Uranium on:
[Wikipedia]
[Google]
[Amazon]
Uranium is a
 When refined, uranium is a silvery white, weakly radioactive
When refined, uranium is a silvery white, weakly radioactive
 The major application of uranium in the military sector is in high-density penetrators. This ammunition consists of depleted uranium (DU) alloyed with 1–2% other elements, such as
The major application of uranium in the military sector is in high-density penetrators. This ammunition consists of depleted uranium (DU) alloyed with 1–2% other elements, such as
 The main use of uranium in the civilian sector is to fuel nuclear power plants. One kilogram of uranium-235 can theoretically produce about 20 terajoules of energy (2 joules), assuming complete fission; as much energy as 1.5 million kilograms (1,500
The main use of uranium in the civilian sector is to fuel nuclear power plants. One kilogram of uranium-235 can theoretically produce about 20 terajoules of energy (2 joules), assuming complete fission; as much energy as 1.5 million kilograms (1,500  Before (and, occasionally, after) the discovery of radioactivity, uranium was primarily used in small amounts for yellow glass and pottery glazes, such as
Before (and, occasionally, after) the discovery of radioactivity, uranium was primarily used in small amounts for yellow glass and pottery glazes, such as 
 Uranium was also used in photographic chemicals (especially uranium nitrate as a toner), in lamp filaments for stage lighting bulbs, to improve the appearance of
Uranium was also used in photographic chemicals (especially uranium nitrate as a toner), in lamp filaments for stage lighting bulbs, to improve the appearance of
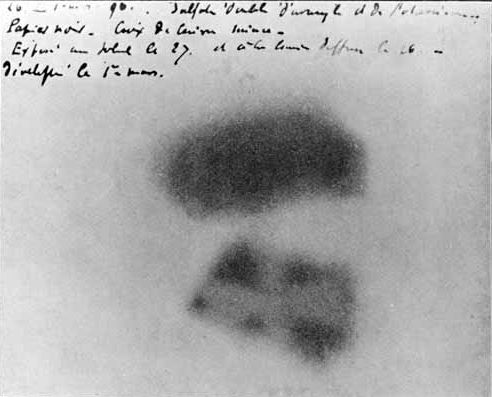 The discovery of the element is credited to the German chemist
The discovery of the element is credited to the German chemist
 A team led by Enrico Fermi in 1934 observed that bombarding uranium with neutrons produces the emission of beta rays (
A team led by Enrico Fermi in 1934 observed that bombarding uranium with neutrons produces the emission of beta rays (
 Two major types of atomic bombs were developed by the United States during
Two major types of atomic bombs were developed by the United States during
 The X-10 Graphite Reactor at Oak Ridge National Laboratory (ORNL) in Oak Ridge, Tennessee, formerly known as the Clinton Pile and X-10 Pile, was the world's second artificial nuclear reactor (after Enrico Fermi's Chicago Pile) and was the first reactor designed and built for continuous operation. Argonne National Laboratory's Experimental Breeder Reactor I, located at the Atomic Energy Commission's National Reactor Testing Station near
The X-10 Graphite Reactor at Oak Ridge National Laboratory (ORNL) in Oak Ridge, Tennessee, formerly known as the Clinton Pile and X-10 Pile, was the world's second artificial nuclear reactor (after Enrico Fermi's Chicago Pile) and was the first reactor designed and built for continuous operation. Argonne National Laboratory's Experimental Breeder Reactor I, located at the Atomic Energy Commission's National Reactor Testing Station near
 Above-ground
Above-ground

 Uranium is a
Uranium is a  Some bacteria, such as ''
Some bacteria, such as ''
 Worldwide production of U3O8 (yellowcake) in 2013 amounted to 70,015
Worldwide production of U3O8 (yellowcake) in 2013 amounted to 70,015
File:U production-demand.png, World uranium production (mines) and demand
File:Yellowcake.jpg, alt=A yellow sand-like rhombic mass on black background.,
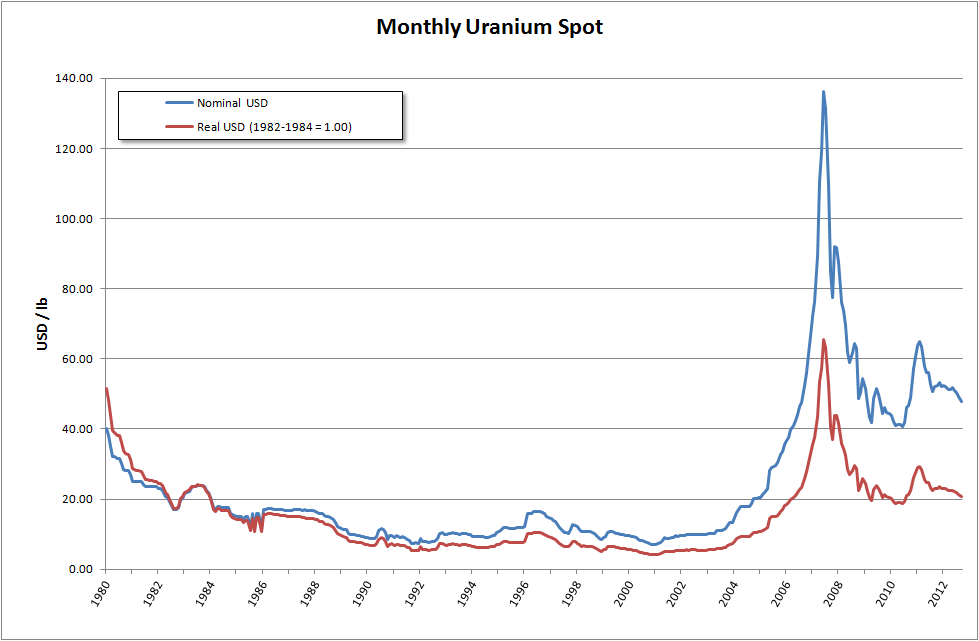
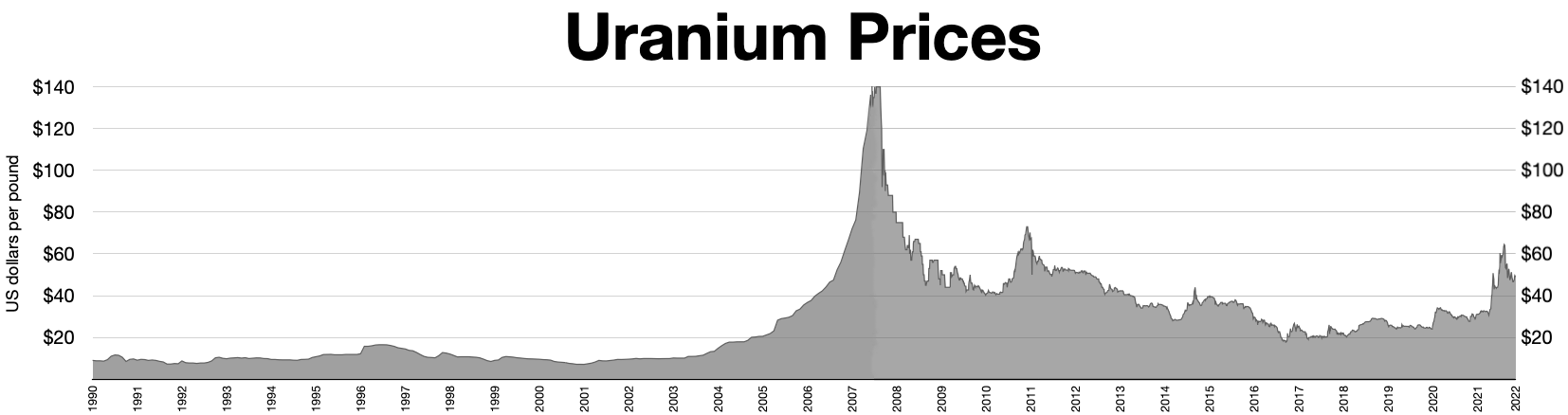 It is estimated that 5.5 million tonnes of uranium exists in ore reserves that are economically viable at US$59 per lb of uranium, while 35 million tonnes are classed as mineral resources (reasonable prospects for eventual economic extraction). Prices went from about $10/lb in May 2003 to $138/lb in July 2007. This has caused a big increase in spending on exploration, with US$200 million being spent worldwide in 2005, a 54% increase on the previous year. This trend continued through 2006, when expenditure on exploration rocketed to over $774 million, an increase of over 250% compared to 2004. The
It is estimated that 5.5 million tonnes of uranium exists in ore reserves that are economically viable at US$59 per lb of uranium, while 35 million tonnes are classed as mineral resources (reasonable prospects for eventual economic extraction). Prices went from about $10/lb in May 2003 to $138/lb in July 2007. This has caused a big increase in spending on exploration, with US$200 million being spent worldwide in 2005, a 54% increase on the previous year. This trend continued through 2006, when expenditure on exploration rocketed to over $774 million, an increase of over 250% compared to 2004. The
 In 2005, seventeen countries produced concentrated uranium oxides:
In 2005, seventeen countries produced concentrated uranium oxides:

 Salts of many oxidation states of uranium are water-
Salts of many oxidation states of uranium are water-
 All uranium fluorides are created using
All uranium fluorides are created using
 In nature, uranium is found as uranium-238 (99.2742%) and uranium-235 (0.7204%).
In nature, uranium is found as uranium-238 (99.2742%) and uranium-235 (0.7204%).
U.S. EPA: Radiation Information for Uranium
from
Nuclear fuel data and analysis
from the U.S. Energy Information Administration
Current market price of uranium
Annotated bibliography for uranium from the Alsos Digital Library
NLM Hazardous Substances Databank—Uranium, Radioactive
Mining Uranium at Namibia's Langer Heinrich Mine
World Nuclear News
ATSDR Case Studies in Environmental Medicine: Uranium Toxicity
U.S.
Uranium
at ''
chemical element
A chemical element is a species of atoms that have a given number of protons in their nuclei, including the pure substance consisting only of that species. Unlike chemical compounds, chemical elements cannot be broken down into simpler sub ...
with the symbol U and atomic number 92. It is a silvery-grey metal
A metal (from Greek μέταλλον ''métallon'', "mine, quarry, metal") is a material that, when freshly prepared, polished, or fractured, shows a lustrous appearance, and conducts electricity and heat relatively well. Metals are typicall ...
in the actinide
The actinide () or actinoid () series encompasses the 15 metallic chemical elements with atomic numbers from 89 to 103, actinium through lawrencium. The actinide series derives its name from the first element in the series, actinium. The info ...
series of the periodic table. A uranium atom has 92 protons and 92 electron
The electron ( or ) is a subatomic particle with a negative one elementary electric charge. Electrons belong to the first generation of the lepton particle family,
and are generally thought to be elementary particles because they have no ...
s, of which 6 are valence electron
In chemistry and physics, a valence electron is an electron in the outer shell associated with an atom, and that can participate in the formation of a chemical bond if the outer shell is not closed. In a single covalent bond, a shared pair form ...
s. Uranium is weakly radioactive because all isotopes of uranium
Uranium (92U) is a naturally occurring radioactive element that has no stable isotope. It has two primordial isotopes, uranium-238 and uranium-235, that have long half-lives and are found in appreciable quantity in the Earth's crust. The d ...
are unstable; the half-lives of its naturally occurring isotopes range between 159,200 years and 4.5 billion years. The most common isotopes in natural uranium are uranium-238 (which has 146 neutron
The neutron is a subatomic particle, symbol or , which has a neutral (not positive or negative) charge, and a mass slightly greater than that of a proton. Protons and neutrons constitute the nuclei of atoms. Since protons and neutrons beh ...
s and accounts for over 99% of uranium on Earth) and uranium-235 (which has 143 neutrons). Uranium has the highest atomic weight
Relative atomic mass (symbol: ''A''; sometimes abbreviated RAM or r.a.m.), also known by the deprecated synonym atomic weight, is a dimensionless physical quantity defined as the ratio of the average mass of atoms of a chemical element in a giv ...
of the primordially occurring elements. Its density
Density (volumetric mass density or specific mass) is the substance's mass per unit of volume. The symbol most often used for density is ''ρ'' (the lower case Greek letter rho), although the Latin letter ''D'' can also be used. Mathematical ...
is about 70% higher than that of lead
Lead is a chemical element with the symbol Pb (from the Latin ) and atomic number 82. It is a heavy metal that is denser than most common materials. Lead is soft and malleable, and also has a relatively low melting point. When freshly cu ...
, and slightly lower than that of gold or tungsten
Tungsten, or wolfram, is a chemical element with the symbol W and atomic number 74. Tungsten is a rare metal found naturally on Earth almost exclusively as compounds with other elements. It was identified as a new element in 1781 and first isol ...
. It occurs naturally in low concentrations of a few parts per million
In science and engineering, the parts-per notation is a set of pseudo-units to describe small values of miscellaneous dimensionless quantities, e.g. mole fraction or mass fraction. Since these fractions are quantity-per-quantity measures, th ...
in soil, rock and water, and is commercially extracted from uranium-bearing mineral
In geology and mineralogy, a mineral or mineral species is, broadly speaking, a solid chemical compound with a fairly well-defined chemical composition and a specific crystal structure that occurs naturally in pure form.John P. Rafferty, ed. (2 ...
s such as uraninite.
In nature, uranium is found as uranium-238 (99.2739–99.2752%), uranium-235 (0.7198–0.7202%), and a very small amount of uranium-234 (0.0050–0.0059%). Uranium decays slowly by emitting an alpha particle. The half-life of uranium-238 is about 4.47 billion
Billion is a word for a large number, and it has two distinct definitions:
*1,000,000,000, i.e. one thousand million, or (ten to the ninth power), as defined on the short scale. This is its only current meaning in English.
* 1,000,000,000,000, i. ...
years and that of uranium-235 is 704 million years, making them useful in dating the age of the Earth.
Many contemporary uses of uranium exploit its unique nuclear properties. Uranium-235 is the only naturally occurring fissile
In nuclear engineering, fissile material is material capable of sustaining a nuclear fission chain reaction. By definition, fissile material can sustain a chain reaction with neutrons of thermal energy. The predominant neutron energy may be t ...
isotope
Isotopes are two or more types of atoms that have the same atomic number (number of protons in their nuclei) and position in the periodic table (and hence belong to the same chemical element), and that differ in nucleon numbers (mass numb ...
, which makes it widely used in nuclear power plants and nuclear weapon
A nuclear weapon is an explosive device that derives its destructive force from nuclear reactions, either fission (fission bomb) or a combination of fission and fusion reactions ( thermonuclear bomb), producing a nuclear explosion. Both bom ...
s. However, because of the tiny amounts found in nature, uranium needs to undergo enrichment so that enough uranium-235 is present. Uranium-238 is fissionable by fast neutrons, and is fertile, meaning it can be transmuted to fissile plutonium-239
Plutonium-239 (239Pu or Pu-239) is an isotope of plutonium. Plutonium-239 is the primary fissile isotope used for the production of nuclear weapons, although uranium-235 is also used for that purpose. Plutonium-239 is also one of the three mai ...
in a nuclear reactor
A nuclear reactor is a device used to initiate and control a fission nuclear chain reaction or nuclear fusion reactions. Nuclear reactors are used at nuclear power plants for electricity generation and in nuclear marine propulsion. Heat fr ...
. Another fissile isotope, uranium-233
Uranium-233 (233U or U-233) is a fissile isotope of uranium that is bred from thorium-232 as part of the thorium fuel cycle. Uranium-233 was investigated for use in nuclear weapons and as a reactor fuel. It has been used successfully in exp ...
, can be produced from natural thorium and is studied for future industrial use in nuclear technology. Uranium-238 has a small probability for spontaneous fission or even induced fission with fast neutrons; uranium-235 and to a lesser degree uranium-233 have a much higher fission cross-section for slow neutrons. In sufficient concentration, these isotopes maintain a sustained nuclear chain reaction. This generates the heat in nuclear power reactors, and produces the fissile material for nuclear weapons. Depleted uranium (238U) is used in kinetic energy penetrators and armor plating
Military vehicles are commonly armoured (or armored; see spelling differences) to withstand the impact of shrapnel, bullets, shells, rockets, and missiles, protecting the personnel inside from enemy fire. Such vehicles include armoured fighti ...
.. Uranium is used as a colorant in uranium glass
Uranium glass is glass which has had uranium, usually in oxide diuranate form, added to a glass mix before melting for colouration. The proportion usually varies from trace levels to about 2% uranium by weight, although some 20th-century piec ...
, producing lemon yellow to green colors. Uranium glass fluoresces green in ultraviolet light. It was also used for tinting and shading in early photography
Photography is the art, application, and practice of creating durable images by recording light, either electronically by means of an image sensor, or chemically by means of a light-sensitive material such as photographic film. It is employe ...
.
The 1789 discovery of uranium in the mineral pitchblende
Uraninite, formerly pitchblende, is a radioactive, uranium-rich mineral and ore with a chemical composition that is largely UO2 but because of oxidation typically contains variable proportions of U3O8. Radioactive decay of the uranium causes t ...
is credited to Martin Heinrich Klaproth
Martin Heinrich Klaproth (1 December 1743 – 1 January 1817) was a German chemist. He trained and worked for much of his life as an apothecary, moving in later life to the university. His shop became the second-largest apothecary in Berlin, and ...
, who named the new element after the recently discovered planet Uranus
Uranus is the seventh planet from the Sun. Its name is a reference to the Greek god of the sky, Uranus ( Caelus), who, according to Greek mythology, was the great-grandfather of Ares (Mars), grandfather of Zeus (Jupiter) and father of ...
. Eugène-Melchior Péligot
Eugène-Melchior Péligot (24 March 1811 – 15 April 1890), also known as Eugène Péligot, was a French chemist who isolated the first sample of uranium metal in 1841.
Péligot proved that the black powder of Martin Heinrich Klaproth was not a ...
was the first person to isolate the metal and its radioactive properties were discovered in 1896 by Henri Becquerel
Antoine Henri Becquerel (; 15 December 1852 – 25 August 1908) was a French engineer, physicist, Nobel laureate, and the first person to discover evidence of radioactivity. For work in this field he, along with Marie Skłodowska-Curie and Pie ...
. Research by Otto Hahn
Otto Hahn (; 8 March 1879 – 28 July 1968) was a German chemist who was a pioneer in the fields of radioactivity and radiochemistry. He is referred to as the father of nuclear chemistry and father of nuclear fission. Hahn and Lise Meitner ...
, Lise Meitner, Enrico Fermi and others, such as J. Robert Oppenheimer
J. Robert Oppenheimer (; April 22, 1904 – February 18, 1967) was an American theoretical physicist. A professor of physics at the University of California, Berkeley, Oppenheimer was the wartime head of the Los Alamos Laboratory and is oft ...
starting in 1934 led to its use as a fuel in the nuclear power
Nuclear power is the use of nuclear reactions to produce electricity. Nuclear power can be obtained from nuclear fission, nuclear decay and nuclear fusion reactions. Presently, the vast majority of electricity from nuclear power is produced ...
industry and in ''Little Boy
"Little Boy" was the type of atomic bomb dropped on the Japanese city of Hiroshima on 6 August 1945 during World War II, making it the first nuclear weapon used in warfare. The bomb was dropped by the Boeing B-29 Superfortress ''Enola Gay'' p ...
'', the first nuclear weapon used in war. An ensuing arms race during the Cold War between the United States
The United States of America (U.S.A. or USA), commonly known as the United States (U.S. or US) or America, is a country primarily located in North America. It consists of 50 states, a federal district, five major unincorporated territori ...
and the Soviet Union
The Soviet Union,. officially the Union of Soviet Socialist Republics. (USSR),. was a List of former transcontinental countries#Since 1700, transcontinental country that spanned much of Eurasia from 1922 to 1991. A flagship communist state, ...
produced tens of thousands of nuclear weapons that used uranium metal and uranium-derived plutonium-239
Plutonium-239 (239Pu or Pu-239) is an isotope of plutonium. Plutonium-239 is the primary fissile isotope used for the production of nuclear weapons, although uranium-235 is also used for that purpose. Plutonium-239 is also one of the three mai ...
. The security of those weapons is closely monitored. Since around 2000, plutonium obtained by dismantling Cold War-era bombs is used as fuel for nuclear reactors. The development and deployment of these nuclear reactor
A nuclear reactor is a device used to initiate and control a fission nuclear chain reaction or nuclear fusion reactions. Nuclear reactors are used at nuclear power plants for electricity generation and in nuclear marine propulsion. Heat fr ...
s continue on a global base as they are powerful sources of CO2-free energy.
Characteristics
metal
A metal (from Greek μέταλλον ''métallon'', "mine, quarry, metal") is a material that, when freshly prepared, polished, or fractured, shows a lustrous appearance, and conducts electricity and heat relatively well. Metals are typicall ...
. It has a Mohs hardness of 6, sufficient to scratch glass and approximately equal to that of titanium
Titanium is a chemical element with the Symbol (chemistry), symbol Ti and atomic number 22. Found in nature only as an oxide, it can be reduced to produce a lustrous transition metal with a silver color, low density, and high strength, resista ...
, rhodium, manganese
Manganese is a chemical element with the symbol Mn and atomic number 25. It is a hard, brittle, silvery metal, often found in minerals in combination with iron. Manganese is a transition metal with a multifaceted array of industrial alloy use ...
and niobium. It is malleable
Ductility is a mechanical property commonly described as a material's amenability to drawing (e.g. into wire). In materials science, ductility is defined by the degree to which a material can sustain plastic deformation under tensile stres ...
, ductile
Ductility is a mechanical property commonly described as a material's amenability to drawing (e.g. into wire). In materials science, ductility is defined by the degree to which a material can sustain plastic deformation under tensile stres ...
, slightly paramagnetic
Paramagnetism is a form of magnetism whereby some materials are weakly attracted by an externally applied magnetic field, and form internal, induced magnetic fields in the direction of the applied magnetic field. In contrast with this behavior, ...
, strongly electropositive
Electronegativity, symbolized as , is the tendency for an atom of a given chemical element to attract shared electrons (or electron density) when forming a chemical bond. An atom's electronegativity is affected by both its atomic number and the ...
and a poor electrical conductor
In physics and electrical engineering, a conductor is an object or type of material that allows the flow of charge (electric current) in one or more directions. Materials made of metal are common electrical conductors. Electric current is gene ...
. Uranium metal has a very high density
Density (volumetric mass density or specific mass) is the substance's mass per unit of volume. The symbol most often used for density is ''ρ'' (the lower case Greek letter rho), although the Latin letter ''D'' can also be used. Mathematical ...
of 19.1 g/cm3, denser than lead
Lead is a chemical element with the symbol Pb (from the Latin ) and atomic number 82. It is a heavy metal that is denser than most common materials. Lead is soft and malleable, and also has a relatively low melting point. When freshly cu ...
(11.3 g/cm3), but slightly less dense than tungsten
Tungsten, or wolfram, is a chemical element with the symbol W and atomic number 74. Tungsten is a rare metal found naturally on Earth almost exclusively as compounds with other elements. It was identified as a new element in 1781 and first isol ...
and gold (19.3 g/cm3).
Uranium metal reacts with almost all non-metal elements (with the exception of the noble gas
The noble gases (historically also the inert gases; sometimes referred to as aerogens) make up a class of chemical elements with similar properties; under standard conditions, they are all odorless, colorless, monatomic gases with very low ch ...
es) and their compounds, with reactivity increasing with temperature. Hydrochloric
Hydrochloric acid, also known as muriatic acid, is an aqueous solution of hydrogen chloride. It is a colorless solution with a distinctive pungent smell. It is classified as a strong acid. It is a component of the gastric acid in the digestiv ...
and nitric acid
Nitric acid is the inorganic compound with the formula . It is a highly corrosive mineral acid. The compound is colorless, but older samples tend to be yellow cast due to decomposition into oxides of nitrogen. Most commercially available nitri ...
s dissolve uranium, but non-oxidizing acids other than hydrochloric acid attack the element very slowly. When finely divided, it can react with cold water; in air, uranium metal becomes coated with a dark layer of uranium oxide
Uranium oxide is an oxide of the element uranium.
The metal uranium forms several oxides:
* Uranium dioxide or uranium(IV) oxide (UO2, the mineral uraninite or pitchblende)
* Diuranium pentoxide or uranium(V) oxide (U2O5)
* Uranium trioxide o ...
. Uranium in ores is extracted chemically and converted into uranium dioxide or other chemical forms usable in industry.
Uranium-235 was the first isotope that was found to be fissile
In nuclear engineering, fissile material is material capable of sustaining a nuclear fission chain reaction. By definition, fissile material can sustain a chain reaction with neutrons of thermal energy. The predominant neutron energy may be t ...
. Other naturally occurring isotopes are fissionable, but not fissile. On bombardment with slow neutrons, its uranium-235 isotope
Isotopes are two or more types of atoms that have the same atomic number (number of protons in their nuclei) and position in the periodic table (and hence belong to the same chemical element), and that differ in nucleon numbers (mass numb ...
will most of the time divide into two smaller nuclei, releasing nuclear binding energy and more neutrons. If too many of these neutrons are absorbed by other uranium-235 nuclei, a nuclear chain reaction occurs that results in a burst of heat or (in special circumstances) an explosion. In a nuclear reactor, such a chain reaction is slowed and controlled by a neutron poison
In applications such as nuclear reactors, a neutron poison (also called a neutron absorber or a nuclear poison) is a substance with a large neutron absorption cross-section. In such applications, absorbing neutrons is normally an undesirable eff ...
, absorbing some of the free neutrons. Such neutron absorbent materials are often part of reactor control rods (see nuclear reactor physics
Nuclear reactor physics is the field of physics that studies and deals with the applied study and engineering applications of chain reaction to induce a controlled rate of fission in a nuclear reactor for the production of energy.van Dam, H., ...
for a description of this process of reactor control).
As little as of uranium-235 can be used to make an atomic bomb. The nuclear weapon detonated over Hiroshima, called ''Little Boy
"Little Boy" was the type of atomic bomb dropped on the Japanese city of Hiroshima on 6 August 1945 during World War II, making it the first nuclear weapon used in warfare. The bomb was dropped by the Boeing B-29 Superfortress ''Enola Gay'' p ...
'', relied on uranium fission. However, the first nuclear bomb (the ''Gadget'' used at Trinity
The Christian doctrine of the Trinity (, from 'threefold') is the central dogma concerning the nature of God in most Christian churches, which defines one God existing in three coequal, coeternal, consubstantial divine persons: God th ...
) and the bomb that was detonated over Nagasaki ('' Fat Man'') were both plutonium bombs.
Uranium metal has three allotropic
Allotropy or allotropism () is the property of some chemical elements to exist in two or more different forms, in the same physical state, known as allotropes of the elements. Allotropes are different structural modifications of an element: th ...
forms:
* α ( orthorhombic) stable up to . Orthorhombic, space group
In mathematics, physics and chemistry, a space group is the symmetry group of an object in space, usually in three dimensions. The elements of a space group (its symmetry operations) are the rigid transformations of an object that leave it uncha ...
No. 63, ''Cmcm'', lattice parameter
A lattice constant or lattice parameter is one of the physical dimensions and angles that determine the geometry of the unit cells in a crystal lattice, and is proportional to the distance between atoms in the crystal. A simple cubic crystal has o ...
s ''a''= 285.4 pm, ''b'' = 587 pm, ''c'' = 495.5 pm.
* β (tetragonal
In crystallography, the tetragonal crystal system is one of the 7 crystal systems. Tetragonal crystal lattices result from stretching a cubic lattice along one of its lattice vectors, so that the cube becomes a rectangular prism with a squar ...
) stable from . Tetragonal, space group ''P''42/''mnm'', ''P''42''nm'', or ''P''4''n''2, lattice parameters ''a'' = 565.6 pm, ''b'' = ''c'' = 1075.9 pm.
* γ (body-centered cubic
In crystallography, the cubic (or isometric) crystal system is a crystal system where the unit cell is in the shape of a cube. This is one of the most common and simplest shapes found in crystals and minerals.
There are three main varieties of ...
) from to melting point—this is the most malleable and ductile state. Body-centered cubic, lattice parameter ''a'' = 352.4 pm.
Applications
Military
 The major application of uranium in the military sector is in high-density penetrators. This ammunition consists of depleted uranium (DU) alloyed with 1–2% other elements, such as
The major application of uranium in the military sector is in high-density penetrators. This ammunition consists of depleted uranium (DU) alloyed with 1–2% other elements, such as titanium
Titanium is a chemical element with the Symbol (chemistry), symbol Ti and atomic number 22. Found in nature only as an oxide, it can be reduced to produce a lustrous transition metal with a silver color, low density, and high strength, resista ...
or molybdenum. At high impact speed, the density, hardness, and pyrophoricity
A substance is pyrophoric (from grc-gre, πυροφόρος, , 'fire-bearing') if it ignites spontaneously in air at or below (for gases) or within 5 minutes after coming into contact with air (for liquids and solids). Examples are organolith ...
of the projectile enable the destruction of heavily armored targets. Tank armor and other removable vehicle armor
Military vehicles are commonly armoured (or armored; see spelling differences) to withstand the impact of shrapnel, bullets, shells, rockets, and missiles, protecting the personnel inside from enemy fire. Such vehicles include armoured fighti ...
can also be hardened with depleted uranium plates. The use of depleted uranium became politically and environmentally contentious after the use of such munitions by the US, UK and other countries during wars in the Persian Gulf and the Balkans raised questions concerning uranium compounds left in the soil (see Gulf War syndrome
Gulf War syndrome or Gulf War illness is a chronic and multi-symptomatic disorder affecting military veterans of both sides of the 1990–1991 Persian Gulf War. A wide range of acute and chronic symptoms have been linked to it, including fatigue ...
).
Depleted uranium is also used as a shielding material in some containers used to store and transport radioactive materials. While the metal itself is radioactive, its high density makes it more effective than lead
Lead is a chemical element with the symbol Pb (from the Latin ) and atomic number 82. It is a heavy metal that is denser than most common materials. Lead is soft and malleable, and also has a relatively low melting point. When freshly cu ...
in halting radiation from strong sources such as radium. Other uses of depleted uranium include counterweights for aircraft control surfaces, as ballast for missile re-entry vehicles and as a shielding material. Due to its high density, this material is found in inertial guidance systems and in gyroscopic compass
A compass is a device that shows the cardinal directions used for navigation and geographic orientation. It commonly consists of a magnetized needle or other element, such as a compass card or compass rose, which can pivot to align itself wit ...
es. Depleted uranium is preferred over similarly dense metals due to its ability to be easily machined and cast as well as its relatively low cost.. The main risk of exposure to depleted uranium is chemical poisoning by uranium oxide
Uranium oxide is an oxide of the element uranium.
The metal uranium forms several oxides:
* Uranium dioxide or uranium(IV) oxide (UO2, the mineral uraninite or pitchblende)
* Diuranium pentoxide or uranium(V) oxide (U2O5)
* Uranium trioxide o ...
rather than radioactivity (uranium being only a weak alpha emitter).
During the later stages of World War II
World War II or the Second World War, often abbreviated as WWII or WW2, was a world war that lasted from 1939 to 1945. It involved the vast majority of the world's countries—including all of the great powers—forming two opposing ...
, the entire Cold War, and to a lesser extent afterwards, uranium-235 has been used as the fissile explosive material to produce nuclear weapons. Initially, two major types of fission bombs were built: a relatively simple device that uses uranium-235 and a more complicated mechanism that uses plutonium-239
Plutonium-239 (239Pu or Pu-239) is an isotope of plutonium. Plutonium-239 is the primary fissile isotope used for the production of nuclear weapons, although uranium-235 is also used for that purpose. Plutonium-239 is also one of the three mai ...
derived from uranium-238. Later, a much more complicated and far more powerful type of fission/fusion bomb ( thermonuclear weapon) was built, that uses a plutonium-based device to cause a mixture of tritium
Tritium ( or , ) or hydrogen-3 (symbol T or H) is a rare and radioactive isotope of hydrogen with half-life about 12 years. The nucleus of tritium (t, sometimes called a ''triton'') contains one proton and two neutrons, whereas the nucleus of ...
and deuterium to undergo nuclear fusion
Nuclear fusion is a reaction in which two or more atomic nuclei are combined to form one or more different atomic nuclei and subatomic particles ( neutrons or protons). The difference in mass between the reactants and products is manife ...
. Such bombs are jacketed in a non-fissile (unenriched) uranium case, and they derive more than half their power from the fission of this material by fast neutron
The neutron detection temperature, also called the neutron energy, indicates a free neutron's kinetic energy, usually given in electron volts. The term ''temperature'' is used, since hot, thermal and cold neutrons are moderated in a medium with ...
s from the nuclear fusion process.
Civilian
 The main use of uranium in the civilian sector is to fuel nuclear power plants. One kilogram of uranium-235 can theoretically produce about 20 terajoules of energy (2 joules), assuming complete fission; as much energy as 1.5 million kilograms (1,500
The main use of uranium in the civilian sector is to fuel nuclear power plants. One kilogram of uranium-235 can theoretically produce about 20 terajoules of energy (2 joules), assuming complete fission; as much energy as 1.5 million kilograms (1,500 tonne
The tonne ( or ; symbol: t) is a unit of mass equal to 1000 kilograms. It is a non-SI unit accepted for use with SI. It is also referred to as a metric ton to distinguish it from the non-metric units of the short ton ( United State ...
s) of coal
Coal is a combustible black or brownish-black sedimentary rock, formed as rock strata called coal seams. Coal is mostly carbon with variable amounts of other elements, chiefly hydrogen, sulfur, oxygen, and nitrogen.
Coal is formed when ...
.
Commercial nuclear power
Nuclear power is the use of nuclear reactions to produce electricity. Nuclear power can be obtained from nuclear fission, nuclear decay and nuclear fusion reactions. Presently, the vast majority of electricity from nuclear power is produced ...
plants use fuel that is typically enriched to around 3% uranium-235. The CANDU and Magnox
Magnox is a type of nuclear power/production reactor that was designed to run on natural uranium with graphite as the moderator and carbon dioxide gas as the heat exchange coolant. It belongs to the wider class of gas-cooled reactors. The n ...
designs are the only commercial reactors capable of using unenriched uranium fuel. Fuel used for United States Navy
The United States Navy (USN) is the maritime service branch of the United States Armed Forces and one of the eight uniformed services of the United States. It is the largest and most powerful navy in the world, with the estimated tonnage ...
reactors is typically highly enriched in uranium-235 (the exact values are classified). In a breeder reactor
A breeder reactor is a nuclear reactor that generates more fissile material than it consumes. Breeder reactors achieve this because their neutron economy is high enough to create more fissile fuel than they use, by irradiation of a fertile mate ...
, uranium-238 can also be converted into plutonium
Plutonium is a radioactive chemical element with the symbol Pu and atomic number 94. It is an actinide metal of silvery-gray appearance that tarnishes when exposed to air, and forms a dull coating when oxidized. The element normally exhibi ...
through the following reaction:
: + n +
 Before (and, occasionally, after) the discovery of radioactivity, uranium was primarily used in small amounts for yellow glass and pottery glazes, such as
Before (and, occasionally, after) the discovery of radioactivity, uranium was primarily used in small amounts for yellow glass and pottery glazes, such as uranium glass
Uranium glass is glass which has had uranium, usually in oxide diuranate form, added to a glass mix before melting for colouration. The proportion usually varies from trace levels to about 2% uranium by weight, although some 20th-century piec ...
and in Fiestaware.
The discovery and isolation of radium in uranium ore (pitchblende) by Marie Curie sparked the development of uranium mining to extract the radium, which was used to make glow-in-the-dark paints for clock and aircraft dials. This left a prodigious quantity of uranium as a waste product, since it takes three tonnes of uranium to extract one gram
The gram (originally gramme; SI unit symbol g) is a unit of mass in the International System of Units (SI) equal to one one thousandth of a kilogram.
Originally defined as of 1795 as "the absolute weight of a volume of pure water equal to th ...
of radium. This waste product was diverted to the glazing industry, making uranium glazes very inexpensive and abundant. Besides the pottery glazes, uranium tile glazes accounted for the bulk of the use, including common bathroom and kitchen tiles which can be produced in green, yellow, mauve, black, blue, red and other colors.

 Uranium was also used in photographic chemicals (especially uranium nitrate as a toner), in lamp filaments for stage lighting bulbs, to improve the appearance of
Uranium was also used in photographic chemicals (especially uranium nitrate as a toner), in lamp filaments for stage lighting bulbs, to improve the appearance of dentures
Dentures (also known as false teeth) are prosthetic devices constructed to replace missing teeth, and are supported by the surrounding soft and hard tissues of the oral cavity. Conventional dentures are removable ( removable partial denture o ...
, and in the leather and wood industries for stains and dyes. Uranium salts are mordants of silk or wool. Uranyl acetate and uranyl formate are used as electron-dense "stains" in transmission electron microscopy, to increase the contrast of biological specimens in ultrathin sections and in negative staining of virus
A virus is a submicroscopic infectious agent that replicates only inside the living cells of an organism. Viruses infect all life forms, from animals and plants to microorganisms, including bacteria and archaea.
Since Dmitri Ivanovsk ...
es, isolated cell organelles and macromolecules.
The discovery of the radioactivity of uranium ushered in additional scientific and practical uses of the element. The long half-life of the isotope uranium-238 (4.47 years) makes it well-suited for use in estimating the age of the earliest igneous rock
Igneous rock (derived from the Latin word ''ignis'' meaning fire), or magmatic rock, is one of the three main rock types, the others being sedimentary and metamorphic. Igneous rock is formed through the cooling and solidification of magma o ...
s and for other types of radiometric dating, including uranium–thorium dating
Uranium–thorium dating, also called thorium-230 dating, uranium-series disequilibrium dating or uranium-series dating, is a radiometric dating technique established in the 1960s which has been used since the 1970s to determine the age of calciu ...
, uranium–lead dating
Uranium–lead dating, abbreviated U–Pb dating, is one of the oldest and most refined of the radiometric dating schemes. It can be used to date rocks that formed and crystallised from about 1 million years to over 4.5 billion years ago with routi ...
and uranium–uranium dating
Uranium–uranium dating is a radiometric dating technique which compares two isotopes of uranium (U) in a sample: uranium-234 (234U) and uranium-238 (238U). It is one of several radiometric dating techniques exploiting the uranium radioactive dec ...
. Uranium metal is used for X-ray
An X-ray, or, much less commonly, X-radiation, is a penetrating form of high-energy electromagnetic radiation. Most X-rays have a wavelength ranging from 10 picometers to 10 nanometers, corresponding to frequencies in the range 30&nb ...
targets in the making of high-energy X-rays.
History
Pre-discovery use
The use of uranium in its natural oxide form dates back to at least the year 79 CE, when it was used in theRoman Empire
The Roman Empire ( la, Imperium Romanum ; grc-gre, Βασιλεία τῶν Ῥωμαίων, Basileía tôn Rhōmaíōn) was the post- Republican period of ancient Rome. As a polity, it included large territorial holdings around the Mediter ...
to add a yellow color to ceramic
A ceramic is any of the various hard, brittle, heat-resistant and corrosion-resistant materials made by shaping and then firing an inorganic, nonmetallic material, such as clay, at a high temperature. Common examples are earthenware, porcelain ...
glazes. Yellow glass with 1% uranium oxide was found in a Roman villa on Cape Posillipo
Posillipo (; nap, Pusilleco ) is an affluent residential quarter of Naples, southern Italy, located along the northern coast of the Gulf of Naples.
From the 1st century BC the Bay of Naples witnessed the rise of villas constructed by elite Roma ...
in the Bay of Naples, Italy, by R. T. Gunther of the University of Oxford
, mottoeng = The Lord is my light
, established =
, endowment = £6.1 billion (including colleges) (2019)
, budget = £2.145 billion (2019–20)
, chancellor ...
in 1912.. Starting in the late Middle Ages
In the history of Europe, the Middle Ages or medieval period lasted approximately from the late 5th to the late 15th centuries, similar to the post-classical period of global history. It began with the fall of the Western Roman Empire ...
, pitchblende was extracted from the Habsburg silver mines in Joachimsthal, Bohemia (now Jáchymov in the Czech Republic), and was used as a coloring agent in the local glass
Glass is a non-crystalline, often transparent, amorphous solid that has widespread practical, technological, and decorative use in, for example, window panes, tableware, and optics. Glass is most often formed by rapid cooling ( quenching ...
making industry. In the early 19th century, the world's only known sources of uranium ore were these mines. Mining for uranium in the Ore Mountains ceased on the German side after the Cold War ended and SDAG Wismut
SAG/SDAG Wismut was a uranium mining company in East Germany
Germany,, officially the Federal Republic of Germany, is a country in Central Europe. It is the second most populous country in Europe after Russia, and the most populo ...
was wound down. On the Czech side there were attempts during the uranium price bubble of 2007 to restart mining, but those were quickly abandoned following a fall in uranium prices.
Discovery
 The discovery of the element is credited to the German chemist
The discovery of the element is credited to the German chemist Martin Heinrich Klaproth
Martin Heinrich Klaproth (1 December 1743 – 1 January 1817) was a German chemist. He trained and worked for much of his life as an apothecary, moving in later life to the university. His shop became the second-largest apothecary in Berlin, and ...
. While he was working in his experimental laboratory in Berlin
Berlin ( , ) is the capital and List of cities in Germany by population, largest city of Germany by both area and population. Its 3.7 million inhabitants make it the European Union's List of cities in the European Union by population within ci ...
in 1789, Klaproth was able to precipitate a yellow compound (likely sodium diuranate
Sodium diuranate, Na2U2O7·6H2O, is a uranium salt also known as the yellow oxide of uranium. Sodium diuranate is commonly referred to by the initials SDU. Along with ammonium diuranate it was a component in early yellowcakes. The ratio of the tw ...
) by dissolving pitchblende
Uraninite, formerly pitchblende, is a radioactive, uranium-rich mineral and ore with a chemical composition that is largely UO2 but because of oxidation typically contains variable proportions of U3O8. Radioactive decay of the uranium causes t ...
in nitric acid
Nitric acid is the inorganic compound with the formula . It is a highly corrosive mineral acid. The compound is colorless, but older samples tend to be yellow cast due to decomposition into oxides of nitrogen. Most commercially available nitri ...
and neutralizing the solution with sodium hydroxide.. Klaproth assumed the yellow substance was the oxide of a yet-undiscovered element and heated it with charcoal to obtain a black powder, which he thought was the newly discovered metal itself (in fact, that powder was an oxide of uranium). He named the newly discovered element after the planet Uranus
Uranus is the seventh planet from the Sun. Its name is a reference to the Greek god of the sky, Uranus ( Caelus), who, according to Greek mythology, was the great-grandfather of Ares (Mars), grandfather of Zeus (Jupiter) and father of ...
(named after the primordial Greek god of the sky), which had been discovered eight years earlier by William Herschel
Frederick William Herschel (; german: Friedrich Wilhelm Herschel; 15 November 1738 – 25 August 1822) was a German-born British astronomer and composer. He frequently collaborated with his younger sister and fellow astronomer Caroline ...
.
In 1841, Eugène-Melchior Péligot
Eugène-Melchior Péligot (24 March 1811 – 15 April 1890), also known as Eugène Péligot, was a French chemist who isolated the first sample of uranium metal in 1841.
Péligot proved that the black powder of Martin Heinrich Klaproth was not a ...
, Professor of Analytical Chemistry at the Conservatoire National des Arts et Métiers
A music school is an educational institution specialized in the study, training, and research of music. Such an institution can also be known as a school of music, music academy, music faculty, college of music, music department (of a larger ins ...
(Central School of Arts and Manufactures) in Paris
Paris () is the Capital city, capital and List of communes in France with over 20,000 inhabitants, most populous city of France, with an estimated population of 2,165,423 residents in 2019 in an area of more than 105 km² (41 sq mi), ma ...
, isolated the first sample of uranium metal by heating uranium tetrachloride
Uranium tetrachloride is an inorganic compound, a salt of uranium and chlorine, with the formula UCl4. It is a hygroscopic olive-green solid. It was used in the electromagnetic isotope separation (EMIS) process of uranium enrichment. It is one o ...
with potassium.
Henri Becquerel
Antoine Henri Becquerel (; 15 December 1852 – 25 August 1908) was a French engineer, physicist, Nobel laureate, and the first person to discover evidence of radioactivity. For work in this field he, along with Marie Skłodowska-Curie and Pie ...
discovered radioactivity by using uranium in 1896. Becquerel made the discovery in Paris by leaving a sample of a uranium salt, K2UO2(SO4)2 (potassium uranyl sulfate), on top of an unexposed photographic plate in a drawer and noting that the plate had become "fogged". He determined that a form of invisible light or rays emitted by uranium had exposed the plate.
During World War I when the Central Powers suffered a shortage of molybdenum to make artillery gun barrels and high speed tool steels they routinely substituted ferrouranium
Ferrouranium, also called ferro-uranium, is a ferroalloy, an alloy of iron and uranium, after World War II usually depleted uranium.
Composition and properties
The alloy contains about 35–50% uranium and 1.5–4.0% carbon. At least two intermeta ...
alloys which present many of the same physical characteristics. When this practice became known in 1916 the USA government requested several prominent universities to research these uses for uranium and tools made with these formulas remained in use for several decades only ending when the Manhattan Project and the Cold War placed a large demand on uranium for fission research and weapon development.
Fission research
 A team led by Enrico Fermi in 1934 observed that bombarding uranium with neutrons produces the emission of beta rays (
A team led by Enrico Fermi in 1934 observed that bombarding uranium with neutrons produces the emission of beta rays (electron
The electron ( or ) is a subatomic particle with a negative one elementary electric charge. Electrons belong to the first generation of the lepton particle family,
and are generally thought to be elementary particles because they have no ...
s or positrons from the elements produced; see beta particle). The fission products were at first mistaken for new elements with atomic numbers 93 and 94, which the Dean of the Faculty of Rome, Orso Mario Corbino, christened '' ausonium'' and '' hesperium'', respectively. The experiments leading to the discovery of uranium's ability to fission (break apart) into lighter elements and release binding energy were conducted by Otto Hahn
Otto Hahn (; 8 March 1879 – 28 July 1968) was a German chemist who was a pioneer in the fields of radioactivity and radiochemistry. He is referred to as the father of nuclear chemistry and father of nuclear fission. Hahn and Lise Meitner ...
and Fritz Strassmann
Friedrich Wilhelm Strassmann (; 22 February 1902 – 22 April 1980) was a German chemist who, with Otto Hahn in December 1938, identified the element barium as a product of the bombardment of uranium with neutrons. Their observation was the ke ...
. in Hahn's laboratory in Berlin. Lise Meitner and her nephew, the physicist Otto Robert Frisch, published the physical explanation in February 1939 and named the process " nuclear fission". Soon after, Fermi hypothesized that the fission of uranium might release enough neutrons to sustain a fission reaction. Confirmation of this hypothesis came in 1939, and later work found that on average about 2.5 neutrons are released by each fission of the rare uranium isotope uranium-235. Fermi urged Alfred O. C. Nier to separate uranium isotopes for determination of the fissile component, and on 29 February 1940, Nier used an instrument he built at the University of Minnesota to separate the world's first uranium-235 sample in the Tate Laboratory. After mailed to Columbia University
Columbia University (also known as Columbia, and officially as Columbia University in the City of New York) is a private research university in New York City. Established in 1754 as King's College on the grounds of Trinity Church in Manhatt ...
's cyclotron
A cyclotron is a type of particle accelerator invented by Ernest O. Lawrence in 1929–1930 at the University of California, Berkeley, and patented in 1932. Lawrence, Ernest O. ''Method and apparatus for the acceleration of ions'', filed: Jan ...
, John Dunning confirmed the sample to be the isolated fissile material on 1 March. Further work found that the far more common uranium-238 isotope can be transmuted into plutonium, which, like uranium-235, is also fissile by thermal neutrons. These discoveries led numerous countries to begin working on the development of nuclear weapons and nuclear power
Nuclear power is the use of nuclear reactions to produce electricity. Nuclear power can be obtained from nuclear fission, nuclear decay and nuclear fusion reactions. Presently, the vast majority of electricity from nuclear power is produced ...
. Despite fission having been discovered in Germany, the '' Uranverein'' ("uranium club") Germany's wartime project to research nuclear power and/or weapons was hampered by limited resources, infighting, the exile or non-involvement of several prominent scientists in the field and several crucial mistakes such as failing to account for impurities in available graphite samples which made it appear less suitable as a neutron moderator than it is in reality. Germany's attempts to build a natural uranium / heavy water reactor had not come close to reaching criticality by the time the Americans reached Haigerloch, the site of the last German wartime reactor experiment.
On 2 December 1942, as part of the Manhattan Project, another team led by Enrico Fermi was able to initiate the first artificial self-sustained nuclear chain reaction, Chicago Pile-1. An initial plan using enriched uranium-235 was abandoned as it was as yet unavailable in sufficient quantities. Working in a lab below the stands of Stagg Field at the University of Chicago
The University of Chicago (UChicago, Chicago, U of C, or UChi) is a private university, private research university in Chicago, Illinois. Its main campus is located in Chicago's Hyde Park, Chicago, Hyde Park neighborhood. The University of Chic ...
, the team created the conditions needed for such a reaction by piling together 400 short tons
The short ton (symbol tn) is a measurement unit equal to . It is commonly used in the United States, where it is known simply as a ton,
although the term is ambiguous, the single word being variously used for short, long, and metric ton.
The vari ...
(360 metric tons) of graphite, 58 short tons (53 metric tons) of uranium oxide
Uranium oxide is an oxide of the element uranium.
The metal uranium forms several oxides:
* Uranium dioxide or uranium(IV) oxide (UO2, the mineral uraninite or pitchblende)
* Diuranium pentoxide or uranium(V) oxide (U2O5)
* Uranium trioxide o ...
, and six short tons (5.5 metric tons) of uranium metal, a majority of which was supplied by Westinghouse Lamp Plant
The Westinghouse Lamp Plant located in Bloomfield, New Jersey, was one of the lamp manufacturing plants of Westinghouse Electric Corporation. The plant had a major involvement in supplying uranium metal for the world's first self-sustaining chain ...
in a makeshift production process.
Nuclear weaponry
 Two major types of atomic bombs were developed by the United States during
Two major types of atomic bombs were developed by the United States during World War II
World War II or the Second World War, often abbreviated as WWII or WW2, was a world war that lasted from 1939 to 1945. It involved the vast majority of the world's countries—including all of the great powers—forming two opposing ...
: a uranium-based device (codenamed "Little Boy
"Little Boy" was the type of atomic bomb dropped on the Japanese city of Hiroshima on 6 August 1945 during World War II, making it the first nuclear weapon used in warfare. The bomb was dropped by the Boeing B-29 Superfortress ''Enola Gay'' p ...
") whose fissile material was highly enriched uranium
Enriched uranium is a type of uranium in which the percent composition of uranium-235 (written 235U) has been increased through the process of isotope separation. Naturally occurring uranium is composed of three major isotopes: uranium-238 (238U ...
, and a plutonium-based device (see Trinity test
Trinity was the code name of the first detonation of a nuclear weapon. It was conducted by the United States Army at 5:29 a.m. on July 16, 1945, as part of the Manhattan Project. The test was conducted in the Jornada del Muerto desert abo ...
and " Fat Man") whose plutonium was derived from uranium-238. The uranium-based Little Boy device became the first nuclear weapon used in war when it was detonated over the Japanese city of Hiroshima on 6 August 1945. Exploding with a yield equivalent to 12,500 tonnes of TNT
Trinitrotoluene (), more commonly known as TNT, more specifically 2,4,6-trinitrotoluene, and by its preferred IUPAC name 2-methyl-1,3,5-trinitrobenzene, is a chemical compound with the formula C6H2(NO2)3CH3. TNT is occasionally used as a reagen ...
, the blast and thermal wave of the bomb destroyed nearly 50,000 buildings and killed approximately 75,000 people (see Atomic bombings of Hiroshima and Nagasaki). Initially it was believed that uranium was relatively rare, and that nuclear proliferation could be avoided by simply buying up all known uranium stocks, but within a decade large deposits of it were discovered in many places around the world.
Reactors
 The X-10 Graphite Reactor at Oak Ridge National Laboratory (ORNL) in Oak Ridge, Tennessee, formerly known as the Clinton Pile and X-10 Pile, was the world's second artificial nuclear reactor (after Enrico Fermi's Chicago Pile) and was the first reactor designed and built for continuous operation. Argonne National Laboratory's Experimental Breeder Reactor I, located at the Atomic Energy Commission's National Reactor Testing Station near
The X-10 Graphite Reactor at Oak Ridge National Laboratory (ORNL) in Oak Ridge, Tennessee, formerly known as the Clinton Pile and X-10 Pile, was the world's second artificial nuclear reactor (after Enrico Fermi's Chicago Pile) and was the first reactor designed and built for continuous operation. Argonne National Laboratory's Experimental Breeder Reactor I, located at the Atomic Energy Commission's National Reactor Testing Station near Arco, Idaho
Arco is a city in Butte County, Idaho, Butte County, Idaho, United States. The population was 879 as of the 2020 United States census, down from 995 at the 2010 United States Census, 2010 census. Arco is the county seat and largest city in Butte ...
, became the first nuclear reactor to create electricity on 20 December 1951. Initially, four 150-watt light bulbs were lit by the reactor, but improvements eventually enabled it to power the whole facility (later, the town of Arco became the first in the world to have all its electricity
Electricity is the set of physical phenomena associated with the presence and motion of matter that has a property of electric charge. Electricity is related to magnetism, both being part of the phenomenon of electromagnetism, as describ ...
come from nuclear power generated by BORAX-III, another reactor designed and operated by Argonne National Laboratory). The world's first commercial scale nuclear power station, Obninsk
Obninsk (russian: О́бнинск) is a city in Kaluga Oblast, Russia, located on the bank of the Protva River southwest of Moscow and northeast of Kaluga. Population:
History
The history of Obninsk began in 1945 when the First Research In ...
in the Soviet Union
The Soviet Union,. officially the Union of Soviet Socialist Republics. (USSR),. was a List of former transcontinental countries#Since 1700, transcontinental country that spanned much of Eurasia from 1922 to 1991. A flagship communist state, ...
, began generation with its reactor AM-1 on 27 June 1954. Other early nuclear power plants were Calder Hall in England, which began generation on 17 October 1956, and the Shippingport Atomic Power Station
The Shippingport Atomic Power Station was (according to the US Nuclear Regulatory Commission) the world's first full-scale atomic electric power plant devoted exclusively to peacetime uses.Though Obninsk Nuclear Power Plant was connected to the M ...
in Pennsylvania
Pennsylvania (; ( Pennsylvania Dutch: )), officially the Commonwealth of Pennsylvania, is a state spanning the Mid-Atlantic, Northeastern, Appalachian, and Great Lakes regions of the United States. It borders Delaware to its southeast, ...
, which began on 26 May 1958. Nuclear power was used for the first time for propulsion by a submarine, the USS ''Nautilus'', in 1954.
Prehistoric naturally occurring fission
In 1972, the French physicist Francis Perrin discovered fifteen ancient and no longer active natural nuclear fission reactors in three separate ore deposits at theOklo mine
Oklo Mine (sometimes Oklo Reactor or Oklo Mines), located in Oklo, Gabon on the west coast of Central Africa, is believed to be the only natural nuclear fission reactor. Oklo consists of 16 sites at which self-sustaining nuclear fission reactio ...
in Gabon
Gabon (; ; snq, Ngabu), officially the Gabonese Republic (french: République gabonaise), is a country on the west coast of Central Africa. Located on the equator, it is bordered by Equatorial Guinea to the northwest, Cameroon to the nort ...
, West Africa
West Africa or Western Africa is the westernmost region of Africa. The United Nations defines Western Africa as the 16 countries of Benin, Burkina Faso, Cape Verde, The Gambia, Ghana, Guinea, Guinea-Bissau, Ivory Coast, Liberia, Mali, M ...
, collectively known as the Oklo Fossil Reactors. The ore deposit is 1.7 billion years old; then, uranium-235 constituted about 3% of the total uranium on Earth. This is high enough to permit a sustained nuclear fission chain reaction to occur, provided other supporting conditions exist. The capacity of the surrounding sediment to contain the nuclear waste
Radioactive waste is a type of hazardous waste that contains radioactive material. Radioactive waste is a result of many activities, including nuclear medicine, nuclear research, nuclear power generation, rare-earth mining, and nuclear weapons ...
products has been cited by the U.S. federal government as supporting evidence for the feasibility to store spent nuclear fuel at the Yucca Mountain nuclear waste repository
The Yucca Mountain Nuclear Waste Repository, as designated by the Nuclear Waste Policy Act amendments of 1987, is a proposed deep geological repository storage facility within Yucca Mountain for spent nuclear fuel and other high-level radio ...
.
Contamination and the Cold War legacy
nuclear tests
Nuclear weapons tests are experiments carried out to determine nuclear weapons' effectiveness, yield, and explosive capability. Testing nuclear weapons offers practical information about how the weapons function, how detonations are affected by ...
by the Soviet Union and the United States in the 1950s and early 1960s and by France
France (), officially the French Republic ( ), is a country primarily located in Western Europe. It also comprises of overseas regions and territories in the Americas and the Atlantic, Pacific and Indian Oceans. Its metropolitan area ...
into the 1970s and 1980s spread a significant amount of fallout from uranium daughter isotope
In nuclear physics, a decay product (also known as a daughter product, daughter isotope, radio-daughter, or daughter nuclide) is the remaining nuclide left over from radioactive decay. Radioactive decay often proceeds via a sequence of steps (de ...
s around the world. Additional fallout and pollution occurred from several nuclear accidents
A nuclear and radiation accident is defined by the International Atomic Energy Agency (IAEA) as "an event that has led to significant consequences to people, the environment or the facility. Examples include lethal effects to individuals, lar ...
.
Uranium miners have a higher incidence of cancer
Cancer is a group of diseases involving abnormal cell growth with the potential to invade or spread to other parts of the body. These contrast with benign tumors, which do not spread. Possible signs and symptoms include a lump, abnormal b ...
. An excess risk of lung cancer among Navajo uranium miners, for example, has been documented and linked to their occupation. The Radiation Exposure Compensation Act
The United States Radiation Exposure Compensation Act (RECA) is a federal statute providing for the monetary compensation of people, including atomic veterans, who contracted cancer and a number of other specified diseases as a direct result of t ...
, a 1990 law in the US, required $100,000 in "compassion payments" to uranium miners diagnosed with cancer or other respiratory ailments.
During the Cold War between the Soviet Union and the United States, huge stockpiles of uranium were amassed and tens of thousands of nuclear weapons were created using enriched uranium and plutonium made from uranium. Since the break-up of the Soviet Union in 1991, an estimated 600 short tons (540 metric tons) of highly enriched weapons grade uranium (enough to make 40,000 nuclear warheads) have been stored in often inadequately guarded facilities in the Russian Federation
Russia (, , ), or the Russian Federation, is a transcontinental country spanning Eastern Europe and Northern Asia. It is the largest country in the world, with its internationally recognised territory covering , and encompassing one-eig ...
and several other former Soviet states. Police in Asia
Asia (, ) is one of the world's most notable geographical regions, which is either considered a continent in its own right or a subcontinent of Eurasia, which shares the continental landmass of Afro-Eurasia with Africa. Asia covers an are ...
, Europe
Europe is a large peninsula conventionally considered a continent in its own right because of its great physical size and the weight of its history and traditions. Europe is also considered a subcontinent of Eurasia and it is located entirel ...
, and South America
South America is a continent entirely in the Western Hemisphere and mostly in the Southern Hemisphere, with a relatively small portion in the Northern Hemisphere at the northern tip of the continent. It can also be described as the sout ...
on at least 16 occasions from 1993 to 2005 have intercepted shipments of smuggled bomb-grade uranium or plutonium, most of which was from ex-Soviet sources. From 1993 to 2005 the Material Protection, Control, and Accounting Program, operated by the federal government of the United States
The federal government of the United States (U.S. federal government or U.S. government) is the national government of the United States, a federal republic located primarily in North America, composed of 50 states, a city within a fe ...
, spent approximately US $550 million to help safeguard uranium and plutonium stockpiles in Russia. This money was used for improvements and security enhancements at research and storage facilities. ''Scientific American'' reported in February 2006 that in some of the facilities security consisted of chain link fences which were in severe states of disrepair. According to an interview from the article, one facility had been storing samples of enriched (weapons grade) uranium in a broom closet before the improvement project; another had been keeping track of its stock of nuclear warheads using index cards kept in a shoe box.
Occurrence
Origin
Along with all elements havingatomic weight
Relative atomic mass (symbol: ''A''; sometimes abbreviated RAM or r.a.m.), also known by the deprecated synonym atomic weight, is a dimensionless physical quantity defined as the ratio of the average mass of atoms of a chemical element in a giv ...
s higher than that of iron
Iron () is a chemical element with Symbol (chemistry), symbol Fe (from la, Wikt:ferrum, ferrum) and atomic number 26. It is a metal that belongs to the first transition series and group 8 element, group 8 of the periodic table. It is, Abundanc ...
, uranium is only naturally formed by the r-process (rapid neutron capture) in supernovae and neutron star merger
A neutron star merger is a type of stellar collision.
It occurs in a fashion similar to the rare brand of type Ia supernovae resulting from merging white dwarf stars.
When two neutron stars orbit each other closely, they gradually spiral i ...
s. Primordial thorium and uranium are only produced in the r-process, because the s-process (slow neutron capture) is too slow and cannot pass the gap of instability after bismuth. Besides the two extant primordial uranium isotopes, 235U and 238U, the r-process also produced significant quantities of 236U, which has a shorter half-life and so is an extinct radionuclide
An extinct radionuclide is a radionuclide that was formed by nucleosynthesis before the formation of the Solar System, about 4.6 billion years ago, but has since decayed to virtually zero abundance and is no longer detectable as a primordial nuc ...
, having long since decayed completely to 232Th. Uranium-236 was itself enriched by the decay of 244Pu, accounting for the observed higher-than-expected abundance of thorium and lower-than-expected abundance of uranium. While the natural abundance of uranium has been supplemented by the decay of extinct 242Pu (half-life 0.375 million years) and 247Cm (half-life 16 million years), producing 238U and 235U respectively, this occurred to an almost negligible extent due to the shorter half-lives of these parents and their lower production than 236U and 244Pu, the parents of thorium: the 247Cm:235U ratio at the formation of the Solar System was .
Biotic and abiotic

naturally occurring
A natural product is a natural compound or substance produced by a living organism—that is, found in nature. In the broadest sense, natural products include any substance produced by life. Natural products can also be prepared by chemical sy ...
element that can be found in low levels within all rock, soil, and water. Uranium is the 51st element in order of abundance in the Earth's crust. Uranium is also the highest-numbered element to be found naturally in significant quantities on Earth and is almost always found combined with other elements. The decay of uranium, thorium, and potassium-40
Potassium-40 (40K) is a radioactive isotope of potassium which has a long half-life of 1.25 billion years. It makes up about 0.012% (120 ppm) of the total amount of potassium found in nature.
Potassium-40 undergoes three types of radioactive d ...
in the Earth's mantle is thought to be the main source of heat that keeps the Earth's outer core
Earth's outer core is a fluid layer about thick, composed of mostly iron and nickel that lies above Earth's solid inner core and below its mantle. The outer core begins approximately beneath Earth's surface at the core-mantle boundary and e ...
in the liquid state and drives mantle convection, which in turn drives plate tectonics.
Uranium's average concentration in the Earth's crust is (depending on the reference) 2 to 4 parts per million, or about 40 times as abundant as silver
Silver is a chemical element with the symbol Ag (from the Latin ', derived from the Proto-Indo-European ''h₂erǵ'': "shiny" or "white") and atomic number 47. A soft, white, lustrous transition metal, it exhibits the highest electrical ...
. The Earth's crust from the surface to 25 km (15 mi) down is calculated to contain 1017 kg (2 lb) of uranium while the ocean
The ocean (also the sea or the world ocean) is the body of salt water that covers approximately 70.8% of the surface of Earth and contains 97% of Earth's water. An ocean can also refer to any of the large bodies of water into which the wo ...
s may contain 1013 kg (2 lb). The concentration of uranium in soil ranges from 0.7 to 11 parts per million (up to 15 parts per million in farmland soil due to use of phosphate fertilizer
A fertilizer (American English) or fertiliser (British English; see spelling differences) is any material of natural or synthetic origin that is applied to soil or to plant tissues to supply plant nutrients. Fertilizers may be distinct from ...
s), and its concentration in sea water is 3 parts per billion.
Uranium is more plentiful than antimony
Antimony is a chemical element with the symbol Sb (from la, stibium) and atomic number 51. A lustrous gray metalloid, it is found in nature mainly as the sulfide mineral stibnite (Sb2S3). Antimony compounds have been known since ancient t ...
, tin, cadmium
Cadmium is a chemical element with the symbol Cd and atomic number 48. This soft, silvery-white metal is chemically similar to the two other stable metals in group 12, zinc and mercury. Like zinc, it demonstrates oxidation state +2 in most of ...
, mercury, or silver, and it is about as abundant as arsenic
Arsenic is a chemical element with the symbol As and atomic number 33. Arsenic occurs in many minerals, usually in combination with sulfur and metals, but also as a pure elemental crystal. Arsenic is a metalloid. It has various allotropes, ...
or molybdenum. Uranium is found in hundreds of minerals, including uraninite (the most common uranium ore
Ore is natural rock or sediment that contains one or more valuable minerals, typically containing metals, that can be mined, treated and sold at a profit.Encyclopædia Britannica. "Ore". Encyclopædia Britannica Online. Retrieved 7 Apr ...
), carnotite
Carnotite is a potassium uranium vanadate radioactive mineral with chemical formula K2( U O2)2( VO4)2·3 H2O. The water content can vary and small amounts of calcium, barium, magnesium, iron, and sodium are often present.
Occurrence
Carnotite ...
, autunite
Autunite (hydrated calcium uranyl phosphate), with formula Ca(UO2)2(PO4)2·10–12H2O, is a yellow-greenish fluorescent phosphate mineral with a hardness of 2–. Autunite crystallizes in the orthorhombic system and often occurs as tabular square ...
, uranophane
Uranophane ( Ca( U O2)2( SiO3O H)2·5 H2O), also known as uranotile, is a rare calcium uranium silicate hydrate mineral that forms from the oxidation of other uranium-bearing minerals. It has a yellow color and is radioactive.
Alice Mary Weeks, a ...
, torbernite
Torbernite is a radioactive, hydrated green copper uranyl phosphate mineral, found in granites and other uranium-bearing deposits as a secondary mineral. Its name derives from the Swedish chemist Torbern Bergman (1735–1784), It is also known ...
, and coffinite
Coffinite is a uranium-bearing silicate mineral with formula: U(SiO4)1−x(OH)4x.
It occurs as black incrustations, dark to pale-brown in thin section. It has a grayish-black streak. It has a brittle to conchoidal fracture. The hardness of coffini ...
. Significant concentrations of uranium occur in some substances such as phosphate
In chemistry, a phosphate is an anion, salt, functional group or ester derived from a phosphoric acid. It most commonly means orthophosphate, a derivative of orthophosphoric acid .
The phosphate or orthophosphate ion is derived from phosph ...
rock deposits, and minerals such as lignite, and monazite
Monazite is a primarily reddish-brown phosphate mineral that contains rare-earth elements. Due to variability in composition, monazite is considered a group of minerals. The most common species of the group is monazite-(Ce), that is, the ceriu ...
sands in uranium-rich ores (it is recovered commercially from sources with as little as 0.1% uranium).
 Some bacteria, such as ''
Some bacteria, such as ''Shewanella putrefaciens
''Shewanella putrefaciens'' is a Gram-negative pleomorphic bacterium. It has been isolated from marine environments, as well as from anaerobic sandstone in the Morrison Formation in New Mexico. ''S. putrefaciens'' is also a facultative anaerobe ...
'', '' Geobacter metallireducens'' and some strains of '' Burkholderia fungorum'', use uranium for their growth and convert U(VI) to U(IV). Recent research suggests that this pathway includes reduction of the soluble U(VI) via an intermediate U(V) pentavalent state.
Other organisms, such as the lichen ''Trapelia involuta'' or microorganisms such as the bacterium
Bacteria (; singular: bacterium) are ubiquitous, mostly free-living organisms often consisting of one biological cell. They constitute a large domain of prokaryotic microorganisms. Typically a few micrometres in length, bacteria were amon ...
'' Citrobacter'', can absorb concentrations of uranium that are up to 300 times the level of their environment. ''Citrobacter'' species absorb uranyl ions when given glycerol phosphate (or other similar organic phosphates). After one day, one gram of bacteria can encrust themselves with nine grams of uranyl phosphate crystals; this creates the possibility that these organisms could be used in bioremediation
Bioremediation broadly refers to any process wherein a biological system (typically bacteria, microalgae, fungi, and plants), living or dead, is employed for removing environmental pollutants from air, water, soil, flue gasses, industrial effluent ...
to decontaminate uranium-polluted water.
The proteobacterium '' Geobacter'' has also been shown to bioremediate uranium in ground water. The mycorrhizal fungus Glomus intraradices
''Rhizophagus irregularis'' (previously known as ''Glomus intraradices'') is an arbuscular mycorrhizal fungus used as a soil inoculant in agriculture and horticulture. ''Rhizophagus irregularis'' is also commonly used in scientific studies of the ...
increases uranium content in the roots of its symbiotic plant.
In nature, uranium(VI) forms highly soluble carbonate complexes at alkaline pH. This leads to an increase in mobility and availability of uranium to groundwater and soil from nuclear wastes which leads to health hazards. However, it is difficult to precipitate uranium as phosphate in the presence of excess carbonate at alkaline pH. A ''Sphingomonas
''Sphingomonas'' was defined in 1990 as a group of Gram-negative, rod-shaped, chemoheterotrophic, strictly aerobic bacteria. They possess ubiquinone 10 as their major respiratory quinone, contain glycosphingolipids (GSLs), specifically ceramide ...
'' sp. strain BSAR-1 has been found to express a high activity alkaline phosphatase (PhoK) that has been applied for bioprecipitation of uranium as uranyl phosphate species from alkaline solutions. The precipitation ability was enhanced by overexpressing PhoK protein in '' E. coli''.
Plant
Plants are predominantly photosynthetic eukaryotes of the kingdom Plantae. Historically, the plant kingdom encompassed all living things that were not animals, and included algae and fungi; however, all current definitions of Plantae exclu ...
s absorb some uranium from soil. Dry weight concentrations of uranium in plants range from 5 to 60 parts per billion, and ash from burnt wood can have concentrations up to 4 parts per million. Dry weight concentrations of uranium in food plants are typically lower with one to two micrograms per day ingested through the food people eat.
Production and mining
tonne
The tonne ( or ; symbol: t) is a unit of mass equal to 1000 kilograms. It is a non-SI unit accepted for use with SI. It is also referred to as a metric ton to distinguish it from the non-metric units of the short ton ( United State ...
s, of which 22,451 t (32%) was mined in Kazakhstan
Kazakhstan, officially the Republic of Kazakhstan, is a transcontinental country located mainly in Central Asia and partly in Eastern Europe. It borders Russia to the north and west, China to the east, Kyrgyzstan to the southeast, Uzbeki ...
. Other important uranium mining countries are Canada
Canada is a country in North America. Its ten provinces and three territories extend from the Atlantic Ocean to the Pacific Ocean and northward into the Arctic Ocean, covering over , making it the world's second-largest country by tot ...
(9,331 t), Australia (6,350 t), Niger (4,518 t), Namibia
Namibia (, ), officially the Republic of Namibia, is a country in Southern Africa. Its western border is the Atlantic Ocean. It shares land borders with Zambia and Angola to the north, Botswana to the east and South Africa to the south and ea ...
(4,323 t) and Russia
Russia (, , ), or the Russian Federation, is a transcontinental country spanning Eastern Europe and Northern Asia. It is the largest country in the world, with its internationally recognised territory covering , and encompassing one-eig ...
(3,135 t).
Uranium ore is mined in several ways: by open pit, underground, in-situ leach
In-situ leaching (ISL), also called in-situ recovery (ISR) or solution mining, is a mining process used to recover minerals such as copper and uranium through boreholes drilled into a deposit, ''in situ''. In situ leach works by artificially disso ...
ing, and borehole mining (see uranium mining). Low-grade uranium ore mined typically contains 0.01 to 0.25% uranium oxides. Extensive measures must be employed to extract the metal from its ore.. High-grade ores found in Athabasca Basin
The Athabasca Basin is a region in the Canadian Shield of northern Saskatchewan and Alberta, Canada. It is best known as the world's leading source of high-grade uranium and currently supplies about 20% of the world's uranium.
The basin i ...
deposits in Saskatchewan
Saskatchewan ( ; ) is a province in western Canada, bordered on the west by Alberta, on the north by the Northwest Territories, on the east by Manitoba, to the northeast by Nunavut, and on the south by the U.S. states of Montana and North Dak ...
, Canada can contain up to 23% uranium oxides on average. Uranium ore is crushed and rendered into a fine powder and then leached with either an acid or alkali. The leachate is subjected to one of several sequences of precipitation, solvent extraction, and ion exchange. The resulting mixture, called yellowcake
Yellowcake (also called urania) is a type of uranium concentrate powder obtained from leach solutions, in an intermediate step in the processing of uranium ores. It is a step in the processing of uranium after it has been mined but before f ...
, contains at least 75% uranium oxides U3O8. Yellowcake is then calcined
Calcination refers to thermal treatment of a solid chemical compound (e.g. mixed carbonate ores) whereby the compound is raised to high temperature without melting under restricted supply of ambient oxygen (i.e. gaseous O2 fraction of air), gener ...
to remove impurities from the milling process before refining and conversion.
Commercial-grade uranium can be produced through the reduction of uranium halides with alkali or alkaline earth metal
The alkaline earth metals are six chemical elements in group 2 of the periodic table. They are beryllium (Be), magnesium (Mg), calcium (Ca), strontium (Sr), barium (Ba), and radium (Ra).. The elements have very similar properties: they are all ...
s. Uranium metal can also be prepared through electrolysis of or
, dissolved in molten calcium chloride
Calcium chloride is an inorganic compound, a salt with the chemical formula . It is a white crystalline solid at room temperature, and it is highly soluble in water. It can be created by neutralising hydrochloric acid with calcium hydroxide.
Ca ...
() and sodium chloride ( NaCl) solution. Very pure uranium is produced through the thermal decomposition
Thermal decomposition, or thermolysis, is a chemical decomposition caused by heat. The decomposition temperature of a substance is the temperature at which the substance chemically decomposes. The reaction is usually endothermic as heat is re ...
of uranium halides on a hot filament.
Yellowcake
Yellowcake (also called urania) is a type of uranium concentrate powder obtained from leach solutions, in an intermediate step in the processing of uranium ores. It is a step in the processing of uranium after it has been mined but before f ...
is a concentrated mixture of uranium oxides that is further refined to extract pure uranium.
Resources and reserves
 It is estimated that 5.5 million tonnes of uranium exists in ore reserves that are economically viable at US$59 per lb of uranium, while 35 million tonnes are classed as mineral resources (reasonable prospects for eventual economic extraction). Prices went from about $10/lb in May 2003 to $138/lb in July 2007. This has caused a big increase in spending on exploration, with US$200 million being spent worldwide in 2005, a 54% increase on the previous year. This trend continued through 2006, when expenditure on exploration rocketed to over $774 million, an increase of over 250% compared to 2004. The
It is estimated that 5.5 million tonnes of uranium exists in ore reserves that are economically viable at US$59 per lb of uranium, while 35 million tonnes are classed as mineral resources (reasonable prospects for eventual economic extraction). Prices went from about $10/lb in May 2003 to $138/lb in July 2007. This has caused a big increase in spending on exploration, with US$200 million being spent worldwide in 2005, a 54% increase on the previous year. This trend continued through 2006, when expenditure on exploration rocketed to over $774 million, an increase of over 250% compared to 2004. The OECD
The Organisation for Economic Co-operation and Development (OECD; french: Organisation de coopération et de développement économiques, ''OCDE'') is an intergovernmental organisation with 38 member countries, founded in 1961 to stimulate e ...
Nuclear Energy Agency
The Nuclear Energy Agency (NEA) is an intergovernmental agency that is organized under the Organisation for Economic Co-operation and Development (OECD). Originally formed on 1 February 1958 with the name European Nuclear Energy Agency (ENEA)— ...
said exploration figures for 2007 would likely match those for 2006.
Australia has 31% of the world's known uranium ore reserves and the world's largest single uranium deposit, located at the Olympic Dam Mine in South Australia
South Australia (commonly abbreviated as SA) is a state in the southern central part of Australia. It covers some of the most arid parts of the country. With a total land area of , it is the fourth-largest of Australia's states and territories ...
. There is a significant reserve of uranium in Bakouma, a sub-prefecture in the prefecture of Mbomou in the Central African Republic
The Central African Republic (CAR; ; , RCA; , or , ) is a landlocked country in Central Africa. It is bordered by Chad to the north, Sudan to the northeast, South Sudan to the southeast, the DR Congo to the south, the Republic of th ...
.
Some nuclear fuel comes from nuclear weapons being dismantled, such as from the Megatons to Megawatts Program.
An additional 4.6 billion tonnes of uranium are estimated to be dissolved in sea water ( Japanese scientists in the 1980s showed that extraction of uranium from sea water using ion exchange
Ion exchange is a reversible interchange of one kind of ion present in an insoluble solid with another of like charge present in a solution surrounding the solid with the reaction being used especially for softening or making water demineralised, ...
rs was technically feasible). There have been experiments to extract uranium from sea water, but the yield has been low due to the carbonate present in the water. In 2012, ORNL researchers announced the successful development of a new absorbent material dubbed HiCap which performs surface retention of solid or gas molecules, atoms or ions and also effectively removes toxic metals from water, according to results verified by researchers at Pacific Northwest National Laboratory.
Supplies
 In 2005, seventeen countries produced concentrated uranium oxides:
In 2005, seventeen countries produced concentrated uranium oxides: Canada
Canada is a country in North America. Its ten provinces and three territories extend from the Atlantic Ocean to the Pacific Ocean and northward into the Arctic Ocean, covering over , making it the world's second-largest country by tot ...
(27.9% of world production), Australia (22.8%), Kazakhstan
Kazakhstan, officially the Republic of Kazakhstan, is a transcontinental country located mainly in Central Asia and partly in Eastern Europe. It borders Russia to the north and west, China to the east, Kyrgyzstan to the southeast, Uzbeki ...
(10.5%), Russia
Russia (, , ), or the Russian Federation, is a transcontinental country spanning Eastern Europe and Northern Asia. It is the largest country in the world, with its internationally recognised territory covering , and encompassing one-eig ...
(8.0%), Namibia
Namibia (, ), officially the Republic of Namibia, is a country in Southern Africa. Its western border is the Atlantic Ocean. It shares land borders with Zambia and Angola to the north, Botswana to the east and South Africa to the south and ea ...
(7.5%), Niger (7.4%), Uzbekistan
Uzbekistan (, ; uz, Ozbekiston, italic=yes / , ; russian: Узбекистан), officially the Republic of Uzbekistan ( uz, Ozbekiston Respublikasi, italic=yes / ; russian: Республика Узбекистан), is a doubly landlocked co ...
(5.5%), the United States
The United States of America (U.S.A. or USA), commonly known as the United States (U.S. or US) or America, is a country primarily located in North America. It consists of 50 states, a federal district, five major unincorporated territori ...
(2.5%), Argentina
Argentina (), officially the Argentine Republic ( es, link=no, República Argentina), is a country in the southern half of South America. Argentina covers an area of , making it the second-largest country in South America after Brazil, th ...
(2.1%), Ukraine
Ukraine ( uk, Україна, Ukraïna, ) is a country in Eastern Europe. It is the second-largest European country after Russia, which it borders to the east and northeast. Ukraine covers approximately . Prior to the ongoing Russian inv ...
(1.9%) and China (1.7%). In 2008 Kazakhstan was forecast to increase production and may have become the world's largest producer of uranium by 2009 with an expected production of 12,826 tonnes, compared to Canada with 11,100 t and Australia with 9,430 t. The predictions have come true. In 2019 Kazakhstan produces the largest share of uranium from mines 42% of world supply, followed by Canada (13%) and Australia (12%), Namibia (10%), Uzbekistan (6%), Niger (5%), Russia (5%), China (3%), Ukraine (1.5%), USA (0.12%), India (0.6%), Iran (0.13%), with total world production 54752 tonnes from mines. However, it should be mentioned that in 2019 uranium was mined not only by conventional underground mining of ores 43% of production (54752 tonnes), where rock mineralised is removed from the ground, breaking it up and treating it to remove the minerals being sought but also by in-situ leaching methods (ISL) 57% of world production (64,566 tonnes).
In the late 1960s, UN geologists also discovered major uranium deposits and other rare mineral reserves in Somalia
Somalia, , Osmanya script: 𐒈𐒝𐒑𐒛𐒐𐒘𐒕𐒖; ar, الصومال, aṣ-Ṣūmāl officially the Federal Republic of SomaliaThe ''Federal Republic of Somalia'' is the country's name per Article 1 of thProvisional Constituti ...
. The find was the largest of its kind, with industry experts estimating the deposits at over 25% of the world's then known uranium reserves of 800,000 tons.
The ultimate available supply is believed to be sufficient for at least the next 85 years, although some studies indicate underinvestment in the late twentieth century may produce supply problems in the 21st century.
Uranium deposits seem to be log-normal distributed. There is a 300-fold increase in the amount of uranium recoverable for each tenfold decrease in ore grade.
In other words, there is little high grade ore and proportionately much more low grade ore available.
Compounds
Oxidation states and oxides
Oxides
Calcined uranium yellowcake, as produced in many large mills, contains a distribution of uranium oxidation species in various forms ranging from most oxidized to least oxidized. Particles with short residence times in a calciner will generally be less oxidized than those with long retention times or particles recovered in the stack scrubber. Uranium content is usually referenced to , which dates to the days of the Manhattan Project when was used as an analytical chemistry reporting standard. Phase relationships in the uranium-oxygen system are complex. The most important oxidation states of uranium are uranium(IV) and uranium(VI), and their two corresponding oxides are, respectively, uranium dioxide () anduranium trioxide
Uranium trioxide (UO3), also called uranyl oxide, uranium(VI) oxide, and uranic oxide, is the hexavalent oxide of uranium. The solid may be obtained by heating uranyl nitrate to 400 °C. Its most commonly encountered polymorph, γ-UO3, is a ...
().. Other uranium oxide
Uranium oxide is an oxide of the element uranium.
The metal uranium forms several oxides:
* Uranium dioxide or uranium(IV) oxide (UO2, the mineral uraninite or pitchblende)
* Diuranium pentoxide or uranium(V) oxide (U2O5)
* Uranium trioxide o ...
s such as uranium monoxide (UO), diuranium pentoxide (), and uranium peroxide () also exist.
The most common forms of uranium oxide are triuranium octoxide
Triuranium octoxide (U3O8) is a compound of uranium. It is present as an olive green to black, odorless solid. It is one of the more popular forms of yellowcake and is shipped between mills and refineries in this form.
U3O8 has potential long-ter ...
() and . Both oxide forms are solids that have low solubility in water and are relatively stable over a wide range of environmental conditions. Triuranium octoxide is (depending on conditions) the most stable compound of uranium and is the form most commonly found in nature. Uranium dioxide is the form in which uranium is most commonly used as a nuclear reactor fuel. At ambient temperatures, will gradually convert to . Because of their stability, uranium oxides are generally considered the preferred chemical form for storage or disposal.
Aqueous chemistry
 Salts of many oxidation states of uranium are water-
Salts of many oxidation states of uranium are water-soluble
In chemistry, solubility is the ability of a substance, the solute, to form a solution with another substance, the solvent. Insolubility is the opposite property, the inability of the solute to form such a solution.
The extent of the solubi ...
and may be studied in aqueous solutions. The most common ionic forms are (brown-red), (green), (unstable), and (yellow), for U(III), U(IV), U(V), and U(VI), respectively.. A few solid and semi-metallic compounds such as UO and US exist for the formal oxidation state uranium(II), but no simple ions are known to exist in solution for that state. Ions of liberate hydrogen
Hydrogen is the chemical element with the symbol H and atomic number 1. Hydrogen is the lightest element. At standard conditions hydrogen is a gas of diatomic molecules having the formula . It is colorless, odorless, tasteless, non-toxic ...
from water
Water (chemical formula ) is an Inorganic compound, inorganic, transparent, tasteless, odorless, and Color of water, nearly colorless chemical substance, which is the main constituent of Earth's hydrosphere and the fluids of all known living ...
and are therefore considered to be highly unstable. The ion represents the uranium(VI) state and is known to form compounds such as uranyl carbonate
Uranyl carbonate refers to the inorganic compound with the formula UO2CO3. Also known by its mineral name rutherfordine, this material consists of uranyl (UO22+) and carbonate (CO32-). Like most uranyl salts, the compound is a polymeric, each uran ...
, uranyl chloride and uranyl sulfate
Uranyl sulfate describes a family of inorganic compounds with the formula UO2SO4(H2O)n. These salts consist of sulfate, the uranyl ion, and water. They are lemon-yellow solids. Uranyl sulfates are intermediates in some extraction methods used for ...
. also forms complexes with various organic chelating
Chelation is a type of bonding of ions and molecules to metal ions. It involves the formation or presence of two or more separate coordinate bonds between a polydentate (multiple bonded) ligand and a single central metal atom. These ligands are ...
agents, the most commonly encountered of which is uranyl acetate
Uranyl acetate is the acetate salt of uranium oxide, a toxic yellow-green powder useful in certain laboratory tests. Structurally, it is a coordination polymer with formula UO2(CH3CO2)2(H2O)·H2O.
Structure
In the polymer, uranyl (UO22+) ...
.
Unlike the uranyl salts of uranium and polyatomic ion uranium-oxide cationic forms, the uranate
A uranate is a ternary oxide involving the element uranium in one of the oxidation states 4, 5 or 6. A typical chemical formula is MxUyOz, where M represents a cation. The uranium atom in uranates(VI) has two short collinear U–O bonds and eithe ...
s, salts containing a polyatomic uranium-oxide anion, are generally not water-soluble.
Carbonates
The interactions of carbonate anions with uranium(VI) cause the Pourbaix diagram to change greatly when the medium is changed from water to a carbonate containing solution. While the vast majority of carbonates are insoluble in water (students are often taught that all carbonates other than those of alkali metals are insoluble in water), uranium carbonates are often soluble in water. This is because a U(VI) cation is able to bind two terminal oxides and three or more carbonates to form anionic complexes.Effects of pH
The uranium fraction diagrams in the presence of carbonate illustrate this further: when the pH of a uranium(VI) solution increases, the uranium is converted to a hydrated uranium oxide hydroxide and at high pHs it becomes an anionic hydroxide complex. When carbonate is added, uranium is converted to a series of carbonate complexes if the pH is increased. One effect of these reactions is increased solubility of uranium in the pH range 6 to 8, a fact that has a direct bearing on the long term stability of spent uranium dioxide nuclear fuels.Hydrides, carbides and nitrides
Uranium metal heated to reacts withhydrogen
Hydrogen is the chemical element with the symbol H and atomic number 1. Hydrogen is the lightest element. At standard conditions hydrogen is a gas of diatomic molecules having the formula . It is colorless, odorless, tasteless, non-toxic ...
to form uranium hydride
Uranium hydride, also called uranium trihydride (UH3), is an inorganic compound and a hydride of uranium.
Properties
Uranium hydride is a highly toxic, brownish grey to brownish black pyrophoric powder or brittle solid. Its density at 20 ° ...
. Even higher temperatures will reversibly remove the hydrogen. This property makes uranium hydrides convenient starting materials to create reactive uranium powder along with various uranium carbide, nitride
In chemistry, a nitride is an inorganic compound of nitrogen. The "nitride" anion, N3- ion, is very elusive but compounds of nitride are numerous, although rarely naturally occuring. Some nitrides have a find applications, such as wear-resistant ...
, and halide compounds.. Two crystal modifications of uranium hydride exist: an α form that is obtained at low temperatures and a β form that is created when the formation temperature is above 250 °C.
Uranium carbides and uranium nitride
Uranium nitrides is any of a family of several ceramic materials: uranium mononitride (UN), uranium sesquinitride (U2N3) and uranium dinitride (UN2). The word nitride refers to the −3 oxidation state of the nitrogen bound to the uranium.
Uranium ...
s are both relatively inert semimetal
A semimetal is a material with a very small overlap between the bottom of the conduction band and the top of the valence band.
According to electronic band theory, solids can be classified as insulators, semiconductors, semimetals, or metals ...
lic compounds that are minimally soluble in acids, react with water, and can ignite in air
The atmosphere of Earth is the layer of gases, known collectively as air, retained by Earth's gravity that surrounds the planet and forms its planetary atmosphere. The atmosphere of Earth protects life on Earth by creating pressure allowing f ...
to form . Carbides of uranium include uranium monocarbide (U C), uranium dicarbide (), and diuranium tricarbide (). Both UC and are formed by adding carbon to molten uranium or by exposing the metal to carbon monoxide
Carbon monoxide (chemical formula CO) is a colorless, poisonous, odorless, tasteless, flammable gas that is slightly less dense than air. Carbon monoxide consists of one carbon atom and one oxygen atom connected by a triple bond. It is the simple ...
at high temperatures. Stable below 1800 °C, is prepared by subjecting a heated mixture of UC and to mechanical stress.. Uranium nitrides obtained by direct exposure of the metal to nitrogen
Nitrogen is the chemical element with the symbol N and atomic number 7. Nitrogen is a nonmetal and the lightest member of group 15 of the periodic table, often called the pnictogens. It is a common element in the universe, estimated at se ...
include uranium mononitride (UN), uranium dinitride (), and diuranium trinitride ().
Halides
 All uranium fluorides are created using
All uranium fluorides are created using uranium tetrafluoride
Uranium tetrafluoride is the inorganic compound with the formula UF4. It is a green solid with an insignificant vapor pressure and low solubility in water. Uranium in its tetravalent ( uranous) state is important in various technological process ...
(); itself is prepared by hydrofluorination of uranium dioxide. Reduction of with hydrogen at 1000 °C produces uranium trifluoride (). Under the right conditions of temperature and pressure, the reaction of solid with gaseous uranium hexafluoride
Uranium hexafluoride (), (sometimes called "hex") is an inorganic compound with the formula UF6. Uranium hexafluoride is a volatile white solid that reacts with water, releasing corrosive hydrofluoric acid. The compound reacts mildly with alumin ...
() can form the intermediate fluorides of , , and .
At room temperatures, has a high vapor pressure
Vapor pressure (or vapour pressure in English-speaking countries other than the US; see spelling differences) or equilibrium vapor pressure is defined as the pressure exerted by a vapor in thermodynamic equilibrium with its condensed phas ...
, making it useful in the gaseous diffusion
Gaseous diffusion is a technology used to produce enriched uranium by forcing gaseous uranium hexafluoride (UF6) through semipermeable membranes. This produces a slight separation between the molecules containing uranium-235 (235U) and uranium-2 ...
process to separate the rare uranium-235 from the common uranium-238 isotope. This compound can be prepared from uranium dioxide and uranium hydride by the following process:
: + 4 HF → + 2 (500 °C, endothermic)
: + → (350 °C, endothermic)
The resulting , a white solid, is highly reactive
Reactive may refer to:
*Generally, capable of having a reaction (disambiguation)
*An adjective abbreviation denoting a bowling ball coverstock made of reactive resin
*Reactivity (chemistry)
*Reactive mind
*Reactive programming
See also
*Reactanc ...
(by fluorination), easily sublimes (emitting a vapor that behaves as a nearly ideal gas
An ideal gas is a theoretical gas composed of many randomly moving point particles that are not subject to interparticle interactions. The ideal gas concept is useful because it obeys the ideal gas law, a simplified equation of state, and is a ...
), and is the most volatile compound of uranium known to exist.
One method of preparing uranium tetrachloride
Uranium tetrachloride is an inorganic compound, a salt of uranium and chlorine, with the formula UCl4. It is a hygroscopic olive-green solid. It was used in the electromagnetic isotope separation (EMIS) process of uranium enrichment. It is one o ...
() is to directly combine chlorine
Chlorine is a chemical element with the Symbol (chemistry), symbol Cl and atomic number 17. The second-lightest of the halogens, it appears between fluorine and bromine in the periodic table and its properties are mostly intermediate betwee ...
with either uranium metal or uranium hydride. The reduction of by hydrogen produces uranium trichloride () while the higher chlorides of uranium are prepared by reaction with additional chlorine. All uranium chlorides react with water and air.
Bromide
A bromide ion is the negatively charged form (Br−) of the element bromine, a member of the halogens group on the periodic table. Most bromides are colorless. Bromides have many practical roles, being found in anticonvulsants, flame-retardant ...
s and iodide
An iodide ion is the ion I−. Compounds with iodine in formal oxidation state −1 are called iodides. In everyday life, iodide is most commonly encountered as a component of iodized salt, which many governments mandate. Worldwide, iodine de ...
s of uranium are formed by direct reaction of, respectively, bromine
Bromine is a chemical element with the symbol Br and atomic number 35. It is the third-lightest element in group 17 of the periodic table (halogens) and is a volatile red-brown liquid at room temperature that evaporates readily to form a simila ...
and iodine
Iodine is a chemical element with the symbol I and atomic number 53. The heaviest of the stable halogens, it exists as a semi-lustrous, non-metallic solid at standard conditions that melts to form a deep violet liquid at , and boils to a vi ...
with uranium or by adding to those element's acids. Known examples include: , , , and . has never been prepared. Uranium oxyhalides are water-soluble and include , , , and . Stability of the oxyhalides decrease as the atomic weight
Relative atomic mass (symbol: ''A''; sometimes abbreviated RAM or r.a.m.), also known by the deprecated synonym atomic weight, is a dimensionless physical quantity defined as the ratio of the average mass of atoms of a chemical element in a giv ...
of the component halide increases.
Isotopes
Natural concentrations
Natural uranium consists of three major isotopes: uranium-238 (99.28% natural abundance), uranium-235 (0.71%), and uranium-234 (0.0054%). All three are radioactive, emitting alpha particles, with the exception that all three of these isotopes have small probabilities of undergoing spontaneous fission. There are also four other trace isotopes: uranium-239, which is formed when 238U undergoes spontaneous fission, releasing neutrons that are captured by another 238U atom; uranium-237, which is formed when 238U captures a neutron but emits two more, which then decays toneptunium-237
Neptunium (93Np) is usually considered an artificial element, although trace quantities are found in nature, so a standard atomic weight cannot be given. Like all trace or artificial elements, it has no stable isotopes. The first isotope to be sy ...
; uranium-236
Uranium-236 (236U) is an isotope of uranium that is neither fissile with thermal neutrons, nor very good fertile material, but is generally considered a nuisance and long-lived radioactive waste. It is found in spent nuclear fuel and in the re ...
, which occurs in trace quantities due to neutron capture on 235U and as a decay product of plutonium-244
Plutonium-244 (244Pu) is an isotope of plutonium that has a half-life of 80 million years. This is longer than any of the other isotopes of plutonium and longer than any other actinide isotope except for the three naturally abundant ones: uranium ...
; and finally, uranium-233, which is formed in the decay chain
In nuclear science, the decay chain refers to a series of radioactive decays of different radioactive decay products as a sequential series of transformations. It is also known as a "radioactive cascade". Most radioisotopes do not decay dire ...
of neptunium-237. It is also expected that thorium-232
Thorium-232 () is the main naturally occurring isotope of thorium, with a relative abundance of 99.98%. It has a half life of 14 billion years, which makes it the longest-lived isotope of thorium. It decays by alpha decay to radium-228; its decay ...
should be able to undergo double beta decay
In nuclear physics, double beta decay is a type of radioactive decay in which two neutrons are simultaneously transformed into two protons, or vice versa, inside an atomic nucleus. As in single beta decay, this process allows the atom to move clos ...
, which would produce uranium-232, but this has not yet been observed experimentally. The only significant deviation from the U-235 to U-238 ratio in any known natural samples occurs in Oklo
Oklo is a region near the town of Franceville, in the Haut-Ogooué province of the Central African country of Gabon. Several natural nuclear fission reactors were discovered in the uranium mines in the region in 1972.
History
Gabon was a Fren ...
, Gabon
Gabon (; ; snq, Ngabu), officially the Gabonese Republic (french: République gabonaise), is a country on the west coast of Central Africa. Located on the equator, it is bordered by Equatorial Guinea to the northwest, Cameroon to the nort ...
where natural nuclear fission reactor
A natural nuclear fission reactor is a uranium deposit where self-sustaining nuclear chain reactions occur. The conditions under which a natural nuclear reactor could exist had been predicted in 1956 by Japanese American chemist Paul Kuroda. ...
s consumed some of the U-235 some two billion years ago when the ratio of U-235 to U-238 was more akin to that of low enriched uranium
Enriched uranium is a type of uranium in which the percent composition of uranium-235 (written 235U) has been increased through the process of isotope separation. Naturally occurring uranium is composed of three major isotopes: uranium-238 (238 ...
allowing regular ("light") water to act as a neutron moderator akin to the process in humanmade light water reactor
The light-water reactor (LWR) is a type of thermal-neutron reactor that uses normal water, as opposed to heavy water, as both its coolant and neutron moderator; furthermore a solid form of fissile elements is used as fuel. Thermal-neutron react ...
s. The existence of such natural fission reactors which had been theoretically predicted beforehand was proven as the slight deviation of U-235 concentration from the expected values were discovered during uranium enrichment
Enriched uranium is a type of uranium in which the percent composition of uranium-235 (written 235U) has been increased through the process of isotope separation. Naturally occurring uranium is composed of three major isotopes: uranium-238 (238 ...
in France. Subsequent investigations to rule out any nefarious human action (such as stealing of U-235) confirmed the theory by finding isotope ratios of common fission products (or rather their stable daughter nuclides) in line with the values expected for fission but deviating from the values expected for non-fission derived samples of those elements.
Uranium-238 is the most stable isotope of uranium, with a half-life of about 4.468 years, roughly the age of the Earth. Uranium-235 has a half-life of about 7.13 years, and uranium-234 has a half-life of about 2.48 years.. Uranium-238 is usually an alpha emitter (occasionally, it undergoes spontaneous fission), decaying through the uranium series
In nuclear science, the decay chain refers to a series of radioactive decays of different radioactive decay products as a sequential series of transformations. It is also known as a "radioactive cascade". Most radioisotopes do not decay direct ...
, which has 18 members, into lead-206
Lead (82Pb) has four stable isotopes: 204Pb, 206Pb, 207Pb, 208Pb. Lead-204 is entirely a primordial nuclide and is not a radiogenic nuclide. The three isotopes lead-206, lead-207, and lead-208 represent the ends of three decay chains: the urani ...
, by a variety of different decay paths.
The decay chain of 235U, which is called the actinium series
In nuclear science, the decay chain refers to a series of radioactive decays of different radioactive decay products as a sequential series of transformations. It is also known as a "radioactive cascade". Most radioisotopes do not decay directly ...
, has 15 members and eventually decays into lead-207. The constant rates of decay in these decay series makes the comparison of the ratios of parent to daughter elements useful in radiometric dating.
Uranium-234, which is a member of the uranium series (the decay chain of uranium-238), decays to lead-206 through a series of relatively short-lived isotopes.
Uranium-233 is made from thorium-232
Thorium-232 () is the main naturally occurring isotope of thorium, with a relative abundance of 99.98%. It has a half life of 14 billion years, which makes it the longest-lived isotope of thorium. It decays by alpha decay to radium-228; its decay ...
by neutron bombardment, usually in a nuclear reactor, and 233U is also fissile. Its decay chain forms part of the neptunium series
In nuclear science, the decay chain refers to a series of radioactive decays of different radioactive decay products as a sequential series of transformations. It is also known as a "radioactive cascade". Most radioisotopes do not decay directly ...
and ends at bismuth-209
Bismuth-209 (209Bi) is the isotope of bismuth with the longest known half-life of any radioisotope that undergoes α-decay (alpha decay). It has 83 protons and a magic number of 126 neutrons, and an atomic mass of 208.9803987 amu (atomic mass un ...
and thallium
Thallium is a chemical element with the Symbol (chemistry), symbol Tl and atomic number 81. It is a gray post-transition metal that is not found free in nature. When isolated, thallium resembles tin, but discolors when exposed to air. Chemists W ...
-205.
Uranium-235 is important for both nuclear reactor
A nuclear reactor is a device used to initiate and control a fission nuclear chain reaction or nuclear fusion reactions. Nuclear reactors are used at nuclear power plants for electricity generation and in nuclear marine propulsion. Heat fr ...
s and nuclear weapon
A nuclear weapon is an explosive device that derives its destructive force from nuclear reactions, either fission (fission bomb) or a combination of fission and fusion reactions ( thermonuclear bomb), producing a nuclear explosion. Both bom ...
s, because it is the only uranium isotope existing in nature on Earth in any significant amount that is fissile. This means that it can be split into two or three fragments ( fission products) by thermal neutrons.
Uranium-238 is not fissile, but is a fertile isotope, because after neutron activation
Neutron activation is the process in which neutron radiation induces radioactivity in materials, and occurs when atomic nuclei capture free neutrons, becoming heavier and entering excited states. The excited nucleus decays immediately by emit ...
it can be converted to plutonium-239, another fissile isotope. Indeed, the 238U nucleus can absorb one neutron to produce the radioactive isotope uranium-239
Uranium (92U) is a naturally occurring radioactive element that has no stable isotope. It has two primordial isotopes, uranium-238 and uranium-235, that have long half-lives and are found in appreciable quantity in the Earth's crust. The decay ...
. 239U decays by beta emission
In nuclear physics, beta decay (β-decay) is a type of radioactive decay in which a beta particle (fast energetic electron or positron) is emitted from an atomic nucleus, transforming the original nuclide to an isobar of that nuclide. For exam ...
to neptunium
Neptunium is a chemical element with the symbol Np and atomic number 93. A radioactive actinide metal, neptunium is the first transuranic element. Its position in the periodic table just after uranium, named after the planet Uranus, led to it bein ...
-239, also a beta-emitter, that decays in its turn, within a few days into plutonium-239. 239Pu was used as fissile material in the first atomic bomb detonated in the "Trinity test" on 15 July 1945 in New Mexico
)
, population_demonym = New Mexican ( es, Neomexicano, Neomejicano, Nuevo Mexicano)
, seat = Santa Fe
, LargestCity = Albuquerque
, LargestMetro = Tiguex
, OfficialLang = None
, Languages = English, Spanish ( New Mexican), Navajo, Ker ...
.
Enrichment
 In nature, uranium is found as uranium-238 (99.2742%) and uranium-235 (0.7204%).
In nature, uranium is found as uranium-238 (99.2742%) and uranium-235 (0.7204%). Isotope separation
Isotope separation is the process of concentrating specific isotopes of a chemical element by removing other isotopes. The use of the nuclides produced is varied. The largest variety is used in research (e.g. in chemistry where atoms of "marker" n ...
concentrates (enriches) the fissionable uranium-235 for nuclear weapons and most nuclear power plants, except for gas cooled reactor
A gas-cooled reactor (GCR) is a nuclear reactor that uses graphite as a neutron moderator and a gas (carbon dioxide or helium in extant designs) as coolant. Although there are many other types of reactor cooled by gas, the terms ''GCR'' and to a ...
s and pressurised heavy water reactor
A pressurized heavy-water reactor (PHWR) is a nuclear reactor that uses heavy water ( deuterium oxide D2O) as its coolant and neutron moderator. PHWRs frequently use natural uranium as fuel, but sometimes also use very low enriched uranium. The ...
s. Most neutrons released by a fissioning atom of uranium-235 must impact other uranium-235 atoms to sustain the nuclear chain reaction. The concentration and amount of uranium-235 needed to achieve this is called a 'critical mass
In nuclear engineering, a critical mass is the smallest amount of fissile material needed for a sustained nuclear chain reaction. The critical mass of a fissionable material depends upon its nuclear properties (specifically, its nuclear fi ...
'.
To be considered 'enriched', the uranium-235 fraction should be between 3% and 5%. This process produces huge quantities of uranium that is depleted of uranium-235 and with a correspondingly increased fraction of uranium-238, called depleted uranium or 'DU'. To be considered 'depleted', the uranium-235 isotope concentration should be no more than 0.3%. The price of uranium has risen since 2001, so enrichment tailings containing more than 0.35% uranium-235 are being considered for re-enrichment, driving the price of depleted uranium hexafluoride above $130 per kilogram in July 2007 from $5 in 2001.
The gas centrifuge
A gas centrifuge is a device that performs isotope separation of gases. A centrifuge relies on the principles of centrifugal force accelerating molecules so that particles of different masses are physically separated in a gradient along the radiu ...
process, where gaseous uranium hexafluoride
Uranium hexafluoride (), (sometimes called "hex") is an inorganic compound with the formula UF6. Uranium hexafluoride is a volatile white solid that reacts with water, releasing corrosive hydrofluoric acid. The compound reacts mildly with alumin ...
() is separated by the difference in molecular weight between 235UF6 and 238UF6 using high-speed centrifuge
A centrifuge is a device that uses centrifugal force to separate various components of a fluid. This is achieved by spinning the fluid at high speed within a container, thereby separating fluids of different densities (e.g. cream from milk) or ...
s, is the cheapest and leading enrichment process.. The gaseous diffusion
Gaseous diffusion is a technology used to produce enriched uranium by forcing gaseous uranium hexafluoride (UF6) through semipermeable membranes. This produces a slight separation between the molecules containing uranium-235 (235U) and uranium-2 ...
process had been the leading method for enrichment and was used in the Manhattan Project. In this process, uranium hexafluoride is repeatedly diffused
Molecular diffusion, often simply called diffusion, is the thermal motion of all (liquid or gas) particles at temperatures above absolute zero. The rate of this movement is a function of temperature, viscosity of the fluid and the size (mass) of ...
through a silver
Silver is a chemical element with the symbol Ag (from the Latin ', derived from the Proto-Indo-European ''h₂erǵ'': "shiny" or "white") and atomic number 47. A soft, white, lustrous transition metal, it exhibits the highest electrical ...
-zinc
Zinc is a chemical element with the symbol Zn and atomic number 30. Zinc is a slightly brittle metal at room temperature and has a shiny-greyish appearance when oxidation is removed. It is the first element in group 12 (IIB) of the periodi ...
membrane, and the different isotopes of uranium are separated by diffusion rate (since uranium-238 is heavier it diffuses slightly slower than uranium-235). The molecular laser isotope separation
Molecular laser isotope separation (MLIS) is a method of isotope separation, where specially tuned lasers are used to separate isotopes of uranium using selective ionization of hyperfine transitions of uranium hexafluoride molecules. It is simila ...
method employs a laser
A laser is a device that emits light through a process of optical amplification based on the stimulated emission of electromagnetic radiation. The word "laser" is an acronym for "light amplification by stimulated emission of radiation". The fir ...
beam of precise energy to sever the bond between uranium-235 and fluorine. This leaves uranium-238 bonded to fluorine and allows uranium-235 metal to precipitate from the solution. An alternative laser method of enrichment is known as atomic vapor laser isotope separation
Atomic vapor laser isotope separation, or AVLIS, is a method by which specially tuned lasers are used to separate isotopes of uranium using selective ionization of hyperfine transitions. A similar technology, using molecules instead of atoms, is ...
(AVLIS) and employs visible tunable laser
A tunable laser is a laser whose wavelength of operation can be altered in a controlled manner. While all laser gain media allow small shifts in output wavelength, only a few types of lasers allow continuous tuning over a significant wavelength ran ...
s such as dye laser
A dye laser is a laser that uses an organic dye as the lasing medium, usually as a liquid solution. Compared to gases and most solid state lasing media, a dye can usually be used for a much wider range of wavelengths, often spanning 50 to 100 ...
s. Another method used is liquid thermal diffusion.
Human exposure
A person can be exposed to uranium (or its radioactive daughters, such asradon
Radon is a chemical element with the symbol Rn and atomic number 86. It is a radioactive, colourless, odourless, tasteless noble gas. It occurs naturally in minute quantities as an intermediate step in the normal radioactive decay chains through ...
) by inhaling dust in air or by ingesting contaminated water and food. The amount of uranium in air is usually very small; however, people who work in factories that process phosphate
In chemistry, a phosphate is an anion, salt, functional group or ester derived from a phosphoric acid. It most commonly means orthophosphate, a derivative of orthophosphoric acid .
The phosphate or orthophosphate ion is derived from phosph ...
fertilizer
A fertilizer (American English) or fertiliser (British English; see spelling differences) is any material of natural or synthetic origin that is applied to soil or to plant tissues to supply plant nutrients. Fertilizers may be distinct from ...
s, live near government facilities that made or tested nuclear weapons, live or work near a modern battlefield where depleted uranium weapons
A weapon, arm or armament is any implement or device that can be used to deter, threaten, inflict physical damage, harm, or kill. Weapons are used to increase the efficacy and efficiency of activities such as hunting, crime, law enforcement, s ...
have been used, or live or work near a coal
Coal is a combustible black or brownish-black sedimentary rock, formed as rock strata called coal seams. Coal is mostly carbon with variable amounts of other elements, chiefly hydrogen, sulfur, oxygen, and nitrogen.
Coal is formed when ...
-fired power plant, facilities that mine or process uranium ore, or enrich uranium for reactor fuel, may have increased exposure to uranium. Houses or structures that are over uranium deposits (either natural or man-made slag deposits) may have an increased incidence of exposure to radon gas. The Occupational Safety and Health Administration
The Occupational Safety and Health Administration'' (OSHA ) is a large regulatory agency of the United States Department of Labor that originally had federal visitorial powers to inspect and examine workplaces. Congress established the agenc ...
(OSHA) has set the permissible exposure limit
The permissible exposure limit (PEL or OSHA PEL) is a legal limit in the United States for exposure of an employee to a chemical substance or physical agent such as high level noise. Permissible exposure limits are established by the Occupational ...
for uranium exposure in the workplace as 0.25 mg/m3 over an 8-hour workday. The National Institute for Occupational Safety and Health
The National Institute for Occupational Safety and Health (NIOSH, ) is the United States federal agency responsible for conducting research and making recommendations for the prevention of work-related injury and illness. NIOSH is part of the C ...
(NIOSH) has set a recommended exposure limit
A recommended exposure limit (REL) is an occupational exposure limit that has been recommended by the United States National Institute for Occupational Safety and Health. The REL is a level that NIOSH believes would be protective of worker safet ...
(REL) of 0.2 mg/m3 over an 8-hour workday and a short-term limit of 0.6 mg/m3. At levels of 10 mg/m3, uranium is immediately dangerous to life and health
The term immediately dangerous to life or health (IDLH) is defined by the US National Institute for Occupational Safety and Health (NIOSH) as exposure to airborne contaminants that is "likely to cause death or immediate or delayed permanent advers ...
.
Most ingested uranium is excreted during digestion
Digestion is the breakdown of large insoluble food molecules into small water-soluble food molecules so that they can be absorbed into the watery blood plasma. In certain organisms, these smaller substances are absorbed through the small intest ...
. Only 0.5% is absorbed when insoluble forms of uranium, such as its oxide, are ingested, whereas absorption of the more soluble uranyl ion can be up to 5%. However, soluble uranium compounds tend to quickly pass through the body, whereas insoluble uranium compounds, especially when inhaled by way of dust into the lung
The lungs are the primary organs of the respiratory system in humans and most other animals, including some snails and a small number of fish. In mammals and most other vertebrates, two lungs are located near the backbone on either side of t ...
s, pose a more serious exposure hazard. After entering the bloodstream, the absorbed uranium tends to bioaccumulate
Bioaccumulation is the gradual accumulation of substances, such as pesticides or other chemicals, in an organism. Bioaccumulation occurs when an organism absorbs a substance at a rate faster than that at which the substance is lost or eliminated ...
and stay for many years in bone
A bone is a Stiffness, rigid Organ (biology), organ that constitutes part of the skeleton in most vertebrate animals. Bones protect the various other organs of the body, produce red blood cell, red and white blood cells, store minerals, provid ...
tissue because of uranium's affinity for phosphates. Uranium is not absorbed through the skin, and alpha particles released by uranium cannot penetrate the skin.
Incorporated uranium becomes uranyl ions, which accumulate in bone, liver, kidney, and reproductive tissues. Uranium can be decontaminated from steel surfaces and aquifer
An aquifer is an underground layer of water-bearing, permeable rock, rock fractures, or unconsolidated materials ( gravel, sand, or silt). Groundwater from aquifers can be extracted using a water well. Aquifers vary greatly in their characteris ...
s.
Effects and precautions
Normal functioning of thekidney
The kidneys are two reddish-brown bean-shaped organs found in vertebrates. They are located on the left and right in the retroperitoneal space, and in adult humans are about in length. They receive blood from the paired renal arteries; blood ...
, brain
A brain is an organ that serves as the center of the nervous system in all vertebrate and most invertebrate animals. It is located in the head, usually close to the sensory organs for senses such as vision. It is the most complex organ in a v ...
, liver
The liver is a major Organ (anatomy), organ only found in vertebrates which performs many essential biological functions such as detoxification of the organism, and the Protein biosynthesis, synthesis of proteins and biochemicals necessary for ...
, heart
The heart is a muscular organ in most animals. This organ pumps blood through the blood vessels of the circulatory system. The pumped blood carries oxygen and nutrients to the body, while carrying metabolic waste such as carbon dioxide t ...
, and other systems can be affected by uranium exposure, because, besides being weakly radioactive, uranium is a toxic metal
Metal toxicity or metal poisoning is the toxic effect of certain metals in certain forms and doses on life. Some metals are toxic when they form poisonous soluble compounds. Certain metals have no biological role, i.e. are not essential minerals, o ...
. Uranium is also a reproductive toxicant. Radiological effects are generally local because alpha radiation, the primary form of 238U decay, has a very short range, and will not penetrate skin. Alpha radiation from inhaled uranium has been demonstrated to cause lung cancer in exposed nuclear workers. Uranyl
The uranyl ion is an oxycation of uranium in the oxidation state +6, with the chemical formula . It has a linear structure with short U–O bonds, indicative of the presence of multiple bonds between uranium and oxygen. Four or more ligands may ...
() ions, such as from uranium trioxide
Uranium trioxide (UO3), also called uranyl oxide, uranium(VI) oxide, and uranic oxide, is the hexavalent oxide of uranium. The solid may be obtained by heating uranyl nitrate to 400 °C. Its most commonly encountered polymorph, γ-UO3, is a ...
or uranyl nitrate
Uranyl nitrate is a water-soluble yellow uranium salt with the formula . The hexa-, tri-, and dihydrates are known. The compound is mainly of interest because it is an intermediate in the preparation of nuclear fuels.
Uranyl nitrate can be prepa ...
and other hexavalent uranium compounds, have been shown to cause birth defects and immune system damage in laboratory animals. While the CDC has published one study that no human cancer
Cancer is a group of diseases involving abnormal cell growth with the potential to invade or spread to other parts of the body. These contrast with benign tumors, which do not spread. Possible signs and symptoms include a lump, abnormal b ...
has been seen as a result of exposure to natural or depleted uranium, exposure to uranium and its decay products, especially radon
Radon is a chemical element with the symbol Rn and atomic number 86. It is a radioactive, colourless, odourless, tasteless noble gas. It occurs naturally in minute quantities as an intermediate step in the normal radioactive decay chains through ...
, are widely known and significant health threats. Exposure to strontium-90
Strontium-90 () is a radioactive isotope of strontium produced by nuclear fission, with a half-life of 28.8 years. It undergoes β− decay into yttrium-90, with a decay energy of 0.546 MeV. Strontium-90 has applications in medicine and ...
, iodine-131
Iodine-131 (131I, I-131) is an important radioisotope of iodine discovered by Glenn Seaborg and John Livingood in 1938 at the University of California, Berkeley. It has a radioactive decay half-life of about eight days. It is associated with nu ...
, and other fission products is unrelated to uranium exposure, but may result from medical procedures or exposure to spent reactor fuel or fallout from nuclear weapons.
Although accidental inhalation exposure to a high concentration of uranium hexafluoride
Uranium hexafluoride (), (sometimes called "hex") is an inorganic compound with the formula UF6. Uranium hexafluoride is a volatile white solid that reacts with water, releasing corrosive hydrofluoric acid. The compound reacts mildly with alumin ...
has
resulted in human fatalities, those deaths were associated with the generation of highly toxic hydrofluoric acid and uranyl fluoride
Uranyl fluoride is the inorganic compound with the formula UO2F2. As shown by x-ray crystallography, the uranyl (UO22+) centers are complemented by six fluoride ligands.
This salt is very soluble in water as well as hygroscopic
Hygroscopy is t ...
rather than with uranium itself. Finely divided uranium metal presents a fire hazard because uranium is pyrophoric
A substance is pyrophoric (from grc-gre, πυροφόρος, , 'fire-bearing') if it ignites spontaneously in air at or below (for gases) or within 5 minutes after coming into contact with air (for liquids and solids). Examples are organolit ...
; small grains will ignite spontaneously in air at room temperature.
Uranium metal is commonly handled with gloves as a sufficient precaution. Uranium concentrate is handled and contained so as to ensure that people do not inhale or ingest it.
See also
*K-65 residues K-65 residues are the very radioactive mill residues resulting from the uniquely concentrated uranium ore discovered before WW II in Katanga province (Shinkolobwe mine) of the Democratic Republic of the Congo (formerly Belgian Congo).
According t ...
* List of countries by uranium production
This contains lists of countries by uranium production. The first two lists are compiled by the World Nuclear Association, and measures uranium production by tonnes mined. The last list is compiled by TradeTech, a consulting company which specializ ...
* List of countries by uranium reserves
Uranium reserves are reserves of recoverable uranium, regardless of isotope, based on a set market price. The list given here is based on ''Uranium 2020: Resources, Production and Demand'', a joint report by the OECD Nuclear Energy Agency and th ...
* List of uranium projects
Uranium production is carried out in about 13 countries around the world, in 2017 producing a cumulative total of 59,462 tonnes of uranium (tU). The international producers were Kazakhstan (39%), Canada (22%), Australia (10%), Namibia (7.1%), Nig ...
* Lists of nuclear disasters and radioactive incidents
These are lists of nuclear disasters and radioactive incidents.
Main lists
* List of attacks on nuclear plants
* List of Chernobyl-related articles
* List of civilian nuclear accidents
* List of civilian radiation accidents
* List of ...
* Nuclear and radiation accidents and incidents
A nuclear and radiation accident is defined by the International Atomic Energy Agency (IAEA) as "an event that has led to significant consequences to people, the environment or the facility. Examples include lethal effects to individuals, lar ...
* Nuclear engineering
* Nuclear fuel cycle
The nuclear fuel cycle, also called nuclear fuel chain, is the progression of nuclear fuel through a series of differing stages. It consists of steps in the ''front end'', which are the preparation of the fuel, steps in the ''service period'' in w ...
* Nuclear physics
Nuclear physics is the field of physics that studies atomic nuclei and their constituents and interactions, in addition to the study of other forms of nuclear matter.
Nuclear physics should not be confused with atomic physics, which studies the ...
* Quintuple bond
A quintuple bond in chemistry is an unusual type of chemical bond, first reported in 2005 for a dichromium compound. Single bonds, double bonds, and triple bonds are commonplace in chemistry. Quadruple bonds are rarer but are currently known only ...
(earlier thought to be a Phi bond), in the molecule U2
* Thorium fuel cycle
The thorium fuel cycle is a nuclear fuel cycle that uses an isotope of thorium, , as the fertile material. In the reactor, is transmuted into the fissile artificial uranium isotope which is the nuclear fuel. Unlike natural uranium, natural ...
* World Uranium Hearing
The World Uranium Hearing was held in Salzburg, Austria in September 1992. Anti-nuclear speakers from all continents, including indigenous speakers and scientists, testified to the health and environmental problems of uranium mining and processing ...
Notes
References
* *External links
U.S. EPA: Radiation Information for Uranium
from
World Nuclear Association
World Nuclear Association is the international organization that promotes nuclear power and supports the companies that comprise the global nuclear industry. Its members come from all parts of the nuclear fuel cycle, including uranium mining, ur ...
Nuclear fuel data and analysis
from the U.S. Energy Information Administration
Current market price of uranium
Annotated bibliography for uranium from the Alsos Digital Library
NLM Hazardous Substances Databank—Uranium, Radioactive
Mining Uranium at Namibia's Langer Heinrich Mine
World Nuclear News
ATSDR Case Studies in Environmental Medicine: Uranium Toxicity
U.S.
Department of Health and Human Services
The United States Department of Health and Human Services (HHS) is a cabinet-level executive branch department of the U.S. federal government created to protect the health of all Americans and providing essential human services. Its motto is ...
Uranium
at ''
The Periodic Table of Videos
''Periodic Videos'' (also known as ''The Periodic Table of Videos'') is a video project and YouTube channel on chemistry. It consists of a series of videos about chemical elements and the periodic table, with additional videos on other topics i ...
'' (University of Nottingham)
{{Authority control
Chemical elements
Actinides
Nuclear fuels
Nuclear materials
Suspected male-mediated teratogens
Manhattan Project