Lyme disease on:
[Wikipedia]
[Google]
[Amazon]
Lyme disease, also known as Lyme borreliosis, is a
 Lyme disease can affect several body systems and produce a broad range of symptoms. Not everyone with Lyme disease has all of the symptoms and many of the symptoms are not specific to Lyme disease but can occur with other diseases as well.
The
Lyme disease can affect several body systems and produce a broad range of symptoms. Not everyone with Lyme disease has all of the symptoms and many of the symptoms are not specific to Lyme disease but can occur with other diseases as well.
The
 Lyme arthritis occurs in up to 60% of untreated people, typically starting about six months after infection. It usually affects only one or a few joints, often a knee or possibly the
Lyme arthritis occurs in up to 60% of untreated people, typically starting about six months after infection. It usually affects only one or a few joints, often a knee or possibly the
 Lyme disease is caused by
Lyme disease is caused by

 There are three stages in the life cycle of a tick. Larva, nymph, and adult. During the nymph stage, ticks most frequently transmit Lyme disease and are usually most active in late spring and early summer in regions where the climate is mild. During the adult stage, Lyme disease transmission is less common due to the fact that it is less possible for adult ticks to bite humans and because they tend to be larger in size and therefore can be easily seen and removed.
There are three stages in the life cycle of a tick. Larva, nymph, and adult. During the nymph stage, ticks most frequently transmit Lyme disease and are usually most active in late spring and early summer in regions where the climate is mild. During the adult stage, Lyme disease transmission is less common due to the fact that it is less possible for adult ticks to bite humans and because they tend to be larger in size and therefore can be easily seen and removed.
 In Europe, the main vector is ''
In Europe, the main vector is ''
 Attached ticks should be removed promptly. Risk of infection increases with time of attachment, but in North America risk of Lyme disease is small if the tick is removed within 36 hours.
Attached ticks should be removed promptly. Risk of infection increases with time of attachment, but in North America risk of Lyme disease is small if the tick is removed within 36 hours.
 Lyme disease Endemic (epidemiology), occurs regularly in
Lyme disease Endemic (epidemiology), occurs regularly in
 In 2019, according to the CDC, there were approximately 35,000 confirmed and probable cases in the U.S. with incidences occurring in almost every state. The CDC is currently conducting research on evaluation and diagnostics of the disease and preliminary results suggest the number of new cases to be around 300,000.
Lyme disease is the most common tick-borne disease in North America and Europe, and one of the fastest-growing infectious diseases in the United States. Of cases reported to the United States CDC, the ratio of Lyme disease infection is 7.9 cases for every 100,000 persons. In the ten states where Lyme disease is most common, the average was 31.6 cases for every 100,000 persons for the year 2005.
In 2019, according to the CDC, there were approximately 35,000 confirmed and probable cases in the U.S. with incidences occurring in almost every state. The CDC is currently conducting research on evaluation and diagnostics of the disease and preliminary results suggest the number of new cases to be around 300,000.
Lyme disease is the most common tick-borne disease in North America and Europe, and one of the fastest-growing infectious diseases in the United States. Of cases reported to the United States CDC, the ratio of Lyme disease infection is 7.9 cases for every 100,000 persons. In the ten states where Lyme disease is most common, the average was 31.6 cases for every 100,000 persons for the year 2005.
vector-borne disease
In epidemiology, a disease vector is any living agent that carries and transmits an infectious pathogen to another living organism; agents regarded as vectors are organisms, such as parasites or microbes. The first major discovery of a disease ve ...
caused by the ''Borrelia
''Borrelia'' is a genus of bacteria of the spirochete phylum. Several species cause Lyme disease, also called Lyme borreliosis, a zoonotic, vector-borne disease transmitted by ticks. Other species of ''Borrelia'' cause relapsing fever, and are t ...
'' bacterium, which is spread by ticks
Ticks (order Ixodida) are parasitic arachnids that are part of the mite superorder Parasitiformes. Adult ticks are approximately 3 to 5 mm in length depending on age, sex, species, and "fullness". Ticks are external parasites, living by ...
in the genus ''Ixodes
''Ixodes'' is a genus of hard-bodied ticks (family Ixodidae). It includes important disease vectors of animals and humans ( tick-borne disease), and some species (notably '' Ixodes holocyclus'') inject toxins that can cause paralysis. Som ...
''. The most common sign of infection is an expanding red rash
A rash is a change of the human skin which affects its color, appearance, or texture.
A rash may be localized in one part of the body, or affect all the skin. Rashes may cause the skin to change color, itch, become warm, bumpy, chapped, dry, cr ...
, known as erythema migrans
Erythema migrans or erythema chronicum migrans is an expanding rash often seen in the early stage of Lyme disease, and can also (but less commonly) be caused by southern tick-associated rash illness (STARI). Page last reviewed: October 22, 2015 ...
(EM), which appears at the site of the tick bite about a week afterwards. The rash is typically neither itchy nor painful. Approximately 70–80% of infected people develop a rash. Early diagnosis can be difficult. Other early symptoms may include fever, headaches and tiredness. If untreated, symptoms may include loss of the ability to move one or both sides of the face, joint pains, severe headaches with neck stiffness or heart palpitation
Palpitations are perceived abnormalities of the heartbeat characterized by awareness of cardiac muscle contractions in the chest, which is further characterized by the hard, fast and/or irregular beatings of the heart.
Symptoms include a rap ...
s. Months to years later repeated episodes of joint pain and swelling may occur. Occasionally shooting pains or tingling in the arms and legs may develop. Despite appropriate treatment about 10 to 20% of those affected develop joint pains, memory problems and tiredness for at least six months.
Lyme disease is transmitted to humans by the bites of infected ticks of the genus ''Ixodes
''Ixodes'' is a genus of hard-bodied ticks (family Ixodidae). It includes important disease vectors of animals and humans ( tick-borne disease), and some species (notably '' Ixodes holocyclus'') inject toxins that can cause paralysis. Som ...
''. In the United States ticks of concern are usually of the ''Ixodes scapularis
''Ixodes scapularis'' is commonly known as the deer tick or black-legged tick (although some people reserve the latter term for ''Ixodes pacificus'', which is found on the west coast of the US), and in some parts of the US as the bear tick. It wa ...
'' type and must be attached for at least 36 hours before the bacteria can spread. In Europe ''Ixodes ricinus
''Ixodes ricinus'', the castor bean tick, is a chiefly European species of hard-bodied tick. It may reach a length of when engorged with a blood meal, and can transmit both bacterial and viral pathogens such as the causative agents of Lyme disea ...
'' ticks may spread the bacteria more quickly. In North America the bacteria ''Borrelia burgdorferi
''Borrelia burgdorferi'' is a bacterial species of the spirochete class in the genus '' Borrelia'', and is one of the causative agents of Lyme disease in humans. Along with a few similar genospecies, some of which also cause Lyme disease, it mak ...
''and ''B. mayonii'' cause Lyme disease. In Europe and Asia'' Borrelia afzelii
''Borrelia afzelii'' is a species of '' Borrelia'' a bacterium that can infect various species of vertebrates and invertebrates.
Among 30 ''Borrelia'' known species, it is one of four which are likely to infect humans causing a variant of Lyme d ...
, Borrelia garinii
''Borrelia garinii'' is a spirochete
A spirochaete () or spirochete is a member of the phylum Spirochaetota (), (synonym Spirochaetes) which contains distinctive diderm (double-membrane) gram-negative bacteria, most of which have long, ...
, B. spielmanii'' and four other species also cause the disease. The disease does not appear to be transmissible between people, by other animals nor through food. Diagnosis is based on a combination of symptoms, history of tick exposure and possibly testing for specific antibodies
An antibody (Ab), also known as an immunoglobulin (Ig), is a large, Y-shaped protein used by the immune system to identify and neutralize foreign objects such as pathogenic bacteria and viruses. The antibody recognizes a unique molecule of the ...
in the blood. Blood tests are often negative in the early stages of the disease. Testing of individual ticks is not typically useful.
Prevention includes efforts to prevent tick bites by wearing clothing to cover the arms and legs and using DEET or picaridin
Icaridin, also known as picaridin, is an insect repellent which can be used directly on skin or clothing. It has broad efficacy against various arthropods such as mosquitos, ticks, gnats, flies and fleas, and is almost colorless and odorless. A s ...
-based insect repellent
An insect repellent (also commonly called "bug spray") is a substance applied to skin, clothing, or other surfaces to discourage insects (and arthropods in general) from landing or climbing on that surface. Insect repellents help prevent and cont ...
s. Using pesticide
Pesticides are substances that are meant to control pests. This includes herbicide, insecticide, nematicide, molluscicide, piscicide, avicide, rodenticide, bactericide, insect repellent, animal repellent, microbicide, fungicide, and lampri ...
s to reduce tick numbers may also be effective. Ticks can be removed using tweezer
Tweezers are small hand tools used for grasping objects too small to be easily handled with the human fingers. Tweezers are thumb-driven forceps most likely derived from tongs used to grab or hold hot objects since the dawn of recorded history. ...
s. If the removed tick is full of blood a single dose of doxycycline
Doxycycline is a broad-spectrum tetracycline class antibiotic used in the treatment of infections caused by bacteria and certain parasites. It is used to treat bacterial pneumonia, acne, chlamydia infections, Lyme disease, cholera, typhus, an ...
may be used to prevent the development of infection but is not generally recommended since the development of infection is rare. If an infection develops, a number of antibiotics are effective, including doxycycline, amoxicillin
Amoxicillin is an antibiotic medication used to treat a number of bacterial infections. These include middle ear infection, strep throat, pneumonia, skin infections, and urinary tract infections among others. It is taken by mouth, or less c ...
and cefuroxime
Cefuroxime, sold under the brand name Zinacef among others, is a second-generation cephalosporin antibiotic used to treat and prevent a number of bacterial infections. These include pneumonia, meningitis, otitis media, sepsis, urinary tract inf ...
. Standard treatment usually lasts for two or three weeks. Some people develop a fever and muscle and joint pains from treatment, which may last for one or two days. In those who develop persistent symptoms, long-term antibiotic therapy has not been found to be useful.
Lyme disease is the most common disease spread by ticks in the Northern Hemisphere
The Northern Hemisphere is the half of Earth that is north of the Equator. For other planets in the Solar System, north is defined as being in the same celestial hemisphere relative to the invariable plane of the solar system as Earth's Nort ...
. An estimated 476,000 people a year are diagnosed and treated for the disease in the United States, and over 200,000 people a year in Western Europe. Infections are most common in the spring and early summer. Lyme disease was diagnosed as a separate condition for the first time in 1975 in Lyme, Connecticut
Lyme is a New England town, town in New London County, Connecticut, New London County, Connecticut, United States, situated on the eastern side of the Connecticut River. The population was 2,352 at the 2020 United States Census, 2020 census. Lyme i ...
. It was originally mistaken for juvenile rheumatoid arthritis
Juvenile may refer to:
*Juvenile status, or minor (law), prior to adulthood
* Juvenile (organism)
*Juvenile (rapper) (born 1975), American rapper
* ''Juvenile'' (2000 film), Japanese film
* ''Juvenile'' (2017 film)
*Juvenile (greyhounds), a greyh ...
. The bacterium involved was first described in 1981 by Willy Burgdorfer
Wilhelm Burgdorfer (June 27, 1925 – November 17, 2014) was an American scientist born and educated in Basel, Switzerland, considered an international leader in the field of medical entomology. He discovered the bacterial pathogen that causes Ly ...
. Chronic symptoms following treatment are known as "post-treatment Lyme disease syndrome" (PTLDS). PTLDS is different from chronic Lyme disease
Chronic Lyme disease (CLD) is the name used by some people with "a broad array of illnesses or symptom complexes for which there is no reproducible or convincing scientific evidence of any relationship to ''Borrelia burgdorferi'' infection" to ...
, a term no longer supported by scientists and used in different ways by different groups. Some healthcare providers claim that PTLDS is caused by persistent infection but this is not believed to be true because no evidence of persistent infection can be found after standard treatment.
No human vaccines for Lyme disease are currently available, although research is ongoing. Multiple vaccines are available for the prevention of Lyme disease in dogs.
Signs and symptoms
 Lyme disease can affect several body systems and produce a broad range of symptoms. Not everyone with Lyme disease has all of the symptoms and many of the symptoms are not specific to Lyme disease but can occur with other diseases as well.
The
Lyme disease can affect several body systems and produce a broad range of symptoms. Not everyone with Lyme disease has all of the symptoms and many of the symptoms are not specific to Lyme disease but can occur with other diseases as well.
The incubation period
Incubation period (also known as the latent period or latency period) is the time elapsed between exposure to a pathogenic organism, a chemical, or radiation, and when symptoms and signs are first apparent. In a typical infectious disease, the i ...
from infection to the onset of symptoms is usually one to two weeks but can be much shorter (days) or much longer (months to years). Lyme symptoms most often occur from May to September in the northern hemisphere because the nymphal stage of the tick is responsible for most cases. Asymptomatic
In medicine, any disease is classified asymptomatic if a patient tests as carrier for a disease or infection but experiences no symptoms. Whenever a medical condition fails to show noticeable symptoms after a diagnosis it might be considered asy ...
infection exists but occurs in less than 7% of infected individuals in the United States. Asymptomatic infection may be much more common among those infected in Europe.
Early localized infection
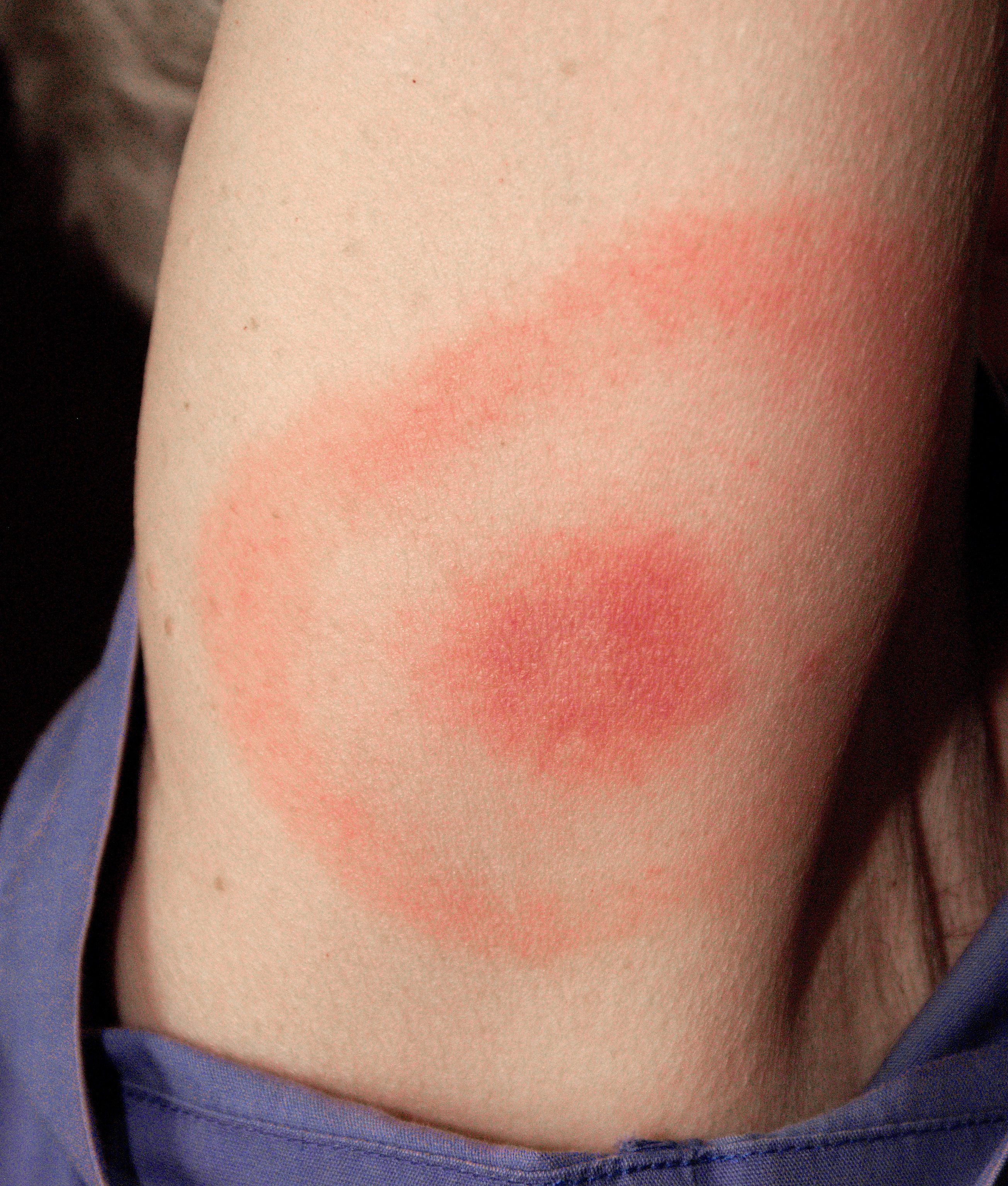
Early localized infection can occur when the infection has not yet spread throughout the body. Only the site where the infection has first come into contact with the skin is affected. The initial sign of about 80% of Lyme infections is an ''erythema migrans
Erythema migrans or erythema chronicum migrans is an expanding rash often seen in the early stage of Lyme disease, and can also (but less commonly) be caused by southern tick-associated rash illness (STARI). Page last reviewed: October 22, 2015 ...
'' (EM) rash at the site of a tick bite, often near skin folds such as the armpit
The axilla (also, armpit, underarm or oxter) is the area on the human body directly under the shoulder joint. It includes the axillary space, an anatomical space within the shoulder girdle between the arm and the thoracic cage, bounded superior ...
, groin
In human anatomy, the groin (the adjective is ''inguinal'', as in inguinal canal) is the junctional area (also known as the inguinal region) between the abdomen and the thigh on either side of the pubic bone. This is also known as the medial comp ...
or back of the knee, on the trunk, under clothing straps, or in children's hair, ears or neck. Most people who get infected do not remember seeing a tick or a bite. The rash appears typically one or two weeks (range 3–32 days) after the bite and expands 2–3 cm per day up to a diameter of 5–70 cm (median 16 cm). The rash is usually circular or oval, red or bluish, and may have an elevated or darker center. In about 79% of cases in Europe but only 19% of cases in endemic areas of the U.S. the rash gradually clears from the center toward the edges, possibly forming a "bull's eye" pattern. The rash may feel warm but usually is not itchy, is rarely tender or painful and takes up to four weeks to resolve if untreated.
The EM rash is often accompanied by symptoms of a viral-like illness, including fatigue, headache, body aches, fever and chills but usually not nausea or upper-respiratory problems. These symptoms may also appear without a rash or linger after the rash has disappeared. Lyme can progress to later stages without these symptoms or a rash.
People with high fever for more than two days or whose other symptoms of viral-like illness do not improve despite antibiotic treatment for Lyme disease, or who have abnormally low levels of white
White is the lightest color and is achromatic (having no hue). It is the color of objects such as snow, chalk, and milk, and is the opposite of black. White objects fully reflect and scatter all the visible wavelengths of light. White on ...
or red
Red is the color at the long wavelength end of the visible spectrum of light, next to orange and opposite violet. It has a dominant wavelength of approximately 625–740 nanometres. It is a primary color in the RGB color model and a secondar ...
cells or platelets
Platelets, also called thrombocytes (from Greek θρόμβος, "clot" and κύτος, "cell"), are a component of blood whose function (along with the coagulation factors) is to react to bleeding from blood vessel injury by clumping, thereby ini ...
in the blood, should be investigated for possible coinfection with other tick-borne diseases such as ehrlichiosis
Ehrlichiosis is a tick-borne
bacterial infection, caused by bacteria of the family Anaplasmataceae, genera '' Ehrlichia'' and ''Anaplasma''. These obligate intracellular bacteria infect and kill white blood cells.
The average reported annual in ...
and babesiosis
Babesiosis or piroplasmosis is a malaria-like parasitic disease caused by infection with a eukaryotic parasite in the order Piroplasmida, typically a ''Babesia'' or '' Theileria'', in the phylum Apicomplexa. Human babesiosis transmission via ...
.
Early disseminated infection
Within days to weeks after the onset of local infection the ''Borrelia
''Borrelia'' is a genus of bacteria of the spirochete phylum. Several species cause Lyme disease, also called Lyme borreliosis, a zoonotic, vector-borne disease transmitted by ticks. Other species of ''Borrelia'' cause relapsing fever, and are t ...
'' bacteria may spread through the lymphatic system or bloodstream. In 10–20% of untreated cases EM rashes develop at sites across the body that bear no relation to the original tick bite. Transient muscle pains
Myalgia (also called muscle pain and muscle ache in layman's terms) is the medical term for muscle pain. Myalgia is a symptom of many diseases. The most common cause of acute myalgia is the overuse of a muscle or group of muscles; another lik ...
and joint pains are also common.
In about 10–15% of untreated people Lyme causes neurological problems known as neuroborreliosis
Neuroborreliosis is a disorder of the central nervous system. A neurological manifestation of Lyme disease, neuroborreliosis is caused by a systemic infection of spirochetes of the genus ''Borrelia.'' Symptoms of the disease include erythema migr ...
. Early neuroborreliosis typically appears 4–6 weeks (range 1–12 weeks) after the tick bite and involves some combination of lymphocytic meningitis, cranial neuritis, radiculopathy and/or mononeuritis multiplex. Lymphocytic meningitis causes characteristic changes in the cerebrospinal fluid
Cerebrospinal fluid (CSF) is a clear, colorless body fluid found within the tissue that surrounds the brain and spinal cord of all vertebrates.
CSF is produced by specialised ependymal cells in the choroid plexus of the ventricles of the bra ...
(CSF) and may be accompanied for several weeks by variable headache and, less commonly, usually mild meningitis signs such as inability to flex the neck fully and intolerance to bright lights but typically no or only very low fever. After several months neuroborreliosis can also present otolaryngological symptoms. Up to 76.5% of these otolaryngological present as tinnitus, the most common symptom. Vertigo and dizziness (53.7%) and hearing loss (16.7%) were the next most common symptoms. In children partial loss of vision may also occur. Cranial neuritis
Neuritis () is inflammation of a nerve or the general inflammation of the peripheral nervous system. Inflammation, and frequently concomitant demyelination, cause impaired transmission of neural signals and leads to aberrant nerve function. Neuri ...
is an inflammation of cranial nerves
Cranial nerves are the nerves that emerge directly from the brain (including the brainstem), of which there are conventionally considered twelve pairs. Cranial nerves relay information between the brain and parts of the body, primarily to and f ...
. When due to Lyme it most typically causes facial palsy
Facial nerve paralysis is a common problem that involves the paralysis of any structures innervated by the facial nerve. The pathway of the facial nerve is long and relatively convoluted, so there are a number of causes that may result in facial ...
, impairing blinking, smiling and chewing on one or both sides of the face. It may also cause intermittent double vision
Diplopia is the simultaneous perception of two images of a single object that may be displaced horizontally or vertically in relation to each other. Also called double vision, it is a loss of visual focus under regular conditions, and is often v ...
. Lyme radiculopathy
Radiculopathy, also commonly referred to as pinched nerve, refers to a set of conditions in which one or more nerves are affected and do not work properly (a neuropathy). Radiculopathy can result in pain (radicular pain), weakness, altered sens ...
is an inflammation of spinal nerve root A nerve root (Latin: ''radix nervi'') is the initial segment of a nerve leaving the central nervous system. Nerve roots can be classified as:
*cranial nerves, Cranial nerve roots: the initial or proximal segment of one of the twelve pairs of crania ...
s that often causes pain
Pain is a distressing feeling often caused by intense or damaging stimuli. The International Association for the Study of Pain defines pain as "an unpleasant sensory and emotional experience associated with, or resembling that associated with, ...
and less often weakness
Weakness is a symptom of a number of different conditions. The causes are many and can be divided into conditions that have true or perceived muscle weakness. True muscle weakness is a primary symptom of a variety of skeletal muscle diseases, i ...
, numbness
Hypoesthesia or numbness is a common side effect of various medical conditions that manifests as a reduced sense of touch or sensation, or a partial loss of sensitivity to sensory stimuli. In everyday speech this is generally referred to as num ...
, or altered sensation in the areas of the body served by nerves connected to the affected roots, e.g. limb(s) or part(s) of trunk. The pain is often described as unlike any other previously felt, excruciating, migrating, worse at night, rarely symmetrical, and often accompanied by extreme sleep disturbance. Mononeuritis multiplex
Peripheral neuropathy, often shortened to neuropathy, is a general term describing disease affecting the peripheral nerves, meaning nerves beyond the brain and spinal cord. Damage to peripheral nerves may impair sensation, movement, gland, or or ...
is an inflammation causing similar symptoms in one or more unrelated peripheral nerves. Rarely, early neuroborreliosis may involve inflammation of the brain
A brain is an organ that serves as the center of the nervous system in all vertebrate and most invertebrate animals. It is located in the head, usually close to the sensory organs for senses such as vision. It is the most complex organ in a v ...
or spinal cord
The spinal cord is a long, thin, tubular structure made up of nervous tissue, which extends from the medulla oblongata in the brainstem to the lumbar region of the vertebral column (backbone). The backbone encloses the central canal of the spi ...
, with symptoms such as confusion, abnormal gait, ocular movements, or speech, impaired movement, impaired motor planning, or shaking.
In North America, facial palsy is the typical early neuroborreliosis presentation, occurring in 5–10% of untreated people, in about 75% of cases accompanied by lymphocytic meningitis. Lyme radiculopathy is reported half as frequently, but many cases may be unrecognized. In European adults, the most common presentation is a combination of lymphocytic meningitis and radiculopathy known as Bannwarth syndrome, accompanied in 36-89% of cases by facial palsy. In this syndrome, radicular pain tends to start in the same body region as the initial erythema migrans
Erythema migrans or erythema chronicum migrans is an expanding rash often seen in the early stage of Lyme disease, and can also (but less commonly) be caused by southern tick-associated rash illness (STARI). Page last reviewed: October 22, 2015 ...
rash, if there was one, and precedes possible facial palsy
Facial nerve paralysis is a common problem that involves the paralysis of any structures innervated by the facial nerve. The pathway of the facial nerve is long and relatively convoluted, so there are a number of causes that may result in facial ...
and other impaired movement. In extreme cases, permanent impairment of motor or sensory function of the lower limbs may occur. In European children, the most common manifestations are facial palsy (in 55%), other cranial neuritis, and lymphocytic meningitis (in 27%).
In about 4–10% of untreated cases in the U.S. and 0.3–4% of untreated cases in Europe, typically between June and December, about one month (range 4 days-7 months) after the tick bite, the infection may cause heart complications known as Lyme carditis
Carditis (pl. carditides) is the inflammation of the heart.
It is usually studied and treated by specifying it as:
* Pericarditis is the inflammation of the pericardium
* Myocarditis is the inflammation of the heart muscle
* Endocarditis is the ...
. Symptoms may include heart palpitations
Palpitations are perceived abnormalities of the heartbeat characterized by awareness of cardiac muscle contractions in the chest, which is further characterized by the hard, fast and/or irregular beatings of the heart.
Symptoms include a rapi ...
(in 69% of people), dizziness
Dizziness is an imprecise term that can refer to a sense of disorientation in space, vertigo, or lightheadedness. It can also refer to disequilibrium or a non-specific feeling, such as giddiness or foolishness.
Dizziness is a common medical c ...
, fainting
Syncope, commonly known as fainting, or passing out, is a loss of consciousness and muscle strength characterized by a fast onset, short duration, and spontaneous recovery. It is caused by a decrease in blood flow to the brain, typically from ...
, shortness of breath
Shortness of breath (SOB), also medically known as dyspnea (in AmE) or dyspnoea (in BrE), is an uncomfortable feeling of not being able to breathe well enough. The American Thoracic Society defines it as "a subjective experience of breathing disc ...
, and chest pain
Chest pain is pain or discomfort in the chest, typically the front of the chest. It may be described as sharp, dull, pressure, heaviness or squeezing. Associated symptoms may include pain in the shoulder, arm, upper abdomen, or jaw, along with n ...
. Other symptoms of Lyme disease may also be present, such as EM rash, joint aches, facial palsy
Facial nerve paralysis is a common problem that involves the paralysis of any structures innervated by the facial nerve. The pathway of the facial nerve is long and relatively convoluted, so there are a number of causes that may result in facial ...
, headaches
Headache is the symptom of pain in the face, head, or neck. It can occur as a migraine, tension-type headache, or cluster headache. There is an increased risk of depression in those with severe headaches.
Headaches can occur as a result of m ...
, or radicular pain
Radicular pain, or radiculitis, is pain "radiated" along the dermatome (sensory distribution) of a nerve due to inflammation or other irritation of the nerve root (radiculopathy) at its connection to the spinal column. A common form of radiculit ...
. In some people, however, carditis may be the first manifestation of Lyme disease. Lyme carditis in 19–87% of people adversely impacts the heart's electrical conduction system, causing atrioventricular block
Block or blocked may refer to:
Arts, entertainment and media Broadcasting
* Block programming, the result of a programming strategy in broadcasting
* W242BX, a radio station licensed to Greenville, South Carolina, United States known as ''96.3 ...
that often manifests as heart rhythms that alternate within minutes between abnormally slow and abnormally fast. In 10–15% of people, Lyme causes myocardial
Cardiac muscle (also called heart muscle, myocardium, cardiomyocytes and cardiac myocytes) is one of three types of vertebrate muscle tissues, with the other two being skeletal muscle and smooth muscle. It is an involuntary, striated muscle that ...
complications such as cardiomegaly
Cardiomegaly (sometimes megacardia or megalocardia) is a medical condition in which the heart is enlarged. As such, it is more commonly referred to simply as "having an enlarged heart". It is usually the result of underlying conditions that make t ...
, left ventricular dysfunction, or congestive heart failure.
Another skin condition, found in Europe but not in North America, is borrelial lymphocytoma, a purplish lump that develops on the ear lobe, nipple, or scrotum
The scrotum or scrotal sac is an anatomical male reproductive structure located at the base of the penis that consists of a suspended dual-chambered sac of skin and smooth muscle. It is present in most terrestrial male mammals. The scrotum cont ...
.
Late disseminated infection
 Lyme arthritis occurs in up to 60% of untreated people, typically starting about six months after infection. It usually affects only one or a few joints, often a knee or possibly the
Lyme arthritis occurs in up to 60% of untreated people, typically starting about six months after infection. It usually affects only one or a few joints, often a knee or possibly the hip
In vertebrate anatomy, hip (or "coxa"Latin ''coxa'' was used by Celsus in the sense "hip", but by Pliny the Elder in the sense "hip bone" (Diab, p 77) in medical terminology) refers to either an anatomical region or a joint.
The hip region is ...
, other large joints, or the temporomandibular joint
In anatomy, the temporomandibular joints (TMJ) are the two joints connecting the jawbone to the skull. It is a bilateral synovial articulation between the temporal bone of the skull above and the mandible below; it is from these bones that it ...
. There is usually large joint effusion
A joint effusion is the presence of increased intra-articular fluid. It may affect any joint. Commonly it involves the knee.
Diagnostic approach
The approach to diagnosis depends on the joint involved. While aspiration of the joint is conside ...
and swelling, but only mild or moderate pain. Without treatment, swelling and pain typically resolve over time but periodically return. Baker's cyst
A Baker's cyst, also known as a popliteal cyst, is a type of fluid collection behind the knee. Often there are no symptoms. If symptoms do occur these may include swelling and pain behind the knee, or knee stiffness. If the cyst breaks open, pain ...
s may form and rupture.
In early US studies of Lyme disease, a rare peripheral neuropathy
Peripheral neuropathy, often shortened to neuropathy, is a general term describing disease affecting the peripheral nerves, meaning nerves beyond the brain and spinal cord. Damage to peripheral nerves may impair sensation, movement, gland, or or ...
was described that included numbness, tingling or burning starting at the feet or hands and over time possibly moving up the limbs. In a later analysis that discovered poor documentation of this manifestation, experts wondered if it exists at all in the US or is merely very rare.
A neurologic syndrome called Lyme encephalopathy is associated with subtle memory and cognitive difficulties, insomnia
Insomnia, also known as sleeplessness, is a sleep disorder in which people have trouble sleeping. They may have difficulty falling asleep, or staying asleep as long as desired. Insomnia is typically followed by daytime sleepiness, low energy, ...
, a general sense of feeling unwell, and changes in personality. However, problems such as depression and fibromyalgia
Fibromyalgia (FM) is a medical condition defined by the presence of chronic widespread pain, fatigue, waking unrefreshed, cognitive symptoms, lower abdominal pain or cramps, and depression. Other symptoms include insomnia and a general hyp ...
are as common in people with Lyme disease as in the general population.
'' Acrodermatitis chronica atrophicans'' (ACA) is a chronic skin disorder observed primarily in Europe among the elderly. ACA begins as a reddish-blue patch of discolored skin, often on the backs of the hands or feet. The lesion slowly atrophies over several weeks or months, with the skin becoming first thin and wrinkled and then, if untreated, completely dry and hairless. ACA is also associated with peripheral neuropathy.
Cause
 Lyme disease is caused by
Lyme disease is caused by spirochete
A spirochaete () or spirochete is a member of the phylum Spirochaetota (), (synonym Spirochaetes) which contains distinctive diderm (double-membrane) gram-negative bacteria, most of which have long, helically coiled (corkscrew-shaped or s ...
s, spiral bacteria
Spiral bacteria, bacteria of spiral ( helical) shape, form the third major morphological category of prokaryotes along with the rod-shaped bacilli and round cocci. Spiral bacteria can be subclassified by the number of twists per cell, cell thick ...
from the genus
Genus ( plural genera ) is a taxonomic rank used in the biological classification of extant taxon, living and fossil organisms as well as Virus classification#ICTV classification, viruses. In the hierarchy of biological classification, genus com ...
''Borrelia
''Borrelia'' is a genus of bacteria of the spirochete phylum. Several species cause Lyme disease, also called Lyme borreliosis, a zoonotic, vector-borne disease transmitted by ticks. Other species of ''Borrelia'' cause relapsing fever, and are t ...
''. Spirochetes are surrounded by peptidoglycan
Peptidoglycan or murein is a unique large macromolecule, a polysaccharide, consisting of sugars and amino acids that forms a mesh-like peptidoglycan layer outside the plasma membrane, the rigid cell wall (murein sacculus) characteristic of most ...
and flagella
A flagellum (; ) is a hairlike appendage that protrudes from certain plant and animal sperm cells, and from a wide range of microorganisms to provide motility. Many protists with flagella are termed as flagellates.
A microorganism may have f ...
, along with an outer membrane similar to Gram-negative bacteria. Because of their double-membrane envelope, ''Borrelia'' bacteria are often mistakenly described as Gram negative
The gram (originally gramme; SI unit symbol g) is a unit of mass in the International System of Units (SI) equal to one one thousandth of a kilogram.
Originally defined as of 1795 as "the absolute weight of a volume of pure water equal to th ...
despite the considerable differences in their envelope components from Gram-negative bacteria. The Lyme-related ''Borrelia'' species are collectively known as ''Borrelia burgdorferi
''Borrelia burgdorferi'' is a bacterial species of the spirochete class in the genus '' Borrelia'', and is one of the causative agents of Lyme disease in humans. Along with a few similar genospecies, some of which also cause Lyme disease, it mak ...
sensu lato
''Sensu'' is a Latin word meaning "in the sense of". It is used in a number of fields including biology, geology, linguistics, semiotics, and law. Commonly it refers to how strictly or loosely an expression is used in describing any particular c ...
'', and show a great deal of genetic diversity.
''B. burgdorferi sensu lato'' is a species complex made up of 20 accepted and three proposed genospecies. Eight species are known to cause Lyme disease: ''B. mayonii'' (found in North America), ''B. burgdorferi sensu stricto
''Sensu'' is a Latin word meaning "in the sense of". It is used in a number of fields including biology, geology, linguistics, semiotics, and law. Commonly it refers to how strictly or loosely an expression is used in describing any particular co ...
'', (found in North America and Europe), ''B. afzelii'', ''B. garinii, B. spielmanii, and B. lusitaniae'' (all found in Eurasia
Eurasia (, ) is the largest continental area on Earth, comprising all of Europe and Asia. Primarily in the Northern and Eastern Hemispheres, it spans from the British Isles and the Iberian Peninsula in the west to the Japanese archipelago a ...
). Some studies have also proposed that ''B. valaisiana'' may sometimes infect humans, but this species does not seem to be important causes of disease.
Tick life cycle
 There are three stages in the life cycle of a tick. Larva, nymph, and adult. During the nymph stage, ticks most frequently transmit Lyme disease and are usually most active in late spring and early summer in regions where the climate is mild. During the adult stage, Lyme disease transmission is less common due to the fact that it is less possible for adult ticks to bite humans and because they tend to be larger in size and therefore can be easily seen and removed.
There are three stages in the life cycle of a tick. Larva, nymph, and adult. During the nymph stage, ticks most frequently transmit Lyme disease and are usually most active in late spring and early summer in regions where the climate is mild. During the adult stage, Lyme disease transmission is less common due to the fact that it is less possible for adult ticks to bite humans and because they tend to be larger in size and therefore can be easily seen and removed.
Transmission
Lyme disease is classified as azoonosis
A zoonosis (; plural zoonoses) or zoonotic disease is an infectious disease of humans caused by a pathogen (an infectious agent, such as a bacterium, virus, parasite or prion) that has jumped from a non-human (usually a vertebrate) to a human. ...
, as it is transmitted to humans from a natural reservoir
In infectious disease ecology and epidemiology, a natural reservoir, also known as a disease reservoir or a reservoir of infection, is the population of organisms or the specific environment in which an infectious pathogen naturally lives and r ...
among small mammals and birds by tick
Ticks (order Ixodida) are parasitic arachnids that are part of the mite superorder Parasitiformes. Adult ticks are approximately 3 to 5 mm in length depending on age, sex, species, and "fullness". Ticks are external parasites, living by ...
s that feed on both sets of hosts
A host is a person responsible for guests at an event or for providing hospitality during it.
Host may also refer to:
Places
*Host, Pennsylvania, a village in Berks County
People
*Jim Host (born 1937), American businessman
*Michel Host ( ...
. Hard-bodied ticks of the genus ''Ixodes
''Ixodes'' is a genus of hard-bodied ticks (family Ixodidae). It includes important disease vectors of animals and humans ( tick-borne disease), and some species (notably '' Ixodes holocyclus'') inject toxins that can cause paralysis. Som ...
'' are the vectors of Lyme disease (also the vector for ''Babesia
''Babesia'', also called ''Nuttallia'', is an apicomplexan parasite that infects red blood cells and is transmitted by ticks. Originally discovered by the Romanian bacteriologist Victor Babeș in 1888, over 100 species of ''Babesia'' have since ...
''). Most infections are caused by ticks in the nymphal stage, because they are very small and thus may feed for long periods of time undetected. Nymphal ticks are generally the size of a poppy seed and sometimes with a dark head and a translucent body. Or, the nymphal ticks can be darker. (The younger larval ticks are very rarely infected.) Although deer are the preferred hosts of adult deer ticks, and tick populations are much lower in the absence of deer, ticks generally do not acquire ''Borrelia'' from deer, instead they obtain them from infected small mammals such as the white-footed mouse
The white-footed mouse (''Peromyscus leucopus'') is a rodent native to North America from Ontario, Quebec, Labrador, and the Maritime Provinces (excluding the island of Newfoundland) to the southwestern United States and Mexico. In the Maritimes, ...
, and occasionally birds. Areas where Lyme is common are expanding.
Within the tick midgut, the ''Borrelia''s outer surface protein A (OspA) binds to the tick receptor for OspA, known as TROSPA. When the tick feeds, the'' Borrelia'' downregulates OspA and upregulates OspC, another surface protein. After the bacteria migrate from the midgut to the salivary glands, OspC binds to Salp15, a tick salivary protein that appears to have immunosuppressive effects that enhance infection. Successful infection of the mammalian host depends on bacterial expression of OspC.
Tick bites often go unnoticed because of the small size of the tick in its nymphal stage, as well as tick secretions that prevent the host from feeling any itch or pain from the bite. However, transmission is quite rare, with only about 1.2 to 1.4 percent of recognized tick bites resulting in Lyme disease.
 In Europe, the main vector is ''
In Europe, the main vector is ''Ixodes ricinus
''Ixodes ricinus'', the castor bean tick, is a chiefly European species of hard-bodied tick. It may reach a length of when engorged with a blood meal, and can transmit both bacterial and viral pathogens such as the causative agents of Lyme disea ...
'', which is also called the sheep tick or castor bean
''Ricinus communis'', the castor bean or castor oil plant, is a species of perennial flowering plant in the spurge family, Euphorbiaceae. It is the sole species in the monotypic genus, ''Ricinus'', and subtribe, Ricininae. The evolution of ...
tick. In China, '' Ixodes persulcatus'' (the taiga tick) is probably the most important vector. In North America, the black-legged tick or deer tick (''Ixodes scapularis
''Ixodes scapularis'' is commonly known as the deer tick or black-legged tick (although some people reserve the latter term for ''Ixodes pacificus'', which is found on the west coast of the US), and in some parts of the US as the bear tick. It wa ...
'') is the main vector on the East Coast.
The lone star tick (''Amblyomma americanum
''Amblyomma americanum'', also known as the lone star tick, the northeastern water tick, or the turkey tick, or the "Cricker Tick", is a type of tick indigenous to much of the eastern United States and Mexico, that bites painlessly and commonly ...
''), which is found throughout the Southeastern United States as far west as Texas
Texas (, ; Spanish language, Spanish: ''Texas'', ''Tejas'') is a state in the South Central United States, South Central region of the United States. At 268,596 square miles (695,662 km2), and with more than 29.1 million residents in 2 ...
, is unlikely to transmit the Lyme disease spirochete
A spirochaete () or spirochete is a member of the phylum Spirochaetota (), (synonym Spirochaetes) which contains distinctive diderm (double-membrane) gram-negative bacteria, most of which have long, helically coiled (corkscrew-shaped or s ...
s, though it may be implicated in a related syndrome called southern tick-associated rash illness
Southern tick-associated rash illness (STARI) is an emerging infectious disease related to Lyme disease that occurs in southeastern and south-central United States. It is spread by tick bites and it was hypothesized that the illness was caused by ...
, which resembles a mild form of Lyme disease.
On the West Coast of the United States
The West Coast of the United States, also known as the Pacific Coast, Pacific states, and the western seaboard, is the coastline along which the Western United States meets the North Pacific Ocean. The term typically refers to the contiguous U.S ...
, the main vector is the western black-legged tick (''Ixodes pacificus
''Ixodes pacificus'', the western black-legged tick, is a species of parasitic tick found on the western coast of North America. ''I. pacificus'' is a member of the ''Ixodidae'' (hard-bodied) family. It is the principal vector of Lyme disease in ...
''). The tendency of this tick species to feed predominantly on host species such as the Western Fence Lizard
The western fence lizard (''Sceloporus occidentalis'') is a common lizard of Arizona, New Mexico, California, Idaho, Nevada, Oregon, Utah, Washington, Northern Mexico, and the surrounding area. As the ventral abdomen of an adult is characterist ...
that are resistant to ''Borrelia'' infection appears to diminish transmission of Lyme disease in the West.
Transmission can occur across the placenta
The placenta is a temporary embryonic and later fetal organ that begins developing from the blastocyst shortly after implantation. It plays critical roles in facilitating nutrient, gas and waste exchange between the physically separate mater ...
during pregnancy and as with a number of other spirochetal diseases, adverse pregnancy outcomes are possible with untreated infection; prompt treatment with antibiotics reduces or eliminates this risk.
There is no scientific evidence to support Lyme disease transmission via blood transfusion, sexual contact, or breast milk.
Tick-borne co-infections
Ticks that transmit ''B. burgdorferi'' to humans can also carry and transmit several other microbes, such as ''Babesia microti
Babesia microti is a parasitic blood-borne piroplasm transmitted by deer ticks. ''B. microti'' is responsible for the disease babesiosis, a malaria-like disease which also causes fever and hemolysis.
Life cycle
The life cycle of ''B. microti ...
'' and ''Anaplasma phagocytophilum
''Anaplasma phagocytophilum'' (formerly ''Ehrlichia phagocytophilum'') is a Gram-negative bacterium that is unusual in its tropism to neutrophils. It causes anaplasmosis in sheep and cattle, also known as tick-borne fever and pasture fever, and ...
'', which cause the diseases babesiosis
Babesiosis or piroplasmosis is a malaria-like parasitic disease caused by infection with a eukaryotic parasite in the order Piroplasmida, typically a ''Babesia'' or '' Theileria'', in the phylum Apicomplexa. Human babesiosis transmission via ...
and human granulocytic anaplasmosis
Human granulocytic anaplasmosis (HGA) is a tick-borne, infectious disease caused by ''Anaplasma phagocytophilum'', an obligate intracellular bacterium that is typically transmitted to humans by ticks of the '' Ixodes ricinus'' species complex, i ...
(HGA), respectively. Among people with early Lyme disease, depending on their location, 2–12% will also have HGA and 2–10% will have babesiosis. Ticks in certain regions also transmit viruses that cause tick-borne encephalitis
Tick-borne encephalitis (TBE) is a viral infectious disease involving the central nervous system. The disease most often manifests as meningitis, encephalitis or meningoencephalitis. Myelitis and spinal paralysis also occurs. In about one thir ...
and Powassan virus disease. Co-infections of Lyme disease may not require additional treatment, since they may resolve on their own or—as in the case of HGA—can be treated with the doxycycline prescribed for Lyme. Persistent fever or compatible anomalous laboratory findings may be indicative of a co-infection.
Pathophysiology
''B. burgdorferi'' can spread throughout the body during the course of the disease, and has been found in the skin, heart, joints, peripheral nervous system, and central nervous system. ''B. Burgdorferi'' does not produce toxins. Therefore, many of the signs and symptoms of Lyme disease are a consequence of the immune response to spirochete in those tissues. ''B. burgdorferi'' is injected into the skin by the bite of an infected ''Ixodes'' tick. Tick saliva, which accompanies the spirochete into the skin during the feeding process, contains substances that disrupt the immune response at the site of the bite. This provides a protective environment where the spirochete can establish infection. The spirochetes multiply and migrate outward within thedermis
The dermis or corium is a layer of skin between the epidermis (with which it makes up the cutis) and subcutaneous tissues, that primarily consists of dense irregular connective tissue and cushions the body from stress and strain. It is divided i ...
. The host inflammatory response to the bacteria in the skin causes the characteristic circular EM lesion. Neutrophils, however, which are necessary to eliminate the spirochetes from the skin, fail to appear in necessary numbers in the developing EM lesion because tick saliva inhibits neutrophil function. This allows the bacteria to survive and eventually spread throughout the body.
Days to weeks following the tick bite, the spirochetes spread via the bloodstream to joints, heart, nervous system, and distant skin sites, where their presence gives rise to the variety of symptoms of the disseminated disease. The spread of ''B. burgdorferi'' is aided by the attachment of the host protease plasmin
Plasmin is an important enzyme () present in blood that degrades many blood plasma proteins, including fibrin clots. The degradation of fibrin is termed fibrinolysis. In humans, the plasmin protein (in the zymogen form of plasminogen) is encode ...
to the surface of the spirochete.
If untreated, the bacteria may persist in the body for months or even years, despite the production of ''B. burgdorferi'' antibodies by the immune system. The spirochetes may avoid the immune response by decreasing expression of surface proteins that are targeted by antibodies, antigenic variation
Antigenic variation or antigenic alteration refers to the mechanism by which an infectious agent such as a protozoan, bacterium or virus alters the proteins or carbohydrates on its surface and thus avoids a host immune response, making it one of ...
of the VlsE surface protein, inactivating key immune components such as complement
A complement is something that completes something else.
Complement may refer specifically to:
The arts
* Complement (music), an interval that, when added to another, spans an octave
** Aggregate complementation, the separation of pitch-clas ...
, and hiding in the extracellular matrix
In biology, the extracellular matrix (ECM), also called intercellular matrix, is a three-dimensional network consisting of extracellular macromolecules and minerals, such as collagen, enzymes, glycoproteins and hydroxyapatite that provide stru ...
, which may interfere with the function of immune factors.
In the brain, ''B. burgdorferi'' may induce astrocytes
Astrocytes (from Ancient Greek , , "star" + , , "cavity", "cell"), also known collectively as astroglia, are characteristic star-shaped glial cells in the brain and spinal cord. They perform many functions, including biochemical control of endo ...
to undergo astrogliosis
Astrogliosis (also known as astrocytosis or referred to as reactive astrogliosis) is an abnormal increase in the number of astrocytes due to the destruction of nearby neurons from central nervous system (CNS) trauma, infection, ischemia, stroke, a ...
(proliferation followed by apoptosis
Apoptosis (from grc, ἀπόπτωσις, apóptōsis, 'falling off') is a form of programmed cell death that occurs in multicellular organisms. Biochemical events lead to characteristic cell changes (morphology) and death. These changes incl ...
), which may contribute to neurodysfunction. The spirochetes may also induce host cells to secrete quinolinic acid
Quinolinic acid (abbreviated QUIN or QA), also known as pyridine-2,3-dicarboxylic acid, is a dicarboxylic acid with a pyridine backbone. It is a colorless solid. It is the biosynthetic precursor to niacin.
Quinolinic acid is a downstream produ ...
, which stimulates the NMDA receptor on nerve cells, which may account for the fatigue and malaise observed with Lyme encephalopathy
Encephalopathy (; from grc, ἐνκέφαλος "brain" + πάθος "suffering") means any disorder or disease of the brain, especially chronic degenerative conditions. In modern usage, encephalopathy does not refer to a single disease, but r ...
. In addition, diffuse white matter pathology during Lyme encephalopathy may disrupt gray matter
Grey matter is a major component of the central nervous system, consisting of neuronal cell bodies, neuropil (dendrites and unmyelinated axons), glial cells (astrocytes and oligodendrocytes), synapses, and capillaries. Grey matter is distingui ...
connections, and could account for deficits in attention, memory, visuospatial ability, complex cognition, and emotional status. White matter disease may have a greater potential for recovery than gray matter disease, perhaps because neuronal loss is less common. Resolution of MRI white matter hyperintensities after antibiotic treatment has been observed.
Tryptophan
Tryptophan (symbol Trp or W)
is an α-amino acid that is used in the biosynthesis of proteins. Tryptophan contains an α-amino group, an α- carboxylic acid group, and a side chain indole, making it a polar molecule with a non-polar aromatic ...
, a precursor to serotonin
Serotonin () or 5-hydroxytryptamine (5-HT) is a monoamine neurotransmitter. Its biological function is complex and multifaceted, modulating mood, cognition, reward, learning, memory, and numerous physiological processes such as vomiting and vas ...
, appears to be reduced within the central nervous system
The central nervous system (CNS) is the part of the nervous system consisting primarily of the brain and spinal cord. The CNS is so named because the brain integrates the received information and coordinates and influences the activity of all par ...
in a number of infectious diseases that affect the brain, including Lyme. Researchers are investigating if this neurohormone secretion is the cause of neuropsychiatric
Neuropsychiatry or Organic Psychiatry is a branch of medicine that deals with psychiatry as it relates to neurology, in an effort to understand and attribute behavior to the interaction of neurobiology and social psychology factors. Within neurop ...
disorders developing in some people with borreliosis.
Immunological studies
Exposure to the ''Borrelia'' bacterium during Lyme disease possibly causes a long-lived and damaginginflammatory response
Inflammation (from la, inflammatio) is part of the complex biological response of body tissues to harmful stimuli, such as pathogens, damaged cells, or irritants, and is a protective response involving immune cells, blood vessels, and molecu ...
, a form of pathogen-induced autoimmune
In immunology, autoimmunity is the system of immune responses of an organism against its own healthy cells, tissues and other normal body constituents. Any disease resulting from this type of immune response is termed an "autoimmune disease". ...
disease. The production of this reaction might be due to a form of molecular mimicry Molecular mimicry is defined as the theoretical possibility that sequence similarities between foreign and self-peptides are sufficient to result in the cross-activation of autoreactive T or B cells by pathogen-derived peptides. Despite the preva ...
, where ''Borrelia'' avoids being killed by the immune system by resembling normal parts of the body's tissues.
Chronic symptoms from an autoimmune reaction could explain why some symptoms persist even after the spirochetes have been eliminated from the body. This hypothesis may explain why chronic arthritis persists after antibiotic therapy, similar to rheumatic fever
Rheumatic fever (RF) is an inflammatory disease that can involve the heart, joints, skin, and brain. The disease typically develops two to four weeks after a streptococcal throat infection. Signs and symptoms include fever, multiple painful jo ...
, but its wider application is controversial.
Diagnosis
Lyme disease is diagnosed based on symptoms, objective physical findings (such aserythema migrans
Erythema migrans or erythema chronicum migrans is an expanding rash often seen in the early stage of Lyme disease, and can also (but less commonly) be caused by southern tick-associated rash illness (STARI). Page last reviewed: October 22, 2015 ...
(EM) rash, facial palsy
Facial nerve paralysis is a common problem that involves the paralysis of any structures innervated by the facial nerve. The pathway of the facial nerve is long and relatively convoluted, so there are a number of causes that may result in facial ...
, or arthritis
Arthritis is a term often used to mean any disorder that affects joints. Symptoms generally include joint pain and stiffness. Other symptoms may include redness, warmth, swelling, and decreased range of motion of the affected joints. In som ...
), history of possible exposure to infected ticks
Ticks (order Ixodida) are parasitic arachnids that are part of the mite superorder Parasitiformes. Adult ticks are approximately 3 to 5 mm in length depending on age, sex, species, and "fullness". Ticks are external parasites, living by ...
, and possibly laboratory tests
A laboratory (; ; colloquially lab) is a facility that provides controlled conditions in which scientific or technological research, experiments, and measurement may be performed. Laboratory services are provided in a variety of settings: physici ...
. People with symptoms of early Lyme disease should have a total body skin examination for EM rashes and asked whether EM-type rashes had manifested within the last 1–2 months. Presence of an EM rash and recent tick exposure (i.e., being outdoors in a likely tick habitat where Lyme is common
Common may refer to:
Places
* Common, a townland in County Tyrone, Northern Ireland
* Boston Common, a central public park in Boston, Massachusetts
* Cambridge Common, common land area in Cambridge, Massachusetts
* Clapham Common, originally com ...
, within 30 days of the appearance of the rash) are sufficient for Lyme diagnosis; no laboratory confirmation is needed or recommended. Most people who get infected do not remember a tick or a bite, and the EM rash need not look like a bull's eye (most EM rashes in the U.S. do not) or be accompanied by any other symptoms. In the U.S., Lyme is most common in the New England
New England is a region comprising six states in the Northeastern United States: Connecticut, Maine, Massachusetts, New Hampshire, Rhode Island, and Vermont. It is bordered by the state of New York to the west and by the Canadian provinces ...
and Mid-Atlantic states and parts of Wisconsin
Wisconsin () is a state in the upper Midwestern United States. Wisconsin is the 25th-largest state by total area and the 20th-most populous. It is bordered by Minnesota to the west, Iowa to the southwest, Illinois to the south, Lake M ...
and Minnesota
Minnesota () is a state in the upper midwestern region of the United States. It is the 12th largest U.S. state in area and the 22nd most populous, with over 5.75 million residents. Minnesota is home to western prairies, now given over to ...
, but it is expanding into other areas. Several bordering areas of Canada also have high Lyme risk.
In the absence of an EM rash or history of tick exposure, Lyme diagnosis depends on laboratory confirmation. The bacteria that cause Lyme disease are difficult to observe directly in body tissues and also difficult and too time-consuming to grow in the laboratory. The most widely used tests look instead for presence of antibodies
An antibody (Ab), also known as an immunoglobulin (Ig), is a large, Y-shaped protein used by the immune system to identify and neutralize foreign objects such as pathogenic bacteria and viruses. The antibody recognizes a unique molecule of the ...
against those bacteria in the blood. A positive antibody test result does not by itself prove active infection but can confirm an infection that is suspected because of symptoms, objective findings, and history of tick exposure in a person. Because as many as 5–20% of the normal population have antibodies against Lyme, people without history and symptoms suggestive of Lyme disease should not be tested for Lyme antibodies: a positive result would likely be false, possibly causing unnecessary treatment.
In some cases, when history, signs, and symptoms are strongly suggestive of early disseminated Lyme disease, empiric treatment
Empiric therapy or empirical therapy is medical treatment or therapy based on experience and, more specifically, therapy begun on the basis of a clinical "educated guess" in the absence of complete or perfect information. Thus it is applied before ...
may be started and reevaluated as laboratory test results become available.
Laboratory testing
Tests for antibodies in the blood byELISA
The enzyme-linked immunosorbent assay (ELISA) (, ) is a commonly used analytical biochemistry assay, first described by Eva Engvall and Peter Perlmann in 1971. The assay uses a solid-phase type of enzyme immunoassay (EIA) to detect the presence ...
and Western blot
The western blot (sometimes called the protein immunoblot), or western blotting, is a widely used analytical technique in molecular biology and immunogenetics to detect specific proteins in a sample of tissue homogenate or extract. Besides detect ...
is the most widely used method for Lyme diagnosis. A two-tiered protocol is recommended by the Centers for Disease Control and Prevention
The Centers for Disease Control and Prevention (CDC) is the national public health agency of the United States. It is a United States federal agency, under the Department of Health and Human Services, and is headquartered in Atlanta, Georgi ...
(CDC): the sensitive ELISA test is performed first, and if it is positive or equivocal, then the more specific
Specific may refer to:
* Specificity (disambiguation)
* Specific, a cure or therapy for a specific illness
Law
* Specific deterrence, focussed on an individual
* Specific finding, intermediate verdict used by a jury in determining the fina ...
Western blot is run. The immune system
The immune system is a network of biological processes that protects an organism from diseases. It detects and responds to a wide variety of pathogens, from viruses to parasitic worms, as well as cancer cells and objects such as wood splinte ...
takes some time to produce antibodies in quantity. After Lyme infection onset, antibodies of types IgM
Immunoglobulin M (IgM) is one of several isotypes of antibody (also known as immunoglobulin) that are produced by vertebrates. IgM is the largest antibody, and it is the first antibody to appear in the response to initial exposure to an antig ...
and IgG
Immunoglobulin G (Ig G) is a type of antibody. Representing approximately 75% of serum antibodies in humans, IgG is the most common type of antibody found in blood circulation. IgG molecules are created and released by plasma B cells. Each IgG ...
usually can first be detected respectively at 2–4 weeks and 4–6 weeks, and peak at 6–8 weeks. When an EM rash first appears, detectable antibodies may not be present. Therefore, it is recommended that testing not be performed and diagnosis be based on the presence of the EM rash. Up to 30 days after suspected Lyme infection onset, infection can be confirmed by detection of IgM or IgG antibodies; after that, it is recommended that only IgG antibodies be considered. A positive IgM and negative IgG test result after the first month of infection is generally indicative of a false-positive result. The number of IgM antibodies usually collapses 4–6 months after infection, while IgG antibodies can remain detectable for years.
Other tests may be used in neuroborreliosis cases. In Europe, neuroborreliosis is usually caused by Borrelia garinii
''Borrelia garinii'' is a spirochete
A spirochaete () or spirochete is a member of the phylum Spirochaetota (), (synonym Spirochaetes) which contains distinctive diderm (double-membrane) gram-negative bacteria, most of which have long, ...
and almost always involves lymphocytic pleocytosis
Lymphocytic pleocytosis is an abnormal increase in the amount of lymphocytes in the cerebrospinal fluid (CSF). It is usually considered to be a sign of infection or inflammation within the nervous system, and is encountered in a number of neurol ...
, i.e. the densities of lymphocytes
A lymphocyte is a type of white blood cell (leukocyte) in the immune system of most vertebrates. Lymphocytes include natural killer cells (which function in cell-mediated, cytotoxic innate immunity), T cells (for cell-mediated, cytotoxic adap ...
(infection-fighting cells) and protein in the cerebrospinal fluid
Cerebrospinal fluid (CSF) is a clear, colorless body fluid found within the tissue that surrounds the brain and spinal cord of all vertebrates.
CSF is produced by specialised ependymal cells in the choroid plexus of the ventricles of the bra ...
(CSF) typically rise to characteristically abnormal levels, while glucose level remains normal. Additionally, the immune system produces antibodies against Lyme inside the intrathecal space, which contains the CSF. Demonstration by lumbar puncture
Lumbar puncture (LP), also known as a spinal tap, is a medical procedure in which a needle is inserted into the spinal canal, most commonly to collect cerebrospinal fluid (CSF) for diagnostic testing. The main reason for a lumbar puncture is to ...
and CSF analysis of pleocytosis and intrathecal antibody production are required for definite diagnosis of neuroborreliosis in Europe (except in cases of peripheral neuropathy associated with acrodermatitis chronica atrophicans, which usually is caused by Borrelia afzelii
''Borrelia afzelii'' is a species of '' Borrelia'' a bacterium that can infect various species of vertebrates and invertebrates.
Among 30 ''Borrelia'' known species, it is one of four which are likely to infect humans causing a variant of Lyme d ...
and confirmed by blood antibody tests). In North America, neuroborreliosis is caused by Borrelia burgdorferi
''Borrelia burgdorferi'' is a bacterial species of the spirochete class in the genus '' Borrelia'', and is one of the causative agents of Lyme disease in humans. Along with a few similar genospecies, some of which also cause Lyme disease, it mak ...
and may not be accompanied by the same CSF signs; they confirm a diagnosis of central nervous system (CNS) neuroborreliosis if positive, but do not exclude it if negative. American guidelines consider CSF analysis optional when symptoms appear to be confined to the peripheral nervous system (PNS), e.g. facial palsy without overt meningitis symptoms. Unlike blood and intrathecal antibody tests, CSF pleocytosis tests revert to normal after infection ends and therefore can be used as objective markers of treatment success and inform decisions on whether to retreat. In infection involving the PNS, electromyography
Electromyography (EMG) is a technique for evaluating and recording the electrical activity produced by skeletal muscles. EMG is performed using an instrument called an electromyograph to produce a record called an electromyogram. An electromyog ...
and nerve conduction studies
A nerve conduction study (NCS) is a medical diagnostic test commonly used to evaluate the function, especially the ability of electrical conduction, of the motor and sensory nerves of the human body. These tests may be performed by medical spec ...
can be used to monitor objectively the response to treatment.
In Lyme carditis, electrocardiograms
Electrocardiography is the process of producing an electrocardiogram (ECG or EKG), a recording of the heart's electrical activity. It is an electrogram of the heart which is a graph of voltage versus time of the electrical activity of the hear ...
are used to evidence heart conduction abnormalities, while echocardiography
An echocardiography, echocardiogram, cardiac echo or simply an echo, is an ultrasound of the heart.
It is a type of medical imaging of the heart, using standard ultrasound or Doppler ultrasound.
Echocardiography has become routinely used in t ...
may show myocardial
Cardiac muscle (also called heart muscle, myocardium, cardiomyocytes and cardiac myocytes) is one of three types of vertebrate muscle tissues, with the other two being skeletal muscle and smooth muscle. It is an involuntary, striated muscle that ...
dysfunction. Biopsy
A biopsy is a medical test commonly performed by a surgeon, interventional radiologist, or an interventional cardiologist. The process involves extraction of sample cells or tissues for examination to determine the presence or extent of a diseas ...
and confirmation of Borrelia cells in myocardial tissue may be used in specific cases but are usually not done because of risk of the procedure.
Polymerase chain reaction
The polymerase chain reaction (PCR) is a method widely used to rapidly make millions to billions of copies (complete or partial) of a specific DNA sample, allowing scientists to take a very small sample of DNA and amplify it (or a part of it) t ...
(PCR) tests for Lyme disease have also been developed to detect the genetic material ( DNA) of the Lyme disease spirochete. Culture or PCR are the current means for detecting the presence of the organism, as serologic
Serology is the scientific study of serum and other body fluids. In practice, the term usually refers to the diagnostic identification of antibodies in the serum. Such antibodies are typically formed in response to an infection (against a given mic ...
studies only test for antibodies
An antibody (Ab), also known as an immunoglobulin (Ig), is a large, Y-shaped protein used by the immune system to identify and neutralize foreign objects such as pathogenic bacteria and viruses. The antibody recognizes a unique molecule of the ...
of ''Borrelia''. PCR has the advantage of being much faster than culture. However, PCR tests are susceptible to false positive
A false positive is an error in binary classification in which a test result incorrectly indicates the presence of a condition (such as a disease when the disease is not present), while a false negative is the opposite error, where the test resul ...
results, e.g. by detection of debris of dead Borrelia cells or specimen contamination. Even when properly performed, PCR often shows false-negative results because few Borrelia cells can be found in blood and cerebrospinal fluid
Cerebrospinal fluid (CSF) is a clear, colorless body fluid found within the tissue that surrounds the brain and spinal cord of all vertebrates.
CSF is produced by specialised ependymal cells in the choroid plexus of the ventricles of the bra ...
(CSF) during infection. Hence, PCR tests are recommended only in special cases, e.g. diagnosis of Lyme arthritis, because it is a highly sensitive way of detecting ''ospA'' DNA in synovial fluid. Although sensitivity of PCR in CSF is low, its use may be considered when intrathecal antibody production test results are suspected of being falsely negative, e.g. in very early (< 6 weeks) neuroborreliosis or in immunosuppressed people.
Several other forms of laboratory testing for Lyme disease are available, some of which have not been adequately validated. OspA antigens, shed by live ''Borrelia'' bacteria into urine, are a promising technique being studied. The use of nanotrap particles for their detection is being looked at and the OspA has been linked to active symptoms of Lyme. High titer
Titer (American English) or titre (British English) is a way of expressing concentration. Titer testing employs serial dilution to obtain approximate quantitative information from an analytical procedure that inherently only evaluates as positiv ...
s of either immunoglobulin G (IgG) or immunoglobulin M (IgM) antibodies to ''Borrelia'' antigens indicate disease, but lower titers can be misleading, because the IgM antibodies may remain after the initial infection, and IgG antibodies may remain for years.
The CDC does not recommend urine antigen tests, PCR tests on urine, immunofluorescent staining for cell-wall-deficient forms of ''B. burgdorferi'', and lymphocyte transformation tests.
Imaging
Neuroimaging
Neuroimaging is the use of quantitative (computational) techniques to study the structure and function of the central nervous system, developed as an objective way of scientifically studying the healthy human brain in a non-invasive manner. Incr ...
is controversial in whether it provides specific patterns unique to neuroborreliosis
Neuroborreliosis is a disorder of the central nervous system. A neurological manifestation of Lyme disease, neuroborreliosis is caused by a systemic infection of spirochetes of the genus ''Borrelia.'' Symptoms of the disease include erythema migr ...
, but may aid in differential diagnosis
In healthcare, a differential diagnosis (abbreviated DDx) is a method of analysis of a patient's history and physical examination to arrive at the correct diagnosis. It involves distinguishing a particular disease or condition from others that p ...
and in understanding the pathophysiology of the disease. Though controversial, some evidence shows certain neuroimaging tests can provide data that are helpful in the diagnosis of a person. Magnetic resonance imaging
Magnetic resonance imaging (MRI) is a medical imaging technique used in radiology to form pictures of the anatomy and the physiological processes of the body. MRI scanners use strong magnetic fields, magnetic field gradients, and radio wave ...
(MRI) and single-photon emission computed tomography (SPECT) are two of the tests that can identify abnormalities in the brain of a person affected with this disease. Neuroimaging findings in an MRI include lesions in the periventricular white matter, as well as enlarged ventricles and cortical atrophy. The findings are considered somewhat unexceptional because the lesions have been found to be reversible following antibiotic treatment. Images produced using SPECT show numerous areas where an insufficient amount of blood is being delivered to the cortex and subcortical white matter. However, SPECT images are known to be nonspecific because they show a heterogeneous pattern in the imaging. The abnormalities seen in the SPECT images are very similar to those seen in people with cerebral vacuities and Creutzfeldt–Jakob disease
Creutzfeldt–Jakob disease (CJD), also known as subacute spongiform encephalopathy or neurocognitive disorder due to prion disease, is an invariably fatal degenerative brain disorder. Early symptoms include memory problems, behavioral changes, ...
, which makes them questionable.
Differential diagnosis
Community clinics have been reported to misdiagnose 23–28% ofErythema migrans
Erythema migrans or erythema chronicum migrans is an expanding rash often seen in the early stage of Lyme disease, and can also (but less commonly) be caused by southern tick-associated rash illness (STARI). Page last reviewed: October 22, 2015 ...
(EM) rashes and 83% of other objective manifestations of early Lyme disease. EM rashes are often misdiagnosed as spider bites
A spider bite, also known as arachnidism, is an injury resulting from the bite of a spider. The effects of most bites are not serious. Most bites result in mild symptoms around the area of the bite. Rarely they may produce a necrotic skin wound ...
, cellulitis
Cellulitis is usually a bacterial infection involving the inner layers of the skin. It specifically affects the dermis and subcutaneous fat. Signs and symptoms include an area of redness which increases in size over a few days. The borders of ...
, or shingles
Shingles, also known as zoster or herpes zoster, is a viral disease characterized by a painful skin rash with blisters in a localized area. Typically the rash occurs in a single, wide mark either on the left or right side of the body or face. ...
. Many misdiagnoses are credited to the widespread misconception that EM rashes should look like a bull's eye. Actually, the key distinguishing features of the EM rash are the speed and extent to which it expands, respectively up to 2–3 cm/day and a diameter of at least 5 cm, and in 50% of cases more than 16 cm. The rash expands away from its center, which may or may not look different or be separated by ring-like clearing from the rest of the rash. Compared to EM rashes, spider bites
A spider bite, also known as arachnidism, is an injury resulting from the bite of a spider. The effects of most bites are not serious. Most bites result in mild symptoms around the area of the bite. Rarely they may produce a necrotic skin wound ...
are more common in the limbs, tend to be more painful and itchy or become swollen, and some may cause necrosis
Necrosis () is a form of cell injury which results in the premature death of cells in living tissue by autolysis. Necrosis is caused by factors external to the cell or tissue, such as infection, or trauma which result in the unregulated dige ...
(sinking dark blue patch of dead skin). Cellulitis
Cellulitis is usually a bacterial infection involving the inner layers of the skin. It specifically affects the dermis and subcutaneous fat. Signs and symptoms include an area of redness which increases in size over a few days. The borders of ...
most commonly develops around a wound or ulcer, is rarely circular, and is more likely to become swollen and tender. EM rashes often appear at sites that are unusual for cellulitis, such as the armpit, groin, abdomen, or back of knee. Like Lyme, shingles
Shingles, also known as zoster or herpes zoster, is a viral disease characterized by a painful skin rash with blisters in a localized area. Typically the rash occurs in a single, wide mark either on the left or right side of the body or face. ...
often begins with headache, fever, and fatigue, which are followed by pain or numbness. However, unlike Lyme, in shingles these symptoms are usually followed by appearance of rashes composed of multiple small blisters along with a nerve's dermatome Dermatome may refer to:
* Dermatome (anatomy), an area of skin that is supplied by a single pair of dorsal roots
* Dermatome (embryology), the portion of the embryonic paraxial mesoderm, the somite, which gives rise to dermis
* Dermatome (instrument ...
, and shingles can also be confirmed by quick laboratory tests.
Facial palsy
Facial nerve paralysis is a common problem that involves the paralysis of any structures innervated by the facial nerve. The pathway of the facial nerve is long and relatively convoluted, so there are a number of causes that may result in facial ...
caused by Lyme disease (LDFP) is often misdiagnosed as Bell's palsy. Although Bell's palsy is the most common type of one-sided facial palsy (about 70% of cases), LDFP can account for about 25% of cases of facial palsy in areas where Lyme disease is common. Compared to LDFP, Bell's palsy much less frequently affects both sides of the face. Even though LDFP and Bell's palsy have similar symptoms and evolve similarly if untreated, corticosteroid treatment is beneficial for Bell's Palsy, while being detrimental for LDFP. Recent history of exposure to a likely tick habitat during warmer months, EM rash, viral-like symptoms such as headache and fever, and/or palsy in both sides of the face should be evaluated for the likelihood of LDFP; if it is more than minimal, empiric therapy
Empiric therapy or empirical therapy is medical treatment or therapy based on experience and, more specifically, therapy begun on the basis of a clinical "educated guess" in the absence of complete or perfect information. Thus it is applied before ...
with antibiotics should be initiated, without corticosteroids, and reevaluated upon completion of laboratory tests for Lyme disease.
Unlike viral meningitis
Viral meningitis, also known as aseptic meningitis, is a type of meningitis due to a viral infection. It results in inflammation of the meninges (the membranes covering the brain and spinal cord). Symptoms commonly include headache, fever, sen ...
, Lyme lymphocytic meningitis tends to not cause fever, last longer, and recur. Lymphocytic meningitis is also characterized by possibly co-occurring with EM rash, facial palsy
Facial nerve paralysis is a common problem that involves the paralysis of any structures innervated by the facial nerve. The pathway of the facial nerve is long and relatively convoluted, so there are a number of causes that may result in facial ...
, or partial vision obstruction and having much lower percentage of polymorphonuclear leukocytes in CSF.
Lyme radiculopathy affecting the limbs is often misdiagnosed as a radiculopathy
Radiculopathy, also commonly referred to as pinched nerve, refers to a set of conditions in which one or more nerves are affected and do not work properly (a neuropathy). Radiculopathy can result in pain (radicular pain), weakness, altered sens ...
caused by nerve root compression, such as sciatica
Sciatica is pain going down the leg from the lower back. This pain may go down the back, outside, or front of the leg. Onset is often sudden following activities like heavy lifting, though gradual onset may also occur. The pain is often described ...
. Although most cases of radiculopathy are compressive and resolve with conservative treatment (e.g., rest) within 4–6 weeks, guidelines for managing radiculopathy recommend first evaluating risks of other possible causes that, although less frequent, require immediate diagnosis and treatment, including infections such as Lyme and shingles. A history of outdoor activities in likely tick habitats in the last 3 months possibly followed by a rash or viral-like symptoms, and current headache, other symptoms of lymphocytic meningitis, or facial palsy would lead to suspicion of Lyme disease and recommendation of serological
Serology is the scientific study of serum and other body fluids. In practice, the term usually refers to the diagnostic identification of antibodies in the serum. Such antibodies are typically formed in response to an infection (against a given mi ...
and lumbar puncture
Lumbar puncture (LP), also known as a spinal tap, is a medical procedure in which a needle is inserted into the spinal canal, most commonly to collect cerebrospinal fluid (CSF) for diagnostic testing. The main reason for a lumbar puncture is to ...
tests for confirmation.
Lyme radiculopathy affecting the trunk can be misdiagnosed as myriad other conditions, such as diverticulitis and acute coronary syndrome
Acute coronary syndrome (ACS) is a syndrome (a set of signs and symptoms) due to decreased blood flow in the coronary arteries such that part of the heart muscle is unable to function properly or dies. The most common symptom is centrally loca ...
. Diagnosis of late-stage Lyme disease is often complicated by a multifaceted appearance and nonspecific symptoms, prompting one reviewer to call Lyme the new "great imitator". As all people with later-stage infection will have a positive antibody test, simple blood tests can exclude Lyme disease as a possible cause of a person's symptoms.
Prevention
Tick bites may be prevented by avoiding or reducing time in likely tick habitats and taking precautions while in and when getting out of one. Most Lyme human infections are caused byIxodes
''Ixodes'' is a genus of hard-bodied ticks (family Ixodidae). It includes important disease vectors of animals and humans ( tick-borne disease), and some species (notably '' Ixodes holocyclus'') inject toxins that can cause paralysis. Som ...
nymph bites between April and September. Ticks prefer moist, shaded locations in woodland
A woodland () is, in the broad sense, land covered with trees, or in a narrow sense, synonymous with wood (or in the U.S., the ''plurale tantum'' woods), a low-density forest forming open habitats with plenty of sunlight and limited shade (see ...
s, shrubs, tall grasses and leaf litter or wood piles. Tick densities tend to be highest in woodlands, followed by unmaintained edges between woods and lawns (about half as high), ornamental plants and perennial groundcover (about a quarter), and lawns (about 30 times less). Ixodes larva
A larva (; plural larvae ) is a distinct juvenile form many animals undergo before metamorphosis into adults. Animals with indirect development such as insects, amphibians, or cnidarians typically have a larval phase of their life cycle.
The ...
e and nymphs tend to be abundant also where mice nest, such as stone walls and wood logs. Ixodes larvae and nymphs typically wait for potential hosts
A host is a person responsible for guests at an event or for providing hospitality during it.
Host may also refer to:
Places
*Host, Pennsylvania, a village in Berks County
People
*Jim Host (born 1937), American businessman
*Michel Host ( ...
("quest") on leaves or grasses close to the ground with forelegs outstretched; when a host brushes against its limbs, the tick rapidly clings and climbs on the host looking for a skin location to bite. In Northeastern United States, 69% of tick bites are estimated to happen in residences, 11% in schools or camps, 9% in parks or recreational areas, 4% at work, 3% while hunting, and 4% in other areas. Activities associated with tick bites around residences include yard work, brush clearing, gardening, playing in the yard, and letting into the house dogs or cats that roam outside in woody or grassy areas. In parks, tick bites often happen while hiking or camping. Walking on a mowed lawn or center of a trail without touching adjacent vegetation is less risky than crawling or sitting on a log or stone wall. Pets should not be allowed to roam freely in likely tick habitats.
As a precaution, CDC
The Centers for Disease Control and Prevention (CDC) is the national public health agency of the United States. It is a United States federal agency, under the Department of Health and Human Services, and is headquartered in Atlanta, Georgi ...
recommends soaking or spraying clothes, shoes, and camping gear such as tents, backpacks and sleeping bags with 0.5% permethrin
Permethrin is a medication and an insecticide. As a medication, it is used to treat scabies and lice. It is applied to the skin as a cream or lotion. As an insecticide, it can be sprayed onto clothing or mosquito nets to kill the insects th ...
solution and hanging them to dry before use. Permethrin is odorless and safe for humans but highly toxic to ticks. After crawling on permethrin-treated fabric for as few as 10–20 seconds, tick nymphs become irritated and fall off or die. Permethrin-treated closed-toed shoes and socks reduce by 74 times the number of bites from nymphs that make first contact with a shoe of a person also wearing treated shorts (because nymphs usually quest near the ground, this is a typical contact scenario). Better protection can be achieved by tucking permethrin-treated trousers (pants) into treated socks and a treated long-sleeve shirt into the trousers so as to minimize gaps through which a tick might reach the wearer's skin. Light-colored clothing may make it easier to see ticks and remove them before they bite. Military and outdoor workers' uniforms treated with permethrin have been found to reduce the number of bite cases by 80–95%. Permethrin protection lasts several weeks of wear and washings in customer-treated items and up to 70 washings for factory-treated items. Permethrin should not be used on human skin, underwear or cats.
The EPA
The Environmental Protection Agency (EPA) is an independent executive agency of the United States federal government tasked with environmental protection matters. President Richard Nixon proposed the establishment of EPA on July 9, 1970; it be ...
recommends several tick repellents for use on exposed skin, including DEET, picaridin
Icaridin, also known as picaridin, is an insect repellent which can be used directly on skin or clothing. It has broad efficacy against various arthropods such as mosquitos, ticks, gnats, flies and fleas, and is almost colorless and odorless. A s ...
, IR3535
Ethyl butylacetylaminopropionate (trade name IR3535) is an insect repellent which is applied topically to prevent bites and stings from mosquitos, ticks, and other insects. It is a colorless and almost odorless oil, has efficacy against a broad ran ...
(a derivative of amino acid beta-alanine), oil of lemon eucalyptus (OLE, a natural compound) and OLE's active ingredient para-menthane-diol (PMD). Unlike permethrin, repellents repel but do not kill ticks, protect for only several hours after application, and may be washed off by sweat or water. The most popular repellent is DEET in the U.S. and picaridin in Europe. Unlike DEET, picaridin is odorless and is less likely to irritate the skin or harm fabric or plastics. Repellents with higher concentration may last longer but are not more effective; against ticks, 20% picaridin may work for 8 hours vs. 55–98.11% DEET for 5–6 hours or 30–40% OLE for 6 hours. Repellents should not be used under clothes, on eyes, mouth, wounds or cuts, or on babies younger than 2 months (3 years for OLE or PMD). If sunscreen
Sunscreen, also known as sunblock or sun cream, is a photoprotective topical product for the skin that mainly absorbs, or to a much lesser extent reflects, some of the sun's ultraviolet (UV) radiation and thus helps protect against sunburn and ...
is used, repellent should be applied on top of it. Repellents should not be sprayed directly on a face, but should instead be sprayed on a hand and then rubbed on the face.
After coming indoors, clothes, gear and pets should be checked for ticks. Clothes can be put into a hot dryer for 10 minutes to kill ticks (just washing or warm dryer are not enough). Showering as soon as possible, looking for ticks over the entire body, and removing them reduce risk of infection. Unfed tick nymphs are the size of a poppy seed, but a day or two after biting and attaching themselves to a person, they look like a small blood blister
A blood blister is a type of blister that forms when subdermal tissue (biology), tissues and blood vessels are damaged without piercing the skin. It consists of a pool of lymph, blood and other body fluids trapped beneath the skin. If punctured ...
. The following areas should be checked especially carefully: armpits, between legs, back of knee, bellybutton, trunk, and in children ears, neck and hair.
Tick removal
CDC
The Centers for Disease Control and Prevention (CDC) is the national public health agency of the United States. It is a United States federal agency, under the Department of Health and Human Services, and is headquartered in Atlanta, Georgi ...
recommends inserting a fine-tipped tweezer between the skin and the tick, grasping very firmly, and pulling the closed tweezer straight away from the skin without twisting, jerking, squeezing or crushing the tick. After tick removal, any tick parts remaining in the skin should be removed with a clean tweezer, if possible. The wound and hands should then be cleaned with alcohol or soap and water. The tick may be disposed by placing it in a container with alcohol, sealed bag, tape or flushed down the toilet. The bitten person should write down where and when the bite happened so that this can be informed to a doctor if the person gets a rash or flu-like symptoms in the following several weeks. CDC recommends not using fingers, nail polish, petroleum jelly or heat on the tick to try to remove it.
In Australia, where the Australian paralysis tick is prevalent, the Australasian Society of Clinical Immunology and Allergy
The Australasian Society of Clinical Immunology and Allergy (ASCIA) is the peak professional body of clinical immunologists, allergy specialists, immunology scientists, and other health professionals working in allergy and immunology in Australi ...
recommends not using tweezers to remove ticks, because if the person is allergic, anaphylaxis
Anaphylaxis is a serious, potentially fatal allergic reaction and medical emergency that is rapid in onset and requires immediate medical attention regardless of use of emergency medication on site. It typically causes more than one of the follow ...
could result. Instead, a product should be sprayed on the tick to cause it to freeze and then drop off. A doctor would use liquid nitrogen, but products available from chemists for freezing warts can be used instead. Another method originating from Australia consists in using about 20 cm of dental floss or fishing line for slowly tying an overhand knot between the skin and the tick and then pulling it away from the skin.
Preventive antibiotics
The risk of infectious transmission increases with the duration of tick attachment. It requires between 36 and 48 hours of attachment for the bacteria that causes Lyme to travel from within the tick into its saliva. If a deer tick that is sufficiently likely to be carrying ''Borrelia'' is found attached to a person and removed, and if the tick has been attached for 36 hours or is engorged, a single dose of doxycycline administered within the 72 hours after removal may reduce the risk of Lyme disease. It is not generally recommended for all people bitten, as development of infection is rare: about 50 bitten people would have to be treated this way to prevent one case of erythema migrans (i.e. the typical rash found in about 70–80% of people infected).Garden landscaping
Several landscaping practices may reduce the risk of tick bites in residential yards. These include keeping lawns mowed, removing leaf litter and weeds, and avoiding the use of ground cover. A 3-ft-wide rock or woodchip barrier is recommended to separate lawns from wood piles,woodland
A woodland () is, in the broad sense, land covered with trees, or in a narrow sense, synonymous with wood (or in the U.S., the ''plurale tantum'' woods), a low-density forest forming open habitats with plenty of sunlight and limited shade (see ...
s, stone walls, and shrubs. Without vegetation on the barrier, ticks will tend not to cross it; acaricides
Acaricides are pesticides that kill members of the arachnid subclass ''Acari'', which includes ticks and mites.
Acaricides are used both in medicine and agriculture, although the desired selective toxicity differs between the two fields.
Termino ...
may also be sprayed on it to kill ticks. A sun-exposed tick-safe zone at least 9 ft from the barrier should concentrate human activity on the yard, including any patios, playgrounds and gardening. Materials such as wood decking, concrete, bricks, gravel or woodchips used on the ground under patios and playgrounds would discourage ticks there. An 8-ft-high fence may be added to keep deer away from the tick-safe zone.
Occupational exposure
Outdoor workers are at risk of Lyme disease if they work at sites with infected ticks. This includes construction, landscaping, forestry, brush clearing, land surveying, farming, railroad work, oil field work, utility line work, park or wildlife management. U.S. workers in the northeastern and north-central states are at highest risk of exposure to infected ticks. Ticks may also transmit other tick-borne diseases to workers in these and other regions of the country. Worksites with woods, bushes, high grass or leaf litter are likely to have more ticks. Outdoor workers should be most careful to protect themselves in the late spring and summer when young ticks are most active.Host animals
Ticks can feed upon the blood of a wide array of possible host species, including lizards,birds
Birds are a group of warm-blooded vertebrates constituting the class Aves (), characterised by feathers, toothless beaked jaws, the laying of hard-shelled eggs, a high metabolic rate, a four-chambered heart, and a strong yet lightweigh ...
, mice
A mouse ( : mice) is a small rodent. Characteristically, mice are known to have a pointed snout, small rounded ears, a body-length scaly tail, and a high breeding rate. The best known mouse species is the common house mouse (''Mus musculus' ...
, cats, dogs
The dog (''Canis familiaris'' or ''Canis lupus familiaris'') is a domesticated descendant of the wolf. Also called the domestic dog, it is derived from the extinct Pleistocene wolf, and the modern wolf is the dog's nearest living relative. Do ...
, deer
Deer or true deer are hoofed ruminant mammals forming the family Cervidae. The two main groups of deer are the Cervinae, including the muntjac, the elk (wapiti), the red deer, and the fallow deer; and the Capreolinae, including the reindeer ...
, cattle
Cattle (''Bos taurus'') are large, domesticated, cloven-hooved, herbivores. They are a prominent modern member of the subfamily Bovinae and the most widespread species of the genus ''Bos''. Adult females are referred to as cows and adult mal ...
and humans
Humans (''Homo sapiens'') are the most abundant and widespread species of primate, characterized by bipedalism and exceptional cognitive skills due to a large and complex brain. This has enabled the development of advanced tools, culture, ...
. The extent to which a tick can feed, reproduce, and spread will depend on the type and availability of its hosts. Whether it will spread disease is also affected by its available hosts. Some species, such as lizards, are referred to as "dilution hosts" because they don't tend to support Lyme disease pathogens and so decrease the likelihood that the disease will be passed on by ticks feeding on them. White-tailed deer
The white-tailed deer (''Odocoileus virginianus''), also known as the whitetail or Virginia deer, is a medium-sized deer native to North America, Central America, and South America as far south as Peru and Bolivia. It has also been introduced t ...
are both a food source and a "reproductive host", where ticks tend to mate. The white-footed mouse
The white-footed mouse (''Peromyscus leucopus'') is a rodent native to North America from Ontario, Quebec, Labrador, and the Maritime Provinces (excluding the island of Newfoundland) to the southwestern United States and Mexico. In the Maritimes, ...
is a reservoir host in which the pathogen for Lyme disease can survive. Availability of hosts can have significant impacts on the transmission of Lyme disease. A greater diversity of hosts, or of those that don't support the pathogen, tends to decrease the likelihood that the disease will be transmitted.
In the United States, one approach to reducing the incidence of Lyme and other deer tick-borne diseases has been to greatly reduce the deer population on which the adult ticks depend for feeding and reproduction. Lyme disease cases fell following deer eradication on an island, Monhegan, Maine
Monhegan () is an island in the Gulf of Maine located in Lincoln County, Maine, United States. A plantation, a minor civil division in the state of Maine falling between unincorporated area and a town, it is located about off the mainland. Th ...
, and following deer control in Mumford Cove, Connecticut. Advocates have suggested reducing the deer population to levels of 8 to 10 deer per square mile, compared to levels of 60 or more deer per square mile in the areas of the country with the highest Lyme disease rates.
Others have noted that while deer are reproductive hosts, they are not ''Borrelia burgdorferi'' reservoirs. Researchers have suggested that smaller, less obviously visible Lyme reservoirs, like white-footed mice
The white-footed mouse (''Peromyscus leucopus'') is a rodent native to North America from Ontario, Quebec, Labrador, and the Maritime Provinces (excluding the island of Newfoundland) to the southwestern United States and Mexico. In the Maritimes, ...
and Eastern chipmunks, may more strongly impact Lyme disease occurrence. Ecosystem studies in New York state suggest that white-footed mice thrive when forests are broken into smaller isolated chunks of woodland with fewer rodent predators. With more rodents harboring the disease, the odds increase that a tick will feed on a disease-harboring rodent and that someone will pick up a disease-carrying tick in their garden or walking in the woods. Data indicates that the smaller the wooded area, the more ticks it will contain and the likely they are to carry Lyme disease, supporting the idea that deforestation
Deforestation or forest clearance is the removal of a forest or stand of trees from land that is then converted to non-forest use. Deforestation can involve conversion of forest land to farms, ranches, or urban use. The most concentrated d ...
and habitat fragmentation affect ticks, hosts and disease transmission.
Tick-borne diseases are estimated to affect ~80 % of cattle
Cattle (''Bos taurus'') are large, domesticated, cloven-hooved, herbivores. They are a prominent modern member of the subfamily Bovinae and the most widespread species of the genus ''Bos''. Adult females are referred to as cows and adult mal ...
worldwide. They also affect cats, dogs, and other pets. Routine veterinary control of ticks of domestic animals
Ticks of domestic animals directly cause poor health and loss of production to their hosts. Ticks also transmit numerous kinds of viruses, bacteria, and protozoa between domestic animals. These microbes cause diseases which can be severely debilita ...
through the use of acaricide
Acaricides are pesticides that kill members of the arachnid subclass ''Acari'', which includes ticks and mites.
Acaricides are used both in medicine and agriculture, although the desired selective toxicity differs between the two fields.
Termino ...
s has been suggested as a way to reduce exposure of humans to ticks. However, chemical control with acaricides is now criticized on a number of grounds. Ticks appear to develop resistance to acaricides; acaricides are costly; and there are concerns over their toxicity and the potential for chemical residues to affect food and the environment.
In Europe, known reservoirs of ''Borrelia burgdorferi'' were 9 small mammals, 7 medium-sized mammals and 16 species of birds (including passerines, sea-birds and pheasants). These animals seem to transmit spirochetes to ticks and thus participate in the natural circulation of B. burgdorferi in Europe. The house mouse is also suspected as well as other species of small rodents, particularly in Eastern Europe and Russia. "The reservoir species that contain the most pathogens are the European roe deer ''Capreolus capreolus
The roe deer (''Capreolus capreolus''), also known as the roe, western roe deer, or European roe, is a species of deer. The male of the species is sometimes referred to as a roebuck. The roe is a small deer, reddish and grey-brown, and well-adapt ...
''; "''it does not appear to serve as a major reservoir of B. burgdorferi''" thought Jaenson & al. (1992) (incompetent host for ''B. burgdorferi'' and TBE virus) but it is important for feeding the ticks, as red deer
The red deer (''Cervus elaphus'') is one of the largest deer species. A male red deer is called a stag or hart, and a female is called a hind. The red deer inhabits most of Europe, the Caucasus Mountains region, Anatolia, Iran, and parts of wes ...
and wild boars
The wild boar (''Sus scrofa''), also known as the wild swine, common wild pig, Eurasian wild pig, or simply wild pig, is a suid native to much of Eurasia and North Africa, and has been introduced to the Americas and Oceania. The species is n ...
('' Sus scrofa''), in which one ''Rickettsia'' and three ''Borrelia'' species were identified", with high risks of coinfection in roe deer. Nevertheless, in the 2000s, in roe deer in Europe "''two species of Rickettsia and two species of Borrelia were identified''".
Vaccination
No human vaccines for Lyme disease are currently available. The only human vaccine to advance to market was LYMErix, which was available briefly before being pulled from the market in 2002. The vaccine candidate VLA15 is scheduled to start a phase 3 trial in the third quarter of 2022, and other research is ongoing. Multiple vaccines are available for the prevention of Lyme disease in dogs.LYMErix
The vaccine LYMErix was available from 1998 to 2002. Therecombinant vaccine
A vaccine is a biological preparation that provides active acquired immunity to a particular infectious or malignant disease. The safety and effectiveness of vaccines has been widely studied and verified.< ...
against Lyme disease, based on the outer surface protein A (OspA) of ''B. burgdorferi'' with aluminum hydroxide as adjuvant, was developed by SmithKline Beecham
GSK plc, formerly GlaxoSmithKline plc, is a British multinational pharmaceutical and biotechnology company with global headquarters in London, England. Established in 2000 by a merger of Glaxo Wellcome and SmithKline Beecham. GSK is the tent ...
. In clinical trial
Clinical trials are prospective biomedical or behavioral research studies on human participants designed to answer specific questions about biomedical or behavioral interventions, including new treatments (such as novel vaccines, drugs, dietar ...
s involving more than 10,000 people, the vaccine was found to confer protective immunity to Lyme disease in 76% of adults after three doses with only mild or moderate and transient adverse effect
An adverse effect is an undesired harmful effect resulting from a medication or other intervention, such as surgery. An adverse effect may be termed a "side effect", when judged to be secondary to a main or therapeutic effect. The term complica ...
s. On 21 December 1998, the Food and Drug Administration
The United States Food and Drug Administration (FDA or US FDA) is a List of United States federal agencies, federal agency of the United States Department of Health and Human Services, Department of Health and Human Services. The FDA is respon ...
(FDA) approved LYMErix on the basis of these trials for persons of ages 15 through 70.
Following approval of the vaccine
A vaccine is a biological Dosage form, preparation that provides active acquired immunity to a particular infectious disease, infectious or cancer, malignant disease. The safety and effectiveness of vaccines has been widely studied and verifie ...
, its entry into clinical practice was slow for a variety of reasons, including its cost, which was often not reimbursed by insurance companies. Subsequently, hundreds of vaccine recipients reported they had developed autoimmune
In immunology, autoimmunity is the system of immune responses of an organism against its own healthy cells, tissues and other normal body constituents. Any disease resulting from this type of immune response is termed an "autoimmune disease". ...
and other side effects. Supported by some advocacy groups, a number of class-action lawsuit
A class action, also known as a class-action lawsuit, class suit, or representative action, is a type of lawsuit where one of the parties is a group of people who are represented collectively by a member or members of that group. The class actio ...
s were filed against GlaxoSmithKline
GSK plc, formerly GlaxoSmithKline plc, is a British multinational pharmaceutical and biotechnology company with global headquarters in London, England. Established in 2000 by a merger of Glaxo Wellcome and SmithKline Beecham. GSK is the ten ...
, alleging the vaccine had caused these health problems. These claims were investigated by the FDA and the Centers for Disease Control, which found no connection between the vaccine and the autoimmune complaints.
Despite the lack of evidence that the complaints were caused by the vaccine, sales plummeted and LYMErix was withdrawn from the U.S. market by GlaxoSmithKline in February 2002, in the setting of negative media coverage and fears of vaccine side effects. The fate of LYMErix was described in the medical literature as a "cautionary tale"; an editorial in ''Nature
Nature, in the broadest sense, is the physics, physical world or universe. "Nature" can refer to the phenomenon, phenomena of the physical world, and also to life in general. The study of nature is a large, if not the only, part of science. ...
'' cited the withdrawal of LYMErix as an instance in which "unfounded public fears place pressures on vaccine developers that go beyond reasonable safety considerations." The original developer of the OspA vaccine at the Max Planck Institute
Max or MAX may refer to:
Animals
* Max (dog) (1983–2013), at one time purported to be the world's oldest living dog
* Max (English Springer Spaniel), the first pet dog to win the PDSA Order of Merit (animal equivalent of OBE)
* Max (gorilla) ...
told ''Nature'': "This just shows how irrational the world can be ... There was no scientific justification for the first OspA vaccine LYMErix being pulled."
VLA15
The hexavalent (OspA) protein subunit-based vaccine candidate VLA15 was developed by Valneva. It was grantedfast track designation
Fast track is a designation by the United States Food and Drug Administration (FDA) of an investigational drug for expedited review to facilitate development of drugs that treat a serious or life-threatening condition and fill an unmet medical ...
by the U.S. Food and Drug Administration
The United States Food and Drug Administration (FDA or US FDA) is a List of United States federal agencies, federal agency of the United States Department of Health and Human Services, Department of Health and Human Services. The FDA is respon ...
in July 2017. In April 2020 Pfizer
Pfizer Inc. ( ) is an American multinational pharmaceutical and biotechnology corporation headquartered on 42nd Street in Manhattan, New York City. The company was established in 1849 in New York by two German entrepreneurs, Charles Pfizer ...
paid $130 million for the rights to the vaccine, and the companies are developing it together, performing multiple phase 2 trials.
A phase 3 trial of VLA15 was scheduled for late 2022, recruiting volunteers at test sites located across the northeastern United States and in Europe. Participants were scheduled to receive an initial three-dose series of vaccines over the course of five to nine months, followed by a booster dose after twelve months, with both the initial series and the booster dose scheduled to be complete before the year's peak Lyme disease season.
Other research
AnmRNA vaccine
An mRNA vaccine is a type of vaccine that uses a copy of a molecule called messenger RNA (mRNA) to produce an immune response. The vaccine delivers molecules of antigen-encoding mRNA into immune cells, which use the designed mRNA as a blueprin ...
designed to cause a strong fast immune response to tick saliva allowed the immune system to detect and remove the ticks from test animals before they were able to transmit the infectious bacteria. The vaccine contains mRNAs for the body to build 19 proteins in tick saliva which, by enabling quick development of erythema
Erythema (from the Greek , meaning red) is redness of the skin or mucous membranes, caused by hyperemia (increased blood flow) in superficial capillaries. It occurs with any skin injury, infection, or inflammation. Examples of erythema not asso ...
(itchy redness) at the bite site, protects guinea pigs against Lyme disease. It also protected the test animals if the tick is not removed if only one tick, but not three, remain attached.
Canine vaccines
Canine vaccines have been formulated and approved for the prevention of Lyme disease in dogs. Currently, three Lyme disease vaccines are available. LymeVax, formulated by Fort Dodge Laboratories, contains intact dead spirochetes which expose the host to the organism. Galaxy Lyme, Intervet-Schering-Plough
Schering-Plough Corporation was an American pharmaceutical company. It was originally the U.S. subsidiary of the German company Schering AG, which was founded in 1851 by Ernst Christian Friedrich Schering. As a result of nationalization, it beca ...
's vaccine, targets proteins OspC and OspA. The OspC antibodies kill any of the bacteria that have not been killed by the OspA antibodies. Canine Recombinant Lyme, formulated by Merial
Boehringer Ingelheim Animal Health is a multinational animal health company, formed in January 2017 when Merial was acquired by Boehringer Ingelheim and merged with Boehringer Ingelheim's existing animal health assets.
Overview
In August 1997, ...
, generates antibodies against the OspA protein so a tick feeding on a vaccinated dog draws in blood full of anti-OspA antibodies, which kill the spirochetes in the tick's gut before they are transmitted to the dog.
Treatment
Antibiotics
An antibiotic is a type of antimicrobial substance active against bacteria. It is the most important type of antibacterial agent for fighting bacterial infections, and antibiotic medications are widely used in the treatment and prevention o ...
are the primary treatment. The specific approach to their use is dependent on the individual affected and the stage of the disease. For most people with early localized infection, oral administration of doxycycline
Doxycycline is a broad-spectrum tetracycline class antibiotic used in the treatment of infections caused by bacteria and certain parasites. It is used to treat bacterial pneumonia, acne, chlamydia infections, Lyme disease, cholera, typhus, an ...
is widely recommended as the first choice, as it is effective against not only ''Borrelia'' bacteria but also a variety of other illnesses carried by ticks. People taking doxycycline should avoid sun exposure because of higher risk of sunburns. Doxycycline is contraindicated in children younger than eight years of age and women who are pregnant or breastfeeding; alternatives to doxycycline are amoxicillin
Amoxicillin is an antibiotic medication used to treat a number of bacterial infections. These include middle ear infection, strep throat, pneumonia, skin infections, and urinary tract infections among others. It is taken by mouth, or less c ...
, cefuroxime axetil
Cefuroxime axetil, sold under the brand name Ceftin among others, is a second generation oral cephalosporin antibiotic.
It is an acetoxyethyl ester prodrug of cefuroxime which is effective orally. The activity depends on ''in vivo'' hydrolysis a ...
, and azithromycin
Azithromycin, sold under the brand names Zithromax (in oral form) and Azasite (as an eye drop), is an antibiotic medication used for the treatment of a number of bacterial infections. This includes middle ear infections, strep throat, pneumon ...
. Azithromycin is recommended only in case of intolerance to the other antibiotics. The standard treatment for cellulitis
Cellulitis is usually a bacterial infection involving the inner layers of the skin. It specifically affects the dermis and subcutaneous fat. Signs and symptoms include an area of redness which increases in size over a few days. The borders of ...
, cephalexin
Cefalexin, also spelled cephalexin, is an antibiotic that can treat a number of bacterial infections. It kills gram-positive and some gram-negative bacteria by disrupting the growth of the bacterial cell wall. Cefalexin is a beta-lactam antibi ...
, is not useful for Lyme disease. When it is unclear if a rash is caused by Lyme or cellulitis, the Infectious Diseases Society of America, IDSA recommends treatment with cefuroxime
Cefuroxime, sold under the brand name Zinacef among others, is a second-generation cephalosporin antibiotic used to treat and prevent a number of bacterial infections. These include pneumonia, meningitis, otitis media, sepsis, urinary tract inf ...
or amoxicillin/clavulanic acid, as these are effective against both infections. Individuals with early disseminated or late Lyme infection may have symptomatic cardiac disease, Lyme arthritis, or neurologic symptoms like facial palsy
Facial nerve paralysis is a common problem that involves the paralysis of any structures innervated by the facial nerve. The pathway of the facial nerve is long and relatively convoluted, so there are a number of causes that may result in facial ...
, radiculopathy
Radiculopathy, also commonly referred to as pinched nerve, refers to a set of conditions in which one or more nerves are affected and do not work properly (a neuropathy). Radiculopathy can result in pain (radicular pain), weakness, altered sens ...
, meningitis, or peripheral neuropathy
Peripheral neuropathy, often shortened to neuropathy, is a general term describing disease affecting the peripheral nerves, meaning nerves beyond the brain and spinal cord. Damage to peripheral nerves may impair sensation, movement, gland, or or ...
. Intravenous administration of ceftriaxone is recommended as the first choice in these cases; cefotaxime and doxycycline are available as alternatives.
Treatment regimens for Lyme disease range from 14 days in early localized disease, to 14–21 days in early disseminated disease to 14–28 days in late disseminated disease. Neuroborreliosis, Neurologic complications of Lyme disease may be treated with doxycycline
Doxycycline is a broad-spectrum tetracycline class antibiotic used in the treatment of infections caused by bacteria and certain parasites. It is used to treat bacterial pneumonia, acne, chlamydia infections, Lyme disease, cholera, typhus, an ...
as it can be taken by mouth and has a lower cost, although in North America evidence of efficacy is only indirect. In case of failure, guidelines recommend retreatment with injectable ceftriaxone. Several months after treatment for Lyme arthritis, if joint swelling persists or returns, a second round of antibiotics may be considered; intravenous antibiotics are preferred for retreatment in case of poor response to oral antibiotics. Outside of that, a prolonged antibiotic regimen lasting more than 28 days is not recommended as no evidence shows it to be effective. IgM and IgG antibody levels may be elevated for years even after successful treatment with antibiotics. As antibody levels are not indicative of treatment success, testing for them is not recommended.
Facial palsy may resolve without treatment; however, antibiotic treatment is recommended to stop other Lyme complications. Corticosteroids are not recommended when facial palsy is caused by Lyme disease. In those with facial palsy, frequent use of artificial tears while awake is recommended, along with ointment and a patch or taping the eye closed when sleeping.
About a third of people with Lyme carditis need a temporary Artificial cardiac pacemaker, pacemaker until their heart conduction abnormality resolves, and 21% need to be hospitalized. Lyme carditis should not be treated with corticosteroids.
People with Lyme arthritis should limit their level of physical activity to avoid damaging affected joints, and in case of limping should use crutches. Pain associated with Lyme disease may be treated with nonsteroidal anti-inflammatory drugs (NSAIDs). Corticosteroid joint injections are not recommended for Lyme arthritis that is being treated with antibiotics. People with Lyme arthritis treated with intravenous antibiotics or two months of oral antibiotics who continue to have synovitis, joint swelling two months after treatment and have negative Polymerase chain reaction, PCR test for Borrelia
''Borrelia'' is a genus of bacteria of the spirochete phylum. Several species cause Lyme disease, also called Lyme borreliosis, a zoonotic, vector-borne disease transmitted by ticks. Other species of ''Borrelia'' cause relapsing fever, and are t ...
DNA in the synovial fluid are said to have antibiotic-refractory Lyme arthritis; this is more common after infection by certain Borrelia strains in people with certain genetic and immunologic characteristics. Antibiotic-refractory Lyme arthritis may be symptomatically treated with NSAIDs, disease-modifying antirheumatic drugs (DMARDs), or arthroscopic synovectomy. Physical therapy is recommended for adults after resolution of Lyme arthritis.
People receiving treatment should be advised that reinfection is possible and #Prevention, how to prevent it.
Prognosis
Lyme disease's typical first sign, theerythema migrans
Erythema migrans or erythema chronicum migrans is an expanding rash often seen in the early stage of Lyme disease, and can also (but less commonly) be caused by southern tick-associated rash illness (STARI). Page last reviewed: October 22, 2015 ...
(EM) rash, resolves within several weeks even without treatment. However, in untreated people, the infection often disseminates to the nervous system, heart, or joints, possibly causing permanent damage to body tissues.
People who receive recommended antibiotic treatment within several days of appearance of an initial EM rash have the best prospects. Recovery may not be total or immediate. The percentage of people achieving full recovery in the United States increases from about 64–71% at end of treatment for EM rash to about 84–90% after 30 months; higher percentages are reported in Europe. Treatment failure, i.e. persistence of original or appearance of new signs of the disease, occurs only in a few people. Remaining people are considered cured but continue to experience subjective symptoms, e.g. arthralgia, joint or muscle pains
Myalgia (also called muscle pain and muscle ache in layman's terms) is the medical term for muscle pain. Myalgia is a symptom of many diseases. The most common cause of acute myalgia is the overuse of a muscle or group of muscles; another lik ...
or fatigue. These symptoms usually are mild and nondisabling.
People treated only after nervous system manifestations of the disease may end up with objective neurological deficits, in addition to subjective symptoms. In Europe, an average of 32–33 months after initial Lyme symptoms in people treated mostly with doxycycline 200 mg for 14–21 days, the percentage of people with lingering symptoms was much higher among those diagnosed with neuroborreliosis
Neuroborreliosis is a disorder of the central nervous system. A neurological manifestation of Lyme disease, neuroborreliosis is caused by a systemic infection of spirochetes of the genus ''Borrelia.'' Symptoms of the disease include erythema migr ...
(50%) than among those with only an EM rash (16%). In another European study, 5 years after treatment for neuroborreliosis, lingering symptoms were less common among children (15%) than adults (30%), and in the latter was less common among those treated within 30 days of the first symptom (16%) than among those treated later (39%); among those with lingering symptoms, 54% had daily activities restricted and 19% were on sick leave or incapacitated.
Some data suggest that about 90% of Lyme Facial palsy, facial palsies treated with antibiotics recover fully a median of 24 days after appearing and most of the rest recover with only mild abnormality. However, in Europe 41% of people treated for facial palsy had other lingering symptoms at followup up to 6 months later, including 28% with numbness
Hypoesthesia or numbness is a common side effect of various medical conditions that manifests as a reduced sense of touch or sensation, or a partial loss of sensitivity to sensory stimuli. In everyday speech this is generally referred to as num ...
or altered sensation and 14% with fatigue or Attentional control, concentration problems. Palsies in both sides of the face are associated with worse and longer time to recovery. Historical data suggests that untreated people with facial palsies recover at nearly the same rate, but 88% subsequently have Lyme arthritis. Other research shows that synkinesis (involuntary movement of a facial muscle when another one is voluntarily moved) can become evident only 6–12 months after facial palsy appears to be resolved, as damaged nerves regrow and sometimes connect to incorrect muscles. Synkinesis is associated with corticosteroid use. In longer-term follow-up, 16–23% of Lyme facial palsies do not fully recover.
In Europe, about a quarter of people with Bannwarth syndrome (Lyme radiculopathy
Radiculopathy, also commonly referred to as pinched nerve, refers to a set of conditions in which one or more nerves are affected and do not work properly (a neuropathy). Radiculopathy can result in pain (radicular pain), weakness, altered sens ...
and Aseptic meningitis, lymphocytic meningitis) treated with intravenous ceftriaxone for 14 days an average of 30 days after first symptoms had to be retreated 3–6 months later because of unsatisfactory clinical response or continued Lymphocytic pleocytosis, objective markers of infection in cerebrospinal fluid
Cerebrospinal fluid (CSF) is a clear, colorless body fluid found within the tissue that surrounds the brain and spinal cord of all vertebrates.
CSF is produced by specialised ependymal cells in the choroid plexus of the ventricles of the bra ...
; after 12 months, 64% recovered fully, 31% had nondisabling mild or infrequent symptoms that did not require regular use of analgesics, and 5% had symptoms that were disabling or required substantial use of analgesics. The most common lingering nondisabling symptoms were headache, fatigue, altered sensation, joint pains, memory disturbances, malaise, Radiculitis, radicular pain, sleep disturbances, muscle pains
Myalgia (also called muscle pain and muscle ache in layman's terms) is the medical term for muscle pain. Myalgia is a symptom of many diseases. The most common cause of acute myalgia is the overuse of a muscle or group of muscles; another lik ...
, and Attentional control, concentration disturbances. Lingering disabling symptoms included facial palsy
Facial nerve paralysis is a common problem that involves the paralysis of any structures innervated by the facial nerve. The pathway of the facial nerve is long and relatively convoluted, so there are a number of causes that may result in facial ...
and other impaired movement.
Recovery from late neuroborreliosis tends to take longer and be less complete than from early neuroborreliosis, probably because of irreversible neurologic damage.
About half the people with Lyme carditis progress to complete heart block, but it usually resolves in a week. Other Lyme heart conduction abnormalities resolve typically within 6 weeks. About 94% of people have full recovery, but 5% need a permanent Artificial cardiac pacemaker, pacemaker and 1% end up with persistent heart block (the actual percentage may be higher because of unrecognized cases). Lyme myocardial
Cardiac muscle (also called heart muscle, myocardium, cardiomyocytes and cardiac myocytes) is one of three types of vertebrate muscle tissues, with the other two being skeletal muscle and smooth muscle. It is an involuntary, striated muscle that ...
complications usually are mild and self-limiting. However, in some cases Lyme carditis can be fatal.
Recommended antibiotic treatments are effective in about 90% of Lyme arthritis cases, although it can take several months for inflammation to resolve and a second round of antibiotics is often necessary. Antibiotic-refractory Lyme arthritis also eventually resolves, typically within 9–14 months (range 4 months – 4 years); DMARDs or synovectomy can accelerate recovery.
Reinfection is not uncommon. In a U.S. study, 6–11% of people treated for an EM rash had another EM rash within 30 months. The second rash typically is due to infection by a different Borrelia
''Borrelia'' is a genus of bacteria of the spirochete phylum. Several species cause Lyme disease, also called Lyme borreliosis, a zoonotic, vector-borne disease transmitted by ticks. Other species of ''Borrelia'' cause relapsing fever, and are t ...
strain.
People who have nonspecific, subjective symptoms such as fatigue, joint and muscle aches, or cognitive difficulties for more than six months after recommended treatment for Lyme disease are said to have post-treatment Lyme disease syndrome (PTLDS). As of 2016 the reason for the lingering symptoms was not known; the condition is generally managed similarly to fibromyalgia
Fibromyalgia (FM) is a medical condition defined by the presence of chronic widespread pain, fatigue, waking unrefreshed, cognitive symptoms, lower abdominal pain or cramps, and depression. Other symptoms include insomnia and a general hyp ...
or chronic fatigue syndrome.
Epidemiology
 Lyme disease Endemic (epidemiology), occurs regularly in
Lyme disease Endemic (epidemiology), occurs regularly in Northern Hemisphere
The Northern Hemisphere is the half of Earth that is north of the Equator. For other planets in the Solar System, north is defined as being in the same celestial hemisphere relative to the invariable plane of the solar system as Earth's Nort ...
temperate regions. There is a suggestion that tick populations and Lyme disease occurrence are increasing and spreading into new areas, due in part to the warming temperatures of climate change. However tick-borne disease systems are complex, and determining whether changes are due to climate change or other drivers can be difficult.
Africa
In northern Africa, ''B. burgdorferi sensu lato'' has been identified in Morocco, Algeria, Egypt and Tunisia. Lyme disease in sub-Saharan Africa is presently unknown, but evidence indicates it may occur in humans in this region. The abundance of hosts and tick vectors would favor the establishment of Lyme infection in Africa. In East Africa, two cases of Lyme disease have been reported in Kenya.Asia
''B. burgdorferi sensu lato''-infested ticks are being found more frequently in Japan, as well as in northwest China, Nepal, Thailand and far eastern Russia. ''Borrelia'' has also been isolated in Mongolia.Europe
In Europe, Lyme disease is caused by infection with one or more pathogenic European genospecies of the spirochaete ''B. burgdorferi sensu lato'', mainly transmitted by the tick ''Ixodes ricinus
''Ixodes ricinus'', the castor bean tick, is a chiefly European species of hard-bodied tick. It may reach a length of when engorged with a blood meal, and can transmit both bacterial and viral pathogens such as the causative agents of Lyme disea ...
''. Cases of ''B. burgdorferi sensu lato''-infected ticks are found predominantly in central Europe, particularly in Slovenia and Austria, but have been isolated in almost every country on the continent. Number of cases in southern Europe, such as Italy and Portugal, is much lower. Diagnosed cases in some Western countries, such as Iceland, are rising. Lyme disease is rare in Iceland. On average around 6 to 7 cases are diagnosed every year, primarily localised infections presenting as erythema migrans. None of the cases had a definitive Icelandic origin and the yearly number of cases has not been increasing.
United Kingdom
In the United Kingdom the number of laboratory confirmed cases of Lyme disease has been rising steadily since voluntary reporting was introduced in 1986 when 68 cases were recorded in the UK and Ireland combined. In the UK there were 23 confirmed cases in 1988 and 19 in 1990, but 973 in 2009 and 953 in 2010. Provisional figures for the first 3 quarters of 2011 show a 26% increase on the same period in 2010. It is thought, however, that the actual number of cases is significantly higher than suggested by the above figures, with the UK's Health Protection Agency estimating that there are between 2,000 and 3,000 cases per year, (with an average of around 15% of the infections acquired overseas), while Dr Darrel Ho-Yen, Director of the Scottish Toxoplasma Reference Laboratory and National Lyme Disease Testing Service, believes that the number of confirmed cases should be multiplied by 10 "to take account of wrongly diagnosed cases, tests giving false results, sufferers who weren't tested, people who are infected but not showing symptoms, failures to notify and infected individuals who don't consult a doctor." Despite Lyme disease (Borrelia burgdorferi infection) being a notifiable disease in Scotland since January 1990 which should therefore be reported on the basis of clinical suspicion, it is believed that many General practitioner, GPs are unaware of the requirement. Mandatory reporting, limited to laboratory test results only, was introduced throughout the UK in October 2010, under the Health Protection (Notification) Regulations 2010. Although there is a greater number of cases of Lyme disease in the New Forest, Salisbury Plain, Exmoor, the South Downs, parts of Wiltshire and Berkshire, Thetford Forest and the West coast and islands of Scotland, infected ticks are widespread, and can even be found in the parks of London. A 1989 report found that 25% of forestry workers in the New Forest were seropositive, as were between 2% and 4–5% of the general local population of the area. Tests on pet dogs carried out throughout the country in 2009 indicated that around 2.5% of ticks in the UK may be infected, considerably higher than previously thought. It is speculated that global warming may lead to an increase in tick activity in the future, as well as an increase in the amount of time that people spend in public parks, thus increasing the risk of infection. However no published research has proven this to be so.North America
Many studies in North America have examined ecological and environmental correlates of the number of people affected by Lyme disease. A 2005 study using climate suitability modelling of ''I. scapularis'' projected that Effects of climate change on human health, climate change would cause an overall 213% increase in suitable vector habitat by 2080, with northward expansions in Canada, increased suitability in the central U.S., and decreased suitable habitat and vector retraction in the southern U.S. A 2008 review of published studies concluded that the presence of forests or forested areas was the only variable that consistently elevated the risk of Lyme disease whereas other environmental variables showed little or no concordance between studies. The authors argued that the factors influencing tick density and human risk between sites are still poorly understood, and that future studies should be conducted over longer time periods, become more standardized across regions, and incorporate existing knowledge of regional Lyme disease ecology.Canada
The range of ticks able to carry Lyme disease has expanded from a limited area of Ontario to include areas of southern Quebec, Manitoba, northern Ontario, southern New Brunswick, southwest Nova Scotia and limited parts of Saskatchewan and Alberta, as well as British Columbia. Cases have been reported as far east as the island of Newfoundland. A model-based prediction by Leighton ''et al.'' (2012) suggests that the range of the ''I. scapularis'' tick will expand into Canada by 46 km/year over the next decade, with warming climatic temperatures as the main driver of increased speed of spread.Mexico
A 2007 study suggests ''Borrelia burgdorferi'' infections are endemic to Mexico, from four cases reported between 1999 and 2000.United States
 In 2019, according to the CDC, there were approximately 35,000 confirmed and probable cases in the U.S. with incidences occurring in almost every state. The CDC is currently conducting research on evaluation and diagnostics of the disease and preliminary results suggest the number of new cases to be around 300,000.
Lyme disease is the most common tick-borne disease in North America and Europe, and one of the fastest-growing infectious diseases in the United States. Of cases reported to the United States CDC, the ratio of Lyme disease infection is 7.9 cases for every 100,000 persons. In the ten states where Lyme disease is most common, the average was 31.6 cases for every 100,000 persons for the year 2005.
In 2019, according to the CDC, there were approximately 35,000 confirmed and probable cases in the U.S. with incidences occurring in almost every state. The CDC is currently conducting research on evaluation and diagnostics of the disease and preliminary results suggest the number of new cases to be around 300,000.
Lyme disease is the most common tick-borne disease in North America and Europe, and one of the fastest-growing infectious diseases in the United States. Of cases reported to the United States CDC, the ratio of Lyme disease infection is 7.9 cases for every 100,000 persons. In the ten states where Lyme disease is most common, the average was 31.6 cases for every 100,000 persons for the year 2005.