|
Borabenzene
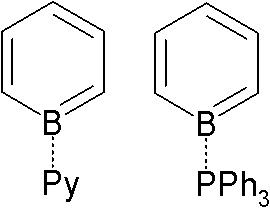
Borabenzene is a hypothetical organoboron compound with the formula C5H5B. Unlike the related but highly stable benzene molecule, borabenzene would be electron-deficient. Related derivatives are the boratabenzene anions, including the parent 5H5BHsup>−. Adducts Adducts of borabenzene with Lewis bases are isolatable. Since borabenzene is unavailable, these adducts require indirect methods. 4-Silyl-1-methoxyboracyclohexadiene is used as a precursor to the borabenzene: : + → + MeOSiMe3 The pyridine adduct is structurally related to biphenyl. It is a yellow whereas biphenyl is colorless, indicating distinct electronic structures. The pyridine ligand is tightly bound: no exchange is observed with free pyridine, even at elevated temperatures. : The borabenzene-pyridine adduct behaves like a diene, not an analog of biphenyl, and will undergo Diels-Alder reactions.{{cite journal , doi=10.1021/ol061201w , title=1-Borabarrelene Derivatives via Diels−Alder Additions to Boraben ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Borabenzene Adducts
Borabenzene is a hypothetical organoboron compound with the formula C5H5B. Unlike the related but highly stable benzene molecule, borabenzene would be electron-deficient. Related derivatives are the boratabenzene anions, including the parent 5H5BHsup>−. Adducts Adducts of borabenzene with Lewis bases are isolatable. Since borabenzene is unavailable, these adducts require indirect methods. 4-Silyl-1-methoxyboracyclohexadiene is used as a precursor to the borabenzene: : + → + MeOSiMe3 The pyridine adduct is structurally related to biphenyl. It is a yellow whereas biphenyl is colorless, indicating distinct electronic structures. The pyridine ligand is tightly bound: no exchange is observed with free pyridine, even at elevated temperatures. : The borabenzene-pyridine adduct behaves like a diene, not an analog of biphenyl, and will undergo Diels-Alder reactions.{{cite journal , doi=10.1021/ol061201w , title=1-Borabarrelene Derivatives via Diels−Alder Additions to Borabe ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organoboron Compound
Organoborane or organoboron compounds are chemical compounds of boron and carbon that are Organic compound, organic derivatives of BH3, for example trialkyl boranes. Organoboron chemistry or organoborane chemistry is the chemistry of these compounds. Organoboron compounds are important reagents in organic chemistry enabling many chemical transformations, the most important one called hydroboration. Reactions of organoborates and boranes involve the transfer of a nucleophilic group attached to boron to an electrophilic center either inter- or intramolecularly. α,β-Unsaturated borates, as well as borates with a leaving group at the α position, are highly susceptible to intramolecular 1,2-migration of a group from boron to the electrophilic α position. Oxidation or protonolysis of the resulting organoboranes may generate a variety of organic products, including alcohols, carbonyl compounds, alkenes, and halides. Properties of the B-C bond The C-B bond has low polarity (the diffe ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Boratabenzene
Boratabenzene is the heteroaromatic anion with the formula 5H5BHsup>−. Derivatives of boratabenzene are ligands akin to cyclopentadienyl anion. sandwich or half-sandwich type complexes of many transition metals have been reported.{{cite journal , doi=10.1016/j.ccr.2015.07.014, title=Boratabenzene Rare-earth Metal Complexes, year=2016, last1=Cui, first1=Peng, last2=Chen, first2=Yaofeng, journal=Coordination Chemistry Reviews, volume=314, pages=2–13 Electronically related heterocycles are adducts of borabenzene. The adduct C5H5B.pyridine exhibits properties of boratabenzene anion, i.e., it has the character C5H5B−-N+C5H5. See also * borabenzene, silabenzene, germabenzene, stannabenzene, pyridine, phosphorine, arsabenzene, bismabenzene, pyrylium, thiopyrylium, selenopyrylium, telluropyrylium * Borazine Borazine, also known as borazole, is a non-polar inorganic compound with the chemical formula B3H6N3. In this cyclic compound, the three BH units and three NH units alte ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Telluropyrylium
Telluropyrylium is an aromatic heterocyclic compound consisting of a six member ring with five carbon atoms, and a positively charged tellurium atom. Derivatives of telluropyrylium are important in research of infrared dyes. Naming and numbering Formerly it was named tellurapyrylium. However this is misleading, as "tellura" indicates that tellurium substitutes for carbon atom, but actually tellurium is substituted for the oxygen atom in pyrilium. In the Hantzsch-Widman system it is called tellurinium. This is the name used by Chemical Abstracts. Replacement nomenclature would call this telluroniabenzene. Numbering in telluropyrylium starts with 1 on the tellurium atom and counts up to 6 counter-clockwise on the carbon atoms. The positions adjacent to the chalcogen, numbered 2 and 6 can also be called α, the next two positions 3 and 5 can be termed "β" and the opposite carbon at position 4 can be called "γ". Occurrence Because telluropyrylium is a positively charged cation, it ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Selenopyrylium
Selenopyrylium is an aromatic heterocyclic compound consisting of a six-membered ring with five carbon atoms and a positively charged selenium atom. Naming and numbering Formerly it was named selenapyrylium. However, this is misleading as "selena" indicates that selenium substitutes for a carbon atom, but actually selenium is substituted for the oxygen atom in pyrylium. In the Hantzsch-Widman system of nomenclature, it is called seleninium. This is the name used by Chemical Abstracts. Replacement nomenclature would call this selenoniabenzene. Numbering in selenopyrylium starts with 1 on the selenium atom and counts up to 6 on the carbon atoms. The positions adjacent to the chalcogen, numbered 2 and 6 can also be called α, the next two positions 3 and 5 can be termed "β" and the opposite carbon at position 4 can be called "γ". Occurrence Because selenopyrylium is a positively charged ion, it takes the solid form as a salt with non-nucleophillic anions such as perchlorate, te ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Thiopyrylium
Thiopyrylium is a cation with the chemical formula C5H5S+. It is analogous to the pyrylium cation with the oxygen atom replaced by a sulfur atom. Thiopyrylium salts are less reactive than the analogous pyrylium salts due to the higher polarizability of the sulfur atom. Among the chalcogenic 6-membered unsaturated heterocycles, thiopyrylium is the most aromatic, due to sulfur having the similar Pauling electronegativity as carbon and only a slightly higher covalent radius. In water, thiopyrylium reacts to it and forms a mixture of 2-hydroxythiopyran and 4-hydroxythiopyran. Thiopyrylium salts can be synthesized by hydrogen abstraction from thiopyran by a hydride ion acceptor, such as trityl perchlorate. The thiopyrylium analogue of 2,4,6-trisubstituted pyrylium salts can be synthesized by treatment with sodium sulfide followed by precipitation with acid. This reaction causes the oxygen atom in the pyrylium cation to be substituted with sulfur. See also * 6-membered aromatic ri ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pyrylium
Pyrylium is a cation (positive ion) with formula , consisting of a six-membered ring of five carbon atoms, each with one hydrogen atom, and one positively charged oxygen atom. The bonds in the ring are conjugated as in benzene, giving it an aromatic character. In particular, because of the positive charge, the oxygen atom is trivalent. Pyrilium is a mono-cyclic and heterocyclic compound, one of the oxonium ions. Salts Pyrylium and its derivatives form stable salts with a variety of anions. Derivatives Many important cations are formally derived from pyrylium by substitution of various functional groups for some or all the hydrogens in the ring. The 2,4,6-triphenylpyrilium, referred to as the Katritzky salt, (after Alan R. Katritzky) is an important example used in many modern examples of metal catalyzed cross-couplings. Chemical properties Like other oxonium ions, pyrylium is unstable in neutral water. However, pyrylium is much less reactive than ordinary oxonium ions beca ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Stibabenzene
Stibinin, also known as stibabenzene, is an organic chemical compound. Stibinin has the chemical formula . The molecule, stibinin, is a derivative of benzene, with one of the carbon atoms in the 6-membered ring replaced by an antimony (Sb) atom. Stibinin is a molecule that is considered to be an organoantimony compound due to it containing carbon, hydrogen, and antimony atoms. Laboratory synthesis The synthesis of stibinin can be accomplished in a three step process. The final product can be isolated, even though the molecule is highly labile. The first step of this synthesis involves the treatment of penta-1,4-diyne with dibutylstannane as shown in the figure below. : : The second step of the synthesis involves reacting the product of the first step, 1,1-dibutyl-1,4-dihydrostannine, with antimony trichloride, to yield 1-chloro-1-stibacyclohexa-2,5-diene. : : The final step of the synthesis of stibinin involves treating 1-chloro-1-stibacyclohexa-2,5-diene with a base, such ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Arsabenzene
Arsabenzene (IUPAC name: arsinine) is an organoarsenic heterocyclic compound with the chemical formula C5H5As. It belongs to a group of compounds called heteroarenes that have the general formula C5H5E (E= N, P, As, Sb, Bi). This air sensitive liquid has an onion odor, and it decomposes on heating. Arsabenzene is also an ambidentate ligand that prefers to coordinate using η1(As)- or η6(π)-routes. The study of arsabenzene and related compounds was an important step in the understanding of compounds that contain multiple bonds between carbon and heavier elements. The study of heteroarenes was begun by Märkl, with the synthesis of 2,4,6-triphenylphosphabenzene. This is achieved by treating 2,4,6-trisubstituted pyrylium salt with phosphanes. The first derivative of arsabenzene was 9-arsaanthracene prepared by Jutzi and Bickelhaupt. Structure Arsabenzene is planar. The C—C bond distances of 1.39 Å, the As—C bond has a length of 1.85 Å, this is 6.6% shorter than the norma ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phosphorine
Phosphorine (IUPAC name: phosphinine) is a heavier element analog of pyridine, containing a phosphorus atom instead of an aza- moiety. It is also called phosphabenzene and belongs to the phosphaalkene class. It is a colorless liquid that is mainly of interest in research. Phosphorine is an air-sensitive oil but is otherwise stable when handled using air-free techniques (however, substituted derivatives can often be handled under air without risk of decomposition). In contrast, silabenzene, a related heavy-element analogue of benzene, is not only air- and moisture-sensitive but also thermally unstable without extensive steric protection. History The first phosphorine to be isolated is 2,4,6-triphenylphosphorine. It was synthesized by Gottfried Märkl in 1966 by condensation of the corresponding pyrylium salt and phosphine or its equivalent ( P(CH2OH)3 and P(SiMe3)3).''G. Märkl'', 2,4,6-Triphenylphosphabenzol iAngewandte Chemie 78, 907–908 (1966)/ref> 500px, Synthesis of T ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pyridine
Pyridine is a basic heterocyclic organic compound with the chemical formula . It is structurally related to benzene, with one methine group replaced by a nitrogen atom. It is a highly flammable, weakly alkaline, water-miscible liquid with a distinctive, unpleasant fish-like smell. Pyridine is colorless, but older or impure samples can appear yellow, due to the formation of extended, unsaturated polymeric chains, which show significant electrical conductivity. The pyridine ring occurs in many important compounds, including agrochemicals, pharmaceuticals, and vitamins. Historically, pyridine was produced from coal tar. As of 2016, it is synthesized on the scale of about 20,000 tons per year worldwide. Properties Physical properties The molecular electric dipole moment is 2.2 debyes. Pyridine is diamagnetic and has a diamagnetic susceptibility of −48.7 × 10−6 cm3·mol−1. The standard enthalpy of formation is 100.2 kJ·mol−1 in the liquid phase ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Stannabenzene
Stannabenzene (C5H6Sn) is the parent representative of a group of organotin compounds that are related to benzene with a carbon atom replaced by a tin atom. Stannabenzene itself has been studied by computational chemistry, but has not been isolated. Stable derivatives of stannabenzene Stable derivatives of stannabenzene have been isolated. The 2-stannanaphthalene depicted below is stable in an inert atmosphere at temperatures below 140 °C. The tin to carbon bond in this compound is shielded from potential reactants by two very bulky groups, one tert-butyl group and the even larger 2,4,6-tris is(trimethylsilyl)methylhenyl or Tbt group. The two Sn-C bonds have bond lengths of 202.9 and 208.1 pm which are shorter than those for Sn-C single bonds (214 pm) and comparable to that of known Sn=C double bonds (201.6 pm). The C-C bonds show little variation with bond lengths between 135.6 and 144.3 pm signaling that this compound is aromatic. : Tbt-substituted 9-stannaphenanthren ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |

(C5H5BH)_(OZUFAR).png)

