
In
biology
Biology is the scientific study of life. It is a natural science with a broad scope but has several unifying themes that tie it together as a single, coherent field. For instance, all organisms are made up of cells that process hereditary i ...
, epigenetics is the study of stable phenotypic changes (known as ''marks'') that do not involve alterations in the
DNA sequence
DNA sequencing is the process of determining the nucleic acid sequence – the order of nucleotides in DNA. It includes any method or technology that is used to determine the order of the four bases: adenine, guanine, cytosine, and thymine. Th ...
.
The
Greek
Greek may refer to:
Greece
Anything of, from, or related to Greece, a country in Southern Europe:
*Greeks, an ethnic group.
*Greek language, a branch of the Indo-European language family.
**Proto-Greek language, the assumed last common ancestor ...
prefix ''
epi-'' ( "over, outside of, around") in ''epigenetics'' implies features that are "on top of" or "in addition to" the traditional
gene
In biology, the word gene (from , ; "...Wilhelm Johannsen coined the word gene to describe the Mendelian units of heredity..." meaning ''generation'' or ''birth'' or ''gender'') can have several different meanings. The Mendelian gene is a ba ...
tic basis for inheritance.
Epigenetics most often involves changes that affect the
regulation of gene expression, but the term can also be used to describe any heritable phenotypic change. Such effects on
cellular
Cellular may refer to:
*Cellular automaton, a model in discrete mathematics
* Cell biology, the evaluation of cells work and more
* ''Cellular'' (film), a 2004 movie
*Cellular frequencies, assigned to networks operating in cellular RF bands
*Cell ...
and
physiological
Physiology (; ) is the scientific study of functions and mechanisms in a living system. As a sub-discipline of biology, physiology focuses on how organisms, organ systems, individual organs, cells, and biomolecules carry out the chemical ...
phenotypic trait
A phenotypic trait, simply trait, or character state is a distinct variant of a phenotypic characteristic of an organism; it may be either inherited or determined environmentally, but typically occurs as a combination of the two.Lawrence, Eleano ...
s may result from external or
environmental
A biophysical environment is a biotic and abiotic surrounding of an organism or population, and consequently includes the factors that have an influence in their survival, development, and evolution. A biophysical environment can vary in scale f ...
factors, or be part of normal development.
The term also refers to the mechanism of changes: functionally relevant alterations to the
genome
In the fields of molecular biology and genetics, a genome is all the genetic information of an organism. It consists of nucleotide sequences of DNA (or RNA in RNA viruses). The nuclear genome includes protein-coding genes and non-coding ge ...
that do not involve mutation of the
nucleotide sequence
A nucleic acid sequence is a succession of bases signified by a series of a set of five different letters that indicate the order of nucleotides forming alleles within a DNA (using GACT) or RNA (GACU) molecule. By convention, sequences are usu ...
. Examples of mechanisms that produce such changes are
DNA methylation
DNA methylation is a biological process by which methyl groups are added to the DNA molecule. Methylation can change the activity of a DNA segment without changing the sequence. When located in a gene promoter, DNA methylation typically acts t ...
and
histone modification
In biology, histones are highly basic proteins abundant in lysine and arginine residues that are found in eukaryotic cell nuclei. They act as spools around which DNA winds to create structural units called nucleosomes. Nucleosomes in turn ar ...
, each of which alters how genes are expressed without altering the underlying
DNA sequence. Gene expression can be controlled through the action of
repressor protein
In molecular genetics, a repressor is a DNA- or RNA-binding protein that inhibits the expression of one or more genes by binding to the operator or associated silencers. A DNA-binding repressor blocks the attachment of RNA polymerase to the ...
s that attach to
silencer regions of the DNA. These epigenetic changes may last through
cell division
Cell division is the process by which a parent cell (biology), cell divides into two daughter cells. Cell division usually occurs as part of a larger cell cycle in which the cell grows and replicates its chromosome(s) before dividing. In eukar ...
s for the duration of the cell's life, and may also last for multiple generations, even though they do not involve changes in the underlying DNA sequence of the organism;
instead, non-genetic factors cause the organism's genes to behave (or "express themselves") differently.
One example of an epigenetic change in
eukaryotic
Eukaryotes () are organisms whose cells have a nucleus. All animals, plants, fungi, and many unicellular organisms, are Eukaryotes. They belong to the group of organisms Eukaryota or Eukarya, which is one of the three domains of life. Bacte ...
biology is the process of
cellular differentiation
Cellular differentiation is the process in which a stem cell alters from one type to a differentiated one. Usually, the cell changes to a more specialized type. Differentiation happens multiple times during the development of a multicellular ...
. During
morphogenesis
Morphogenesis (from the Greek ''morphê'' shape and ''genesis'' creation, literally "the generation of form") is the biological process that causes a cell, tissue or organism to develop its shape. It is one of three fundamental aspects of devel ...
,
totipotent Pluripotency: These are the cells that can generate into any of the three Germ layers which imply Endodermal, Mesodermal, and Ectodermal cells except tissues like the placenta.
According to Latin terms, Pluripotentia means the ability for many thin ...
stem cells
In multicellular organisms, stem cells are undifferentiated or partially differentiated cells that can differentiate into various types of cells and proliferate indefinitely to produce more of the same stem cell. They are the earliest type o ...
become the various
pluripotent Pluripotency: These are the cells that can generate into any of the three Germ layers which imply Endodermal, Mesodermal, and Ectodermal cells except tissues like the placenta.
According to Latin terms, Pluripotentia means the ability for many thin ...
cell line
An immortalised cell line is a population of cells from a multicellular organism which would normally not proliferate indefinitely but, due to mutation, have evaded normal cellular senescence and instead can keep undergoing division. The cell ...
s of the
embryo
An embryo is an initial stage of development of a multicellular organism. In organisms that reproduce sexually, embryonic development is the part of the life cycle that begins just after fertilization of the female egg cell by the male spe ...
, which in turn become fully differentiated cells. In other words, as a single fertilized
egg cell
The egg cell, or ovum (plural ova), is the female reproductive cell, or gamete, in most anisogamous organisms (organisms that reproduce sexually with a larger, female gamete and a smaller, male one). The term is used when the female gamete is ...
– the
zygote
A zygote (, ) is a eukaryotic cell formed by a fertilization event between two gametes. The zygote's genome is a combination of the DNA in each gamete, and contains all of the genetic information of a new individual organism.
In multicellula ...
– continues to
divide, the resulting daughter cells change into all the different cell types in an organism, including
neurons
A neuron, neurone, or nerve cell is an electrically excitable cell that communicates with other cells via specialized connections called synapses. The neuron is the main component of nervous tissue in all animals except sponges and placozoa. N ...
,
muscle cells
A muscle cell is also known as a myocyte when referring to either a cardiac muscle cell (cardiomyocyte), or a smooth muscle cell as these are both small cells. A skeletal muscle cell is long and threadlike with many nuclei and is called a mus ...
,
epithelium
Epithelium or epithelial tissue is one of the four basic types of animal tissue, along with connective tissue, muscle tissue and nervous tissue. It is a thin, continuous, protective layer of compactly packed cells with a little intercellul ...
,
endothelium
The endothelium is a single layer of squamous endothelial cells that line the interior surface of blood vessels and lymphatic vessels. The endothelium forms an interface between circulating blood or lymph in the lumen and the rest of the vessel ...
of
blood vessels
The blood vessels are the components of the circulatory system that transport blood throughout the human body. These vessels transport blood cells, nutrients, and oxygen to the tissues of the body. They also take waste and carbon dioxide away f ...
, etc., by activating some genes while inhibiting the expression of others.
Definitions
The term ''epigenetics'' in its contemporary usage emerged in the 1990s, but for some years has been used with somewhat variable meanings.
A definition of the concept of ''epigenetic trait'' as a "stably heritable phenotype resulting from changes in a chromosome without alterations in the DNA sequence" was formulated at a
Cold Spring Harbor meeting in 2008,
although alternate definitions that include non-heritable traits are still being used widely.
The term ''epigenesis'' has a generic meaning of "extra growth" that has been used in English since the 17th century.
Waddington's canalisation, 1940s
The hypothesis of epigenetic changes affecting the expression of
chromosome
A chromosome is a long DNA molecule with part or all of the genetic material of an organism. In most chromosomes the very long thin DNA fibers are coated with packaging proteins; in eukaryotic cells the most important of these proteins are ...
s was put forth by the Russian biologist
Nikolai Koltsov
Nikolai Konstantinovich Koltsov (russian: Николай Константинович Кольцов; July 14, 1872 – December 2, 1940) was a Russian biologist and a pioneer of modern genetics. Among his students were Nikolay Timofeeff-Ressovs ...
. From the generic meaning, and the associated adjective ''epigenetic'', British embryologist
C. H. Waddington
Conrad Hal Waddington (8 November 1905 – 26 September 1975) was a British developmental biologist, paleontologist, geneticist, embryologist and philosopher who laid the foundations for systems biology, epigenetics, and evolutionary devel ...
coined the term ''epigenetics'' in 1942 as pertaining to ''
epigenesis'', in parallel to
Valentin Haecker
Ferdinand Carl Valentin Haecker (15 September 1864 – 19 December 1927) was a German zoologist, reader at Freiburg University from 1892.
In 1900, he became professor at the University of Applied Sciences Stuttgart and in 1909 at Martin Luther ...
's 'phenogenetics' ().
[
"For the purpose of a study of inheritance, the relation between phenotypes and genotypes ..is, from a wider biological point of view, of crucial importance, since it is the kernel of the whole problem of development. Many geneticists have recognized this and attempted to discover the processes involved in the mechanism by which the genes of the genotype bring about phenotypic effects. The first step in such an enterprise is – or rather should be, since it is often omitted by those with undue respect for the powers of reason – to describe what can be seen of the developmental processes. For enquiries of this kind, the word 'phenogenetics' was coined by Haecker 918, The second and more important part of the task is to discover the causal mechanisms at work and to relate them as far as possible to what experimental embryology has already revealed of the mechanics of development. We might use the name 'epigenetics' for such studies, thus emphasizing their relation to the concepts, so strongly favourable to the classical theory of epigenesis, which have been reached by the experimental embryologists. We certainly need to remember that between genotype and phenotype, and connecting them to each other, there lies a whole complex of developmental processes. It is convenient to have a name for this complex: 'epigenotype' seems suitable."
] ''Epigenesis'' in the context of the biology of that period referred to the
differentiation of cells from their initial
totipotent Pluripotency: These are the cells that can generate into any of the three Germ layers which imply Endodermal, Mesodermal, and Ectodermal cells except tissues like the placenta.
According to Latin terms, Pluripotentia means the ability for many thin ...
state during
embryonic development
An embryo is an initial stage of development of a multicellular organism. In organisms that reproduce sexually, embryonic development is the part of the life cycle that begins just after fertilization of the female egg cell by the male sperm ...
.
When Waddington coined the term, the physical nature of
gene
In biology, the word gene (from , ; "...Wilhelm Johannsen coined the word gene to describe the Mendelian units of heredity..." meaning ''generation'' or ''birth'' or ''gender'') can have several different meanings. The Mendelian gene is a ba ...
s and their role in heredity was not known. He used it instead as a conceptual model of how genetic components might interact with their surroundings to produce a
phenotype
In genetics, the phenotype () is the set of observable characteristics or traits of an organism. The term covers the organism's morphology or physical form and structure, its developmental processes, its biochemical and physiological proper ...
; he used the phrase "
epigenetic landscape" as a metaphor for
biological development
Developmental biology is the study of the process by which animals and plants grow and develop. Developmental biology also encompasses the biology of regeneration, asexual reproduction, metamorphosis, and the growth and differentiation of stem c ...
. Waddington held that cell fates were established during development in a process he called
canalisation much as a marble rolls down to the point of
lowest local elevation.
Waddington suggested visualising increasing irreversibility of cell type differentiation as ridges rising between the valleys where the marbles (analogous to cells) are travelling.
In recent times, Waddington's notion of the epigenetic landscape has been rigorously formalized in the context of the
systems dynamics state approach to the study of cell-fate.
Cell-fate determination is predicted to exhibit certain dynamics, such as attractor-convergence (the attractor can be an equilibrium point, limit cycle or
strange attractor
In the mathematical field of dynamical systems, an attractor is a set of states toward which a system tends to evolve, for a wide variety of starting conditions of the system. System values that get close enough to the attractor values remain ...
) or oscillatory.
Contemporary
Robin Holliday
Robin Holliday (6 November 1932 – 9 April 2014) was a British molecular biologist.
Holliday described a mechanism of DNA-strand exchange that attempted to explain gene-conversion events that occur during meiosis in fungi. That model first pro ...
defined in 1990 epigenetics as "the study of the mechanisms of temporal and spatial control of gene activity during the development of complex organisms."
Thus, in its broadest sense, ''epigenetic'' can be used to describe anything other than DNA sequence that influences the development of an organism.
More recent usage of the word in biology follows stricter definitions. As defined by
Arthur Riggs and colleagues, it is "the study of
mitotically
In cell biology, mitosis () is a part of the cell cycle in which replicated chromosomes are separated into two new nuclei. Cell division by mitosis gives rise to genetically identical cells in which the total number of chromosomes is mainta ...
and/or
meiotically heritable changes in gene function that cannot be explained by changes in DNA sequence."
The term has also been used, however, to describe processes which have not been demonstrated to be heritable, such as some forms of histone modification. Consequently, there are attempts to redefine "epigenetics" in broader terms that would avoid the constraints of requiring
heritability
Heritability is a statistic used in the fields of breeding and genetics that estimates the degree of ''variation'' in a phenotypic trait in a population that is due to genetic variation between individuals in that population. The concept of h ...
. For example,
Adrian Bird
Sir Adrian Peter Bird, (born 3 July 1947) is a British geneticist and Buchanan Professor of Genetics at the University of Edinburgh. Bird has spent much of his academic career in Edinburgh, from receiving his PhD in 1970 to working at the MRC ...
defined epigenetics as "the structural adaptation of chromosomal regions so as to register, signal or perpetuate altered activity states."
This definition would be inclusive of transient modifications associated with
DNA repair
DNA repair is a collection of processes by which a cell identifies and corrects damage to the DNA molecules that encode its genome. In human cells, both normal metabolic activities and environmental factors such as radiation can cause DNA dam ...
or
cell-cycle
The cell cycle, or cell-division cycle, is the series of events that take place in a cell that cause it to divide into two daughter cells. These events include the duplication of its DNA (DNA replication) and some of its organelles, and subse ...
phases as well as stable changes maintained across multiple cell generations, but exclude others such as templating of membrane architecture and
prions
Prions are misfolded proteins that have the ability to transmit their misfolded shape onto normal variants of the same protein. They characterize several fatal and transmissible neurodegenerative diseases in humans and many other animals. It i ...
unless they impinge on chromosome function. Such redefinitions however are not universally accepted and are still subject to debate.
The
NIH
The National Institutes of Health, commonly referred to as NIH (with each letter pronounced individually), is the primary agency of the United States government responsible for biomedical and public health research. It was founded in the late ...
"Roadmap Epigenomics Project", ongoing as of 2016, uses the following definition: "For purposes of this program, epigenetics refers to both heritable changes in gene activity and
expression
Expression may refer to:
Linguistics
* Expression (linguistics), a word, phrase, or sentence
* Fixed expression, a form of words with a specific meaning
* Idiom, a type of fixed expression
* Metaphorical expression, a particular word, phrase, o ...
(in the progeny of cells or of individuals) and also stable, long-term alterations in the transcriptional potential of a cell that are not necessarily heritable."
In 2008, a consensus definition of the epigenetic trait, a "stably heritable phenotype resulting from changes in a chromosome without alterations in the DNA sequence," was made at a
Cold Spring Harbor meeting.
The similarity of the word to "genetics" has generated many parallel usages. The "
epigenome
An epigenome consists of a record of the chemical changes to the DNA and histone proteins of an organism; these changes can be passed down to an organism's offspring via transgenerational stranded epigenetic inheritance. Changes to the epigenome ...
" is a parallel to the word "
genome
In the fields of molecular biology and genetics, a genome is all the genetic information of an organism. It consists of nucleotide sequences of DNA (or RNA in RNA viruses). The nuclear genome includes protein-coding genes and non-coding ge ...
," referring to the overall epigenetic state of a cell, and
epigenomics
Epigenomics is the study of the complete set of epigenetic modifications on the genetic material of a cell, known as the epigenome. The field is analogous to genomics and proteomics, which are the study of the genome and proteome of a cell. Epigen ...
refers to global analyses of epigenetic changes across the entire genome.
The phrase "
genetic code
The genetic code is the set of rules used by living cells to translate information encoded within genetic material ( DNA or RNA sequences of nucleotide triplets, or codons) into proteins. Translation is accomplished by the ribosome, which links ...
" has also been adapted – the "
epigenetic code" has been used to describe the set of epigenetic features that create different phenotypes in different cells from the same underlying DNA sequence. Taken to its extreme, the "epigenetic code" could represent the total state of the cell, with the position of each molecule accounted for in an ''epigenomic map'', a diagrammatic representation of the gene expression, DNA methylation and histone modification status of a particular genomic region. More typically, the term is used in reference to systematic efforts to measure specific, relevant forms of epigenetic information such as the
histone code or
DNA methylation
DNA methylation is a biological process by which methyl groups are added to the DNA molecule. Methylation can change the activity of a DNA segment without changing the sequence. When located in a gene promoter, DNA methylation typically acts t ...
patterns.
Developmental psychology
In a sense somewhat unrelated to its use in any biological disciplines, the term "epigenetic" has also been used in
developmental psychology
Developmental psychology is the science, scientific study of how and why humans grow, change, and adapt across the course of their lives. Originally concerned with infants and children, the field has expanded to include adolescence, adult deve ...
to describe psychological development as the result of an ongoing, bi-directional interchange between heredity and the environment. Interactive ideas of development have been discussed in various forms and under various names throughout the 19th and 20th centuries. An early version was proposed, among the founding statements in
embryology
Embryology (from Greek ἔμβρυον, ''embryon'', "the unborn, embryo"; and -λογία, '' -logia'') is the branch of animal biology that studies the prenatal development of gametes (sex cells), fertilization, and development of embryos and ...
, by
Karl Ernst von Baer
Karl Ernst Ritter von Baer Edler von Huthorn ( – ) was a Baltic German scientist and explorer. Baer was a naturalist, biologist, geologist, meteorologist, geographer, and is considered a, or the, founding father of embryology. He was ...
and popularized by
Ernst Haeckel
Ernst Heinrich Philipp August Haeckel (; 16 February 1834 – 9 August 1919) was a German zoologist, naturalist, eugenicist, philosopher, physician, professor, marine biologist and artist. He discovered, described and named thousands of new sp ...
. A radical epigenetic view, known as physiological epigenesis, was developed by
Paul Wintrebert Paul Wintrebert (1867–1966) was a French embryologist and a theoretician of developmental biology.
He coined the term cytoskeleton (''cytosquelette'') in 1931.
He held radical epigenetic views. In his 60s, he published a trilogy in which he d ...
. Another variation, probabilistic epigenesis, was presented by
Gilbert Gottlieb
Gilbert Gottlieb (22 October 1929 – 13 July 2006) was an American psychologist.
After receiving his bachelor's and master's degrees from the University of Miami, he received his Ph.D. in the psychology - zoology program at Duke University. He ...
in 2003. This view encompasses all of the possible developing factors on an organism and how they not only influence the organism and each other but how the organism also influences its own development. Gottlieb gave an example of Rhesus monkeys where infants that did not receive typical maternal care lacked
serotonin
Serotonin () or 5-hydroxytryptamine (5-HT) is a monoamine neurotransmitter. Its biological function is complex and multifaceted, modulating mood, cognition, reward, learning, memory, and numerous physiological processes such as vomiting and vas ...
, which in turn made them more aggressive as they got older. On another note, the long-standing notion "cells that fire together, wire together" derives from
Hebbian theory
Hebbian theory is a neuroscientific theory claiming that an increase in synaptic efficacy arises from a presynaptic cell's repeated and persistent stimulation of a postsynaptic cell. It is an attempt to explain synaptic plasticity, the adaptation ...
which asserts that
synaptogenesis
Synaptogenesis is the formation of synapses between neurons in the nervous system. Although it occurs throughout a healthy person's lifespan, an explosion of synapse formation occurs during early brain development, known as exuberant synaptogenes ...
, a developmental process with great epigenetic precedence, depends on the activity of the respective synapses within a neural network. Where experience alters the excitability of neurons, increased neural activity has been linked to increased demethylation .
The developmental psychologist
Erik Erikson
Erik Homburger Erikson (born Erik Salomonsen; 15 June 1902 – 12 May 1994) was a German-American developmental psychologist and psychoanalyst known for his theory on psychological development of human beings. He coined the phrase identity cr ...
wrote of an ''epigenetic principle'' in his 1968 book
''Identity: Youth and Crisis'', encompassing the notion that we develop through an unfolding of our personality in predetermined stages, and that our environment and surrounding culture influence how we progress through these stages. This biological unfolding in relation to our socio-cultural settings is done in
stages of psychosocial development, where "progress through each stage is in part determined by our success, or lack of success, in all the previous stages."
[Boeree, C. George, (1997/2006)]
''Personality Theories, Erik Erikson''
/ref>
Although empirical studies have yielded discrepant results, epigenetic modifications are thought to be a biological mechanism for transgenerational trauma
Transgenerational trauma is the psychological and physiological effects that the trauma experienced by people has on subsequent generations in that group. The primary modes of transmission are the uterine environment during pregnancy causing epige ...
.
Molecular basis
Epigenetic changes modify the activation of certain genes, but not the genetic code sequence of DNA. The microstructure (not code) of DNA itself or the associated chromatin
Chromatin is a complex of DNA and protein found in eukaryotic cells. The primary function is to package long DNA molecules into more compact, denser structures. This prevents the strands from becoming tangled and also plays important roles in r ...
proteins may be modified, causing activation or silencing. This mechanism enables differentiated cells in a multicellular organism to express only the genes that are necessary for their own activity. Epigenetic changes are preserved when cells divide. Most epigenetic changes only occur within the course of one individual organism's lifetime; however, these epigenetic changes can be transmitted to the organism's offspring through a process called transgenerational epigenetic inheritance
Transgenerational epigenetic inheritance is the transmission of epigenetic markers from one organism to the next (i.e., from parent to child) that affects the traits of offspring without altering the primary structure of DNA (i.e. the sequence of ...
. Moreover, if gene inactivation occurs in a sperm or egg cell that results in fertilization, this epigenetic modification may also be transferred to the next generation.paramutation
In epigenetics, a paramutation is an interaction between two alleles at a single locus, whereby one allele induces a heritable change in the other allele. The change may be in the pattern of DNA methylation or histone modifications. The allele indu ...
, bookmarking
Bookmarking (also "gene bookmarking" or "mitotic bookmarking") refers to a potential mechanism of transmission of gene expression programs through cell division.
During mitosis, gene transcription is silenced and most transcription factors are r ...
, imprinting, gene silencing
Gene silencing is the regulation of gene expression in a cell to prevent the expression of a certain gene. Gene silencing can occur during either transcription or translation and is often used in research. In particular, methods used to silence ge ...
, X chromosome inactivation
X-inactivation (also called Lyonization, after English geneticist Mary Lyon) is a process by which one of the copies of the X chromosome is inactivated in therian female mammals. The inactive X chromosome is silenced by being packaged into a ...
, position effect, DNA methylation reprogramming
In biology, reprogramming refers to erasure and remodeling of epigenetic marks, such as DNA methylation, during mammalian development or in cell culture. Such control is also often associated with alternative covalent modifications of histones.
...
, transvection, maternal effect
A maternal effect is a situation where the phenotype of an organism is determined not only by the environment it experiences and its genotype, but also by the environment and genotype of its mother. In genetics, maternal effects occur when an org ...
s, the progress of carcinogenesis
Carcinogenesis, also called oncogenesis or tumorigenesis, is the formation of a cancer, whereby normal cells are transformed into cancer cells. The process is characterized by changes at the cellular, genetic, and epigenetic levels and abno ...
, many effects of teratogens, regulation of histone
In biology, histones are highly basic proteins abundant in lysine and arginine residues that are found in eukaryotic cell nuclei. They act as spools around which DNA winds to create structural units called nucleosomes. Nucleosomes in turn a ...
modifications and heterochromatin
Heterochromatin is a tightly packed form of DNA or '' condensed DNA'', which comes in multiple varieties. These varieties lie on a continue between the two extremes of constitutive heterochromatin and facultative heterochromatin. Both play a role ...
, and technical limitations affecting parthenogenesis
Parthenogenesis (; from the Greek grc, παρθένος, translit=parthénos, lit=virgin, label=none + grc, γένεσις, translit=génesis, lit=creation, label=none) is a natural form of asexual reproduction in which growth and development ...
and cloning
Cloning is the process of producing individual organisms with identical or virtually identical DNA, either by natural or artificial means. In nature, some organisms produce clones through asexual reproduction. In the field of biotechnology, cl ...
.
DNA damage
DNA damage can also cause epigenetic changes. DNA damage is very frequent, occurring on average about 60,000 times a day per cell of the human body (see DNA damage (naturally occurring)
DNA damage is an alteration in the chemical structure of DNA, such as a break in a strand of DNA, a nucleobase missing from the backbone of DNA, or a chemically changed base such as 8-OHdG. DNA damage can occur naturally or via environmental fac ...
). These damages are largely repaired, however, epigenetic changes can still remain at the site of DNA repair. In particular, a double strand break in DNA can initiate unprogrammed epigenetic gene silencing both by causing DNA methylation as well as by promoting silencing types of histone modifications (chromatin remodeling - see next section). In addition, the enzyme Parp1 (poly(ADP)-ribose polymerase) and its product poly(ADP)-ribose (PAR) accumulate at sites of DNA damage as part of the repair process. This accumulation, in turn, directs recruitment and activation of the chromatin remodeling protein, ALC1, that can cause nucleosome
A nucleosome is the basic structural unit of DNA packaging in eukaryotes. The structure of a nucleosome consists of a segment of DNA wound around eight histone proteins and resembles thread wrapped around a spool. The nucleosome is the fundamen ...
remodeling. Nucleosome remodeling has been found to cause, for instance, epigenetic silencing of DNA repair gene MLH1.benzene
Benzene is an organic chemical compound with the molecular formula C6H6. The benzene molecule is composed of six carbon atoms joined in a planar ring with one hydrogen atom attached to each. Because it contains only carbon and hydrogen atoms, ...
, hydroquinone
Hydroquinone, also known as benzene-1,4-diol or quinol, is an aromatic organic compound that is a type of phenol, a derivative of benzene, having the chemical formula C6H4(OH)2. It has two hydroxyl groups bonded to a benzene ring in a ''para'' ...
, styrene
Styrene () is an organic compound with the chemical formula C6H5CH=CH2. This derivative of benzene is a colorless oily liquid, although aged samples can appear yellowish. The compound evaporates easily and has a sweet smell, although high concen ...
, carbon tetrachloride
Carbon tetrachloride, also known by many other names (such as tetrachloromethane, also IUPAC nomenclature of inorganic chemistry, recognised by the IUPAC, carbon tet in the cleaning industry, Halon-104 in firefighting, and Refrigerant-10 in HVAC ...
and trichloroethylene
The chemical compound trichloroethylene is a halocarbon commonly used as an industrial solvent. It is a clear, colourless non-flammable liquid with a chloroform-like sweet smell. It should not be confused with the similar 1,1,1-trichloroethane, ...
, cause considerable hypomethylation of DNA, some through the activation of oxidative stress pathways.
Foods are known to alter the epigenetics of rats on different diets. Some food components epigenetically increase the levels of DNA repair enzymes such as MGMT
MGMT () is an American indie rock band formed in 2002 in Middletown, Connecticut. It was founded by multi-instrumentalists Andrew VanWyngarden and Ben Goldwasser. Alongside VanWyngarden and Goldwasser, MGMT's live lineup currently consists of ...
and MLH1 and p53
p53, also known as Tumor protein P53, cellular tumor antigen p53 (UniProt name), or transformation-related protein 53 (TRP53) is a regulatory protein that is often mutated in human cancers. The p53 proteins (originally thought to be, and often s ...
. Other food components can reduce DNA damage, such as soy isoflavones. In one study, markers for oxidative stress, such as modified nucleotides that can result from DNA damage, were decreased by a 3-week diet supplemented with soy. A decrease in oxidative DNA damage was also observed 2 h after consumption of anthocyanin
Anthocyanins (), also called anthocyans, are water-soluble vacuolar pigments that, depending on their pH, may appear red, purple, blue, or black. In 1835, the German pharmacist Ludwig Clamor Marquart gave the name Anthokyan to a chemical compo ...
-rich bilberry
Bilberries (), or sometimes European blueberries, are a primarily Eurasian species of low-growing shrubs in the genus ''Vaccinium'' (family Ericaceae), bearing edible, dark blue berries. The species most often referred to is ''Vaccinium myrtillus ...
('' Vaccinium myrtillius'' L.) pomace
Pomace ( ), or marc (; from French ''marc'' ), is the solid remains of grapes, olives, or other fruit after pressing for juice or oil. It contains the skins, pulp, seeds, and stems of the fruit.
Grape pomace has traditionally been used to prod ...
extract.
DNA repair
Damage to DNA is very common and is constantly being repaired. Epigenetic alterations can accompany DNA repair of oxidative damage or double-strand breaks. In human cells, oxidative DNA damage occurs about 10,000 times a day and DNA double-strand breaks occur about 10 to 50 times a cell cycle in somatic replicating cells (see DNA damage (naturally occurring)
DNA damage is an alteration in the chemical structure of DNA, such as a break in a strand of DNA, a nucleobase missing from the backbone of DNA, or a chemically changed base such as 8-OHdG. DNA damage can occur naturally or via environmental fac ...
). The selective advantage of DNA repair is to allow the cell to survive in the face of DNA damage. The selective advantage of epigenetic alterations that occur with DNA repair is not clear.
Repair of oxidative DNA damage can alter epigenetic markers
In the steady state (with endogenous damages occurring and being repaired), there are about 2,400 oxidatively damaged guanines that form 8-oxo-2'-deoxyguanosine
8-Oxo-2'-deoxyguanosine (8-oxo-dG) is an oxidized derivative of deoxyguanosine. 8-Oxo-dG is one of the major products of DNA oxidation. Concentrations of 8-oxo-dG within a cell are a measurement of oxidative stress.
In DNA
Steady-state leve ...
(8-OHdG) in the average mamalian cell DNA.methylated
In the chemical sciences, methylation denotes the addition of a methyl group on a substrate, or the substitution of an atom (or group) by a methyl group. Methylation is a form of alkylation, with a methyl group replacing a hydrogen atom. These ...
CpG site
The CpG sites or CG sites are regions of DNA where a cytosine nucleotide is followed by a guanine nucleotide in the linear sequence of bases along its 5' → 3' direction. CpG sites occur with high frequency in genomic regions called CpG isl ...
(a cytosine followed by guanine along its 5' → 3' direction and where the cytosine is methylated (5-mCpG)). Oxidized guanine has mispairing potential and is mutagenic.
Oxidized guanine has mispairing potential and is mutagenic.CpG site
The CpG sites or CG sites are regions of DNA where a cytosine nucleotide is followed by a guanine nucleotide in the linear sequence of bases along its 5' → 3' direction. CpG sites occur with high frequency in genomic regions called CpG isl ...
it recruits TET1 to the 8-OHdG lesion (see Figure). This allows TET1 to demethylate an adjacent methylated cytosine. Demethylation of cytosine is an epigenetic alteration.
As an example, when human mammary epithelial cells were treated with H2O2 for six hours, 8-OHdG increased about 3.5-fold in DNA and this caused about 80% demethylation of the 5-methylcytosines in the genome.CpG island
The CpG sites or CG sites are regions of DNA where a cytosine nucleotide is followed by a guanine nucleotide in the linear sequence of bases along its 5' → 3' direction. CpG sites occur with high frequency in genomic regions called CpG isl ...
was reduced (an epigenetic alteration) and this allowed about 6.5 fold increase of expression of ''BACE1'' messenger RNA.
While six-hour incubation with H2O2 causes considerable demethylation of 5-mCpG sites, shorter times of H2O2 incubation appear to promote other epigenetic alterations. Treatment of cells with H2O2 for 30 minutes causes the mismatch repair protein heterodimer MSH2-MSH6 to recruit DNA methyltransferase 1 (DNMT1) to sites of some kinds of oxidative DNA damage.HEK 293 cells
Human embryonic kidney 293 cells, also often referred to as HEK 293, HEK-293, 293 cells, or less precisely as HEK cells, are a specific immortalised cell line derived from a spontaneously miscarried or aborted fetus or human embryonic kidney cel ...
with agents causing oxidative DNA damage, (potassium bromate
Potassium bromate (KBrO3), is a bromate of potassium and takes the form of white crystals or powder. It is a strong oxidizing agent.
It is a toxic and carcinogenic compound.
Preparation
Potassium bromate is produced when bromine is passed throug ...
(KBrO3) or potassium chromate
Potassium chromate is the inorganic compound with the formula K2 CrO4. This yellow solid is the potassium salt of the chromate anion. It is a common laboratory chemical, whereas sodium chromate is important industrially.
Structure
Two crystal ...
(K2CrO4)). Base excision repair
Base excision repair (BER) is a cellular mechanism, studied in the fields of biochemistry and genetics, that repairs damaged DNA throughout the cell cycle. It is responsible primarily for removing small, non-helix-distorting base lesions from t ...
(BER) of oxidative damage occurred with the DNA repair enzyme polymerase beta localizing to oxidized guanines. Polymerase beta is the main human polymerase in short-patch BER of oxidative DNA damage. Jiang et al.DNA methyltransferase
In biochemistry, the DNA methyltransferase (DNA MTase, DNMT) family of enzymes catalyze the transfer of a methyl group to DNA. DNA methylation serves a wide variety of biological functions. All the known DNA methyltransferases use S-adenosyl m ...
protein DNMT3b to BER repair sites. They then evaluated the methylation pattern at the single nucleotide level in a small region of DNA including the promoter region and the early transcription region of the BRCA1
Breast cancer type 1 susceptibility protein is a protein that in humans is encoded by the ''BRCA1'' () gene. Orthologs are common in other vertebrate species, whereas invertebrate genomes may encode a more distantly related gene. ''BRCA1'' is a h ...
gene. Oxidative DNA damage from bromate modulated the DNA methylation pattern (caused epigenetic alterations) at CpG sites within the region of DNA studied. In untreated cells, CpGs located at −189, −134, −29, −19, +16, and +19 of the BRCA1 gene had methylated cytosines (where numbering is from the messenger RNA
In molecular biology, messenger ribonucleic acid (mRNA) is a single-stranded molecule of RNA that corresponds to the genetic sequence of a gene, and is read by a ribosome in the process of synthesizing a protein.
mRNA is created during the p ...
transcription start site, and negative numbers indicate nucleotides in the upstream promoter region). Bromate treatment-induced oxidation resulted in the loss of cytosine methylation at −189, −134, +16 and +19 while also leading to the formation of new methylation at the CpGs located at −80, −55, −21 and +8 after DNA repair was allowed.
Homologous recombinational repair alters epigenetic markers
At least four articles report the recruitment of DNA methyltransferase 1 (DNMT1) to sites of DNA double-strand breaks.CpG site
The CpG sites or CG sites are regions of DNA where a cytosine nucleotide is followed by a guanine nucleotide in the linear sequence of bases along its 5' → 3' direction. CpG sites occur with high frequency in genomic regions called CpG isl ...
s downstream of the repaired double-strand break. The other DNA strand loses methylation at about six CpG sites that were previously methylated downstream of the double-strand break, as well as losing methylation at about five CpG sites that were previously methylated upstream of the double-strand break. When the chromosome is replicated, this gives rise to one daughter chromosome that is heavily methylated downstream of the previous break site and one that is unmethylated in the region both upstream and downstream of the previous break site. With respect to the gene that was broken by the double-strand break, half of the progeny cells express that gene at a high level and in the other half of the progeny cells expression of that gene is repressed. When clones of these cells were maintained for three years, the new methylation patterns were maintained over that time period.
Non-homologous end joining can cause some epigenetic marker alterations
Non-homologous end joining
Non-homologous end joining (NHEJ) is a pathway that repairs double-strand breaks in DNA. NHEJ is referred to as "non-homologous" because the break ends are directly ligated without the need for a homologous template, in contrast to homology direc ...
(NHEJ) repair of a double-strand break can cause a small number of demethylations of pre-existing cytosine DNA methylations downstream of the repaired double-strand break.
Techniques used to study epigenetics
Epigenetic research uses a wide range of molecular biological
Molecular biology is the branch of biology that seeks to understand the molecular basis of biological activity in and between cells, including biomolecular synthesis, modification, mechanisms, and interactions. The study of chemical and physi ...
techniques to further understanding of epigenetic phenomena. These techniques include chromatin immunoprecipitation (together with its large-scale variants ChIP-on-chip
ChIP-on-chip (also known as ChIP-chip) is a technology that combines chromatin immunoprecipitation ('ChIP') with DNA microarray (''"chip"''). Like regular ChIP, ChIP-on-chip is used to investigate interactions between proteins and DNA ''in vivo' ...
and ChIP-Seq
ChIP-sequencing, also known as ChIP-seq, is a method used to analyze protein interactions with DNA. ChIP-seq combines chromatin immunoprecipitation (ChIP) with massively parallel DNA sequencing to identify the binding sites of DNA-associated prote ...
), fluorescent in situ hybridization
Fluorescence ''in situ'' hybridization (FISH) is a molecular cytogenetic technique that uses fluorescent probes that bind to only particular parts of a nucleic acid sequence with a high degree of sequence complementarity. It was developed ...
, methylation-sensitive restriction enzymes
A restriction enzyme, restriction endonuclease, REase, ENase or'' restrictase '' is an enzyme that cleaves DNA into fragments at or near specific recognition sites within molecules known as restriction sites. Restriction enzymes are one class o ...
, DNA adenine methyltransferase identification (DamID
DNA adenine methyltransferase identification, often abbreviated DamID, is a molecular biology protocol used to map the binding sites of DNA- and chromatin-binding proteins in eukaryotes. DamID identifies binding sites by expressing the proposed D ...
) and bisulfite sequencing
Bisulfite sequencing (also known as bisulphite sequencing) is the use of bisulfite treatment of DNA before routine sequencing to determine the pattern of methylation. DNA methylation was the first discovered epigenetic mark, and remains the mo ...
.bioinformatics
Bioinformatics () is an interdisciplinary field that develops methods and software tools for understanding biological data, in particular when the data sets are large and complex. As an interdisciplinary field of science, bioinformatics combi ...
methods has a role in computational epigenetics.[
]
Chromatin Immunoprecipitation
Chromatin Immunoprecipitation (ChIP) has helped bridge the gap between DNA and epigenetic interactions.
Fluorescent ''in situ'' hybridization
Fluorescent ''in situ'' hybridization (FISH) is very important to understand epigenetic mechanisms.
Methylation-sensitive restriction enzymes
Methylation sensitive restriction enzymes paired with PCR is a way to evaluate methylation in DNA - specifically the CpG sites.
Bisulfite sequencing
Bisulfite sequencing is another way to evaluate DNA methylation. Cytosine will be changed to uracil from being treated with sodium bisulfite, whereas methylated cytosines will not be affected.
Mechanisms
Epigenetic mechanisms are sensitive to environmental effects, and key participants in shaping an adult phenotype. Several types of epigenetic inheritance systems may play a role in what has become known as cell memory,
Covalent modifications
Covalent
A covalent bond is a chemical bond that involves the sharing of electrons to form electron pairs between atoms. These electron pairs are known as shared pairs or bonding pairs. The stable balance of attractive and repulsive forces between atoms ...
modification of either DNA (e.g. cytosine methylation and hydroxymethylation) or of histone proteins (e.g. lysine acetylation, lysine and arginine methylation, serine and threonine phosphorylation, and lysine ubiquitination and sumoylation) play central roles in many types of epigenetic inheritance. Therefore, the word "epigenetics" is sometimes used as a synonym for these processes. However, this can be misleading. Chromatin remodeling is not always inherited, and not all epigenetic inheritance involves chromatin remodeling. Because the
Because the phenotype
In genetics, the phenotype () is the set of observable characteristics or traits of an organism. The term covers the organism's morphology or physical form and structure, its developmental processes, its biochemical and physiological proper ...
of a cell or individual is affected by which of its genes are transcribed, heritable transcription states can give rise to epigenetic effects. There are several layers of regulation of gene expression
Gene expression is the process by which information from a gene is used in the synthesis of a functional gene product that enables it to produce end products, protein or non-coding RNA, and ultimately affect a phenotype, as the final effect. The ...
. One way that genes are regulated is through the remodeling of chromatin. Chromatin is the complex of DNA and the histone
In biology, histones are highly basic proteins abundant in lysine and arginine residues that are found in eukaryotic cell nuclei. They act as spools around which DNA winds to create structural units called nucleosomes. Nucleosomes in turn a ...
proteins with which it associates. If the way that DNA is wrapped around the histones changes, gene expression can change as well. Chromatin remodeling is accomplished through two main mechanisms:
# The first way is post translational modification
Post-translational modification (PTM) is the covalent and generally enzymatic modification of proteins following protein biosynthesis. This process occurs in the endoplasmic reticulum and the golgi apparatus. Proteins are synthesized by ribosome ...
of the amino acids that make up histone proteins. Histone proteins are made up of long chains of amino acids. If the amino acids that are in the chain are changed, the shape of the histone might be modified. DNA is not completely unwound during replication. It is possible, then, that the modified histones may be carried into each new copy of the DNA. Once there, these histones may act as templates, initiating the surrounding new histones to be shaped in the new manner. By altering the shape of the histones around them, these modified histones would ensure that a lineage-specific transcription program is maintained after cell division.
# The second way is the addition of methyl groups to the DNA, mostly at CpG site
The CpG sites or CG sites are regions of DNA where a cytosine nucleotide is followed by a guanine nucleotide in the linear sequence of bases along its 5' → 3' direction. CpG sites occur with high frequency in genomic regions called CpG isl ...
s, to convert cytosine
Cytosine () ( symbol C or Cyt) is one of the four nucleobases found in DNA and RNA, along with adenine, guanine, and thymine (uracil in RNA). It is a pyrimidine derivative, with a heterocyclic aromatic ring and two substituents attached (an am ...
to 5-methylcytosine. 5-Methylcytosine performs much like a regular cytosine, pairing with a guanine in double-stranded DNA. However, when methylated cytosines are present in CpG site
The CpG sites or CG sites are regions of DNA where a cytosine nucleotide is followed by a guanine nucleotide in the linear sequence of bases along its 5' → 3' direction. CpG sites occur with high frequency in genomic regions called CpG isl ...
s in the promoter and enhancer regions of genes, the genes are often repressed.CpG site
The CpG sites or CG sites are regions of DNA where a cytosine nucleotide is followed by a guanine nucleotide in the linear sequence of bases along its 5' → 3' direction. CpG sites occur with high frequency in genomic regions called CpG isl ...
s in the gene body (in the coding region
The coding region of a gene, also known as the coding sequence (CDS), is the portion of a gene's DNA or RNA that codes for protein. Studying the length, composition, regulation, splicing, structures, and functions of coding regions compared to no ...
excluding the transcription start site) expression of the gene is often enhanced. Transcription of a gene usually depends on a transcription factor
In molecular biology, a transcription factor (TF) (or sequence-specific DNA-binding factor) is a protein that controls the rate of transcription of genetic information from DNA to messenger RNA, by binding to a specific DNA sequence. The fu ...
binding to a (10 base or less) recognition sequence
A recognition sequence is a DNA sequence to which a structural motif of a DNA-binding domain exhibits binding specificity. Recognition sequences are palindromes.
The transcription factor Sp1 for example, binds the sequences 5'-(G/T)GGGCGG(G/A)(G/ ...
at the enhancer that interacts with the promoter region of that gene ( Gene expression # Enhancers, transcription factors, mediator complex and DNA loops in mammalian transcription).methyl-CpG-binding domain
The Methyl-CpG-binding domain (MBD) in molecular biology binds to DNA that contains one or more symmetrically methylated CpGs. MBD has negligible non-specific affinity for unmethylated DNA. In vitro foot-printing with the chromosomal protein MeC ...
(MBD) proteins. All MBDs interact with nucleosome
A nucleosome is the basic structural unit of DNA packaging in eukaryotes. The structure of a nucleosome consists of a segment of DNA wound around eight histone proteins and resembles thread wrapped around a spool. The nucleosome is the fundamen ...
remodeling and histone deacetylase complexes, which leads to gene silencing. In addition, another covalent modification involving methylated cytosine is its demethylation Demethylation is the chemical process resulting in the removal of a methyl group (CH3) from a molecule. A common way of demethylation is the replacement of a methyl group by a hydrogen atom, resulting in a net loss of one carbon and two hydrogen at ...
by TET enzymes
The TET enzymes are a family of ten-eleven translocation (TET) methylcytosine dioxygenases. They are instrumental in DNA demethylation. 5-Methylcytosine (see first Figure) is a methylated form of the DNA base cytosine (C) that often regulates ge ...
. Hundreds of such demethylations occur, for instance, during learning and memory forming events in neuron
A neuron, neurone, or nerve cell is an electrically excitable cell that communicates with other cells via specialized connections called synapses. The neuron is the main component of nervous tissue in all animals except sponges and placozoa. N ...
s.CpG site
The CpG sites or CG sites are regions of DNA where a cytosine nucleotide is followed by a guanine nucleotide in the linear sequence of bases along its 5' → 3' direction. CpG sites occur with high frequency in genomic regions called CpG isl ...
, can also associate with H3K9 methyltransferase
Methyltransferases are a large group of enzymes that all methylate their substrates but can be split into several subclasses based on their structural features. The most common class of methyltransferases is class I, all of which contain a Ross ...
activity to methylate histone 3 at lysine 9. On the other hand, DNA maintenance methylation by DNMT1
DNA (cytosine-5)-methyltransferase 1 is an enzyme that catalyzes the transfer of methyl groups to specific CpG structures in DNA, a process called DNA methylation. In humans, it is encoded by the ''DNMT1'' gene. DNMT1 forms part of the family of ...
appears to partly rely on recognition of histone methylation on the nucleosome present at the DNA site to carry out cytosine methylation on newly synthesized DNA.DNMT3A
DNA (cytosine-5)-methyltransferase 3A is an enzyme that catalyzes the transfer of methyl groups to specific CpG structures in DNA, a process called DNA methylation. The enzyme is encoded in humans by the DNMT3A gene.
This enzyme is responsible f ...
and DNMT3B
DNA (cytosine-5)-methyltransferase 3 beta, is an enzyme that in humans in encoded by the DNMT3B gene. Mutation in this gene are associated with immunodeficiency, centromere instability and facial anomalies syndrome.
Function
CpG methylation i ...
and histone methylation so that there is a correlation between the genome-wide distribution of DNA methylation and histone methylation.DNMT1
DNA (cytosine-5)-methyltransferase 1 is an enzyme that catalyzes the transfer of methyl groups to specific CpG structures in DNA, a process called DNA methylation. In humans, it is encoded by the ''DNMT1'' gene. DNMT1 forms part of the family of ...
) that have a higher affinity for 5-methylcytosine than for cytosine. If this enzyme reaches a "hemimethylated" portion of DNA (where 5-methylcytosine is in only one of the two DNA strands) the enzyme will methylate the other half. However, it is now known that DNMT1 physically interacts with the protein UHRF1
Ubiquitin-like, containing PHD and RING finger domains, 1, also known as UHRF1, is a protein which in humans is encoded by the ''UHRF1'' gene.
Function
This gene encodes a member of a subfamily of RING finger domain, RING-finger type E3 ubiqui ...
. UHRF1 has been recently recognized as essential for DNMT1-mediated maintenance of DNA methylation. UHRF1 is the protein that specifically recognizes hemi-methylated DNA, therefore bringing DNMT1 to its substrate to maintain DNA methylation. Although histone modifications occur throughout the entire sequence, the unstructured N-termini of histones (called histone tails) are particularly highly modified. These modifications include
Although histone modifications occur throughout the entire sequence, the unstructured N-termini of histones (called histone tails) are particularly highly modified. These modifications include acetylation
:
In organic chemistry, acetylation is an organic esterification reaction with acetic acid. It introduces an acetyl group into a chemical compound. Such compounds are termed ''acetate esters'' or simply '' acetates''. Deacetylation is the oppo ...
, methylation
In the chemical sciences, methylation denotes the addition of a methyl group on a substrate, or the substitution of an atom (or group) by a methyl group. Methylation is a form of alkylation, with a methyl group replacing a hydrogen atom. These t ...
, ubiquitylation
Ubiquitin is a small (8.6 kDa) regulatory protein found in most tissues of eukaryotic organisms, i.e., it is found ''ubiquitously''. It was discovered in 1975 by Gideon Goldstein and further characterized throughout the late 1970s and 1980s. Fou ...
, phosphorylation
In chemistry, phosphorylation is the attachment of a phosphate group to a molecule or an ion. This process and its inverse, dephosphorylation, are common in biology and could be driven by natural selection. Text was copied from this source, wh ...
, sumoylation
In molecular biology, SUMO (Small Ubiquitin-like Modifier) proteins are a family of small proteins that are covalently attached to and detached from other proteins in cells to modify their function. This process is called SUMOylation (sometimes w ...
, ribosylation and citrullination. Acetylation is the most highly studied of these modifications. For example, acetylation of the K14 and K9 lysine
Lysine (symbol Lys or K) is an α-amino acid that is a precursor to many proteins. It contains an α-amino group (which is in the protonated form under biological conditions), an α-carboxylic acid group (which is in the deprotonated −C ...
s of the tail of histone H3 by histone acetyltransferase enzymes (HATs) is generally related to transcriptional competence (see Figure).
One mode of thinking is that this tendency of acetylation to be associated with "active" transcription is biophysical in nature. Because it normally has a positively charged nitrogen at its end, lysine can bind the negatively charged phosphates of the DNA backbone. The acetylation event converts the positively charged amine group on the side chain into a neutral amide linkage. This removes the positive charge, thus loosening the DNA from the histone. When this occurs, complexes like SWI/SNF
In molecular biology, SWI/SNF (SWItch/Sucrose Non-Fermentable), is a subfamily of ATP-dependent chromatin remodeling complexes, which is found in eukaryotes. In other words, it is a group of proteins that associate to remodel the way DNA is pack ...
and other transcriptional factors can bind to the DNA and allow transcription to occur. This is the "cis" model of the epigenetic function. In other words, changes to the histone tails have a direct effect on the DNA itself.
Another model of epigenetic function is the "trans" model. In this model, changes to the histone tails act indirectly on the DNA. For example, lysine acetylation may create a binding site for chromatin-modifying enzymes (or transcription machinery as well). This chromatin remodeler can then cause changes to the state of the chromatin. Indeed, a bromodomain – a protein domain that specifically binds acetyl-lysine – is found in many enzymes that help activate transcription, including the SWI/SNF
In molecular biology, SWI/SNF (SWItch/Sucrose Non-Fermentable), is a subfamily of ATP-dependent chromatin remodeling complexes, which is found in eukaryotes. In other words, it is a group of proteins that associate to remodel the way DNA is pack ...
complex. It may be that acetylation acts in this and the previous way to aid in transcriptional activation.
The idea that modifications act as docking modules for related factors is borne out by histone methylation as well. Methylation of lysine 9 of histone H3 has long been associated with constitutively transcriptionally silent chromatin (constitutive heterochromatin
Heterochromatin is a tightly packed form of DNA or '' condensed DNA'', which comes in multiple varieties. These varieties lie on a continue between the two extremes of constitutive heterochromatin and facultative heterochromatin. Both play a role ...
) (see bottom Figure). It has been determined that a chromodomain (a domain that specifically binds methyl-lysine) in the transcriptionally repressive protein HP1 recruits HP1 to K9 methylated regions. One example that seems to refute this biophysical model for methylation is that tri-methylation of histone H3 at lysine 4 is strongly associated with (and required for full) transcriptional activation (see top Figure). Tri-methylation, in this case, would introduce a fixed positive charge on the tail.
It has been shown that the histone lysine methyltransferase (KMT) is responsible for this methylation activity in the pattern of histones H3 & H4. This enzyme utilizes a catalytically active site called the SET domain (Suppressor of variegation, Enhancer of Zeste, Trithorax). The SET domain is a 130-amino acid sequence involved in modulating gene activities. This domain has been demonstrated to bind to the histone tail and causes the methylation of the histone.nucleosome
A nucleosome is the basic structural unit of DNA packaging in eukaryotes. The structure of a nucleosome consists of a segment of DNA wound around eight histone proteins and resembles thread wrapped around a spool. The nucleosome is the fundamen ...
. The idea that multiple dynamic modifications regulate gene transcription in a systematic and reproducible way is called the histone code, although the idea that histone state can be read linearly as a digital information carrier has been largely debunked. One of the best-understood systems that orchestrate chromatin-based silencing is the SIR protein based silencing of the yeast hidden mating-type loci HML and HMR.
DNA methylation frequently occurs in repeated sequences, and helps to suppress the expression and mobility of 'transposable elements
A transposable element (TE, transposon, or jumping gene) is a nucleic acid sequence in DNA that can change its position within a genome, sometimes creating or reversing mutations and altering the cell's genetic identity and genome size. Trans ...
':thymidine
Thymidine (symbol dT or dThd), also known as deoxythymidine, deoxyribosylthymine, or thymine deoxyriboside, is a pyrimidine deoxynucleoside. Deoxythymidine is the DNA nucleoside T, which pairs with deoxyadenosine (A) in double-stranded DNA. I ...
, CpG sites are frequently mutated and become rare in the genome, except at CpG islands
The CpG sites or CG sites are regions of DNA where a cytosine nucleotide is followed by a guanine nucleotide in the linear sequence of bases along its 5' → 3' direction. CpG sites occur with high frequency in genomic regions called CpG isl ...
where they remain unmethylated. Epigenetic changes of this type thus have the potential to direct increased frequencies of permanent genetic mutation. DNA methylation
DNA methylation is a biological process by which methyl groups are added to the DNA molecule. Methylation can change the activity of a DNA segment without changing the sequence. When located in a gene promoter, DNA methylation typically acts t ...
patterns are known to be established and modified in response to environmental factors by a complex interplay of at least three independent DNA methyltransferase
In biochemistry, the DNA methyltransferase (DNA MTase, DNMT) family of enzymes catalyze the transfer of a methyl group to DNA. DNA methylation serves a wide variety of biological functions. All the known DNA methyltransferases use S-adenosyl m ...
s, DNMT1, DNMT3A, and DNMT3B, the loss of any of which is lethal in mice.proliferating cell nuclear antigen
Proliferating cell nuclear antigen (PCNA) is a DNA clamp that acts as a processivity factor for DNA polymerase δ in eukaryotic cells and is essential for replication. PCNA is a homotrimer and achieves its processivity by encircling the DNA, whe ...
(PCNA).DNA replication
In molecular biology, DNA replication is the biological process of producing two identical replicas of DNA from one original DNA molecule. DNA replication occurs in all living organisms acting as the most essential part for biological inheritanc ...
, and therefore is often referred to as the ‘maintenance' methyltransferase.structural
A structure is an arrangement and organization of interrelated elements in a material object or system, or the object or system so organized. Material structures include man-made objects such as buildings and machines and natural objects such ...
) heritable states.nucleosome
A nucleosome is the basic structural unit of DNA packaging in eukaryotes. The structure of a nucleosome consists of a segment of DNA wound around eight histone proteins and resembles thread wrapped around a spool. The nucleosome is the fundamen ...
s recruit enzymes that similarly modify nearby nucleosomes. A simplified stochastic model for this type of epigenetics is found here.Small interfering RNA
Small interfering RNA (siRNA), sometimes known as short interfering RNA or silencing RNA, is a class of double-stranded RNA at first non-coding RNA molecules, typically 20-24 (normally 21) base pairs in length, similar to miRNA, and operating wi ...
s can modulate transcriptional gene expression via epigenetic modulation of targeted promoters.
RNA transcripts
Sometimes a gene, after being turned on, transcribes a product that (directly or indirectly) maintains the activity of that gene. For example, Hnf4
HNF4 (Hepatocyte Nuclear Factor 4) is a nuclear receptor protein mostly expressed in the liver, gut, kidney, and pancreatic beta cells that is critical for liver development. In humans, there are two isoforms of HNF4, HNF4α and HNF4γ, enco ...
and MyoD
MyoD, also known as myoblast determination protein 1, is a protein in animals that plays a major role in regulating muscle differentiation. MyoD, which was discovered in the laboratory of Harold M. Weintraub, belongs to a family of proteins know ...
enhance the transcription of many liver-specific and muscle-specific genes, respectively, including their own, through the transcription factor
In molecular biology, a transcription factor (TF) (or sequence-specific DNA-binding factor) is a protein that controls the rate of transcription of genetic information from DNA to messenger RNA, by binding to a specific DNA sequence. The fu ...
activity of the proteins
Proteins are large biomolecules and macromolecules that comprise one or more long chains of amino acid residues. Proteins perform a vast array of functions within organisms, including catalysing metabolic reactions, DNA replication, respo ...
they encode. RNA signalling includes differential recruitment of a hierarchy of generic chromatin modifying complexes and DNA methyltransferases to specific loci by RNAs during differentiation and development.zygote
A zygote (, ) is a eukaryotic cell formed by a fertilization event between two gametes. The zygote's genome is a combination of the DNA in each gamete, and contains all of the genetic information of a new individual organism.
In multicellula ...
by the mother during oogenesis or via nurse cells, resulting in maternal effect
A maternal effect is a situation where the phenotype of an organism is determined not only by the environment it experiences and its genotype, but also by the environment and genotype of its mother. In genetics, maternal effects occur when an org ...
phenotypes. A smaller quantity of sperm RNA is transmitted from the father, but there is recent evidence that this epigenetic information can lead to visible changes in several generations of offspring.
MicroRNAs
MicroRNAs (miRNAs) are members of non-coding RNAs that range in size from 17 to 25 nucleotides. miRNAs regulate a large variety of biological functions in plants and animals.
mRNA
In 2011, it was demonstrated that the methylation
In the chemical sciences, methylation denotes the addition of a methyl group on a substrate, or the substitution of an atom (or group) by a methyl group. Methylation is a form of alkylation, with a methyl group replacing a hydrogen atom. These t ...
of messenger RNA, mRNA plays a critical role in human energy balance (biology), energy homeostasis. The obesity-associated FTO gene is shown to be able to demethylate N6-methyladenosine in RNA.
sRNAs
Bacterial small RNA, sRNAs are small (50–250 nucleotides), highly structured, non-coding RNA fragments found in bacteria. They control gene expression including virulence genes in pathogens and are viewed as new targets in the fight against drug-resistant bacteria. They play an important role in many biological processes, binding to mRNA and protein targets in prokaryotes. Their phylogenetic analyses, for example through sRNA–mRNA target interactions or protein Hfq binding sRNA, binding properties, are used to build comprehensive databases. sRNA-gene maps based on their targets in microbial genomes are also constructed.
Prions
Prions are Infection, infectious forms of proteins. In general, proteins fold into discrete units that perform distinct cellular functions, but some proteins are also capable of forming an infectious conformational state known as a prion. Although often viewed in the context of Transmissible spongiform encephalopathy, infectious disease, prions are more loosely defined by their ability to catalytically convert other native state versions of the same protein to an infectious conformational state. It is in this latter sense that they can be viewed as epigenetic agents capable of inducing a phenotypic change without a modification of the genome.
Fungal prions are considered by some to be epigenetic because the infectious phenotype caused by the prion can be inherited without modification of the genome. PSI (prion), PSI+ and URE3, discovered in Saccharomyces cerevisiae, yeast in 1965 and 1971, are the two best studied of this type of prion.
Structural inheritance
In ciliates such as ''Tetrahymena'' and ''Paramecium'', genetically identical cells show heritable differences in the patterns of ciliary rows on their cell surface. Experimentally altered patterns can be transmitted to daughter cells. It seems existing structures act as templates for new structures. The mechanisms of such inheritance are unclear, but reasons exist to assume that multicellular organisms also use existing cell structures to assemble new ones.
Nucleosome positioning
Eukaryotic genomes have numerous nucleosomes. Nucleosome position is not random, and determine the accessibility of DNA to regulatory proteins. Promoters active in different tissues have been shown to have different nucleosome positioning features. This determines differences in gene expression and cell differentiation. It has been shown that at least some nucleosomes are retained in sperm cells (where most but not all histones are replaced by protamines). Thus nucleosome positioning is to some degree inheritable. Recent studies have uncovered connections between nucleosome positioning and other epigenetic factors, such as DNA methylation and hydroxymethylation.
Histone variants
Different histone variants are incorporated into specific regions of the genome non-randomly. Their differential biochemical characteristics can affect genome functions via their roles in gene regulation, and maintenance of chromosome structures.
Genomic architecture
The three-dimensional configuration of the genome (the 3D genome) is complex, dynamic and crucial for regulating genomic function and nuclear processes such as DNA replication, transcription and DNA-damage repair.
Functions and consequences
In the brain
Memory
Encoding (memory), Memory formation and maintenance are due to epigenetic alterations that cause the required dynamic changes in gene transcription that create and renew memory in neurons.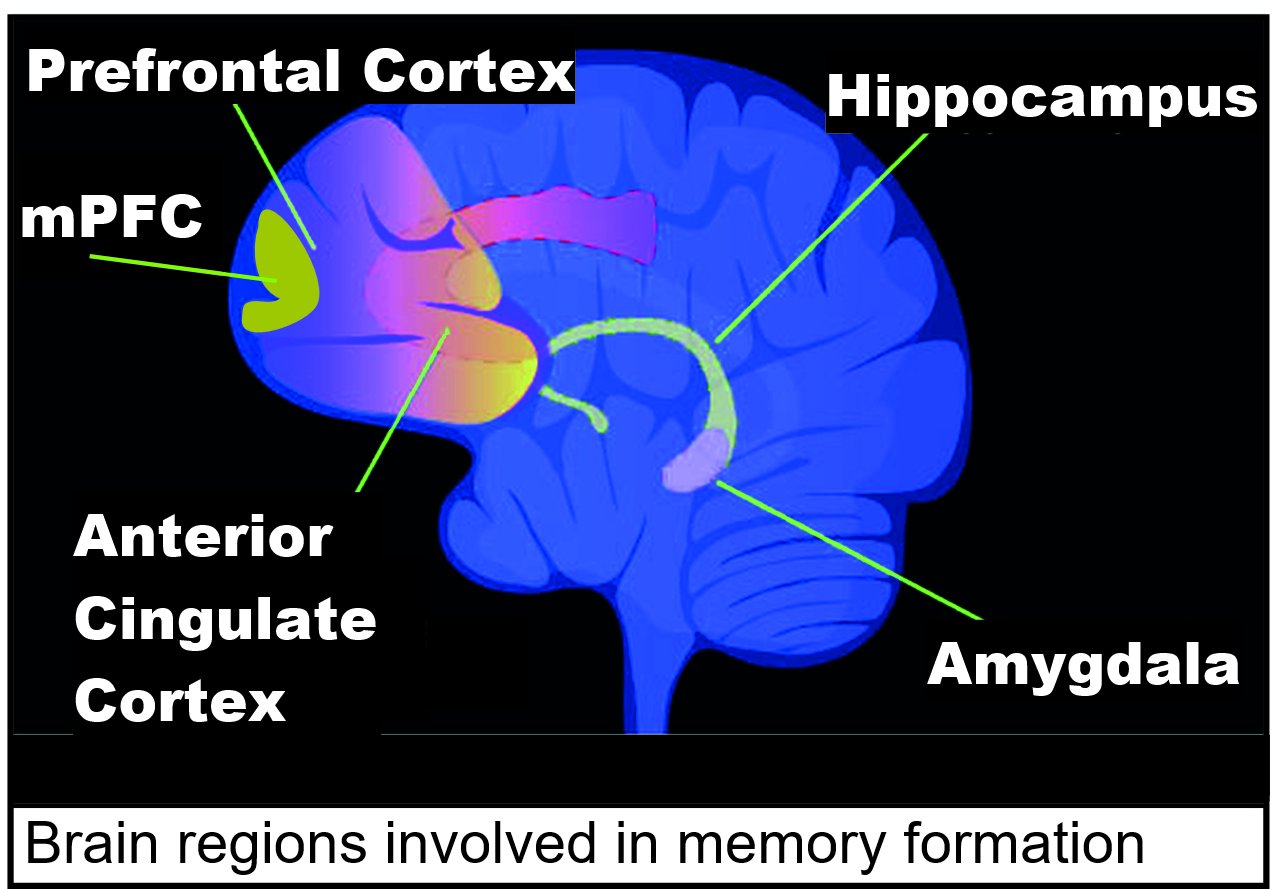 Areas of the brain important in the formation of memories include the hippocampus, medial prefrontal cortex (mPFC), anterior cingulate cortex and amygdala, as shown in the diagram of the human brain in this section.
Areas of the brain important in the formation of memories include the hippocampus, medial prefrontal cortex (mPFC), anterior cingulate cortex and amygdala, as shown in the diagram of the human brain in this section.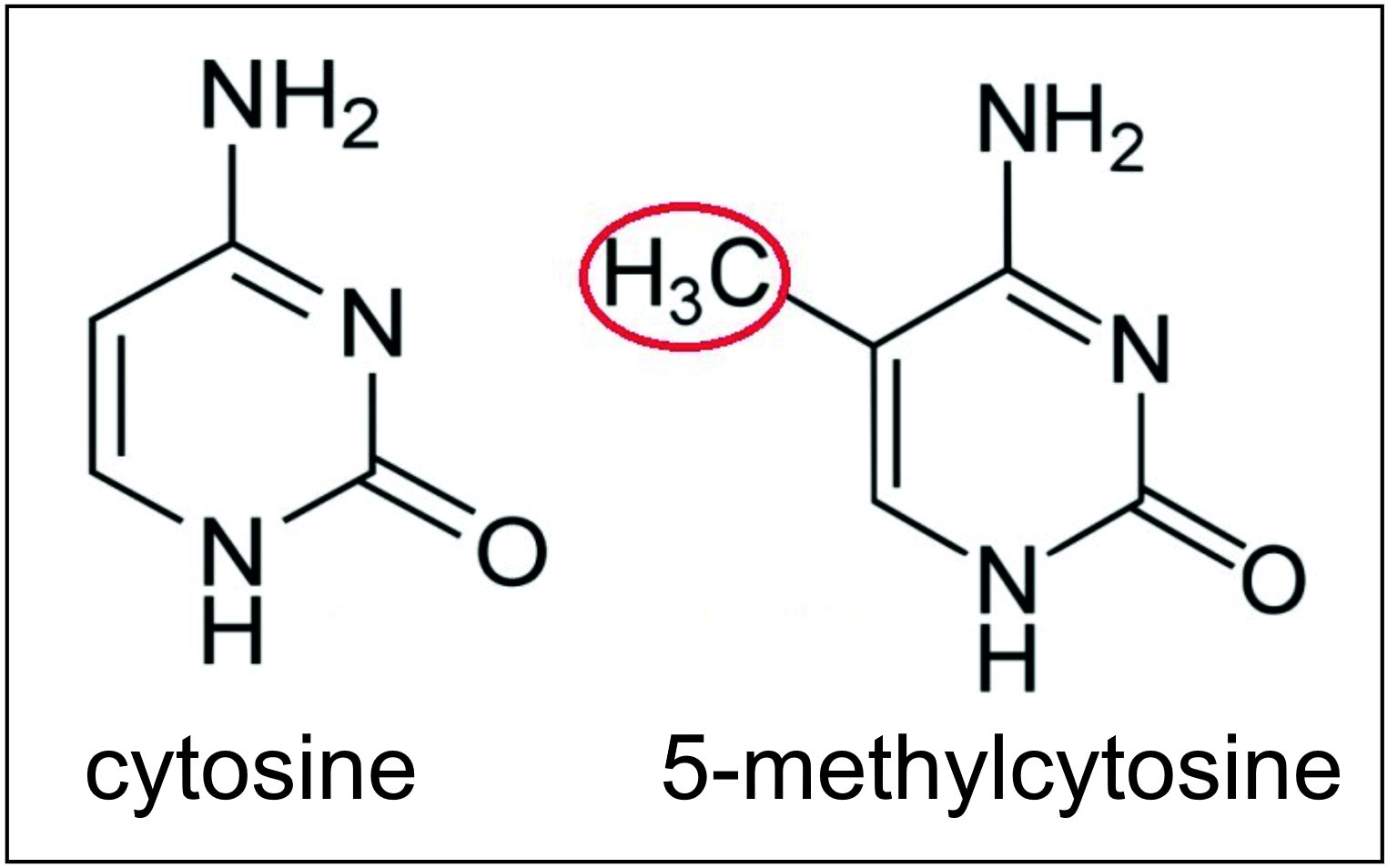 The DNA methyltransferases DNMT3A1, DNMT3A2 and DNMT3B can all methylate cytosines (see image this section) at
The DNA methyltransferases DNMT3A1, DNMT3A2 and DNMT3B can all methylate cytosines (see image this section) at CpG site
The CpG sites or CG sites are regions of DNA where a cytosine nucleotide is followed by a guanine nucleotide in the linear sequence of bases along its 5' → 3' direction. CpG sites occur with high frequency in genomic regions called CpG isl ...
s in or near the promoters of genes. As shown by Manzo et al., CpG site
The CpG sites or CG sites are regions of DNA where a cytosine nucleotide is followed by a guanine nucleotide in the linear sequence of bases along its 5' → 3' direction. CpG sites occur with high frequency in genomic regions called CpG isl ...
s in the promoter regions of genes, the genes are often repressed, and when newly demethylated cytosines are present the genes may be activated.
Other and general
In adulthood, changes in the epigenome are important for various higher cognitive functions. Dysregulation of epigenetic mechanisms is implicated in neurodegenerative disorders and diseases. Epigenetic modifications in neuron
A neuron, neurone, or nerve cell is an electrically excitable cell that communicates with other cells via specialized connections called synapses. The neuron is the main component of nervous tissue in all animals except sponges and placozoa. N ...
s are dynamic and reversible. Epigenetic regulation impacts neuronal action, affecting learning, memory, and other cognitive processes.
Early events, including during embryonic development
An embryo is an initial stage of development of a multicellular organism. In organisms that reproduce sexually, embryonic development is the part of the life cycle that begins just after fertilization of the female egg cell by the male sperm ...
, can influence development, cognition, and health outcomes through epigenetic mechanisms.
Epigenetic mechanisms have been proposed as "a potential molecular mechanism for effects of endogenous hormones on the organization of developing brain circuits".
Nutrients could interact with the epigenome to "protect or boost cognitive processes across the lifespan".
Epigenetics play a major role in brain aging and age-related cognitive decline, with relevance to life extension.
A review suggests neurobiological effects of physical exercise via epigenetics seem "central to building an 'epigenetic memory' to influence long-term brain function and behavior" and may even be heritable.
With the axo-ciliary synapse, there is communication between serotonergic axons and antenna-like primary cilia of Hippocampus anatomy#Basic hippocampal circuit, CA1 Pyramidal cell, pyramidal neuron
A neuron, neurone, or nerve cell is an electrically excitable cell that communicates with other cells via specialized connections called synapses. The neuron is the main component of nervous tissue in all animals except sponges and placozoa. N ...
s that alters the neuron's epigenetic state in the Cell nucleus, nucleus via the signalling distinct from that at the plasma membrane (and longer-term).
Epigenetics also play a major role in the Evolution of the brain#Genetic factors of recent evolution, brain evolution in and to humans.
Development
Developmental epigenetics can be divided into predetermined and probabilistic epigenesis. Predetermined epigenesis is a unidirectional movement from structural development in DNA to the functional maturation of the protein. "Predetermined" here means that development is scripted and predictable. Probabilistic epigenesis on the other hand is a bidirectional structure-function development with experiences and external molding development.nucleosome
A nucleosome is the basic structural unit of DNA packaging in eukaryotes. The structure of a nucleosome consists of a segment of DNA wound around eight histone proteins and resembles thread wrapped around a spool. The nucleosome is the fundamen ...
repositioning, is very important in the development of multicellular eukaryotic organisms.stem cells
In multicellular organisms, stem cells are undifferentiated or partially differentiated cells that can differentiate into various types of cells and proliferate indefinitely to produce more of the same stem cell. They are the earliest type o ...
retaining the ability to differentiate into several cell types ("totipotency" and "multipotency"). In mammals, some stem cells continue producing newly differentiated cells throughout life, such as in Epigenetic Regulation of Neurogenesis, neurogenesis, but mammals are not able to respond to loss of some tissues, for example, the inability to regenerate limbs, which some other animals are capable of. Epigenetic modifications regulate the transition from neural stem cells to glial progenitor cells (for example, differentiation into oligodendrocytes is regulated by the deacetylation and methylation of histones. Unlike animals, plant cells do not terminally differentiate, remaining totipotent with the ability to give rise to a new individual plant. While plants do utilize many of the same epigenetic mechanisms as animals, such as chromatin remodeling, it has been hypothesized that some kinds of plant cells do not use or require "cellular memories", resetting their gene expression patterns using positional information from the environment and surrounding cells to determine their fate.methylation
In the chemical sciences, methylation denotes the addition of a methyl group on a substrate, or the substitution of an atom (or group) by a methyl group. Methylation is a form of alkylation, with a methyl group replacing a hydrogen atom. These t ...
signatures.
Controversial results from one study suggested that traumatic experiences might produce an epigenetic signal that is capable of being passed to future generations. Mice were trained, using foot shocks, to fear a cherry blossom odor. The investigators reported that the mouse offspring had an increased aversion to this specific odor. They suggested epigenetic changes that increase gene expression, rather than in DNA itself, in a gene, M71, that governs the functioning of an odor receptor in the nose that responds specifically to this cherry blossom smell. There were physical changes that correlated with olfactory (smell) function in the brains of the trained mice and their descendants. Several criticisms were reported, including the study's low statistical power as evidence of some irregularity such as bias in reporting results.
Transgenerational
Epigenetic mechanisms were a necessary part of the evolutionary origin of cell differentiation.paramutation
In epigenetics, a paramutation is an interaction between two alleles at a single locus, whereby one allele induces a heritable change in the other allele. The change may be in the pattern of DNA methylation or histone modifications. The allele indu ...
observed in maize). Although most of these multigenerational epigenetic traits are gradually lost over several generations, the possibility remains that multigenerational epigenetics could be another aspect to evolution and adaptation.
As mentioned above, some define epigenetics as heritable.
A sequestered germ line or Weismann barrier is specific to animals, and epigenetic inheritance is more common in plants and microbes. Eva Jablonka, Marion J. Lamb and Étienne Danchin have argued that these effects may require enhancements to the standard conceptual framework of the modern synthesis (20th century), modern synthesis and have called for an extended evolutionary synthesis.transgenerational epigenetic inheritance
Transgenerational epigenetic inheritance is the transmission of epigenetic markers from one organism to the next (i.e., from parent to child) that affects the traits of offspring without altering the primary structure of DNA (i.e. the sequence of ...
phenomena have been reported in a wide range of organisms, including prokaryotes, plants, and animals.
Epigenetics in bacteria
 While epigenetics is of fundamental importance in eukaryotes, especially Multicellular organism, metazoans, it plays a different role in bacteria. Most importantly, eukaryotes use epigenetic mechanisms primarily to regulate gene expression which bacteria rarely do. However, bacteria make widespread use of postreplicative DNA methylation for the epigenetic control of DNA-protein interactions. Bacteria also use DNA adenine methylation (rather than DNA
While epigenetics is of fundamental importance in eukaryotes, especially Multicellular organism, metazoans, it plays a different role in bacteria. Most importantly, eukaryotes use epigenetic mechanisms primarily to regulate gene expression which bacteria rarely do. However, bacteria make widespread use of postreplicative DNA methylation for the epigenetic control of DNA-protein interactions. Bacteria also use DNA adenine methylation (rather than DNA cytosine
Cytosine () ( symbol C or Cyt) is one of the four nucleobases found in DNA and RNA, along with adenine, guanine, and thymine (uracil in RNA). It is a pyrimidine derivative, with a heterocyclic aromatic ring and two substituents attached (an am ...
methylation) as an epigenetic signal. DNA adenine methylation is important in bacteria virulence in organisms such as ''Escherichia coli'', ''Salmonella, Vibrio, Yersinia, Haemophilus'', and ''Brucella''. In ''Alphaproteobacteria'', methylation of adenine regulates the cell cycle and couples gene transcription to DNA replication. In ''Gammaproteobacteria'', adenine methylation provides signals for DNA replication, chromosome segregation, mismatch repair, packaging of bacteriophage, transposase activity and regulation of gene expression.
Medicine
Epigenetics has many and varied potential medical applications.
Twins
Direct comparisons of identical twins constitute an optimal model for interrogating environmental epigenetics. In the case of humans with different environmental exposures, monozygotic (identical) twins were epigenetically indistinguishable during their early years, while older twins had remarkable differences in the overall content and genomic distribution of 5-methylcytosine DNA and histone acetylation.
Genomic imprinting
Some human disorders are associated with genomic imprinting, a phenomenon in mammals where the father and mother contribute different epigenetic patterns for specific genomic loci in their germ cells.[A person's paternal grandson is the son of a son of that person; a maternal grandson is the son of a daughter.] of Swedish men who were exposed during preadolescence to famine in the 19th century were less likely to die of cardiovascular disease. If food was plentiful, then diabetes mortality in the grandchildren increased, suggesting that this was a transgenerational epigenetic inheritance.[ Robert Winston refers to this study in ]
lecture
; see also discussion at Leeds University, her
/ref> The opposite effect was observed for females – the paternal (but not maternal) granddaughters of women who experienced famine while in the womb (and therefore while their eggs were being formed) lived shorter lives on average.
Diabetic wound healing
Epigenetic modifications have given insight into the understanding of the pathophysiology of different disease conditions. Though, they are strongly associated with cancer, their role in other pathological conditions are of equal importance. It appears that the hyperglycaemic environment could imprint such changes at the genomic level, that macrophages are primed towards a pro-inflammatory state and could fail to exhibit any phenotypic alteration towards the pro-healing type. This phenomenon of altered Macrophage Polarization is mostly associated with all the diabetic complications in a clinical set-up. As of 2018, several reports reveal the relevance of different epigenetic modifications with respect to diabetic complications. Sooner or later, with the advancements in biomedical tools, the detection of such biomarkers as prognostic and diagnostic tools in patients could possibly emerge out as alternative approaches. It is noteworthy to mention here that the use of epigenetic modifications as therapeutic targets warrant extensive preclinical as well as clinical evaluation prior to use.
Examples of drugs altering gene expression from epigenetic events
The use of beta-lactam antibiotics can alter glutamate receptor activity and the action of cyclosporine on multiple transcription factors. Additionally, lithium can impact autophagy of aberrant proteins, and opioid drugs via chronic use can increase the expression of genes associated with addictive phenotypes.
In a groundbreaking 2003 report, Caspi and colleagues demonstrated that in a robust cohort of over one-thousand subjects assessed multiple times from preschool to adulthood, subjects who carried one or two copies of the short allele of the serotonin
Serotonin () or 5-hydroxytryptamine (5-HT) is a monoamine neurotransmitter. Its biological function is complex and multifaceted, modulating mood, cognition, reward, learning, memory, and numerous physiological processes such as vomiting and vas ...
transporter promoter polymorphism exhibited higher rates of adult depression and suicidality when exposed to childhood maltreatment when compared to long allele homozygotes with equal ELS exposure.
Addiction
Addiction is a disorder of the brain's reward system which arises through transcriptional and neuroepigenetic mechanisms and occurs over time from chronically high levels of exposure to an addictive stimulus (e.g., morphine, cocaine, sexual intercourse, gambling, etc.).
Depression
Epigenetic inheritance of depression-related phenotypes has also been reported in a preclinical study.
Research
The two forms of heritable information, namely genetic and epigenetic, are collectively called dual inheritance. Members of the APOBEC/AID family of cytosine deaminases may concurrently influence genetic and epigenetic inheritance using similar molecular mechanisms, and may be a point of crosstalk between these conceptually compartmentalized processes.
Epigenome editing
Epigenetic regulation of gene expression that could be altered or used in epigenome editing are or include mRNA modification, mRNA/lncRNA modification, DNA methylation modification and histone modification
In biology, histones are highly basic proteins abundant in lysine and arginine residues that are found in eukaryotic cell nuclei. They act as spools around which DNA winds to create structural units called nucleosomes. Nucleosomes in turn ar ...
.
Pseudoscience
As epigenetics is in the early stages of development as a science and is surrounded by sensationalism in the public media, David Gorski and geneticist Adam Rutherford have advised caution against the proliferation of false and Pseudoscience, pseudoscientific conclusions by new age authors making unfounded suggestions that a person's genes and health can be manipulated by brainwashing, mind control. Misuse of the scientific term by quackery, quack authors has produced misinformation among the general public.["Epigenetics: It doesn't mean what quacks think it means"]
Science-Based Medicine.
See also
* Baldwin effect
* Behavioral epigenetics
* Biological effects of radiation on the epigenome
* Computational epigenetics
* Contribution of epigenetic modifications to evolution
* DAnCER (database), DAnCER database (2010)
* Epigenesis (biology)
* Epigenetics in forensic science
* Epigenetic therapy
* Epigenetics of neurodegenerative diseases
* Genetics
* Lamarckism
* Nutriepigenomics
* Position-effect variegation
* Preformationism
* Somatic epitype
* Synthetic genetic array
* Transcriptional memory
* Transgenerational epigenetic inheritance
References
Further reading
*
External links
*
The Human Epigenome Project (HEP)
The Epigenome Network of Excellence (NoE)
Canadian Epigenetics, Environment and Health Research Consortium (CEEHRC)
The Epigenome Network of Excellence (NoE) – public international site
"DNA Is Not Destiny"
– ''Discover'' magazine cover story
"The Ghost In Your Genes"
''Horizon'' (2005), BBC
at Hopkins Medicine
{{Authority control
Epigenetics,
Genetic mapping
Lamarckism
 In
In  Because the
Because the  Although histone modifications occur throughout the entire sequence, the unstructured N-termini of histones (called histone tails) are particularly highly modified. These modifications include
Although histone modifications occur throughout the entire sequence, the unstructured N-termini of histones (called histone tails) are particularly highly modified. These modifications include  Areas of the brain important in the formation of memories include the hippocampus, medial prefrontal cortex (mPFC), anterior cingulate cortex and amygdala, as shown in the diagram of the human brain in this section.
When a strong memory is created, as in a rat subjected to Fear conditioning, contextual fear conditioning (CFC), one of the earliest events to occur is that more than 100 DNA double-strand breaks are formed by topoisomerase, topoisomerase IIB in neurons of the hippocampus and the medial prefrontal cortex (mPFC). These double-strand breaks are at specific locations that allow activation of transcription of immediate early genes (IEGs) that are important in memory formation, allowing their expression in messenger RNA, mRNA, with peak mRNA transcription at seven to ten minutes after CFC.
Two important IEGs in memory formation are ''EGR1'' and DNA methyltransferase, the alternative promoter variant of ''DNMT3A'', ''DNMT3A2''. EGR1 protein binds to DNA at its binding motifs, 5′-GCGTGGGCG-3′ or 5′-GCGGGGGCGG-3', and there are about 12,000 genome locations at which EGR1 protein can bind. EGR1 protein binds to DNA in gene promoter and enhancer regions. EGR1 recruits the demethylating enzyme TET1 to an association, and brings TET1 to about 600 locations on the genome where TET1 can then demethylate and activate the associated genes.
Areas of the brain important in the formation of memories include the hippocampus, medial prefrontal cortex (mPFC), anterior cingulate cortex and amygdala, as shown in the diagram of the human brain in this section.
When a strong memory is created, as in a rat subjected to Fear conditioning, contextual fear conditioning (CFC), one of the earliest events to occur is that more than 100 DNA double-strand breaks are formed by topoisomerase, topoisomerase IIB in neurons of the hippocampus and the medial prefrontal cortex (mPFC). These double-strand breaks are at specific locations that allow activation of transcription of immediate early genes (IEGs) that are important in memory formation, allowing their expression in messenger RNA, mRNA, with peak mRNA transcription at seven to ten minutes after CFC.
Two important IEGs in memory formation are ''EGR1'' and DNA methyltransferase, the alternative promoter variant of ''DNMT3A'', ''DNMT3A2''. EGR1 protein binds to DNA at its binding motifs, 5′-GCGTGGGCG-3′ or 5′-GCGGGGGCGG-3', and there are about 12,000 genome locations at which EGR1 protein can bind. EGR1 protein binds to DNA in gene promoter and enhancer regions. EGR1 recruits the demethylating enzyme TET1 to an association, and brings TET1 to about 600 locations on the genome where TET1 can then demethylate and activate the associated genes.
 The DNA methyltransferases DNMT3A1, DNMT3A2 and DNMT3B can all methylate cytosines (see image this section) at
The DNA methyltransferases DNMT3A1, DNMT3A2 and DNMT3B can all methylate cytosines (see image this section) at  While epigenetics is of fundamental importance in eukaryotes, especially Multicellular organism, metazoans, it plays a different role in bacteria. Most importantly, eukaryotes use epigenetic mechanisms primarily to regulate gene expression which bacteria rarely do. However, bacteria make widespread use of postreplicative DNA methylation for the epigenetic control of DNA-protein interactions. Bacteria also use DNA adenine methylation (rather than DNA
While epigenetics is of fundamental importance in eukaryotes, especially Multicellular organism, metazoans, it plays a different role in bacteria. Most importantly, eukaryotes use epigenetic mechanisms primarily to regulate gene expression which bacteria rarely do. However, bacteria make widespread use of postreplicative DNA methylation for the epigenetic control of DNA-protein interactions. Bacteria also use DNA adenine methylation (rather than DNA