ions may rotate freely.
Coordination complexes
Alkali metal cations do not usually form coordination complex
A coordination complex consists of a central atom or ion, which is usually metallic and is called the ''coordination centre'', and a surrounding array of bound molecules or ions, that are in turn known as ''ligands'' or complexing agents. Many ...
es with simple Lewis base
A Lewis acid (named for the American physical chemist Gilbert N. Lewis) is a chemical species that contains an empty orbital which is capable of accepting an electron pair from a Lewis base to form a Lewis adduct. A Lewis base, then, is any sp ...
s due to their low charge of just +1 and their relatively large size; thus the Li+ ion forms most complexes and the heavier alkali metal ions form less and less (though exceptions occur for weak complexes).coordination number
In chemistry, crystallography, and materials science, the coordination number, also called ligancy, of a central atom in a molecule or crystal is the number of atoms, molecules or ions bonded to it. The ion/molecule/atom surrounding the central i ...
s from 1 to 12, although octahedral hexacoordination is its preferred mode.aqueous solution
An aqueous solution is a solution in which the solvent is water. It is mostly shown in chemical equations by appending (aq) to the relevant chemical formula. For example, a solution of table salt, or sodium chloride (NaCl), in water would be re ...
, the alkali metal ions exist as octahedral hexahydrate complexes ( (H2O)6)sup>+), with the exception of the lithium ion, which due to its small size forms tetrahedral tetrahydrate complexes ( i(H2O)4)sup>+); the alkali metals form these complexes because their ions are attracted by electrostatic forces of attraction to the polar water molecules. Because of this, anhydrous
A substance is anhydrous if it contains no water. Many processes in chemistry can be impeded by the presence of water; therefore, it is important that water-free reagents and techniques are used. In practice, however, it is very difficult to achie ...
salts containing alkali metal cations are often used as desiccant
A desiccant is a hygroscopic substance that is used to induce or sustain a state of dryness (desiccation) in its vicinity; it is the opposite of a humectant. Commonly encountered pre-packaged desiccants are solids that absorb water. Desiccant ...
s.crown ether
In organic chemistry, crown ethers are cyclic chemical compounds that consist of a ring containing several ether groups (). The most common crown ethers are cyclic oligomers of ethylene oxide, the repeating unit being ethyleneoxy, i.e., . Impo ...
s (e.g. 12-crown-4 for Li+, 15-crown-5 for Na+, 18-crown-6
18-Crown-6 is an organic compound with the formula 2H4O and the IUPAC name of 1,4,7,10,13,16-hexaoxacyclooctadecane. It is a white, hygroscopic crystalline solid with a low melting point. Like other crown ethers, 18-crown-6 functions as a li ...
for K+, and 21-crown-7 for Rb+) and cryptand
In chemistry, cryptands are a family of synthetic, bicyclic and polycyclic, multidentate ligands for a variety of cations. The Nobel Prize for Chemistry in 1987 was given to Donald J. Cram, Jean-Marie Lehn, and Charles J. Pedersen for their e ...
s due to electrostatic attraction.
Ammonia solutions
The alkali metals dissolve slowly in liquid ammonia
Ammonia is an inorganic compound of nitrogen and hydrogen with the formula . A stable binary hydride, and the simplest pnictogen hydride, ammonia is a colourless gas with a distinct pungent smell. Biologically, it is a common nitrogenous was ...
, forming ammoniacal solutions of solvated metal cation M+ and solvated electron
A solvated electron is a free electron in (solvated in) a solution, and is the smallest possible anion. Solvated electrons occur widely. Often, discussions of solvated electrons focus on their solutions in ammonia, which are stable for days, but s ...
e−, which react to form hydrogen gas and the alkali metal amide (MNH2, where M represents an alkali metal): this was first noted by Humphry Davy
Sir Humphry Davy, 1st Baronet, (17 December 177829 May 1829) was a British chemist and inventor who invented the Davy lamp and a very early form of arc lamp. He is also remembered for isolating, by using electricity, several elements for t ...
in 1809 and rediscovered by W. Weyl in 1864. The process may be speeded up by a catalyst
Catalysis () is the process of increasing the rate of a chemical reaction by adding a substance known as a catalyst (). Catalysts are not consumed in the reaction and remain unchanged after it. If the reaction is rapid and the catalyst recyc ...
. Similar solutions are formed by the heavy divalent alkaline earth metal
The alkaline earth metals are six chemical elements in group 2 of the periodic table. They are beryllium (Be), magnesium (Mg), calcium (Ca), strontium (Sr), barium (Ba), and radium (Ra).. The elements have very similar properties: they are al ...
s calcium
Calcium is a chemical element with the symbol Ca and atomic number 20. As an alkaline earth metal, calcium is a reactive metal that forms a dark oxide-nitride layer when exposed to air. Its physical and chemical properties are most similar to ...
, strontium
Strontium is the chemical element with the symbol Sr and atomic number 38. An alkaline earth metal, strontium is a soft silver-white yellowish metallic element that is highly chemically reactive. The metal forms a dark oxide layer when it is ex ...
, barium
Barium is a chemical element with the symbol Ba and atomic number 56. It is the fifth element in group 2 and is a soft, silvery alkaline earth metal. Because of its high chemical reactivity, barium is never found in nature as a free element.
Th ...
, as well as the divalent lanthanide
The lanthanide () or lanthanoid () series of chemical elements comprises the 15 metallic chemical elements with atomic numbers 57–71, from lanthanum through lutetium. These elements, along with the chemically similar elements scandium and yttr ...
s, europium
Europium is a chemical element with the symbol Eu and atomic number 63. Europium is the most reactive lanthanide by far, having to be stored under an inert fluid to protect it from atmospheric oxygen or moisture. Europium is also the softest lanth ...
and ytterbium
Ytterbium is a chemical element with the symbol Yb and atomic number 70. It is a metal, the fourteenth and penultimate element in the lanthanide series, which is the basis of the relative stability of its +2 oxidation state. However, like the othe ...
. The amide salt is quite insoluble and readily precipitates out of solution, leaving intensely coloured ammonia solutions of the alkali metals. In 1907, Charles Krause identified the colour as being due to the presence of solvated electron
A solvated electron is a free electron in (solvated in) a solution, and is the smallest possible anion. Solvated electrons occur widely. Often, discussions of solvated electrons focus on their solutions in ammonia, which are stable for days, but s ...
s, which contribute to the high electrical conductivity of these solutions. At low concentrations (below 3 M), the solution is dark blue and has ten times the conductivity of aqueous sodium chloride
Sodium chloride , commonly known as salt (although sea salt also contains other chemical salts), is an ionic compound with the chemical formula NaCl, representing a 1:1 ratio of sodium and chloride ions. With molar masses of 22.99 and 35.45 g ...
; at higher concentrations (above 3 M), the solution is copper-coloured and has approximately the conductivity of liquid metals like mercury
Mercury commonly refers to:
* Mercury (planet), the nearest planet to the Sun
* Mercury (element), a metallic chemical element with the symbol Hg
* Mercury (mythology), a Roman god
Mercury or The Mercury may also refer to:
Companies
* Merc ...
.diatomic
Diatomic molecules () are molecules composed of only two atoms, of the same or different chemical elements. If a diatomic molecule consists of two atoms of the same element, such as hydrogen () or oxygen (), then it is said to be homonuclear. Ot ...
alkali metal molecules (M2) and alkali metal anions (M−). These are unstable and eventually become the more thermodynamically stable alkali metal amide and hydrogen gas. Solvated electrons are powerful reducing agent
In chemistry, a reducing agent (also known as a reductant, reducer, or electron donor) is a chemical species that "donates" an electron to an (called the , , , or ).
Examples of substances that are commonly reducing agents include the Earth meta ...
s and are often used in chemical synthesis.
Organometallic
Organolithium
 Being the smallest alkali metal, lithium forms the widest variety of and most stable
Being the smallest alkali metal, lithium forms the widest variety of and most stable organometallic compound
Organometallic chemistry is the study of organometallic compounds, chemical compounds containing at least one chemical bond between a carbon atom of an organic molecule and a metal, including alkali, alkaline earth, and transition metals, and so ...
s, which are bonded covalently. Organolithium
In organometallic chemistry
Organometallic chemistry is the study of organometallic compounds, chemical compounds containing at least one chemical bond between a carbon atom of an organic molecule and a metal, including alkali, alkaline earth, ...
compounds are electrically non-conducting volatile solids or liquids that melt at low temperatures, and tend to form oligomer
In chemistry and biochemistry, an oligomer () is a molecule that consists of a few repeating units which could be derived, actually or conceptually, from smaller molecules, monomers.Quote: ''Oligomer molecule: A molecule of intermediate relativ ...
s with the structure (RLi)''x'' where R is the organic group. As the electropositive nature of lithium puts most of the charge density
In electromagnetism, charge density is the amount of electric charge per unit length, surface area, or volume. Volume charge density (symbolized by the Greek letter ρ) is the quantity of charge per unit volume, measured in the SI system in co ...
of the bond on the carbon atom, effectively creating a carbanion
In organic chemistry, a carbanion is an anion in which carbon is trivalent (forms three bonds) and bears a formal negative charge (in at least one significant resonance form).
Formally, a carbanion is the conjugate base of a carbon acid:
:R3C ...
, organolithium compounds are extremely powerful bases and nucleophiles
In chemistry, a nucleophile is a chemical species that forms bonds by donating an electron pair. All molecules and ions with a free pair of electrons or at least one pi bond can act as nucleophiles. Because nucleophiles donate electrons, they are ...
. For use as bases, butyllithium Butyllithium may refer to one of 5 isomeric organolithium reagents of which 3 are commonly used in chemical synthesis:
* ''n''-Butyllithium, abbreviated BuLi or nBuLi
* ''sec''-Butyllithium, abbreviated ''sec''-BuLi or sBuLi, has 2 stereoisomers, ...
s are often used and are commercially available. An example of an organolithium compound is methyllithium
Methyllithium is the simplest organolithium reagent with the empirical formula CH3Li. This s-block organometallic compound adopts an oligomeric structure both in solution and in the solid state. This highly reactive compound, invariably used in so ...
((CH3Li)''x''), which exists in tetrameric (''x'' = 4, tetrahedral) and hexameric (''x'' = 6, octahedral) forms.Grignard reaction
The Grignard reaction () is an organometallic chemical reaction in which alkyl, allyl, vinyl, or aryl-magnesium halides ( Grignard reagent) is added to a carbonyl group in an aldehyde or ketone. This reaction is important for the formation of ...
.aldehyde
In organic chemistry, an aldehyde () is an organic compound containing a functional group with the structure . The functional group itself (without the "R" side chain) can be referred to as an aldehyde but can also be classified as a formyl grou ...
s and ketone
In organic chemistry, a ketone is a functional group with the structure R–C(=O)–R', where R and R' can be a variety of carbon-containing substituents. Ketones contain a carbonyl group –C(=O)– (which contains a carbon-oxygen double bo ...
s by reaction with metal carbonyl
In organic chemistry, a carbonyl group is a functional group composed of a carbon atom double-bonded to an oxygen atom: C=O. It is common to several classes of organic compounds, as part of many larger functional groups. A compound containing a ...
s. The reaction with nickel tetracarbonyl
Nickel carbonyl (IUPAC name: tetracarbonylnickel) is a nickel(0) organometallic compound with the formula Ni(CO)4. This colorless liquid is the principal carbonyl of nickel. It is an intermediate in the Mond process for producing very high-pur ...
, for example, proceeds through an unstable acyl nickel carbonyl complex which then undergoes electrophilic substitution to give the desired aldehyde (using H+ as the electrophile) or ketone (using an alkyl halide) product.amide
In organic chemistry, an amide, also known as an organic amide or a carboxamide, is a compound with the general formula , where R, R', and R″ represent organic groups or hydrogen atoms. The amide group is called a peptide bond when it is ...
s to give aldehydes and ketones, and symmetrical ketones by reacting with carbon monoxide
Carbon monoxide (chemical formula CO) is a colorless, poisonous, odorless, tasteless, flammable gas that is slightly less dense than air. Carbon monoxide consists of one carbon atom and one oxygen atom connected by a triple bond. It is the simple ...
. They thermally decompose to eliminate a β-hydrogen, producing alkene
In organic chemistry, an alkene is a hydrocarbon containing a carbon–carbon double bond.
Alkene is often used as synonym of olefin, that is, any hydrocarbon containing one or more double bonds.H. Stephen Stoker (2015): General, Organic, an ...
s and lithium hydride
Lithium hydride is an inorganic compound with the formula Li H. This alkali metal hydride is a colorless solid, although commercial samples are grey. Characteristic of a salt-like (ionic) hydride, it has a high melting point, and it is not solub ...
: another route is the reaction of ether
In organic chemistry, ethers are a class of compounds that contain an ether group—an oxygen atom connected to two alkyl or aryl groups. They have the general formula , where R and R′ represent the alkyl or aryl groups. Ethers can again be c ...
s with alkyl- and aryllithiums that act as strong bases.carboxylic acid
In organic chemistry, a carboxylic acid is an organic acid that contains a carboxyl group () attached to an R-group. The general formula of a carboxylic acid is or , with R referring to the alkyl, alkenyl, aryl, or other group. Carboxylic ...
s (ArCO2H) and aryl ketones to tertiary carbinols (Ar'2C(Ar)OH). Finally, they may be used to synthesise other organometallic compounds through metal-halogen exchange.
Heavier alkali metals
Unlike the organolithium compounds, the organometallic compounds of the heavier alkali metals are predominantly ionic. The application of organosodium
Organosodium chemistry is the chemistry of organometallic compounds containing a carbon to sodium chemical bond. The application of organosodium compounds in chemistry is limited in part due to competition from organolithium compounds, which are ...
compounds in chemistry is limited in part due to competition from organolithium compound
In organometallic chemistry, organolithium reagents are chemical compounds that contain carbon–lithium (C–Li) bonds. These reagents are important in organic synthesis, and are frequently used to transfer the organic group or the lithium atom ...
s, which are commercially available and exhibit more convenient reactivity. The principal organosodium compound of commercial importance is sodium cyclopentadienide
Sodium cyclopentadienide is an organosodium compound with the formula C5H5Na. The compound is often abbreviated as NaCp, where Cp− is the cyclopentadienide anion. Sodium cyclopentadienide is a colorless solid, although samples often are pi ...
. Sodium tetraphenylborate
Sodium tetraphenylborate is the organic compound with the formula NaB(C6H5)4. It is a salt, wherein the anion consists of four phenyl rings bonded to boron. This white crystalline solid is used to prepare other tetraphenylborate salts, which are o ...
can also be classified as an organosodium compound since in the solid state sodium is bound to the aryl groups. Organometallic compounds of the higher alkali metals are even more reactive than organosodium compounds and of limited utility. A notable reagent is Schlosser's base Schlosser's base (or Lochmann-Schlosser base) describes various superbasic mixtures of an alkyllithium compound and a potassium alkoxide. The reagent is named after Manfred Schlosser, although he uses the term ''LICKOR superbase'' (LIC denoting the ...
, a mixture of ''n''-butyllithium and potassium ''tert''-butoxide. This reagent reacts with propene
Propylene, also known as propene, is an unsaturated organic compound with the chemical formula CH3CH=CH2. It has one double bond, and is the second simplest member of the alkene class of hydrocarbons. It is a colorless gas with a faint petrole ...
to form the compound allylpotassium (KCH2CHCH2). ''cis''-2-Butene and ''trans''-2-butene equilibrate when in contact with alkali metals. Whereas isomerisation is fast with lithium and sodium, it is slow with the heavier alkali metals. The heavier alkali metals also favour the sterically congested conformation. Several crystal structures of organopotassium compounds have been reported, establishing that they, like the sodium compounds, are polymeric.ether
In organic chemistry, ethers are a class of compounds that contain an ether group—an oxygen atom connected to two alkyl or aryl groups. They have the general formula , where R and R′ represent the alkyl or aryl groups. Ethers can again be c ...
s, generating alkoxides. Unlike alkyllithium compounds, alkylsodiums and alkylpotassiums cannot be made by reacting the metals with alkyl halides because Wurtz coupling
In organic chemistry, the Wurtz reaction, named after Charles Adolphe Wurtz, is a coupling reaction whereby two alkyl halides are treated with sodium metal to form a higher alkane.
: 2 R−X + 2 Na → R−R + 2 NaX
The reaction is of little v ...
occurs:nickel arsenide
Nickeline or niccolite is a mineral consisting primarily of nickel arsenide (NiAs). The naturally-occurring mineral contains roughly 43.9% nickel and 56.1% arsenic by mass, but composition of the mineral may vary slightly.
Small quantities of ...
structure with discrete methyl anions and potassium cations.cyclopentadiene
Cyclopentadiene is an organic compound with the chemical formula, formula C5H6.LeRoy H. Scharpen and Victor W. Laurie (1965): "Structure of cyclopentadiene". ''The Journal of Chemical Physics'', volume 43, issue 8, pages 2765-2766. It is often ab ...
s and terminal alkynes, to give salts. Liquid ammonia, ether, or hydrocarbon solvents are used, the most common of which being tetrahydrofuran
Tetrahydrofuran (THF), or oxolane, is an organic compound with the formula (CH2)4O. The compound is classified as heterocyclic compound, specifically a cyclic ether. It is a colorless, water-miscible organic liquid with low viscosity. It is ma ...
. The most important of these compounds is sodium cyclopentadienide
Sodium cyclopentadienide is an organosodium compound with the formula C5H5Na. The compound is often abbreviated as NaCp, where Cp− is the cyclopentadienide anion. Sodium cyclopentadienide is a colorless solid, although samples often are pi ...
, NaC5H5, an important precursor to many transition metal cyclopentadienyl derivatives.cyclooctatetraene
1,3,5,7-Cyclooctatetraene (COT) is an unsaturated derivative of cyclooctane, with the formula C8H8. It is also known as nnulene. This polyunsaturated hydrocarbon is a colorless to light yellow flammable liquid at room temperature. Because of ...
in tetrahydrofuran to give alkali metal cyclooctatetraenide
1,3,5,7-Cyclooctatetraene (COT) is an unsaturated derivative of cyclooctane, with the formula C8H8. It is also known as nnulene. This polyunsaturated hydrocarbon is a colorless to light yellow flammable liquid at room temperature. Because of ...
s; for example, dipotassium cyclooctatetraenide
Dipotassium cyclooctatetraenide, sometimes abbreviated K2COT, is an Organopotassium chemistry, organopotassium compound with the formula K2C8H8. It is a brown solid that is used as a precursor to cyclooctatetraenide complexes, such as uranocene ( ...
(K2C8H8) is an important precursor to many metal cyclooctatetraenyl derivatives, such as uranocene
Uranocene, U(C8H8)2, is an organouranium compound composed of a uranium atom sandwiched between two cyclooctatetraenide rings. It was one of the first organoactinide compounds to be synthesized. It is a green air-sensitive solid that dissolves in ...
.sodium naphthalenide
Sodium naphthalene is an organic salt with the chemical formula Na+. In the research laboratory, it is used as a reductant in the synthesis of organic, organometallic, and inorganic chemistry. It is usually generated in situ. When isolated, it ...
, Na+ 10H8•sup>−, a strong reducing agent.
Representative reactions of alkali metals
''Reaction with oxygen''
Upon reacting with oxygen, alkali metals form oxides
An oxide () is a chemical compound that contains at least one oxygen atom and one other element in its chemical formula. "Oxide" itself is the dianion of oxygen, an O2– (molecular) ion. with oxygen in the oxidation state of −2. Most of the E ...
, peroxides
In chemistry, peroxides are a group of compounds with the structure , where R = any element. The group in a peroxide is called the peroxide group or peroxo group. The nomenclature is somewhat variable.
The most common peroxide is hydrogen p ...
, superoxides
In chemistry, a superoxide is a compound that contains the superoxide ion, which has the chemical formula . The systematic name of the anion is dioxide(1−). The reactive oxygen ion superoxide is particularly important as the product of the ...
and suboxides
Suboxides are a class of oxides wherein the electropositive element is in excess relative to the “normal” oxides. When the electropositive element is a metal, the compounds are sometimes referred to as “metal-rich”. Thus the normal oxide ...
. However, the first three are more common. The table below["Inorganic Chemistry" by Gary L. Miessler and Donald A. Tar, 6th edition, Pearson] shows the types of compounds formed in reaction with oxygen. The compound in brackets represents the minor product of combustion.
The alkali metal peroxides are ionic compounds that are unstable in water. The peroxide anion is weakly bound to the cation, and it is hydrolysed, forming stronger covalent bonds.
:Na2O2 + 2H2O → 2NaOH + H2O2
The other oxygen compounds are also unstable in water.
:2KO2 + 2H2O → 2KOH + H2O2 + O2
:Li2O + H2O → 2LiOH
''Reaction with sulfur''
With sulfur, they form sulfides and polysulfides.
:2Na + 1/8S8 → Na2S + 1/8S8 → Na2S2...Na2S7
Because alkali metal sulfides are essentially salts of a weak acid and a strong base, they form basic solutions.
:S2- + H2O → HS− + HO−
:HS− + H2O → H2S + HO−
''Reaction with nitrogen''
Lithium is the only metal that combines directly with nitrogen at room temperature.
:3Li + 1/3N2 → Li3N
Li3N can react with water to liberate ammonia.
:Li3N + 3H2O → 3LiOH + NH3
''Reaction with hydrogen''
With hydrogen, alkali metals form saline hydrides that hydrolyse in water.
:Na + H2 → NaH (at high temperatures)
:NaH + H2O → NaOH + H2
''Reaction with carbon''
Lithium is the only metal that reacts directly with carbon to give dilithium acetylide. Na and K can react with acetylene
Acetylene (systematic name: ethyne) is the chemical compound with the formula and structure . It is a hydrocarbon and the simplest alkyne. This colorless gas is widely used as a fuel and a chemical building block. It is unstable in its pure ...
to give acetylides.
:2Li + 2C → Li2C2
:Na + C2H2 → NaC2H + 1/2H2 (at 1500C)
:Na + NaC2H → Na2C2 (at 2200C)
''Reaction with water''
On reaction with water, they generate hydroxide ions and hydrogen
Hydrogen is the chemical element with the symbol H and atomic number 1. Hydrogen is the lightest element. At standard conditions hydrogen is a gas of diatomic molecules having the formula . It is colorless, odorless, tasteless, non-toxic, an ...
gas. This reaction is vigorous and highly exothermic and the hydrogen resulted may ignite in air or even explode in the case of Rb and Cs.Titanium
Titanium is a chemical element with the symbol Ti and atomic number 22. Found in nature only as an oxide, it can be reduced to produce a lustrous transition metal with a silver color, low density, and high strength, resistant to corrosion in ...
is produced industrially by the reduction of titanium tetrachloride with Na at 4000C (van Arkel–de Boer process
The van Arkel–de Boer process, also known as the iodide process or crystal-bar process, was the first industrial process for the commercial production of pure ductile titanium, zirconium and some other metals. It was developed by Anton Eduard ...
).
:TiCl4 + 4Na → 4NaCl + Ti
''Reaction with organohalide compounds''
Alkali metals react with halogen derivatives to generate hydrocarbon via the Wurtz reaction
In organic chemistry, the Wurtz reaction, named after Charles Adolphe Wurtz, is a coupling reaction whereby two alkyl halides are treated with sodium metal to form a higher alkane.
: 2 R−X + 2 Na → R−R + 2 NaX
The reaction is of little v ...
.
:2CH3-Cl + 2Na → H3C-CH3 + 2NaCl
''Alkali metals in liquid ammonia''
Alkali metals dissolve in liquid ammonia or other donor solvents like aliphatic amines or hexamethylphosphoramide to give blue solutions. These solutions are believed to contain free electrons.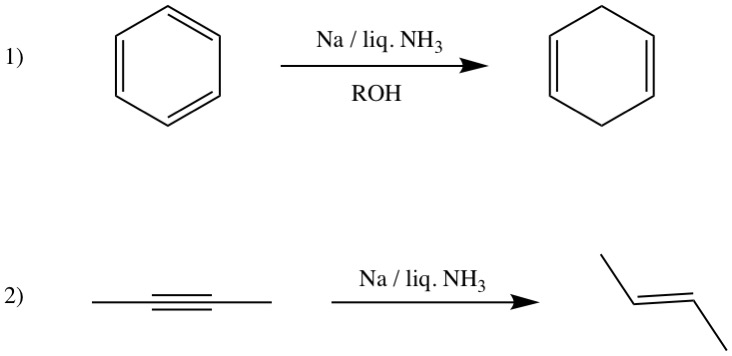 Reaction 1) is known as
Reaction 1) is known as Birch reduction
The Birch reduction is an organic reaction that is used to convert arenes to cyclohexadienes. The reaction is named after the Australian chemist Arthur Birch and involves the organic reduction of aromatic rings in an amine solvent (traditionally ...
.
Other reductions
Extensions
 Although francium is the heaviest alkali metal that has been discovered, there has been some theoretical work predicting the physical and chemical characteristics of hypothetical heavier alkali metals. Being the first period 8 element, the undiscovered element
Although francium is the heaviest alkali metal that has been discovered, there has been some theoretical work predicting the physical and chemical characteristics of hypothetical heavier alkali metals. Being the first period 8 element, the undiscovered element ununennium
Ununennium, also known as eka-francium or element 119, is the hypothetical chemical element with symbol Uue and atomic number 119. ''Ununennium'' and ''Uue'' are the temporary systematic IUPAC name and symbol respectively, which are used until th ...
(element 119) is predicted to be the next alkali metal after francium and behave much like their lighter congeners; however, it is also predicted to differ from the lighter alkali metals in some properties.periodic trends
Periodic trends are specific patterns that are present in the periodic table that illustrate different aspects of a certain element. They were discovered by the Russian chemist Dmitri Mendeleev in the year 1863. Major periodic trends include atom ...
, ignoring relativistic effects would predict ununennium to be even more reactive than caesium and francium. This lowered reactivity is due to the relativistic stabilisation of ununennium's valence electron, increasing ununennium's first ionisation energy and decreasing the metallic
Metallic may be a reference to:
*Metal
* Metalloid, metal-like substance
*Metallic bonding, type of chemical bonding
* Metallicity, in astronomy the proportion of elements other than helium and hydrogen in an object
*Metallic color, a color that ...
and ionic radii
Ionic radius, ''r''ion, is the radius of a monatomic ion in an ionic crystal structure. Although neither atoms nor ions have sharp boundaries, they are treated as if they were hard spheres with radii such that the sum of ionic radii of the cation ...
;electron affinity
The electron affinity (''E''ea) of an atom or molecule is defined as the amount of energy released when an electron attaches to a neutral atom or molecule in the gaseous state to form an anion.
::X(g) + e− → X−(g) + energy
Note that this is ...
far beyond that of caesium and francium; indeed, ununennium is expected to have an electron affinity higher than all the alkali metals lighter than it. Relativistic effects also cause a very large drop in the polarisability
Polarizability usually refers to the tendency of matter, when subjected to an electric field, to acquire an electric dipole moment in proportion to that applied field. It is a property of all matter, considering that matter is made up of elementar ...
of ununennium. The stabilisation of ununennium's valence electron and thus the contraction of the 8s orbital cause its atomic radius to be lowered to 240 pm,
The stabilisation of ununennium's valence electron and thus the contraction of the 8s orbital cause its atomic radius to be lowered to 240 pm,oxidation state
In chemistry, the oxidation state, or oxidation number, is the hypothetical charge of an atom if all of its bonds to different atoms were fully ionic. It describes the degree of oxidation (loss of electrons) of an atom in a chemical compound. C ...
s, which are not seen in any other alkali metal,covalent
A covalent bond is a chemical bond that involves the sharing of electrons to form electron pairs between atoms. These electron pairs are known as shared pairs or bonding pairs. The stable balance of attractive and repulsive forces between atoms ...
character, due to the involvement of the 7p3/2 electrons in the bonding. Not as much work has been done predicting the properties of the alkali metals beyond ununennium. Although a simple extrapolation of the periodic table (by the
Not as much work has been done predicting the properties of the alkali metals beyond ununennium. Although a simple extrapolation of the periodic table (by the aufbau principle
The aufbau principle , from the German ''Aufbauprinzip'' (building-up principle), also called the aufbau rule, states that in the ground state of an atom or ion, electrons fill subshells of the lowest available energy, then they fill subshells o ...
) would put element 169, unhexennium, under ununennium, Dirac-Fock calculations predict that the next element after ununennium with alkali-metal-like properties may be element 165, unhexpentium, which is predicted to have the electron configuration g5g18 6f14 7d10 8s2 8p1/22 9s1.group 11 element
Group 11, by modern IUPAC numbering, is a group of chemical elements in the periodic table, consisting of copper (Cu), silver (Ag), and gold (Au), and roentgenium (Rg), although no chemical experiments have yet been carried out to confirm that ...
, and while its physical and atomic properties would be closer to the former, its chemistry may be closer to that of the latter. Further calculations show that unhexpentium would follow the trend of increasing ionisation energy beyond caesium, having an ionisation energy comparable to that of sodium, and that it should also continue the trend of decreasing atomic radii beyond caesium, having an atomic radius comparable to that of potassium.alkaline earth metal
The alkaline earth metals are six chemical elements in group 2 of the periodic table. They are beryllium (Be), magnesium (Mg), calcium (Ca), strontium (Sr), barium (Ba), and radium (Ra).. The elements have very similar properties: they are al ...
s both being s-block
A block of the periodic table is a set of elements unified by the atomic orbitals their valence electrons or vacancies lie in. The term appears to have been first used by Charles Janet. Each block is named after its characteristic orbital: s-blo ...
elements, these predictions for the trends and properties of ununennium and unhexpentium also mostly hold quite similarly for the corresponding alkaline earth metals unbinilium
Unbinilium, also known as eka-radium or simply element 120, is the hypothetical chemical element in the periodic table with symbol Ubn and atomic number 120. ''Unbinilium'' and ''Ubn'' are the temporary systematic IUPAC name and symbol, which ar ...
(Ubn) and unhexhexium (Uhh).
Pseudo-alkali metals
Many other substances are similar to the alkali metals in their tendency to form monopositive cations. Analogously to the pseudohalogen
Pseudohalogens are polyatomic analogues of halogens, whose chemistry, resembling that of the true halogens, allows them to substitute for halogens in several classes of chemical compounds. Pseudohalogens occur in pseudohalogen molecules, inorganic ...
s, they have sometimes been called "pseudo-alkali metals". These substances include some elements and many more polyatomic ion
A polyatomic ion, also known as a molecular ion, is a covalent bonded set of two or more atoms, or of a metal complex, that can be considered to behave as a single unit and that has a net charge that is not zero. The term molecule may or may not ...
s; the polyatomic ions are especially similar to the alkali metals in their large size and weak polarising power.
Hydrogen
The element hydrogen
Hydrogen is the chemical element with the symbol H and atomic number 1. Hydrogen is the lightest element. At standard conditions hydrogen is a gas of diatomic molecules having the formula . It is colorless, odorless, tasteless, non-toxic, an ...
, with one electron per neutral atom, is usually placed at the top of Group 1 of the periodic table for convenience, but hydrogen is not normally considered to be an alkali metal;diatomic
Diatomic molecules () are molecules composed of only two atoms, of the same or different chemical elements. If a diatomic molecule consists of two atoms of the same element, such as hydrogen () or oxygen (), then it is said to be homonuclear. Ot ...
gas consisting of two atoms per molecule (H2); however, the alkali metals form diatomic molecules (such as dilithium
Dilithium, Li2, is a strongly electrophilic, diatomic molecule comprising two lithium atoms covalently bonded together. Li2 is known in the gas phase.
It has a bond order of 1, an internuclear separation of 267.3 pm and a bond energy o ...
, Li2) only at high temperatures, when they are in the gas
Gas is one of the four fundamental states of matter (the others being solid, liquid, and plasma).
A pure gas may be made up of individual atoms (e.g. a noble gas like neon), elemental molecules made from one type of atom (e.g. oxygen), or ...
eous state.
Hydrogen, like the alkali metals, has one valence electron
In chemistry and physics, a valence electron is an electron in the outer shell associated with an atom, and that can participate in the formation of a chemical bond if the outer shell is not closed. In a single covalent bond, a shared pair forms ...
[ and reacts easily with the ]halogen
The halogens () are a group in the periodic table consisting of five or six chemically related elements: fluorine (F), chlorine (Cl), bromine (Br), iodine (I), astatine (At), and tennessine (Ts). In the modern IUPAC nomenclature, this group is ...
s,[ but the similarities mostly end there because of the small size of a bare proton H+ compared to the alkali metal cations.][ Its placement above lithium is primarily due to its ]electron configuration
In atomic physics and quantum chemistry, the electron configuration is the distribution of electrons of an atom or molecule (or other physical structure) in atomic or molecular orbitals. For example, the electron configuration of the neon atom ...
.fluorine
Fluorine is a chemical element with the symbol F and atomic number 9. It is the lightest halogen and exists at standard conditions as a highly toxic, pale yellow diatomic gas. As the most electronegative reactive element, it is extremely reacti ...
due to their similar chemical properties, though the resemblance is likewise not absolute.[Huheey, J.E.; Keiter, E.A. and Keiter, R.L. (1993) ''Inorganic Chemistry: Principles of Structure and Reactivity'', 4th edition, HarperCollins, New York, USA.][James, A.M. and Lord, M.P. (1992) ''Macmillan's Chemical and Physical Data'', Macmillan, London, UK.] As only one additional electron is required to fill in the outermost shell of the hydrogen atom, hydrogen often behaves like a halogen, forming the negative hydride
In chemistry, a hydride is formally the anion of hydrogen( H−). The term is applied loosely. At one extreme, all compounds containing covalently bound H atoms are called hydrides: water (H2O) is a hydride of oxygen, ammonia is a hydride of ...
ion, and is very occasionally considered to be a halogen on that basis. (The alkali metals can also form negative ions, known as alkalide An alkalide is a chemical compound in which alkali metal atoms are anions (negative ions) with a charge or oxidation state of −1. Until the first discovery of alkalides in the 1970s, alkali metals were known to appear in salts only as cations ...
s, but these are little more than laboratory curiosities, being unstable.)pressure
Pressure (symbol: ''p'' or ''P'') is the force applied perpendicular to the surface of an object per unit area over which that force is distributed. Gauge pressure (also spelled ''gage'' pressure)The preferred spelling varies by country and e ...
s, such as those found at the cores of Jupiter
Jupiter is the fifth planet from the Sun and the List of Solar System objects by size, largest in the Solar System. It is a gas giant with a mass more than two and a half times that of all the other planets in the Solar System combined, but ...
and Saturn
Saturn is the sixth planet from the Sun and the second-largest in the Solar System, after Jupiter. It is a gas giant with an average radius of about nine and a half times that of Earth. It has only one-eighth the average density of Earth; h ...
, hydrogen does become metallic and behaves like an alkali metal; in this phase, it is known as metallic hydrogen
Metallic hydrogen is a phase of hydrogen in which it behaves like an electrical conductor. This phase was predicted in 1935 on theoretical grounds by Eugene Wigner and Hillard Bell Huntington.
At high pressure and temperatures, metallic hydrogen c ...
. The electrical resistivity
Electrical resistivity (also called specific electrical resistance or volume resistivity) is a fundamental property of a material that measures how strongly it resists electric current. A low resistivity indicates a material that readily allows ...
of liquid metallic hydrogen
Metallic hydrogen is a phase of hydrogen in which it behaves like an electrical conductor. This phase was predicted in 1935 on theoretical grounds by Eugene Wigner and Hillard Bell Huntington.
At high pressure and temperatures, metallic hydrogen c ...
at 3000 K is approximately equal to that of liquid rubidium
Rubidium is the chemical element with the symbol Rb and atomic number 37. It is a very soft, whitish-grey solid in the alkali metal group, similar to potassium and caesium. Rubidium is the first alkali metal in the group to have a density higher ...
and caesium
Caesium (IUPAC spelling) (or cesium in American English) is a chemical element with the symbol Cs and atomic number 55. It is a soft, silvery-golden alkali metal with a melting point of , which makes it one of only five elemental metals that a ...
at 2000 K at the respective pressures when they undergo a nonmetal-to-metal transition.
The 1s1 electron configuration of hydrogen, while analogous to that of the alkali metals (ns1), is unique because there is no 1p subshell. Hence it can lose an electron to form the hydron Hydron has the following meanings:
* Hydron (chemistry)
In chemistry, the hydron, informally called proton, is the cationic form of atomic hydrogen, represented with the symbol . The general term "hydron", endorsed by the IUPAC, encompasses cati ...
H+, or gain one to form the hydride
In chemistry, a hydride is formally the anion of hydrogen( H−). The term is applied loosely. At one extreme, all compounds containing covalently bound H atoms are called hydrides: water (H2O) is a hydride of oxygen, ammonia is a hydride of ...
ion H−.ionisation energy
Ionization, or Ionisation is the process by which an atom or a molecule acquires a negative or positive charge by gaining or losing electrons, often in conjunction with other chemical changes. The resulting electrically charged atom or molecule i ...
and electron affinity
The electron affinity (''E''ea) of an atom or molecule is defined as the amount of energy released when an electron attaches to a neutral atom or molecule in the gaseous state to form an anion.
::X(g) + e− → X−(g) + energy
Note that this is ...
, but hydrogen cannot be tetravalent. Thus none of the three placements are entirely satisfactory, although group 1 is the most common placement (if one is chosen) because the hydron is by far the most important of all monatomic hydrogen species, being the foundation of acid-base chemistry.hydrogen bond
In chemistry, a hydrogen bond (or H-bond) is a primarily electrostatic force of attraction between a hydrogen (H) atom which is covalently bound to a more electronegative "donor" atom or group (Dn), and another electronegative atom bearing a ...
s, which are an effect of charge-transfer, electrostatic
Electrostatics is a branch of physics that studies electric charges at rest (static electricity).
Since classical times, it has been known that some materials, such as amber, attract lightweight particles after rubbing. The Greek word for amber ...
, and electron correlative contributing phenomena.
Ammonium and derivatives
 The
The ammonium
The ammonium cation is a positively-charged polyatomic ion with the chemical formula or . It is formed by the protonation of ammonia (). Ammonium is also a general name for positively charged or protonated substituted amines and quaternary a ...
ion () has very similar properties to the heavier alkali metals, acting as an alkali metal intermediate between potassium and rubidium,salts
In chemistry, a salt is a chemical compound consisting of an ionic assembly of positively charged cations and negatively charged anions, which results in a compound with no net electric charge. A common example is table salt, with positively cha ...
are soluble
In chemistry, solubility is the ability of a substance, the solute, to form a solution with another substance, the solvent. Insolubility is the opposite property, the inability of the solute to form such a solution.
The extent of the solubil ...
in water, a property which ammonium salts share. Ammonium is expected to behave stably as a metal ( ions in a sea of delocalised electrons) at very high pressures (though less than the typical pressure where transitions from insulating to metallic behaviour occur around, 100 GPa
Grading in education is the process of applying standardized measurements for varying levels of achievements in a course. Grades can be assigned as letters (usually A through F), as a range (for example, 1 to 6), as a percentage, or as a numbe ...
), and could possibly occur inside the ice giants
An ice giant is a giant planet composed mainly of elements heavier than hydrogen and helium, such as oxygen, carbon, nitrogen, and sulfur. There are two ice giants in the Solar System: Uranus and Neptune.
In astrophysics and planetary science th ...
Uranus
Uranus is the seventh planet from the Sun. Its name is a reference to the Greek god of the sky, Uranus (mythology), Uranus (Caelus), who, according to Greek mythology, was the great-grandfather of Ares (Mars (mythology), Mars), grandfather ...
and Neptune
Neptune is the eighth planet from the Sun and the farthest known planet in the Solar System. It is the fourth-largest planet in the Solar System by diameter, the third-most-massive planet, and the densest giant planet. It is 17 times ...
, which may have significant impacts on their interior magnetic fields.ammonia
Ammonia is an inorganic compound of nitrogen and hydrogen with the formula . A stable binary hydride, and the simplest pnictogen hydride, ammonia is a colourless gas with a distinct pungent smell. Biologically, it is a common nitrogenous was ...
and dihydrogen molecules to metallic ammonium may occur at pressures just below 25 GPa.alkylammonium In organic chemistry, alkylammonium refers to cations of the formula 4−nNHnsup>+, where R = alkyl and 1≤ n ≤ 4. The cations with ''four'' alkyl substituents, i.e., 4Nsup>+, are further classified as quaternary ammonium cations and are discu ...
cations, in which some of the hydrogen atoms in the ammonium cation are replaced by alkyl or aryl groups. In particular, the quaternary ammonium cation
In chemistry, quaternary ammonium cations, also known as quats, are positively charged polyatomic ions of the structure , R being an alkyl group or an aryl group. Unlike the ammonium ion () and the primary, secondary, or tertiary ammonium cations ...
s () are very useful since they are permanently charged, and they are often used as an alternative to the expensive Cs+ to stabilise very large and very easily polarisable anions such as .pnictogen
A pnictogen ( or ; from grc, πνῑ́γω "to choke" and -gen, "generator") is any of the chemical elements in group 15 of the periodic table. Group 15 is also known as the nitrogen group or nitrogen family. Group 15 consists of the ele ...
s), creating a phosphonium
In polyatomic cations with the chemical formula (where R is a hydrogen or an alkyl, aryl, or halide group). These cations have tetrahedral structures. The salts are generally colorless or take the color of the anions.
Types of phosphonium ...
() or arsonium
The arsonium cation is a positively charged polyatomic ion with the chemical formula . An arsonium salt is a salt containing either the arsonium () cation, such as arsonium bromide () and arsonium iodide (), which can be synthesized by reacting ...
() cation that can itself be substituted similarly; while stibonium () itself is not known, some of its organic derivatives are characterised.
Cobaltocene and derivatives
Cobaltocene
Cobaltocene, known also as bis(cyclopentadienyl)cobalt(II) or even "bis Cp cobalt", is an organocobalt compound with the formula Co(C5H5)2. It is a dark purple solid that sublimes readily slightly above room temperature. Cobaltocene was discovere ...
, Co(C5H5)2, is a metallocene
A metallocene is a compound typically consisting of two cyclopentadienyl anions (, abbreviated Cp) bound to a metallic element, metal center (M) in the oxidation state II, with the resulting general formula Closely related to the metallocenes are ...
, the cobalt
Cobalt is a chemical element with the symbol Co and atomic number 27. As with nickel, cobalt is found in the Earth's crust only in a chemically combined form, save for small deposits found in alloys of natural meteoric iron. The free element, pr ...
analogue of ferrocene
Ferrocene is an organometallic compound with the formula . The molecule is a complex consisting of two cyclopentadienyl rings bound to a central iron atom. It is an orange solid with a camphor-like odor, that sublimes above room temperature, a ...
. It is a dark purple solid. Cobaltocene has 19 valence electrons, one more than usually found in organotransition metal complexes, such as its very stable relative, ferrocene, in accordance with the 18-electron rule. This additional electron occupies an orbital that is antibonding with respect to the Co–C bonds. Consequently, many chemical reactions of Co(C5H5)2 are characterized by its tendency to lose this "extra" electron, yielding a very stable 18-electron cation known as cobaltocenium. Many cobaltocenium salts coprecipitate with caesium salts, and cobaltocenium hydroxide is a strong base that absorbs atmospheric carbon dioxide to form cobaltocenium carbonate.decamethylcobaltocene
Decamethylcobaltocene is an organocobalt compound with the formula Co(C5(CH3)5)2, abbreviated CoCp{{su, p=∗, b=2. It is a dark brown solid. This compound is used as a strong reducing agent in organometallic chemistry.
Synthesis
Decamethylco ...
is stronger still due to the combined inductive effect
In chemistry, the inductive effect in a molecule is a local change in the electron density due to electron-withdrawing or electron-donating groups elsewhere in the molecule, resulting in a permanent dipole in a bond.
It is present in a σ (sigm ...
of the ten methyl groups. Cobalt may be substituted by its heavier congener rhodium
Rhodium is a chemical element with the symbol Rh and atomic number 45. It is a very rare, silvery-white, hard, corrosion-resistant transition metal. It is a noble metal and a member of the platinum group. It has only one naturally occurring isoto ...
to give rhodocene
Rhodocene is a chemical compound with the formula . Each molecule contains an atom of rhodium bound between two planar aromatic systems of five carbon atoms known as cyclopentadienyl rings in a sandwich arrangement. It is an organometallic compo ...
, an even stronger reducing agent. Iridocene
Organoiridium chemistry is the chemistry of organometallic compounds containing an iridium-carbon chemical bond. Organoiridium compounds are relevant to many important processes including olefin hydrogenation and the industrial synthesis of ...
(involving iridium
Iridium is a chemical element with the symbol Ir and atomic number 77. A very hard, brittle, silvery-white transition metal of the platinum group, it is considered the second-densest naturally occurring metal (after osmium) with a density of ...
) would presumably be still more potent, but is not very well-studied due to its instability.
Thallium

Thallium
Thallium is a chemical element with the Symbol (chemistry), symbol Tl and atomic number 81. It is a gray post-transition metal that is not found free in nature. When isolated, thallium resembles tin, but discolors when exposed to air. Chemists W ...
is the heaviest stable element in group 13 of the periodic table. At the bottom of the periodic table, the inert pair effect The inert-pair effect is the tendency of the two electrons in the outermost atomic ''s''-orbital to remain unshared in compounds of post-transition metals. The term ''inert-pair effect'' is often used in relation to the increasing stability of oxi ...
is quite strong, because of the relativistic stabilisation of the 6s orbital and the decreasing bond energy as the atoms increase in size so that the amount of energy released in forming two more bonds is not worth the high ionisation energies of the 6s electrons.oxidation state
In chemistry, the oxidation state, or oxidation number, is the hypothetical charge of an atom if all of its bonds to different atoms were fully ionic. It describes the degree of oxidation (loss of electrons) of an atom in a chemical compound. C ...
oxidation state
In chemistry, the oxidation state, or oxidation number, is the hypothetical charge of an atom if all of its bonds to different atoms were fully ionic. It describes the degree of oxidation (loss of electrons) of an atom in a chemical compound. C ...
closely resemble the corresponding potassium or silver
Silver is a chemical element with the Symbol (chemistry), symbol Ag (from the Latin ', derived from the Proto-Indo-European wikt:Reconstruction:Proto-Indo-European/h₂erǵ-, ''h₂erǵ'': "shiny" or "white") and atomic number 47. A soft, whi ...
compounds stoichiometrically due to the similar ionic radii of the Tl+ (164 pm), K+ (152 pm) and Ag+ (129 pm) ions.continental Europe
Continental Europe or mainland Europe is the contiguous continent of Europe, excluding its surrounding islands. It can also be referred to ambiguously as the European continent, – which can conversely mean the whole of Europe – and, by ...
(but not in England) in the years immediately following its discovery,Dmitri Mendeleev
Dmitri Ivanovich Mendeleev (sometimes transliterated as Mendeleyev or Mendeleef) ( ; russian: links=no, Дмитрий Иванович Менделеев, tr. , ; 8 February Old_Style_and_New_Style_dates">O.S._27_January.html" ;"title="O ...
's 1869 periodic table
The periodic table, also known as the periodic table of the (chemical) elements, is a rows and columns arrangement of the chemical elements. It is widely used in chemistry, physics, and other sciences, and is generally seen as an icon of ch ...
and Julius Lothar Meyer
Julius Lothar Meyer (19 August 1830 – 11 April 1895) was a German chemist. He was one of the pioneers in developing the earliest versions of the periodic table of the chemical elements. Russian chemist Dmitri Mendeleev (his chief rival) and he ...
's 1868 periodic table.boron group
The boron group are the chemical elements in group 13 of the periodic table, comprising boron (B), aluminium (Al), gallium (Ga), indium (In), thallium (Tl), and nihonium (Nh). The elements in the boron group are characterized by having three vale ...
and left the space below caesium blank.)[.] While Tl+ is stabilised by the inert pair effect, this inert pair of 6s electrons is still able to participate chemically, so that these electrons are stereochemically active in aqueous solution. Additionally, the thallium halides (except TlF) are quite insoluble in water, and TlI has an unusual structure because of the presence of the stereochemically active inert pair in thallium.
Copper, silver, and gold
The group 11 metals (or coinage metals), copper
Copper is a chemical element with the symbol Cu (from la, cuprum) and atomic number 29. It is a soft, malleable, and ductile metal with very high thermal and electrical conductivity. A freshly exposed surface of pure copper has a pinkis ...
, silver
Silver is a chemical element with the Symbol (chemistry), symbol Ag (from the Latin ', derived from the Proto-Indo-European wikt:Reconstruction:Proto-Indo-European/h₂erǵ-, ''h₂erǵ'': "shiny" or "white") and atomic number 47. A soft, whi ...
, and gold
Gold is a chemical element with the symbol Au (from la, aurum) and atomic number 79. This makes it one of the higher atomic number elements that occur naturally. It is a bright, slightly orange-yellow, dense, soft, malleable, and ductile met ...
, are typically categorised as transition metals given they can form ions with incomplete d-shells. Physically, they have the relatively low melting points and high electronegativity values associated with post-transition metal
The metallic elements in the periodic table located between the transition metals and the chemically weak nonmetallic metalloids have received many names in the literature, such as ''post-transition metals'', ''poor metals'', ''other metals'', ...
s. "The filled ''d'' subshell and free ''s'' electron of Cu, Ag, and Au contribute to their high electrical and thermal conductivity. Transition metals to the left of group 11 experience interactions between ''s'' electrons and the partially filled ''d'' subshell that lower electron mobility." Chemically, the group 11 metals behave like main-group metals in their +1 valence states, and are hence somewhat related to the alkali metals: this is one reason for their previously being labelled as "group IB", paralleling the alkali metals' "group IA". They are occasionally classified as post-transition metals. Their spectra are analogous to those of the alkali metals.paramagnetic
Paramagnetism is a form of magnetism whereby some materials are weakly attracted by an externally applied magnetic field, and form internal, induced magnetic fields in the direction of the applied magnetic field. In contrast with this behavior, d ...
and contribute no colour to their salts, like those of the alkali metals.
In Mendeleev's 1871 periodic table, copper, silver, and gold are listed twice, once under group VIII (with the iron triad
Iron () is a chemical element with symbol Fe (from la, ferrum) and atomic number 26. It is a metal that belongs to the first transition series and group 8 of the periodic table. It is, by mass, the most common element on Earth, right in front ...
and platinum group metal
The platinum-group metals (abbreviated as the PGMs; alternatively, the platinoids, platinides, platidises, platinum group, platinum metals, platinum family or platinum-group elements (PGEs)) are six noble, precious metallic elements clustered t ...
s), and once under group IB. Group IB was nonetheless parenthesised to note that it was tentative. Mendeleev's main criterion for group assignment was the maximum oxidation state of an element: on that basis, the group 11 elements could not be classified in group IB, due to the existence of copper(II) and gold(III) compounds being known at that time.stoichiometries
Stoichiometry refers to the relationship between the quantities of reactants and products before, during, and following chemical reactions.
Stoichiometry is founded on the law of conservation of mass where the total mass of the reactants equals ...
of the +1 compounds of both groups, and not their chemical properties.ammonia
Ammonia is an inorganic compound of nitrogen and hydrogen with the formula . A stable binary hydride, and the simplest pnictogen hydride, ammonia is a colourless gas with a distinct pungent smell. Biologically, it is a common nitrogenous was ...
, as well as having more covalent character in their compounds.electrochemical series
The data values of standard electrode potentials (''E''°) are given in the table below, in volts relative to the standard hydrogen electrode, and are for the following conditions:
* A temperature of .
* An effective concentration of 1 mol ...
, whereas the coinage metals are almost at the very bottom.transition metal
In chemistry, a transition metal (or transition element) is a chemical element in the d-block of the periodic table (groups 3 to 12), though the elements of group 12 (and less often group 3) are sometimes excluded. They are the elements that can ...
character.pseudohalogen
Pseudohalogens are polyatomic analogues of halogens, whose chemistry, resembling that of the true halogens, allows them to substitute for halogens in several classes of chemical compounds. Pseudohalogens occur in pseudohalogen molecules, inorganic ...
because its 5d106s1 configuration has one electron less than the quasi-closed shell 5d106s2 configuration of mercury
Mercury commonly refers to:
* Mercury (planet), the nearest planet to the Sun
* Mercury (element), a metallic chemical element with the symbol Hg
* Mercury (mythology), a Roman god
Mercury or The Mercury may also refer to:
Companies
* Merc ...
.
Production and isolation
The production of pure alkali metals is somewhat complicated due to their extreme reactivity with commonly used substances, such as water.silicate
In chemistry, a silicate is any member of a family of polyatomic anions consisting of silicon and oxygen, usually with the general formula , where . The family includes orthosilicate (), metasilicate (), and pyrosilicate (, ). The name is al ...
ores, all the stable alkali metals may be obtained the same way: sulfuric acid
Sulfuric acid (American spelling and the preferred IUPAC name) or sulphuric acid ( Commonwealth spelling), known in antiquity as oil of vitriol, is a mineral acid composed of the elements sulfur, oxygen and hydrogen, with the molecular formu ...
is first used to dissolve the desired alkali metal ion and aluminium
Aluminium (aluminum in American and Canadian English) is a chemical element with the symbol Al and atomic number 13. Aluminium has a density lower than those of other common metals, at approximately one third that of steel. I ...
(III) ions from the ore (leaching), whereupon basic precipitation removes aluminium ions from the mixture by precipitating it as the hydroxide
Hydroxide is a diatomic anion with chemical formula OH−. It consists of an oxygen and hydrogen atom held together by a single covalent bond, and carries a negative electric charge. It is an important but usually minor constituent of water. I ...
. The remaining insoluble alkali metal carbonate
A carbonate is a salt of carbonic acid (H2CO3), characterized by the presence of the carbonate ion, a polyatomic ion with the formula . The word ''carbonate'' may also refer to a carbonate ester, an organic compound containing the carbonate g ...
is then precipitated selectively; the salt is then dissolved in hydrochloric acid
Hydrochloric acid, also known as muriatic acid, is an aqueous solution of hydrogen chloride. It is a colorless solution with a distinctive pungent smell. It is classified as a strong acid
Acid strength is the tendency of an acid, symbol ...
to produce the chloride. The result is then left to evaporate and the alkali metal can then be isolated.calcium chloride
Calcium chloride is an inorganic compound, a salt with the chemical formula . It is a white crystalline solid at room temperature, and it is highly soluble in water. It can be created by neutralising hydrochloric acid with calcium hydroxide.
Ca ...
typically added to lower the melting point of the mixture. The heavier alkali metals, however, are more typically isolated in a different way, where a reducing agent (typically sodium for potassium and magnesium
Magnesium is a chemical element with the symbol Mg and atomic number 12. It is a shiny gray metal having a low density, low melting point and high chemical reactivity. Like the other alkaline earth metals (group 2 of the periodic ta ...
or calcium
Calcium is a chemical element with the symbol Ca and atomic number 20. As an alkaline earth metal, calcium is a reactive metal that forms a dark oxide-nitride layer when exposed to air. Its physical and chemical properties are most similar to ...
for the heaviest alkali metals) is used to reduce the alkali metal chloride. The liquid or gaseous product (the alkali metal) then undergoes fractional distillation
Fractional distillation is the separation of a mixture into its component parts, or fractions. Chemical compounds are separated by heating them to a temperature at which one or more fractions of the mixture will vaporize. It uses distillation to ...
for purification.pyrolysis
The pyrolysis (or devolatilization) process is the thermal decomposition of materials at elevated temperatures, often in an inert atmosphere. It involves a change of chemical composition. The word is coined from the Greek-derived elements ''py ...
of the corresponding alkali metal azide
In chemistry, azide is a linear, polyatomic anion with the formula and structure . It is the conjugate base of hydrazoic acid . Organic azides are organic compounds with the formula , containing the azide functional group. The dominant applic ...
, which yields the metal for sodium, potassium, rubidium, and caesium and the nitride for lithium.[
Lithium salts have to be extracted from the water of ]mineral spring
Mineral springs are naturally occurring springs that produces hard water, water that contains dissolved minerals. Salts, sulfur compounds, and gases are among the substances that can be dissolved in the spring water during its passage underg ...
s, brine
Brine is a high-concentration solution of salt (NaCl) in water (H2O). In diverse contexts, ''brine'' may refer to the salt solutions ranging from about 3.5% (a typical concentration of seawater, on the lower end of that of solutions used for br ...
pools, and brine deposits. The metal is produced electrolytically from a mixture of fused lithium chloride
Lithium chloride is a chemical compound with the formula Li Cl. The salt is a typical ionic compound (with certain covalent characteristics), although the small size of the Li+ ion gives rise to properties not seen for other alkali metal chlor ...
and potassium chloride
Potassium chloride (KCl, or potassium salt) is a metal halide salt composed of potassium and chlorine. It is odorless and has a white or colorless vitreous crystal appearance. The solid dissolves readily in water, and its solutions have a salt ...
.seabed
The seabed (also known as the seafloor, sea floor, ocean floor, and ocean bottom) is the bottom of the ocean. All floors of the ocean are known as 'seabeds'.
The structure of the seabed of the global ocean is governed by plate tectonics. Most of ...
,electrolysis
In chemistry and manufacturing, electrolysis is a technique that uses direct electric current (DC) to drive an otherwise non-spontaneous chemical reaction. Electrolysis is commercially important as a stage in the separation of elements from n ...
of sodium chloride
Sodium chloride , commonly known as salt (although sea salt also contains other chemical salts), is an ionic compound with the chemical formula NaCl, representing a 1:1 ratio of sodium and chloride ions. With molar masses of 22.99 and 35.45 g ...
by lowering the melting point of the substance to below 700 °C through the use of a Downs cell.sylvite
Sylvite, or sylvine, is potassium chloride (KCl) in natural mineral form. It forms crystals in the isometric system very similar to normal rock salt, halite ( NaCl). The two are, in fact, isomorphous. Sylvite is colorless to white with shades of ...
(potassium chloride
Potassium chloride (KCl, or potassium salt) is a metal halide salt composed of potassium and chlorine. It is odorless and has a white or colorless vitreous crystal appearance. The solid dissolves readily in water, and its solutions have a salt ...
).potassium chloride
Potassium chloride (KCl, or potassium salt) is a metal halide salt composed of potassium and chlorine. It is odorless and has a white or colorless vitreous crystal appearance. The solid dissolves readily in water, and its solutions have a salt ...
or potassium hydroxide
Potassium hydroxide is an inorganic compound with the formula K OH, and is commonly called caustic potash.
Along with sodium hydroxide (NaOH), KOH is a prototypical strong base. It has many industrial and niche applications, most of which exp ...
, found extensively in places such as Canada, Russia, Belarus, Germany, Israel, United States, and Jordan, in a method similar to how sodium was produced in the late 1800s and early 1900s.seawater
Seawater, or salt water, is water from a sea or ocean. On average, seawater in the world's oceans has a salinity of about 3.5% (35 g/L, 35 ppt, 600 mM). This means that every kilogram (roughly one liter by volume) of seawater has appro ...
.fractional distillation
Fractional distillation is the separation of a mixture into its component parts, or fractions. Chemical compounds are separated by heating them to a temperature at which one or more fractions of the mixture will vaporize. It uses distillation to ...
technique. Because metallic caesium is too reactive to handle, it is normally offered as caesium azide
Caesium azide or cesium azide is an inorganic compound of caesium and nitrogen. It is a salt of azide with the formula .
Structure
adopts the same structure as , , and , crystallizing in a tetragonal distorted caesium chloride structure where ...
(CsN3). Caesium hydroxide
Caesium hydroxide is a strong base (pKa= 15.76) containing the highly reactive alkali metal caesium, much like the other alkali metal hydroxides such as sodium hydroxide and potassium hydroxide. Caesium hydroxide is corrosive enough to quickly ...
is formed when caesium interacts aggressively with water
Water (chemical formula ) is an inorganic, transparent, tasteless, odorless, and nearly colorless chemical substance, which is the main constituent of Earth's hydrosphere and the fluids of all known living organisms (in which it acts as a ...
and ice
Ice is water frozen into a solid state, typically forming at or below temperatures of 0 degrees Celsius or Depending on the presence of impurities such as particles of soil or bubbles of air, it can appear transparent or a more or less opaq ...
(CsOH).
Rubidium
Rubidium is the chemical element with the symbol Rb and atomic number 37. It is a very soft, whitish-grey solid in the alkali metal group, similar to potassium and caesium. Rubidium is the first alkali metal in the group to have a density higher ...
is the 16th most prevalent element in the earth's crust, however it is quite rare. Some minerals found in North America, South Africa, Russia, and Canada contain rubidium. Some potassium minerals (lepidolite
Lepidolite is a lilac-gray or rose-colored member of the mica group of minerals with chemical formula . It is the most abundant lithium-bearing mineral and is a secondary source of this metal. It is the major source of the alkali metal rubidium.
...
s, biotite
Biotite is a common group of phyllosilicate minerals within the mica group, with the approximate chemical formula . It is primarily a solid-solution series between the iron-endmember annite, and the magnesium-endmember phlogopite; more alumino ...
s, feldspar
Feldspars are a group of rock-forming aluminium tectosilicate minerals, also containing other cations such as sodium, calcium, potassium, or barium. The most common members of the feldspar group are the ''plagioclase'' (sodium-calcium) feldsp ...
, carnallite
Carnallite (also carnalite) is an evaporite mineral, a hydrated potassium magnesium chloride with formula KMgCl3·6(H2O). It is variably colored yellow to white, reddish, and sometimes colorless or blue. It is usually massive to fibrous with rare ...
) contain it, together with caesium. Pollucite
Pollucite is a zeolite mineral with the formula with iron, calcium, rubidium and potassium as common substituting elements. It is important as a significant ore of caesium and sometimes rubidium. It forms a solid solution series with analcime. I ...
, carnallite
Carnallite (also carnalite) is an evaporite mineral, a hydrated potassium magnesium chloride with formula KMgCl3·6(H2O). It is variably colored yellow to white, reddish, and sometimes colorless or blue. It is usually massive to fibrous with rare ...
, leucite
Leucite is a rock-forming mineral of the feldspathoid group, silica-undersaturated and composed of potassium and aluminium tectosilicate KAlSi2O6. Crystals have the form of cubic icositetrahedra but, as first observed by Sir David Brewster in 18 ...
, and lepidolite
Lepidolite is a lilac-gray or rose-colored member of the mica group of minerals with chemical formula . It is the most abundant lithium-bearing mineral and is a secondary source of this metal. It is the major source of the alkali metal rubidium.
...
are all minerals that contain rubidium. As a by-product of lithium extraction, it is commercially obtained from lepidolite
Lepidolite is a lilac-gray or rose-colored member of the mica group of minerals with chemical formula . It is the most abundant lithium-bearing mineral and is a secondary source of this metal. It is the major source of the alkali metal rubidium.
...
. Rubidium is also found in potassium rocks and brine
Brine is a high-concentration solution of salt (NaCl) in water (H2O). In diverse contexts, ''brine'' may refer to the salt solutions ranging from about 3.5% (a typical concentration of seawater, on the lower end of that of solutions used for br ...
s, which is a commercial supply. The majority of rubidium is now obtained as a byproduct of refining lithium. Rubidium is used in vacuum tube
A vacuum tube, electron tube, valve (British usage), or tube (North America), is a device that controls electric current flow in a high vacuum between electrodes to which an electric voltage, potential difference has been applied.
The type kn ...
s as a getter
A getter is a deposit of reactive material that is placed inside a vacuum system to complete and maintain the vacuum. When gas molecules strike the getter material, they combine with it chemically or by . Thus the getter removes small amounts of ...
, a material that combines with and removes trace gases from vacuum tubes. For several years in the 1950s and 1960s, a by-product of the potassium production called Alkarb was a main source for rubidium. Alkarb contained 21% rubidium while the rest was potassium and a small fraction of caesium. Today the largest producers of caesium, for example the
For several years in the 1950s and 1960s, a by-product of the potassium production called Alkarb was a main source for rubidium. Alkarb contained 21% rubidium while the rest was potassium and a small fraction of caesium. Today the largest producers of caesium, for example the Tanco Mine
Tanco Mine or Bernic Lake mine is an underground caesium and tantalum mine, owned and since 2019 owned and operated by Sinomine Resource Group on the north west shore of Bernic Lake, Manitoba, Canada. The mine has the largest known deposit of ...
in Manitoba, Canada, produce rubidium as by-product from pollucite
Pollucite is a zeolite mineral with the formula with iron, calcium, rubidium and potassium as common substituting elements. It is important as a significant ore of caesium and sometimes rubidium. It forms a solid solution series with analcime. I ...
.alum
An alum () is a type of chemical compound, usually a hydrated double salt, double sulfate salt (chemistry), salt of aluminium with the general chemical formula, formula , where is a valence (chemistry), monovalent cation such as potassium or a ...
( Cs, Rb) Al( SO4)2·12 H2O, which yields pure rubidium alum after approximately 30 recrystallisations.tonne
The tonne ( or ; symbol: t) is a unit of mass equal to 1000 kilograms. It is a non-SI unit accepted for use with SI. It is also referred to as a metric ton to distinguish it from the non-metric units of the short ton ( United State ...
s per year.pollucite
Pollucite is a zeolite mineral with the formula with iron, calcium, rubidium and potassium as common substituting elements. It is important as a significant ore of caesium and sometimes rubidium. It forms a solid solution series with analcime. I ...
ore is the main method of obtaining pure caesium, extracted from the ore mainly by three methods: acid digestion, alkaline decomposition, and direct reduction.calcium
Calcium is a chemical element with the symbol Ca and atomic number 20. As an alkaline earth metal, calcium is a reactive metal that forms a dark oxide-nitride layer when exposed to air. Its physical and chemical properties are most similar to ...
metal at 750 °C and low pressure.francium-210
Francium (87Fr) has no stable isotopes. A standard atomic weight cannot be given. Its most stable isotope is 223Fr with a half-life of 22 minutes, occurring in trace quantities as an intermediate decay product of 235U.
Of elements whose most sta ...
, and francium-211
Francium (87Fr) has no stable isotopes. A standard atomic weight cannot be given. Its most stable isotope is 223Fr with a half-life of 22 minutes, occurring in trace quantities as an intermediate decay product of 235U.
Of elements whose most sta ...
. The greatest quantity of francium ever assembled to date is about 300,000 neutral atoms,
Applications
 Lithium, sodium, and potassium have many applications, while rubidium and caesium are very useful in academic contexts but do not have many applications yet.
Lithium, sodium, and potassium have many applications, while rubidium and caesium are very useful in academic contexts but do not have many applications yet.lithium-ion batteries
A lithium-ion or Li-ion battery is a type of rechargeable battery which uses the reversible reduction of lithium ions to store energy. It is the predominant battery type used in portable consumer electronics and electric vehicles. It also se ...
, and lithium oxide
Lithium oxide ( O) or lithia is an inorganic chemical compound. It is a white solid. Although not specifically important, many materials are assessed on the basis of their Li2O content. For example, the Li2O content of the principal lithium miner ...
can help process silica. Lithium stearate
Lithium stearate is a chemical compound with the formula LiO2C(CH2)16CH3. It is formally classified as a soap (a salt of a fatty acid). Lithium stearate is a white soft solid, prepared by the reaction of lithium hydroxide and stearic acid.
Lithiu ...
is a thickener and can be used to make lubricating greases; it is produced from lithium hydroxide, which is also used to absorb carbon dioxide
Carbon dioxide (chemical formula ) is a chemical compound made up of molecules that each have one carbon atom covalently double bonded to two oxygen atoms. It is found in the gas state at room temperature. In the air, carbon dioxide is transpar ...
in space capsules and submarines.Lithium chloride
Lithium chloride is a chemical compound with the formula Li Cl. The salt is a typical ionic compound (with certain covalent characteristics), although the small size of the Li+ ion gives rise to properties not seen for other alkali metal chlor ...
is used as a brazing alloy for aluminium parts. Metallic lithium is used in alloys with magnesium and aluminium to give very tough and light alloys.table salt
Salt is a mineral composed primarily of sodium chloride (NaCl), a chemical compound belonging to the larger class of salts; salt in the form of a natural crystalline mineral is known as rock salt or halite. Salt is present in vast quantitie ...
. Sodium salts of fatty acid
In chemistry, particularly in biochemistry, a fatty acid is a carboxylic acid with an aliphatic chain, which is either saturated or unsaturated. Most naturally occurring fatty acids have an unbranched chain of an even number of carbon atoms, fr ...
s are used as soap. Pure sodium metal also has many applications, including use in sodium-vapour lamps, which produce very efficient light compared to other types of lighting,titanium
Titanium is a chemical element with the symbol Ti and atomic number 22. Found in nature only as an oxide, it can be reduced to produce a lustrous transition metal with a silver color, low density, and high strength, resistant to corrosion in ...
and zirconium
Zirconium is a chemical element with the symbol Zr and atomic number 40. The name ''zirconium'' is taken from the name of the mineral zircon, the most important source of zirconium. The word is related to Persian '' zargun'' (zircon; ''zar-gun'', ...
, from their chlorides. Furthermore, it is very useful as a heat-exchange liquid in fast breeder nuclear reactor
A breeder reactor is a nuclear reactor that generates more fissile material than it consumes. Breeder reactors achieve this because their neutron economy is high enough to create more fissile fuel than they use, by irradiation of a fertile mate ...
s due to its low melting point, viscosity, and cross-section towards neutron absorption.fertiliser
A fertilizer (American English) or fertiliser (British English; see spelling differences) is any material of natural or synthetic origin that is applied to soil or to plant tissues to supply plant nutrients. Fertilizers may be distinct from ...
sPotassium hydroxide
Potassium hydroxide is an inorganic compound with the formula K OH, and is commonly called caustic potash.
Along with sodium hydroxide (NaOH), KOH is a prototypical strong base. It has many industrial and niche applications, most of which exp ...
is a very strong base, and is used to control the pH of various substances. Potassium nitrate
Potassium nitrate is a chemical compound with the chemical formula . This alkali metal nitrate salt is also known as Indian saltpetre (large deposits of which were historically mined in India). It is an ionic salt of potassium ions K+ and nitrat ...
and potassium permanganate
Potassium permanganate is an inorganic compound with the chemical formula KMnO4. It is a purplish-black crystalline salt, that dissolves in water as K+ and , an intensely pink to purple solution.
Potassium permanganate is widely used in the c ...
are often used as powerful oxidising agents.Potassium superoxide
Potassium superoxide is an inorganic compound with the formula KO2. It is a yellow paramagnetic solid that decomposes in moist air. It is a rare example of a stable salt of the superoxide anion. It is used as a scrubber, dehumidifier, and gen ...
is used in breathing masks, as it reacts with carbon dioxide to give potassium carbonate and oxygen gas. Pure potassium metal is not often used, but its alloys with sodium may substitute for pure sodium in fast breeder nuclear reactors.atomic clock
An atomic clock is a clock that measures time by monitoring the resonant frequency of atoms. It is based on atoms having different energy levels. Electron states in an atom are associated with different energy levels, and in transitions betwee ...
s.firework
Fireworks are a class of low explosive pyrotechnic devices used for aesthetic and entertainment purposes. They are most commonly used in fireworks displays (also called a fireworks show or pyrotechnics), combining a large number of devices in ...
s, and caesium is often used in drilling fluids in the petroleum industry.atomic structure
Every atom is composed of a nucleus and one or more electrons bound to the nucleus. The nucleus is made of one or more protons and a number of neutrons. Only the most common variety of hydrogen has no neutrons.
Every solid, liquid, gas, an ...
, among other things, it has been used in spectroscopy
Spectroscopy is the field of study that measures and interprets the electromagnetic spectra that result from the interaction between electromagnetic radiation and matter as a function of the wavelength or frequency of the radiation. Matter wa ...
experiments, leading to more information regarding energy level
A quantum mechanical system or particle that is bound—that is, confined spatially—can only take on certain discrete values of energy, called energy levels. This contrasts with classical particles, which can have any amount of energy. The te ...
s and the coupling constants between subatomic particle
In physical sciences, a subatomic particle is a particle that composes an atom. According to the Standard Model of particle physics, a subatomic particle can be either a composite particle, which is composed of other particles (for example, a pr ...
s. Studies on the light emitted by laser-trapped francium-210 ions have provided accurate data on transitions between atomic energy levels, similar to those predicted by quantum theory
Quantum theory may refer to:
Science
*Quantum mechanics, a major field of physics
*Old quantum theory, predating modern quantum mechanics
* Quantum field theory, an area of quantum mechanics that includes:
** Quantum electrodynamics
** Quantum ...
.
Biological role and precautions
Metals
Pure alkali metals are dangerously reactive with air and water and must be kept away from heat, fire, oxidising agents, acids, most organic compounds, halocarbon
Halocarbon compounds are chemicals in which one or more carbon atoms are linked by covalent bonds with one or more halogen atoms (fluorine, chlorine, bromine or iodine – ) resulting in the formation of organofluorine compounds, organochlori ...
s, plastic
Plastics are a wide range of synthetic or semi-synthetic materials that use polymers as a main ingredient. Their plasticity makes it possible for plastics to be moulded, extruded or pressed into solid objects of various shapes. This adaptab ...
s, and moisture. They also react with carbon dioxide and carbon tetrachloride, so that normal fire extinguishers are counterproductive when used on alkali metal fires.fume hood
A fume hood (sometimes called a fume cupboard or fume closet) is a type of local ventilation device that is designed to limit exposure to hazardous or toxic fumes, vapors or dusts.
Description
A fume hood is typically a large piece of equipmen ...
. Small quantities of lithium may be disposed of by reaction with cool water, but the heavier alkali metals should be dissolved in the less reactive isopropanol
Isopropyl alcohol (IUPAC name propan-2-ol and also called isopropanol or 2-propanol) is a colorless, flammable organic compound with a pungent alcoholic odor. As an isopropyl group linked to a hydroxyl group (chemical formula ) it is the simple ...
.mineral oil
Mineral oil is any of various colorless, odorless, light mixtures of higher alkanes from a mineral source, particularly a distillate of petroleum, as distinct from usually edible vegetable oils.
The name 'mineral oil' by itself is imprecise, ...
or an inert atmosphere. The inert atmosphere used may be argon
Argon is a chemical element with the symbol Ar and atomic number 18. It is in group 18 of the periodic table and is a noble gas. Argon is the third-most abundant gas in Earth's atmosphere, at 0.934% (9340 ppmv). It is more than twice as abu ...
or nitrogen gas, except for lithium, which reacts with nitrogen.
Ions
 The bioinorganic chemistry of the alkali metal ions has been extensively reviewed.
Solid state crystal structures have been determined for many complexes of alkali metal ions in small peptides, nucleic acid constituents, carbohydrates and ionophore complexes.
Lithium naturally only occurs in traces in biological systems and has no known biological role, but does have effects on the body when ingested.
The bioinorganic chemistry of the alkali metal ions has been extensively reviewed.
Solid state crystal structures have been determined for many complexes of alkali metal ions in small peptides, nucleic acid constituents, carbohydrates and ionophore complexes.
Lithium naturally only occurs in traces in biological systems and has no known biological role, but does have effects on the body when ingested.Lithium carbonate
Lithium carbonate is an inorganic compound, the lithium salt (chemistry), salt of carbonate with the chemical formula, formula . This white Salt (chemistry), salt is widely used in the processing of metal oxides. It is listed on the World Health O ...
is used as a mood stabiliser
A mood stabilizer is a psychiatric medication used to treat mood disorders characterized by intense and sustained mood shifts, such as bipolar disorder and the bipolar type of schizoaffective disorder.
Uses
Mood stabilizers are best known for th ...
in psychiatry
Psychiatry is the medical specialty devoted to the diagnosis, prevention, and treatment of mental disorders. These include various maladaptations related to mood, behaviour, cognition, and perceptions. See glossary of psychiatry.
Initial psych ...
to treat bipolar disorder
Bipolar disorder, previously known as manic depression, is a mental disorder characterized by periods of depression and periods of abnormally elevated mood that last from days to weeks each. If the elevated mood is severe or associated with ...
( manic-depression) in daily doses of about 0.5 to 2 grams, although there are side-effects.poison
Poison is a chemical substance that has a detrimental effect to life. The term is used in a wide range of scientific fields and industries, where it is often specifically defined. It may also be applied colloquially or figuratively, with a broa ...
s the central nervous system
The central nervous system (CNS) is the part of the nervous system consisting primarily of the brain and spinal cord. The CNS is so named because the brain integrates the received information and coordinates and influences the activity of all par ...
,trace element
__NOTOC__
A trace element, also called minor element, is a chemical element whose concentration (or other measure of amount) is very low (a "trace amount"). They are classified into two groups: essential and non-essential. Essential trace elements ...
, although the natural biological function of lithium in humans has yet to be identified.
Sodium and potassium occur in all known biological systems, generally functioning as electrolytes
An electrolyte is a medium containing ions that is electrically conducting through the movement of those ions, but not conducting electrons. This includes most soluble salts, acids, and bases dissolved in a polar solvent, such as water. Upon di ...
inside and outside cells.Sodium chloride
Sodium chloride , commonly known as salt (although sea salt also contains other chemical salts), is an ionic compound with the chemical formula NaCl, representing a 1:1 ratio of sodium and chloride ions. With molar masses of 22.99 and 35.45 g ...
(also known as common salt) is the principal source of sodium in the diet, and is used as seasoning and preservative, such as for pickling
Pickling is the process of preserving or extending the shelf life of food by either anaerobic fermentation in brine or immersion in vinegar. The pickling procedure typically affects the food's texture and flavor. The resulting food is called a ...
and jerky
Jerky is lean trimmed meat cut into strips and dried (dehydrated) to prevent spoilage. Normally, this drying includes the addition of salt to prevent bacteria growth before the meat has finished the dehydrating process. The word "jerky" derive ...
; most of it comes from processed foods. The Dietary Reference Intake
The Dietary Reference Intake (DRI) is a system of nutrition recommendations from the National Academy of Medicine (NAM) of the National Academies (United States). It was introduced in 1997 in order to broaden the existing guidelines known as Reco ...
for sodium is 1.5 grams per day, but most people in the United States consume more than 2.3 grams per day, the minimum amount that promotes hypertension; this in turn causes 7.6 million premature deaths worldwide.
Potassium is the major cation
An ion () is an atom or molecule with a net electrical charge.
The charge of an electron is considered to be negative by convention and this charge is equal and opposite to the charge of a proton, which is considered to be positive by convent ...
(positive ion) inside animal cells
The cell is the basic structural and functional unit of life forms. Every cell consists of a cytoplasm enclosed within a membrane, and contains many biomolecules such as proteins, DNA and RNA, as well as many small molecules of nutrients and ...
,concentration
In chemistry, concentration is the abundance of a constituent divided by the total volume of a mixture. Several types of mathematical description can be distinguished: '' mass concentration'', ''molar concentration'', ''number concentration'', an ...
differences of these charged particles causes a difference in electric potential
The electric potential (also called the ''electric field potential'', potential drop, the electrostatic potential) is defined as the amount of work energy needed to move a unit of electric charge from a reference point to the specific point in ...
between the inside and outside of cells, known as the membrane potential
Membrane potential (also transmembrane potential or membrane voltage) is the difference in electric potential between the interior and the exterior of a biological cell. That is, there is a difference in the energy required for electric charges ...
. The balance between potassium and sodium is maintained by ion transporter
In biology, a transporter is a transmembrane protein that moves ions (or other small molecules) across a biological membrane to accomplish many different biological functions including, cellular communication, maintaining homeostasis, energy produc ...
proteins in the cell membrane
The cell membrane (also known as the plasma membrane (PM) or cytoplasmic membrane, and historically referred to as the plasmalemma) is a biological membrane that separates and protects the interior of all cells from the outside environment ( ...
.action potential
An action potential occurs when the membrane potential of a specific cell location rapidly rises and falls. This depolarization then causes adjacent locations to similarly depolarize. Action potentials occur in several types of animal cells, ...
—a "spike" of electrical discharge. The ability of cells to produce electrical discharge is critical for body functions such as neurotransmission
Neurotransmission (Latin: ''transmissio'' "passage, crossing" from ''transmittere'' "send, let through") is the process by which signaling molecules called neurotransmitters are released by the axon terminal of a neuron (the presynaptic neuron), ...
, muscle contraction, and heart function.hyperkalemia
Hyperkalemia is an elevated level of potassium (K+) in the blood. Normal potassium levels are between 3.5 and 5.0mmol/L (3.5 and 5.0mEq/L) with levels above 5.5mmol/L defined as hyperkalemia. Typically hyperkalemia does not cause symptoms. Occasi ...
strongly influencing the cardiovascular system.United States
The United States of America (U.S.A. or USA), commonly known as the United States (U.S. or US) or America, is a country primarily located in North America. It consists of 50 states, a federal district, five major unincorporated territorie ...
for lethal injection
Lethal injection is the practice of injecting one or more drugs into a person (typically a barbiturate, paralytic, and potassium solution) for the express purpose of causing rapid death. The main application for this procedure is capital puni ...
executions. Due to their similar atomic radii, rubidium and caesium in the body mimic potassium and are taken up similarly. Rubidium has no known biological role, but may help stimulate
Due to their similar atomic radii, rubidium and caesium in the body mimic potassium and are taken up similarly. Rubidium has no known biological role, but may help stimulate metabolism
Metabolism (, from el, μεταβολή ''metabolē'', "change") is the set of life-sustaining chemical reactions in organisms. The three main functions of metabolism are: the conversion of the energy in food to energy available to run cell ...
,hypokalemia
Hypokalemia is a low level of potassium (K+) in the blood serum. Mild low potassium does not typically cause symptoms. Symptoms may include feeling tired, leg cramps, weakness, and constipation. Low potassium also increases the risk of an abno ...
, arrythmia
Arrhythmias, also known as cardiac arrhythmias, heart arrhythmias, or dysrhythmias, are irregularities in the heartbeat, including when it is too fast or too slow. A resting heart rate that is too fast – above 100 beats per minute in adults ...
, and acute cardiac arrest
Cardiac arrest is when the heart suddenly and unexpectedly stops beating. It is a medical emergency that, without immediate medical intervention, will result in sudden cardiac death within minutes. Cardiopulmonary resuscitation (CPR) and possib ...
, but such amounts would not ordinarily be encountered in natural sources.[ The ]median lethal dose
In toxicology, the median lethal dose, LD50 (abbreviation for "lethal dose, 50%"), LC50 (lethal concentration, 50%) or LCt50 is a toxic unit that measures the lethal dose of a toxin, radiation, or pathogen. The value of LD50 for a substance is the ...
(LD50) value for caesium chloride
Caesium chloride or cesium chloride is the inorganic compound with the formula Cs Cl. This colorless salt is an important source of caesium ions in a variety of niche applications. Its crystal structure forms a major structural type where each ...
in mice is 2.3 g per kilogram, which is comparable to the LD50 values of potassium chloride
Potassium chloride (KCl, or potassium salt) is a metal halide salt composed of potassium and chlorine. It is odorless and has a white or colorless vitreous crystal appearance. The solid dissolves readily in water, and its solutions have a salt ...
and sodium chloride
Sodium chloride , commonly known as salt (although sea salt also contains other chemical salts), is an ionic compound with the chemical formula NaCl, representing a 1:1 ratio of sodium and chloride ions. With molar masses of 22.99 and 35.45 g ...
. Caesium chloride has been promoted as an alternative cancer therapy, but has been linked to the deaths of over 50 patients, on whom it was used as part of a scientifically unvalidated cancer treatment.[Wood, Leonie. ]
Radioisotope
A radionuclide (radioactive nuclide, radioisotope or radioactive isotope) is a nuclide that has excess nuclear energy, making it unstable. This excess energy can be used in one of three ways: emitted from the nucleus as gamma radiation; transferr ...
s of caesium require special precautions: the improper handling of caesium-137 gamma ray
A gamma ray, also known as gamma radiation (symbol γ or \gamma), is a penetrating form of electromagnetic radiation arising from the radioactive decay of atomic nuclei. It consists of the shortest wavelength electromagnetic waves, typically ...
sources can lead to release of this radioisotope and radiation injuries. Perhaps the best-known case is the Goiânia accident of 1987, in which an improperly-disposed-of radiation therapy system from an abandoned clinic in the city of Goiânia
Goiânia (; ) is the capital and largest city of the Brazilian state of Goiás. With a population of 1,536,097, it is the second-largest city in the Central-West Region and the 10th-largest in the country. Its metropolitan area has a population ...
, Brazil
Brazil ( pt, Brasil; ), officially the Federative Republic of Brazil (Portuguese: ), is the largest country in both South America and Latin America. At and with over 217 million people, Brazil is the world's fifth-largest country by area ...
, was scavenged from a junkyard, and the glowing caesium salt sold to curious, uneducated buyers. This led to four deaths and serious injuries from radiation exposure. Together with caesium-134
Caesium (55Cs) has 40 known isotopes, making it, along with barium and mercury, one of the elements with the most isotopes. The atomic masses of these isotopes range from 112 to 151. Only one isotope, 133Cs, is stable. The longest-lived radioisot ...
, iodine-131
Iodine-131 (131I, I-131) is an important radioisotope of iodine discovered by Glenn Seaborg and John Livingood in 1938 at the University of California, Berkeley. It has a radioactive decay half-life of about eight days. It is associated with nuc ...
, and strontium-90
Strontium-90 () is a radioactive isotope of strontium produced by nuclear fission, with a half-life of 28.8 years. It undergoes β− decay into yttrium-90, with a decay energy of 0.546 MeV. Strontium-90 has applications in medicine and i ...
, caesium-137 was among the isotopes distributed by the Chernobyl disaster
The Chernobyl disaster was a nuclear accident that occurred on 26 April 1986 at the No. 4 reactor in the Chernobyl Nuclear Power Plant, near the city of Pripyat in the north of the Ukrainian SSR in the Soviet Union. It is one of only two nuc ...
which constitute the greatest risk to health.
Notes
References
{{Authority control
A
Groups (periodic table)
Periodic table
Articles containing video clips
 Sodium compounds have been known since ancient times; salt (
Sodium compounds have been known since ancient times; salt (
 Rubidium and caesium were the first elements to be discovered using the
Rubidium and caesium were the first elements to be discovered using the  After 1869,
After 1869,  The
The  The
The  Electronegativity is a
Electronegativity is a  All the alkali metals react vigorously or explosively with cold water, producing an
All the alkali metals react vigorously or explosively with cold water, producing an  The alkali metals form many
The alkali metals form many  Lithium, the lightest of the alkali metals, is the only alkali metal which reacts with
Lithium, the lightest of the alkali metals, is the only alkali metal which reacts with  Being the smallest alkali metal, lithium forms the widest variety of and most stable
Being the smallest alkali metal, lithium forms the widest variety of and most stable  Reaction 1) is known as
Reaction 1) is known as  The
The 
 For several years in the 1950s and 1960s, a by-product of the potassium production called Alkarb was a main source for rubidium. Alkarb contained 21% rubidium while the rest was potassium and a small fraction of caesium. Today the largest producers of caesium, for example the
For several years in the 1950s and 1960s, a by-product of the potassium production called Alkarb was a main source for rubidium. Alkarb contained 21% rubidium while the rest was potassium and a small fraction of caesium. Today the largest producers of caesium, for example the  Lithium, sodium, and potassium have many applications, while rubidium and caesium are very useful in academic contexts but do not have many applications yet. Lithium is often used in
Lithium, sodium, and potassium have many applications, while rubidium and caesium are very useful in academic contexts but do not have many applications yet. Lithium is often used in  The bioinorganic chemistry of the alkali metal ions has been extensively reviewed.
Solid state crystal structures have been determined for many complexes of alkali metal ions in small peptides, nucleic acid constituents, carbohydrates and ionophore complexes.
Lithium naturally only occurs in traces in biological systems and has no known biological role, but does have effects on the body when ingested.
The bioinorganic chemistry of the alkali metal ions has been extensively reviewed.
Solid state crystal structures have been determined for many complexes of alkali metal ions in small peptides, nucleic acid constituents, carbohydrates and ionophore complexes.
Lithium naturally only occurs in traces in biological systems and has no known biological role, but does have effects on the body when ingested.  Due to their similar atomic radii, rubidium and caesium in the body mimic potassium and are taken up similarly. Rubidium has no known biological role, but may help stimulate
Due to their similar atomic radii, rubidium and caesium in the body mimic potassium and are taken up similarly. Rubidium has no known biological role, but may help stimulate