Titanium on:
[Wikipedia]
[Google]
[Amazon]
Titanium is a
As a
Commercially pure (99.2% pure) grades of titanium have ultimate tensile strength of about 434 MPa (63,000 psi), equal to that of common, low-grade steel alloys, but are less dense. Titanium is 60% denser than aluminium, but more than twice as strong as the most commonly used 6061-T6 aluminium alloy. Certain titanium alloys (e.g., Beta C) achieve tensile strengths of over 1,400 MPa (200,000 psi). However, titanium loses strength when heated above .
Titanium is not as hard as some grades of heat-treated steel; it is non-magnetic and a poor conductor of heat and electricity. Machining requires precautions, because the material can

 Like
Like
 The +4
The +4
 Titanium tetrachloride (titanium(IV) chloride, TiCl4) is a colorless volatile liquid (commercial samples are yellowish) that, in air, hydrolyzes with spectacular emission of white clouds. Via the Kroll process, TiCl4 is used in the conversion of titanium ores to titanium metal. Titanium tetrachloride is also used to make titanium dioxide, e.g., for use in white paint. It is widely used in
Titanium tetrachloride (titanium(IV) chloride, TiCl4) is a colorless volatile liquid (commercial samples are yellowish) that, in air, hydrolyzes with spectacular emission of white clouds. Via the Kroll process, TiCl4 is used in the conversion of titanium ores to titanium metal. Titanium tetrachloride is also used to make titanium dioxide, e.g., for use in white paint. It is widely used in
 Titanium was discovered in 1791 by the
Titanium was discovered in 1791 by the  Titanium of very high purity was made in small quantities when Anton Eduard van Arkel and Jan Hendrik de Boer discovered the iodide process in 1925, by reacting with iodine and decomposing the formed vapors over a hot filament to pure metal.
In the 1950s and 1960s, the Soviet Union pioneered the use of titanium in military and submarine applications ( Alfa class and Mike class) as part of programs related to the Cold War. Starting in the early 1950s, titanium came into use extensively in military aviation, particularly in high-performance jets, starting with aircraft such as the F-100 Super Sabre and Lockheed A-12 and SR-71.
Throughout the Cold War period, titanium was considered a strategic material by the U.S. government, and a large stockpile of titanium sponge (a porous form of the pure metal) was maintained by the Defense National Stockpile Center, until the stockpile was dispersed in the 2000s. As of 2021, the four leading producers of titanium sponge were China (52%), Japan (24%), Russia (16%) and Kazakhstan (7%).
Titanium of very high purity was made in small quantities when Anton Eduard van Arkel and Jan Hendrik de Boer discovered the iodide process in 1925, by reacting with iodine and decomposing the formed vapors over a hot filament to pure metal.
In the 1950s and 1960s, the Soviet Union pioneered the use of titanium in military and submarine applications ( Alfa class and Mike class) as part of programs related to the Cold War. Starting in the early 1950s, titanium came into use extensively in military aviation, particularly in high-performance jets, starting with aircraft such as the F-100 Super Sabre and Lockheed A-12 and SR-71.
Throughout the Cold War period, titanium was considered a strategic material by the U.S. government, and a large stockpile of titanium sponge (a porous form of the pure metal) was maintained by the Defense National Stockpile Center, until the stockpile was dispersed in the 2000s. As of 2021, the four leading producers of titanium sponge were China (52%), Japan (24%), Russia (16%) and Kazakhstan (7%).

 The processing of titanium metal occurs in four major steps: reduction of titanium ore into "sponge", a porous form; melting of sponge, or sponge plus a master alloy to form an ingot; primary fabrication, where an ingot is converted into general mill products such as billet, bar, plate, sheet, strip, and
The processing of titanium metal occurs in four major steps: reduction of titanium ore into "sponge", a porous form; melting of sponge, or sponge plus a master alloy to form an ingot; primary fabrication, where an ingot is converted into general mill products such as billet, bar, plate, sheet, strip, and 2FeTiO3 + 7Cl2 + 6C -> 00^oC2FeCl3 + 2TiCl4 + 6CO
:TiCl4 + 2Mg -> 100^oCTi + 2MgCl2
 All
All
 Common titanium alloys are made by reduction. For example, cuprotitanium (rutile with
Common titanium alloys are made by reduction. For example, cuprotitanium (rutile with
 Titanium is used in steel as an alloying element ( ferro-titanium) to reduce
Titanium is used in steel as an alloying element ( ferro-titanium) to reduce
 Because titanium alloys have high
Because titanium alloys have high
 Welded titanium pipe and process equipment (heat exchangers, tanks, process vessels, valves) are used in the chemical and petrochemical industries primarily for corrosion resistance. Specific alloys are used in oil and gas downhole applications and nickel hydrometallurgy for their high strength (e. g.: titanium beta C alloy), corrosion resistance, or both. The pulp and paper industry uses titanium in process equipment exposed to corrosive media, such as sodium hypochlorite or wet chlorine gas (in the bleachery). Other applications include ultrasonic welding, wave soldering, and sputtering targets.
Titanium tetrachloride (TiCl4), a colorless liquid, is important as an intermediate in the process of making TiO2 and is also used to produce the Ziegler–Natta catalyst. Titanium tetrachloride is also used to iridize glass and, because it fumes strongly in moist air, it is used to make smoke screens.
Welded titanium pipe and process equipment (heat exchangers, tanks, process vessels, valves) are used in the chemical and petrochemical industries primarily for corrosion resistance. Specific alloys are used in oil and gas downhole applications and nickel hydrometallurgy for their high strength (e. g.: titanium beta C alloy), corrosion resistance, or both. The pulp and paper industry uses titanium in process equipment exposed to corrosive media, such as sodium hypochlorite or wet chlorine gas (in the bleachery). Other applications include ultrasonic welding, wave soldering, and sputtering targets.
Titanium tetrachloride (TiCl4), a colorless liquid, is important as an intermediate in the process of making TiO2 and is also used to produce the Ziegler–Natta catalyst. Titanium tetrachloride is also used to iridize glass and, because it fumes strongly in moist air, it is used to make smoke screens.
 Because of its durability, titanium has become more popular for designer jewelry (particularly, titanium rings). Its inertness makes it a good choice for those with allergies or those who will be wearing the jewelry in environments such as swimming pools. Titanium is also alloyed with gold to produce an alloy that can be marketed as 24-karat gold because the 1% of alloyed Ti is insufficient to require a lesser mark. The resulting alloy is roughly the hardness of 14-karat gold and is more durable than pure 24-karat gold.
Titanium's durability, light weight, and dent and corrosion resistance make it useful for
Because of its durability, titanium has become more popular for designer jewelry (particularly, titanium rings). Its inertness makes it a good choice for those with allergies or those who will be wearing the jewelry in environments such as swimming pools. Titanium is also alloyed with gold to produce an alloy that can be marketed as 24-karat gold because the 1% of alloyed Ti is insufficient to require a lesser mark. The resulting alloy is roughly the hardness of 14-karat gold and is more durable than pure 24-karat gold.
Titanium's durability, light weight, and dent and corrosion resistance make it useful for
 Titanium has the inherent ability to osseointegrate, enabling use in dental implants that can last for over 30 years. This property is also useful for orthopedic implant applications. These benefit from titanium's lower modulus of elasticity (
Titanium has the inherent ability to osseointegrate, enabling use in dental implants that can last for over 30 years. This property is also useful for orthopedic implant applications. These benefit from titanium's lower modulus of elasticity (
 An unknown mechanism in
An unknown mechanism in
"Titanium: Our Next Major Metal"
in ''
Titanium
at '' Periodic Videos'' (University of Nottingham)
Titanium.org
official website of the International Titanium Association, an industry association
Metallurgy of Titanium and its Alloys
- slide presentations, movies, and other material from Harshad Bhadeshia and other
chemical element
A chemical element is a chemical substance whose atoms all have the same number of protons. The number of protons is called the atomic number of that element. For example, oxygen has an atomic number of 8: each oxygen atom has 8 protons in its ...
; it has symbol
A symbol is a mark, Sign (semiotics), sign, or word that indicates, signifies, or is understood as representing an idea, physical object, object, or wikt:relationship, relationship. Symbols allow people to go beyond what is known or seen by cr ...
Ti and atomic number
The atomic number or nuclear charge number (symbol ''Z'') of a chemical element is the charge number of its atomic nucleus. For ordinary nuclei composed of protons and neutrons, this is equal to the proton number (''n''p) or the number of pro ...
22. Found in nature only as an oxide
An oxide () is a chemical compound containing at least one oxygen atom and one other element in its chemical formula. "Oxide" itself is the dianion (anion bearing a net charge of −2) of oxygen, an O2− ion with oxygen in the oxidation st ...
, it can be reduced to produce a lustrous transition metal with a silver color
Color (or colour in English in the Commonwealth of Nations, Commonwealth English; American and British English spelling differences#-our, -or, see spelling differences) is the visual perception based on the electromagnetic spectrum. Though co ...
, low density
Density (volumetric mass density or specific mass) is the ratio of a substance's mass to its volume. The symbol most often used for density is ''ρ'' (the lower case Greek letter rho), although the Latin letter ''D'' (or ''d'') can also be u ...
, and high strength, resistant to corrosion
Corrosion is a natural process that converts a refined metal into a more chemically stable oxide. It is the gradual deterioration of materials (usually a metal) by chemical or electrochemical reaction with their environment. Corrosion engine ...
in sea water, aqua regia
Aqua regia (; from Latin, "regal water" or "royal water") is a mixture of nitric acid and hydrochloric acid, optimally in a molar concentration, molar ratio of 1:3. Aqua regia is a fuming liquid. Freshly prepared aqua regia is colorless, but i ...
, and chlorine
Chlorine is a chemical element; it has Symbol (chemistry), symbol Cl and atomic number 17. The second-lightest of the halogens, it appears between fluorine and bromine in the periodic table and its properties are mostly intermediate between ...
.
Titanium was discovered in Cornwall
Cornwall (; or ) is a Ceremonial counties of England, ceremonial county in South West England. It is also one of the Celtic nations and the homeland of the Cornish people. The county is bordered by the Atlantic Ocean to the north and west, ...
, Great Britain
Great Britain is an island in the North Atlantic Ocean off the north-west coast of continental Europe, consisting of the countries England, Scotland, and Wales. With an area of , it is the largest of the British Isles, the List of European ...
, by William Gregor in 1791 and was named by Martin Heinrich Klaproth after the Titans
In Greek mythology, the Titans ( ; ) were the pre-Twelve Olympians, Olympian gods. According to the ''Theogony'' of Hesiod, they were the twelve children of the primordial parents Uranus (mythology), Uranus (Sky) and Gaia (Earth). The six male ...
of Greek mythology
Greek mythology is the body of myths originally told by the Ancient Greece, ancient Greeks, and a genre of ancient Greek folklore, today absorbed alongside Roman mythology into the broader designation of classical mythology. These stories conc ...
. The element occurs within a number of mineral
In geology and mineralogy, a mineral or mineral species is, broadly speaking, a solid substance with a fairly well-defined chemical composition and a specific crystal structure that occurs naturally in pure form.John P. Rafferty, ed. (2011): Mi ...
s, principally rutile
Rutile is an oxide mineral composed of titanium dioxide (TiO2), the most common natural form of TiO2. Rarer polymorphs of TiO2 are known, including anatase, akaogiite, and brookite.
Rutile has one of the highest refractive indices at vis ...
and ilmenite, which are widely distributed in the Earth's crust
Earth's crust is its thick outer shell of rock, referring to less than one percent of the planet's radius and volume. It is the top component of the lithosphere, a solidified division of Earth's layers that includes the crust and the upper ...
and lithosphere
A lithosphere () is the rigid, outermost rocky shell of a terrestrial planet or natural satellite. On Earth, it is composed of the crust and the lithospheric mantle, the topmost portion of the upper mantle that behaves elastically on time ...
; it is found in almost all living things, as well as bodies of water, rocks, and soils. The metal is extracted from its principal mineral ores by the Kroll and Hunter processes. The most common compound, titanium dioxide (TiO2), is a popular photocatalyst and is used in the manufacture of white pigments. Other compounds include titanium tetrachloride (TiCl4), a component of smoke screen
A smoke screen is smoke released to mask the movement or location of military units such as infantry, tanks, aircraft, or ships.
Smoke screens are commonly deployed either by a canister (such as a grenade) or generated by a vehicle (such as ...
s and catalysts; and titanium trichloride (TiCl3), which is used as a catalyst in the production of polypropylene
Polypropylene (PP), also known as polypropene, is a thermoplastic polymer used in a wide variety of applications. It is produced via chain-growth polymerization from the monomer Propene, propylene.
Polypropylene belongs to the group of polyolefin ...
.
Titanium can be alloy
An alloy is a mixture of chemical elements of which in most cases at least one is a metal, metallic element, although it is also sometimes used for mixtures of elements; herein only metallic alloys are described. Metallic alloys often have prop ...
ed with iron
Iron is a chemical element; it has symbol Fe () and atomic number 26. It is a metal that belongs to the first transition series and group 8 of the periodic table. It is, by mass, the most common element on Earth, forming much of Earth's o ...
, aluminium
Aluminium (or aluminum in North American English) is a chemical element; it has chemical symbol, symbol Al and atomic number 13. It has a density lower than that of other common metals, about one-third that of steel. Aluminium has ...
, vanadium, and molybdenum
Molybdenum is a chemical element; it has Symbol (chemistry), symbol Mo (from Neo-Latin ''molybdaenum'') and atomic number 42. The name derived from Ancient Greek ', meaning lead, since its ores were confused with lead ores. Molybdenum minerals hav ...
, among other elements. The resulting titanium alloys are strong, lightweight, and versatile, with applications including aerospace (jet engine
A jet engine is a type of reaction engine, discharging a fast-moving jet (fluid), jet of heated gas (usually air) that generates thrust by jet propulsion. While this broad definition may include Rocket engine, rocket, Pump-jet, water jet, and ...
s, missile
A missile is an airborne ranged weapon capable of self-propelled flight aided usually by a propellant, jet engine or rocket motor.
Historically, 'missile' referred to any projectile that is thrown, shot or propelled towards a target; this ...
s, and spacecraft
A spacecraft is a vehicle that is designed spaceflight, to fly and operate in outer space. Spacecraft are used for a variety of purposes, including Telecommunications, communications, Earth observation satellite, Earth observation, Weather s ...
), military, industrial processes (chemicals and petrochemicals, desalination plant
Desalination is a process that removes mineral components from saline water. More generally, desalination is the removal of salts and minerals from a substance. One example is soil desalination. This is important for agriculture. It is possible ...
s, pulp, and paper
Paper is a thin sheet material produced by mechanically or chemically processing cellulose fibres derived from wood, Textile, rags, poaceae, grasses, Feces#Other uses, herbivore dung, or other vegetable sources in water. Once the water is dra ...
), automotive, agriculture
Agriculture encompasses crop and livestock production, aquaculture, and forestry for food and non-food products. Agriculture was a key factor in the rise of sedentary human civilization, whereby farming of domesticated species created ...
(farming), sporting goods, jewelry, and consumer electronics
Consumer electronics, also known as home electronics, are electronic devices intended for everyday household use. Consumer electronics include those used for entertainment, Communication, communications, and recreation. Historically, these prod ...
. Titanium is also considered one of the most biocompatible metals, leading to a range of medical applications including prostheses, orthopedic implants, dental implants, and surgical instrument
A surgical instrument is a medical device for performing specific actions or carrying out desired effects during a surgery or operation, such as modifying biological tissue, or to provide access for viewing it. Over time, many different kinds of ...
s.
The two most useful properties of the metal are corrosion resistance and strength-to-density ratio, the highest of any metallic element. In its unalloyed condition, titanium is as strong as some steel
Steel is an alloy of iron and carbon that demonstrates improved mechanical properties compared to the pure form of iron. Due to steel's high Young's modulus, elastic modulus, Yield (engineering), yield strength, Fracture, fracture strength a ...
s, but less dense. There are two allotropic forms and five naturally occurring isotope
Isotopes are distinct nuclear species (or ''nuclides'') of the same chemical element. They have the same atomic number (number of protons in their Atomic nucleus, nuclei) and position in the periodic table (and hence belong to the same chemica ...
s of this element, Ti through Ti, with Ti being the most abundant (73.8%).
Characteristics
Physical properties
metal
A metal () is a material that, when polished or fractured, shows a lustrous appearance, and conducts electrical resistivity and conductivity, electricity and thermal conductivity, heat relatively well. These properties are all associated wit ...
, titanium is recognized for its high strength-to-weight ratio. It is a strong metal with low density
Density (volumetric mass density or specific mass) is the ratio of a substance's mass to its volume. The symbol most often used for density is ''ρ'' (the lower case Greek letter rho), although the Latin letter ''D'' (or ''d'') can also be u ...
that is quite ductile (especially in an oxygen
Oxygen is a chemical element; it has chemical symbol, symbol O and atomic number 8. It is a member of the chalcogen group (periodic table), group in the periodic table, a highly reactivity (chemistry), reactive nonmetal (chemistry), non ...
-free environment), lustrous, and metallic-white in color
Color (or colour in English in the Commonwealth of Nations, Commonwealth English; American and British English spelling differences#-our, -or, see spelling differences) is the visual perception based on the electromagnetic spectrum. Though co ...
. Due to its relatively high melting point (1,668 °C or 3,034 °F) it has sometimes been described as a refractory metal, but this is not the case. It is paramagnetic and has fairly low electrical
Electricity is the set of physical phenomena associated with the presence and motion of matter possessing an electric charge. Electricity is related to magnetism, both being part of the phenomenon of electromagnetism, as described by Maxwel ...
and thermal conductivity
The thermal conductivity of a material is a measure of its ability to heat conduction, conduct heat. It is commonly denoted by k, \lambda, or \kappa and is measured in W·m−1·K−1.
Heat transfer occurs at a lower rate in materials of low ...
compared to other metals. Titanium is superconducting when cooled below its critical temperature of 0.49 K.gall
Galls (from the Latin , 'oak-apple') or ''cecidia'' (from the Greek , anything gushing out) are a kind of swelling growth on the external tissues of plants. Plant galls are abnormal outgrowths of plant tissues, similar to benign tumors or war ...
unless sharp tools and proper cooling methods are used. Like steel structures, those made from titanium have a fatigue limit
The fatigue limit or endurance limit is the stress (mechanics), stress level below which an infinite number of loading cycles can be applied to a material without causing fatigue (material), fatigue failure. Some metals such as ferrous alloys and ...
that guarantees longevity in some applications.
The metal is a dimorphic allotrope of a hexagonal close packed α form that changes into a body-centered cubic (lattice) β form at . The specific heat of the α form increases dramatically as it is heated to this transition temperature but then falls and remains fairly constant for the β form regardless of temperature.
Chemical properties

 Like
Like aluminium
Aluminium (or aluminum in North American English) is a chemical element; it has chemical symbol, symbol Al and atomic number 13. It has a density lower than that of other common metals, about one-third that of steel. Aluminium has ...
and magnesium
Magnesium is a chemical element; it has Symbol (chemistry), symbol Mg and atomic number 12. It is a shiny gray metal having a low density, low melting point and high chemical reactivity. Like the other alkaline earth metals (group 2 ...
, the surface of titanium metal and its alloys oxidize immediately upon exposure to air to form a thin non-porous passivation layer that protects the bulk metal from further oxidation or corrosion. When it first forms, this protective layer is only 1–2 nm thick but it continues to grow slowly, reaching a thickness of 25 nm in four years. This layer gives titanium excellent resistance to corrosion against oxidizing acids, but it will dissolve in dilute hydrofluoric acid, hot hydrochloric acid, and hot sulfuric acid.
Titanium is capable of withstanding attack by dilute sulfuric and hydrochloric acids at room temperature, chloride solutions, and most organic acids. However, titanium is corroded by concentrated acids. Titanium is a very reactive metal that burns in normal air at lower temperatures than the melting point. Melting is possible only in an inert atmosphere or vacuum. At , it combines with chlorine. It also reacts with the other halogens and absorbs hydrogen.
Titanium readily reacts with oxygen at in air, and at in pure oxygen, forming titanium dioxide. Titanium is one of the few elements that burns in pure nitrogen gas, reacting at to form titanium nitride, which causes embrittlement. Because of its high reactivity with oxygen, nitrogen, and many other gases, titanium that is evaporated from filaments is the basis for titanium sublimation pumps, in which titanium serves as a scavenger for these gases by chemically binding to them. Such pumps inexpensively produce extremely low pressures in ultra-high vacuum systems.
Occurrence
Titanium is the ninth-most abundant element inEarth
Earth is the third planet from the Sun and the only astronomical object known to Planetary habitability, harbor life. This is enabled by Earth being an ocean world, the only one in the Solar System sustaining liquid surface water. Almost all ...
's crust (0.63% by mass
Mass is an Intrinsic and extrinsic properties, intrinsic property of a physical body, body. It was traditionally believed to be related to the physical quantity, quantity of matter in a body, until the discovery of the atom and particle physi ...
) and the seventh-most abundant metal. It is present as oxides in most igneous rock
Igneous rock ( ), or magmatic rock, is one of the three main rock types, the others being sedimentary and metamorphic. Igneous rocks are formed through the cooling and solidification of magma or lava.
The magma can be derived from partial ...
s, in sediments
Sediment is a solid material that is transported to a new location where it is deposited. It occurs naturally and, through the processes of weathering and erosion, is broken down and subsequently sediment transport, transported by the action of ...
derived from them, in living things, and natural bodies of water. Of the 801 types of igneous rocks analyzed by the United States Geological Survey
The United States Geological Survey (USGS), founded as the Geological Survey, is an agency of the U.S. Department of the Interior whose work spans the disciplines of biology, geography, geology, and hydrology. The agency was founded on Mar ...
, 784 contained titanium. Its proportion in soils is approximately 0.5–1.5%.
Common titanium-containing mineral
In geology and mineralogy, a mineral or mineral species is, broadly speaking, a solid substance with a fairly well-defined chemical composition and a specific crystal structure that occurs naturally in pure form.John P. Rafferty, ed. (2011): Mi ...
s are anatase, brookite, ilmenite, perovskite, rutile
Rutile is an oxide mineral composed of titanium dioxide (TiO2), the most common natural form of TiO2. Rarer polymorphs of TiO2 are known, including anatase, akaogiite, and brookite.
Rutile has one of the highest refractive indices at vis ...
, and titanite (sphene). Akaogiite is an extremely rare mineral consisting of titanium dioxide. Of these minerals, only rutile and ilmenite have economic importance, yet even they are difficult to find in high concentrations. About 6.0 and 0.7 million tonnes of those minerals were mined in 2011, respectively. Significant titanium-bearing ilmenite deposits exist in Australia
Australia, officially the Commonwealth of Australia, is a country comprising mainland Australia, the mainland of the Australia (continent), Australian continent, the island of Tasmania and list of islands of Australia, numerous smaller isl ...
, Canada
Canada is a country in North America. Its Provinces and territories of Canada, ten provinces and three territories extend from the Atlantic Ocean to the Pacific Ocean and northward into the Arctic Ocean, making it the world's List of coun ...
, China
China, officially the People's Republic of China (PRC), is a country in East Asia. With population of China, a population exceeding 1.4 billion, it is the list of countries by population (United Nations), second-most populous country after ...
, India
India, officially the Republic of India, is a country in South Asia. It is the List of countries and dependencies by area, seventh-largest country by area; the List of countries by population (United Nations), most populous country since ...
, Mozambique
Mozambique, officially the Republic of Mozambique, is a country located in Southeast Africa bordered by the Indian Ocean to the east, Tanzania to the north, Malawi and Zambia to the northwest, Zimbabwe to the west, and Eswatini and South Afr ...
, New Zealand
New Zealand () is an island country in the southwestern Pacific Ocean. It consists of two main landmasses—the North Island () and the South Island ()—and List of islands of New Zealand, over 600 smaller islands. It is the List of isla ...
, Norway
Norway, officially the Kingdom of Norway, is a Nordic countries, Nordic country located on the Scandinavian Peninsula in Northern Europe. The remote Arctic island of Jan Mayen and the archipelago of Svalbard also form part of the Kingdom of ...
, Sierra Leone
Sierra Leone, officially the Republic of Sierra Leone, is a country on the southwest coast of West Africa. It is bordered to the southeast by Liberia and by Guinea to the north. Sierra Leone's land area is . It has a tropical climate and envi ...
, South Africa
South Africa, officially the Republic of South Africa (RSA), is the Southern Africa, southernmost country in Africa. Its Provinces of South Africa, nine provinces are bounded to the south by of coastline that stretches along the Atlantic O ...
, and Ukraine
Ukraine is a country in Eastern Europe. It is the List of European countries by area, second-largest country in Europe after Russia, which Russia–Ukraine border, borders it to the east and northeast. Ukraine also borders Belarus to the nor ...
. About 210,000 tonnes of titanium metal sponge were produced in 2020, mostly in China (110,000 t), Japan (50,000 t), Russia (33,000 t) and Kazakhstan (15,000 t). Total reserves of anatase, ilmenite, and rutile are estimated to exceed 2 billion tonnes.
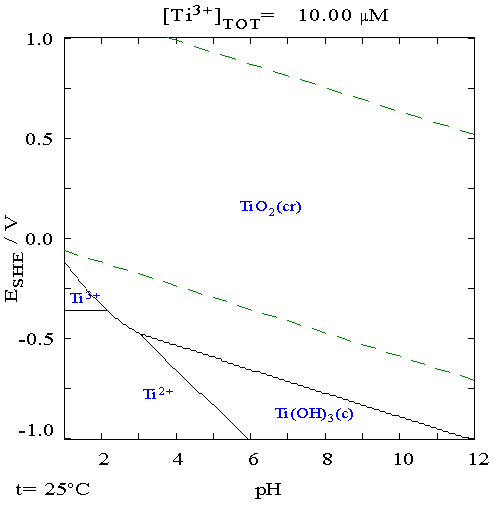
The concentration of titanium is about 4 picomolar in the ocean. At 100 °C, the concentration of titanium in water is estimated to be less than 10−7 M at pH 7. The identity of titanium species in aqueous solution remains unknown because of its low solubility and the lack of sensitive spectroscopic methods, although only the 4+ oxidation state is stable in air. No evidence exists for a biological role, although rare organisms are known to accumulate high concentrations of titanium.
Titanium is contained in meteorite
A meteorite is a rock (geology), rock that originated in outer space and has fallen to the surface of a planet or Natural satellite, moon. When the original object enters the atmosphere, various factors such as friction, pressure, and chemical ...
s, and it has been detected in the Sun and in M-type star
A star is a luminous spheroid of plasma (physics), plasma held together by Self-gravitation, self-gravity. The List of nearest stars and brown dwarfs, nearest star to Earth is the Sun. Many other stars are visible to the naked eye at night sk ...
s (the coolest type) with a surface temperature of . Rocks brought back from the Moon
The Moon is Earth's only natural satellite. It Orbit of the Moon, orbits around Earth at Lunar distance, an average distance of (; about 30 times Earth diameter, Earth's diameter). The Moon rotation, rotates, with a rotation period (lunar ...
during the Apollo 17
Apollo 17 (December 7–19, 1972) was the eleventh and final mission of NASA's Apollo program, the sixth and most recent time humans have set foot on the Moon. Commander Gene Cernan and Lunar Module Pilot Harrison Schmitt walked on the Moon, ...
mission are composed of 12.1% TiO2. Native titanium (pure metallic) is very rare.
Isotopes
Naturally occurring titanium is composed of five stableisotope
Isotopes are distinct nuclear species (or ''nuclides'') of the same chemical element. They have the same atomic number (number of protons in their Atomic nucleus, nuclei) and position in the periodic table (and hence belong to the same chemica ...
s: 46Ti, 47Ti, 48Ti, 49Ti, and 50Ti, with 48Ti being the most abundant (73.8% natural abundance). At least 21 radioisotopes have been characterized, the most stable of which are 44Ti with a half-life Half-life is a mathematical and scientific description of exponential or gradual decay.
Half-life, half life or halflife may also refer to:
Film
* Half-Life (film), ''Half-Life'' (film), a 2008 independent film by Jennifer Phang
* ''Half Life: ...
of 63 years; 45Ti, 184.8 minutes; 51Ti, 5.76 minutes; and 52Ti, 1.7 minutes. All other radioactive isotopes have half-lives less than 33 seconds, with the majority less than half a second.
The isotopes of titanium range in atomic weight from (39Ti) to (64Ti). The primary decay mode for isotopes lighter than 46Ti is positron emission
Positron emission, beta plus decay, or β+ decay is a subtype of radioactive decay called beta decay, in which a proton inside a radionuclide nucleus is converted into a neutron while releasing a positron and an electron neutrino (). Positron emi ...
(with the exception of 44Ti which undergoes electron capture
Electron capture (K-electron capture, also K-capture, or L-electron capture, L-capture) is a process in which the proton-rich nucleus of an electrically neutral atom absorbs an inner atomic electron, usually from the K or L electron shells. Th ...
), leading to isotopes of scandium, and the primary mode for isotopes heavier than 50Ti is beta emission, leading to isotopes of vanadium.
Titanium becomes radioactive upon bombardment with deuterons, emitting mainly positrons and hard gamma rays
A gamma ray, also known as gamma radiation (symbol ), is a penetrating form of electromagnetic radiation arising from high energy interactions like the radioactive decay of atomic nuclei or astronomical events like solar flares. It consists o ...
.
Compounds
 The +4
The +4 oxidation state
In chemistry, the oxidation state, or oxidation number, is the hypothetical Electrical charge, charge of an atom if all of its Chemical bond, bonds to other atoms are fully Ionic bond, ionic. It describes the degree of oxidation (loss of electrons ...
dominates titanium chemistry, but compounds in the +3 oxidation state are also numerous. Commonly, titanium adopts an octahedral coordination geometry in its complexes, but tetrahedral TiCl4 is a notable exception. Because of its high oxidation state, titanium(IV) compounds exhibit a high degree of covalent bond
A covalent bond is a chemical bond that involves the sharing of electrons to form electron pairs between atoms. These electron pairs are known as shared pairs or bonding pairs. The stable balance of attractive and repulsive forces between atom ...
ing.
Oxides, sulfides, and alkoxides
The most important oxide is TiO2, which exists in three important polymorphs; anatase, brookite, and rutile. All three are white diamagnetic solids, although mineral samples can appear dark (seerutile
Rutile is an oxide mineral composed of titanium dioxide (TiO2), the most common natural form of TiO2. Rarer polymorphs of TiO2 are known, including anatase, akaogiite, and brookite.
Rutile has one of the highest refractive indices at vis ...
). They adopt polymeric structures in which Ti is surrounded by six oxide
An oxide () is a chemical compound containing at least one oxygen atom and one other element in its chemical formula. "Oxide" itself is the dianion (anion bearing a net charge of −2) of oxygen, an O2− ion with oxygen in the oxidation st ...
ligands that link to other Ti centers.
The term '' titanates'' usually refers to titanium(IV) compounds, as represented by barium titanate (BaTiO3). With a perovskite structure, this material exhibits piezoelectric
Piezoelectricity (, ) is the electric charge that accumulates in certain solid materials—such as crystals, certain ceramics, and biological matter such as bone, DNA, and various proteins—in response to applied stress (mechanics), mechanical s ...
properties and is used as a transducer in the interconversion of sound
In physics, sound is a vibration that propagates as an acoustic wave through a transmission medium such as a gas, liquid or solid.
In human physiology and psychology, sound is the ''reception'' of such waves and their ''perception'' by the br ...
and electricity
Electricity is the set of physical phenomena associated with the presence and motion of matter possessing an electric charge. Electricity is related to magnetism, both being part of the phenomenon of electromagnetism, as described by Maxwel ...
. Many minerals are titanates, such as ilmenite (FeTiO3). Star sapphires and rubies get their asterism (star-forming shine) from the presence of titanium dioxide impurities.
A variety of reduced oxides ( suboxides) of titanium are known, mainly reduced stoichiometries of titanium dioxide obtained by atmospheric plasma spraying. Ti3O5, described as a Ti(IV)-Ti(III) species, is a purple semiconductor produced by reduction of TiO2 with hydrogen at high temperatures, and is used industrially when surfaces need to be vapor-coated with titanium dioxide: it evaporates as pure TiO, whereas TiO2 evaporates as a mixture of oxides and deposits coatings with variable refractive index. Also known is Ti2O3, with the corundum structure, and TiO, with the rock salt structure, although often nonstoichiometric.
The alkoxides of titanium(IV), prepared by treating TiCl4 with alcohol
Alcohol may refer to:
Common uses
* Alcohol (chemistry), a class of compounds
* Ethanol, one of several alcohols, commonly known as alcohol in everyday life
** Alcohol (drug), intoxicant found in alcoholic beverages
** Alcoholic beverage, an alco ...
s, are colorless compounds that convert to the dioxide on reaction with water. They are industrially useful for depositing solid TiO2 via the sol-gel process. Titanium isopropoxide is used in the synthesis of chiral organic compounds via the Sharpless epoxidation.
Titanium forms a variety of sulfides, but only TiS2 has attracted significant interest. It adopts a layered structure and was used as a cathode in the development of lithium batteries. Because Ti(IV) is a "hard cation", the sulfides of titanium are unstable and tend to hydrolyze to the oxide with release of hydrogen sulfide
Hydrogen sulfide is a chemical compound with the formula . It is a colorless chalcogen-hydride gas, and is toxic, corrosive, and flammable. Trace amounts in ambient atmosphere have a characteristic foul odor of rotten eggs. Swedish chemist ...
.
Nitrides and carbides
Titanium nitride (TiN) is a refractory solid exhibiting extreme hardness, thermal/electrical conductivity, and a high melting point. TiN has a hardness equivalent tosapphire
Sapphire is a precious gemstone, a variety of the mineral corundum, consisting of aluminium oxide () with trace amounts of elements such as iron, titanium, cobalt, lead, chromium, vanadium, magnesium, boron, and silicon. The name ''sapphire ...
and carborundum (9.0 on the Mohs scale
The Mohs scale ( ) of mineral hardness is a qualitative ordinal scale, from 1 to 10, characterizing scratch resistance of minerals through the ability of harder material to scratch softer material.
The scale was introduced in 1812 by the Ger ...
), and is often used to coat cutting tools, such as drill bits. It is also used as a gold-colored decorative finish and as a barrier layer in semiconductor fabrication. Titanium carbide (TiC), which is also very hard, is found in cutting tools and coatings.
Halides
 Titanium tetrachloride (titanium(IV) chloride, TiCl4) is a colorless volatile liquid (commercial samples are yellowish) that, in air, hydrolyzes with spectacular emission of white clouds. Via the Kroll process, TiCl4 is used in the conversion of titanium ores to titanium metal. Titanium tetrachloride is also used to make titanium dioxide, e.g., for use in white paint. It is widely used in
Titanium tetrachloride (titanium(IV) chloride, TiCl4) is a colorless volatile liquid (commercial samples are yellowish) that, in air, hydrolyzes with spectacular emission of white clouds. Via the Kroll process, TiCl4 is used in the conversion of titanium ores to titanium metal. Titanium tetrachloride is also used to make titanium dioxide, e.g., for use in white paint. It is widely used in organic chemistry
Organic chemistry is a subdiscipline within chemistry involving the science, scientific study of the structure, properties, and reactions of organic compounds and organic matter, organic materials, i.e., matter in its various forms that contain ...
as a Lewis acid, for example in the Mukaiyama aldol condensation. In the van Arkel–de Boer process, titanium tetraiodide (TiI4) is generated in the production of high purity titanium metal.
Titanium(III) and titanium(II) also form stable chlorides. A notable example is titanium(III) chloride (TiCl3), which is used as a catalyst for production of polyolefin
A polyolefin is a type of polymer with the general formula (CH2CHR)n where R is an alkyl group. They are usually derived from a small set of simple olefins (alkenes). Dominant in a commercial sense are polyethylene and polypropylene. More speciali ...
s (see Ziegler–Natta catalyst) and a reducing agent in organic chemistry.
Organometallic complexes
Owing to the important role of titanium compounds aspolymerization
In polymer chemistry, polymerization (American English), or polymerisation (British English), is a process of reacting monomer molecules together in a chemical reaction to form polymer chains or three-dimensional networks. There are many fo ...
catalyst, compounds with Ti-C bonds have been intensively studied. The most common organotitanium complex is titanocene dichloride
Titanocene dichloride is the organotitanium compound with the formula (hapticity, ''η''5-C5H5)2TiCl2, commonly abbreviated as Cp2TiCl2. This metallocene is a common reagent in organometallic and organic synthesis. It exists as a bright red solid t ...
((C5H5)2TiCl2). Related compounds include Tebbe's reagent and Petasis reagent. Titanium forms carbonyl complexes, e.g. (C5H5)2Ti(CO)2.
Anticancer therapy studies
Following the success of platinum-based chemotherapy, titanium(IV) complexes were among the first non-platinum compounds to be tested for cancer treatment. The advantage of titanium compounds lies in their high efficacy and low toxicity '' in vivo''. In biological environments, hydrolysis leads to the safe and inert titanium dioxide. Despite these advantages the first candidate compounds failed clinical trials due to insufficient efficacy to toxicity ratios and formulation complications. Further development resulted in the creation of potentially effective, selective, and stable titanium-based drugs.History
 Titanium was discovered in 1791 by the
Titanium was discovered in 1791 by the clergy
Clergy are formal leaders within established religions. Their roles and functions vary in different religious traditions, but usually involve presiding over specific rituals and teaching their religion's doctrines and practices. Some of the ter ...
man and geologist
A geologist is a scientist who studies the structure, composition, and History of Earth, history of Earth. Geologists incorporate techniques from physics, chemistry, biology, mathematics, and geography to perform research in the Field research, ...
William Gregor as an inclusion of a mineral in Cornwall
Cornwall (; or ) is a Ceremonial counties of England, ceremonial county in South West England. It is also one of the Celtic nations and the homeland of the Cornish people. The county is bordered by the Atlantic Ocean to the north and west, ...
, Great Britain. Gregor recognized the presence of a new element in ilmenite when he found black sand by a stream and noticed the sand was attracted by a magnet. Analyzing the sand, he determined the presence of two metal oxides: iron oxide
An iron oxide is a chemical compound composed of iron and oxygen. Several iron oxides are recognized. Often they are non-stoichiometric. Ferric oxyhydroxides are a related class of compounds, perhaps the best known of which is rust.
Iron ...
(explaining the attraction to the magnet) and 45.25% of a white metallic oxide he could not identify. Realizing that the unidentified oxide contained a metal that did not match any known element, in 1791 Gregor reported his findings in both German and French science journals: '' Crell's Annalen'' and ''Observations et Mémoires sur la Physique''. He named this oxide manaccanite.
Around the same time, Franz-Joseph Müller von Reichenstein produced a similar substance, but could not identify it. The oxide was independently rediscovered in 1795 by Prussia
Prussia (; ; Old Prussian: ''Prūsija'') was a Germans, German state centred on the North European Plain that originated from the 1525 secularization of the Prussia (region), Prussian part of the State of the Teutonic Order. For centuries, ...
n chemist Martin Heinrich Klaproth in rutile from Boinik (the German name of Bajmócska), a village in Hungary (now Bojničky in Slovakia).
Klaproth found that it contained a new element and named it for the Titans
In Greek mythology, the Titans ( ; ) were the pre-Twelve Olympians, Olympian gods. According to the ''Theogony'' of Hesiod, they were the twelve children of the primordial parents Uranus (mythology), Uranus (Sky) and Gaia (Earth). The six male ...
of Greek mythology
Greek mythology is the body of myths originally told by the Ancient Greece, ancient Greeks, and a genre of ancient Greek folklore, today absorbed alongside Roman mythology into the broader designation of classical mythology. These stories conc ...
. After hearing about Gregor's earlier discovery, he obtained a sample of manaccanite and confirmed that it contained titanium.
The currently known processes for extracting titanium from its various ores are laborious and costly; it is not possible to reduce the ore by heating with carbon
Carbon () is a chemical element; it has chemical symbol, symbol C and atomic number 6. It is nonmetallic and tetravalence, tetravalent—meaning that its atoms are able to form up to four covalent bonds due to its valence shell exhibiting 4 ...
(as in iron smelting) because titanium combines with the carbon to produce titanium carbide. An extraction of 95% pure titanium was achieved by Lars Fredrik Nilson and Otto Petterson. To achieve this they chlorinated titanium oxide in a carbon monoxide atmosphere with chlorine gas before reducing it to titanium metal by the use of sodium. Pure metallic titanium (99.9%) was first prepared in 1910 by Matthew A. Hunter at Rensselaer Polytechnic Institute by heating TiCl4 with sodium
Sodium is a chemical element; it has Symbol (chemistry), symbol Na (from Neo-Latin ) and atomic number 11. It is a soft, silvery-white, highly reactive metal. Sodium is an alkali metal, being in group 1 element, group 1 of the peri ...
at under great pressure in a batch process known as the Hunter process. Titanium metal was not used outside the laboratory until 1932 when William Justin Kroll produced it by reducing titanium tetrachloride (TiCl4) with calcium. Eight years later he refined this process with magnesium and with sodium in what became known as the Kroll process. Although research continues to seek cheaper and more efficient routes, such as the FFC Cambridge process, the Kroll process is still predominantly used for commercial production.
 Titanium of very high purity was made in small quantities when Anton Eduard van Arkel and Jan Hendrik de Boer discovered the iodide process in 1925, by reacting with iodine and decomposing the formed vapors over a hot filament to pure metal.
In the 1950s and 1960s, the Soviet Union pioneered the use of titanium in military and submarine applications ( Alfa class and Mike class) as part of programs related to the Cold War. Starting in the early 1950s, titanium came into use extensively in military aviation, particularly in high-performance jets, starting with aircraft such as the F-100 Super Sabre and Lockheed A-12 and SR-71.
Throughout the Cold War period, titanium was considered a strategic material by the U.S. government, and a large stockpile of titanium sponge (a porous form of the pure metal) was maintained by the Defense National Stockpile Center, until the stockpile was dispersed in the 2000s. As of 2021, the four leading producers of titanium sponge were China (52%), Japan (24%), Russia (16%) and Kazakhstan (7%).
Titanium of very high purity was made in small quantities when Anton Eduard van Arkel and Jan Hendrik de Boer discovered the iodide process in 1925, by reacting with iodine and decomposing the formed vapors over a hot filament to pure metal.
In the 1950s and 1960s, the Soviet Union pioneered the use of titanium in military and submarine applications ( Alfa class and Mike class) as part of programs related to the Cold War. Starting in the early 1950s, titanium came into use extensively in military aviation, particularly in high-performance jets, starting with aircraft such as the F-100 Super Sabre and Lockheed A-12 and SR-71.
Throughout the Cold War period, titanium was considered a strategic material by the U.S. government, and a large stockpile of titanium sponge (a porous form of the pure metal) was maintained by the Defense National Stockpile Center, until the stockpile was dispersed in the 2000s. As of 2021, the four leading producers of titanium sponge were China (52%), Japan (24%), Russia (16%) and Kazakhstan (7%).
Production

Mineral beneficiation processes
* The Becher process is an industrial process used to produce syntheticrutile
Rutile is an oxide mineral composed of titanium dioxide (TiO2), the most common natural form of TiO2. Rarer polymorphs of TiO2 are known, including anatase, akaogiite, and brookite.
Rutile has one of the highest refractive indices at vis ...
, a form of titanium dioxide, from the ore ilmenite.
* The Chloride process.
* The Sulfate process: "relies on sulfuric acid
Sulfuric acid (American spelling and the preferred IUPAC name) or sulphuric acid (English in the Commonwealth of Nations, Commonwealth spelling), known in antiquity as oil of vitriol, is a mineral acid composed of the elements sulfur, oxygen, ...
(H2SO4) to leach titanium from ilmenite ore (FeTiO3). The resulting reaction produces titanyl sulfate (TiOSO4). A secondary hydrolysis stage is used to break the titanyl sulfate into hydrated TiO2 and H2SO4. Finally, heat is used to remove the water and create the end product - pure TiO2."
Purification processes
Hunter process
The Hunter process was the first industrial process to produce pure metallic titanium. It was invented in 1910 by Matthew A. Hunter, achemist
A chemist (from Greek ''chēm(ía)'' alchemy; replacing ''chymist'' from Medieval Latin ''alchemist'') is a graduated scientist trained in the study of chemistry, or an officially enrolled student in the field. Chemists study the composition of ...
born in New Zealand who worked in the United States. The process involves reducing titanium tetrachloride (TiCl4) with sodium
Sodium is a chemical element; it has Symbol (chemistry), symbol Na (from Neo-Latin ) and atomic number 11. It is a soft, silvery-white, highly reactive metal. Sodium is an alkali metal, being in group 1 element, group 1 of the peri ...
(Na) in a batch reactor with an inert atmosphere at a temperature of 1,000 °C. Dilute hydrochloric acid is then used to leach the salt from the product.
:TiCl4(g) + 4 Na(l) → 4 NaCl(l) + Ti(s)
Kroll process
 The processing of titanium metal occurs in four major steps: reduction of titanium ore into "sponge", a porous form; melting of sponge, or sponge plus a master alloy to form an ingot; primary fabrication, where an ingot is converted into general mill products such as billet, bar, plate, sheet, strip, and
The processing of titanium metal occurs in four major steps: reduction of titanium ore into "sponge", a porous form; melting of sponge, or sponge plus a master alloy to form an ingot; primary fabrication, where an ingot is converted into general mill products such as billet, bar, plate, sheet, strip, and tube
Tube or tubes may refer to:
* ''Tube'' (2003 film), a 2003 Korean film
* "Tubes" (Peter Dale), performer on the Soccer AM television show
* Tube (band), a Japanese rock band
* Tube & Berger, the alias of dance/electronica producers Arndt Rör ...
; and secondary fabrication of finished shapes from mill products.
Because it cannot be readily produced by reduction of titanium dioxide, titanium metal is obtained by reduction of titanium tetrachloride (TiCl4) with magnesium metal in the Kroll process. The complexity of this batch production in the Kroll process explains the relatively high market value of titanium, despite the Kroll process being less expensive than the Hunter process. To produce the TiCl4 required by the Kroll process, the dioxide is subjected to carbothermic reduction in the presence of chlorine
Chlorine is a chemical element; it has Symbol (chemistry), symbol Cl and atomic number 17. The second-lightest of the halogens, it appears between fluorine and bromine in the periodic table and its properties are mostly intermediate between ...
. In this process, the chlorine gas is passed over a red-hot mixture of rutile or ilmenite in the presence of carbon.
After extensive purification by fractional distillation, the TiCl4 is reduced with molten magnesium in an argon
Argon is a chemical element; it has symbol Ar and atomic number 18. It is in group 18 of the periodic table and is a noble gas. Argon is the third most abundant gas in Earth's atmosphere, at 0.934% (9340 ppmv). It is more than twice as abu ...
atmosphere.
:Arkel-Boer process
The van Arkel–de Boer process was the first semi-industrial process for pure Titanium. It involves thermal decomposition of titanium tetraiodide.Armstrong process
Titanium powder is manufactured using a flow production process known as the Armstrong process that is similar to the batch production Hunter process. A stream of titanium tetrachloride gas is added to a stream of molten sodium; the products (sodium chloride salt and titanium particles) is filtered from the extra sodium. Titanium is then separated from the salt by water washing. Both sodium and chlorine are recycled to produce and process more titanium tetrachloride.Pilot plants
Methods for electrolytic production of Ti metal from using molten salt electrolytes have been researched and tested at laboratory and small pilot plant scales. The lead author of an impartial review published in 2017 considered his own process "ready for scaling up." A 2023 review "discusses theelectrochemical
Electrochemistry is the branch of physical chemistry concerned with the relationship between electrical potential difference and identifiable chemical change. These reactions involve electrons moving via an electronically conducting phase (typi ...
principles involved in the recovery of metals from aqueous solutions and fused salt electrolytes", with particular attention paid to titanium. While some metals such as nickel and copper
Copper is a chemical element; it has symbol Cu (from Latin ) and atomic number 29. It is a soft, malleable, and ductile metal with very high thermal and electrical conductivity. A freshly exposed surface of pure copper has a pinkish-orang ...
can be refined by electrowinning at room temperature, titanium must be in the molten state and "there is a strong chance of attack of the refractory
In materials science, a refractory (or refractory material) is a material that is resistant to decomposition by heat or chemical attack and that retains its strength and rigidity at high temperatures. They are inorganic, non-metallic compound ...
lining by molten titanium." Zhang et al concluded their Perspective on Thermochemical and Electrochemical Processes for Titanium Metal Production in 2017 that "Even though there are strong interests in the industry for finding a better method to produce Ti metal, and a large number of new concepts and improvements have been investigated at the laboratory or even at pilot plant scales, there is no new process to date that can replace the Kroll process commercially."
The Hydrogen assisted magnesiothermic reduction (HAMR) process uses titanium dihydride.
Fabrication
 All
All welding
Welding is a fabrication (metal), fabrication process that joins materials, usually metals or thermoplastics, primarily by using high temperature to melting, melt the parts together and allow them to cool, causing Fusion welding, fusion. Co ...
of titanium must be done in an inert atmosphere of argon or helium
Helium (from ) is a chemical element; it has chemical symbol, symbol He and atomic number 2. It is a colorless, odorless, non-toxic, inert gas, inert, monatomic gas and the first in the noble gas group in the periodic table. Its boiling point is ...
to shield it from contamination with atmospheric gases (oxygen, nitrogen, and hydrogen). Contamination causes a variety of conditions, such as embrittlement, which reduce the integrity of the assembly welds and lead to joint failure.
Titanium is very difficult to solder directly, and hence a solderable metal or alloy such as steel is coated on titanium prior to soldering. Titanium metal can be machined with the same equipment and the same processes as stainless steel
Stainless steel, also known as inox, corrosion-resistant steel (CRES), or rustless steel, is an iron-based alloy that contains chromium, making it resistant to rust and corrosion. Stainless steel's resistance to corrosion comes from its chromi ...
.
Titanium alloys
 Common titanium alloys are made by reduction. For example, cuprotitanium (rutile with
Common titanium alloys are made by reduction. For example, cuprotitanium (rutile with copper
Copper is a chemical element; it has symbol Cu (from Latin ) and atomic number 29. It is a soft, malleable, and ductile metal with very high thermal and electrical conductivity. A freshly exposed surface of pure copper has a pinkish-orang ...
added), ferrocarbon titanium (ilmenite reduced with coke in an electric furnace), and manganotitanium (rutile with manganese or manganese oxides) are reduced.
About fifty grades of titanium alloys are designed and currently used, although only a couple of dozen are readily available commercially. The ASTM International
ASTM International, formerly known as American Society for Testing and Materials, is a standards organization that develops and publishes voluntary consensus technical international standards for a wide range of materials, products, systems and s ...
recognizes 31 grades of titanium metal and alloys, of which grades one through four are commercially pure (unalloyed). Those four vary in tensile strength as a function of oxygen content, with grade 1 being the most ductile (lowest tensile strength with an oxygen content of 0.18%), and grade 4 the least ductile (highest tensile strength with an oxygen content of 0.40%). The remaining grades are alloys, each designed for specific properties of ductility, strength, hardness, electrical resistivity, creep resistance, specific corrosion resistance, and combinations thereof.
In addition to the ASTM specifications, titanium alloys are also produced to meet aerospace and military specifications (SAE-AMS, MIL-T), ISO standards, and country-specific specifications, as well as proprietary end-user specifications for aerospace, military, medical, and industrial applications.
Forming and forging
Commercially pure flat product (sheet, plate) can be formed readily, but processing must take into account of the tendency of the metal to springback. This is especially true of certain high-strength alloys. Exposure to the oxygen in air at the elevated temperatures used in forging results in formation of a brittle oxygen-rich metallic surface layer called " alpha case" that worsens the fatigue properties, so it must be removed by milling, etching, or electrochemical treatment. The working of titanium is very complicated, and may include Friction welding, cryo-forging, and Vacuum arc remelting.Applications
 Titanium is used in steel as an alloying element ( ferro-titanium) to reduce
Titanium is used in steel as an alloying element ( ferro-titanium) to reduce grain size
Grain size (or particle size) is the diameter of individual grains of sediment, or the lithified particles in clastic rocks. The term may also be applied to other granular materials. This is different from the crystallite size, which ...
and as a deoxidizer, and in stainless steel to reduce carbon content. Titanium is often alloyed with aluminium (to refine grain size), vanadium, copper (to harden), iron
Iron is a chemical element; it has symbol Fe () and atomic number 26. It is a metal that belongs to the first transition series and group 8 of the periodic table. It is, by mass, the most common element on Earth, forming much of Earth's o ...
, manganese
Manganese is a chemical element; it has Symbol (chemistry), symbol Mn and atomic number 25. It is a hard, brittle, silvery metal, often found in minerals in combination with iron. Manganese was first isolated in the 1770s. It is a transition m ...
, molybdenum
Molybdenum is a chemical element; it has Symbol (chemistry), symbol Mo (from Neo-Latin ''molybdaenum'') and atomic number 42. The name derived from Ancient Greek ', meaning lead, since its ores were confused with lead ores. Molybdenum minerals hav ...
, and other metals. Titanium mill products (sheet, plate, bar, wire, forgings, castings) find application in industrial, aerospace, recreational, and emerging markets. Powdered titanium is used in pyrotechnics
Pyrotechnics is the science and craft of creating fireworks, but also includes safety matches, oxygen candles, Pyrotechnic fastener, explosive bolts (and other fasteners), parts of automotive airbags, as well as gas-pressure blasting in mining, q ...
as a source of bright-burning particles.
Pigments, additives, and coatings
About 95% of all titanium ore is destined for refinement into titanium dioxide (), an intensely white permanentpigment
A pigment is a powder used to add or alter color or change visual appearance. Pigments are completely or nearly solubility, insoluble and reactivity (chemistry), chemically unreactive in water or another medium; in contrast, dyes are colored sub ...
used in paints, paper, toothpaste, and plastics. It is also used in cement, in gemstones, and as an optical opacifier in paper.
pigment is chemically inert, resists fading in sunlight, and is very opaque: it imparts a pure and brilliant white color to the brown or grey chemicals that form the majority of household plastics. In nature, this compound is found in the minerals anatase, brookite, and rutile. Paint made with titanium dioxide does well in severe temperatures and marine environments. Pure titanium dioxide has a very high index of refraction and an optical dispersion higher than diamond
Diamond is a Allotropes of carbon, solid form of the element carbon with its atoms arranged in a crystal structure called diamond cubic. Diamond is tasteless, odourless, strong, brittle solid, colourless in pure form, a poor conductor of e ...
. Titanium dioxide is used in sunscreens because it reflects and absorbs UV light.
Aerospace and marine
 Because titanium alloys have high
Because titanium alloys have high tensile strength
Ultimate tensile strength (also called UTS, tensile strength, TS, ultimate strength or F_\text in notation) is the maximum stress that a material can withstand while being stretched or pulled before breaking. In brittle materials, the ultimate ...
to density ratio, high corrosion resistance, fatigue resistance, high crack resistance, and ability to withstand moderately high temperatures without creeping, they are used in aircraft, armor plating, naval ships, spacecraft, and missiles. For these applications, titanium is alloyed with aluminium, zirconium, nickel, vanadium, and other elements to manufacture a variety of components including critical structural parts, landing gear
Landing gear is the undercarriage of an aircraft or spacecraft that is used for taxiing, takeoff or landing. For aircraft, it is generally needed for all three of these. It was also formerly called ''alighting gear'' by some manufacturers, s ...
, firewalls, exhaust ducts (helicopters), and hydraulic systems. In fact, about two thirds of all titanium metal produced is used in aircraft engines and frames. The titanium 6AL-4V alloy accounts for almost 50% of all alloys used in aircraft applications.
The Lockheed A-12 and the SR-71 "Blackbird" were two of the first aircraft frames where titanium was used, paving the way for much wider use in modern military and commercial aircraft. A large amount of titanium mill products are used in the production of many aircraft, such as (following values are amount of raw mill products used, only a fraction of this ends up in the finished aircraft): 116 metric tons are used in the Boeing 787, 77 in the Airbus A380, 59 in the Boeing 777, 45 in the Boeing 747
The Boeing 747 is a long-range wide-body aircraft, wide-body airliner designed and manufactured by Boeing Commercial Airplanes in the United States between 1968 and 2023.
After the introduction of the Boeing 707, 707 in October 1958, Pan Am ...
, 32 in the Airbus A340, 18 in the Boeing 737
The Boeing 737 is an American narrow-body aircraft, narrow-body aircraft produced by Boeing at its Boeing Renton Factory, Renton factory in Washington (state), Washington.
Developed to supplement the Boeing 727 on short and thin routes, the t ...
, 18 in the Airbus A330, and 12 in the Airbus A320
The Airbus A320 family is a series of narrow-body airliners developed and produced by Airbus.
The A320 was launched in March 1984, Maiden flight, first flew on 22 February 1987, and was introduced in April 1988 by Air France.
The first membe ...
. In aero engine applications, titanium is used for rotors, compressor blades, hydraulic system components, and nacelle
A nacelle ( ) is a streamlined container for aircraft parts such as Aircraft engine, engines, fuel or equipment. When attached entirely outside the airframe, it is sometimes called a pod, in which case it is attached with a Hardpoint#Pylon, pylo ...
s. An early use in jet engines was for the Orenda Iroquois
The Orenda PS.13 Iroquois was an advanced turbojet engine designed for military use. It was developed by the Canadian aircraft engine manufacturer Orenda Engines, a part of the Avro Canada group. Intended for the CF-105 Arrow interceptor, ...
in the 1950s.
Because titanium is resistant to corrosion by sea water, it is used to make propeller shafts, rigging, heat exchangers in desalination plant
Desalination is a process that removes mineral components from saline water. More generally, desalination is the removal of salts and minerals from a substance. One example is soil desalination. This is important for agriculture. It is possible ...
s, heater-chillers for salt water aquariums, fishing line and leader, and divers' knives. Titanium is used in the housings and components of ocean-deployed surveillance and monitoring devices for science and military. The former Soviet Union
The Union of Soviet Socialist Republics. (USSR), commonly known as the Soviet Union, was a List of former transcontinental countries#Since 1700, transcontinental country that spanned much of Eurasia from 1922 until Dissolution of the Soviet ...
developed techniques for making submarines with hulls of titanium alloys, forging titanium in huge vacuum tubes.
Industrial
 Welded titanium pipe and process equipment (heat exchangers, tanks, process vessels, valves) are used in the chemical and petrochemical industries primarily for corrosion resistance. Specific alloys are used in oil and gas downhole applications and nickel hydrometallurgy for their high strength (e. g.: titanium beta C alloy), corrosion resistance, or both. The pulp and paper industry uses titanium in process equipment exposed to corrosive media, such as sodium hypochlorite or wet chlorine gas (in the bleachery). Other applications include ultrasonic welding, wave soldering, and sputtering targets.
Titanium tetrachloride (TiCl4), a colorless liquid, is important as an intermediate in the process of making TiO2 and is also used to produce the Ziegler–Natta catalyst. Titanium tetrachloride is also used to iridize glass and, because it fumes strongly in moist air, it is used to make smoke screens.
Welded titanium pipe and process equipment (heat exchangers, tanks, process vessels, valves) are used in the chemical and petrochemical industries primarily for corrosion resistance. Specific alloys are used in oil and gas downhole applications and nickel hydrometallurgy for their high strength (e. g.: titanium beta C alloy), corrosion resistance, or both. The pulp and paper industry uses titanium in process equipment exposed to corrosive media, such as sodium hypochlorite or wet chlorine gas (in the bleachery). Other applications include ultrasonic welding, wave soldering, and sputtering targets.
Titanium tetrachloride (TiCl4), a colorless liquid, is important as an intermediate in the process of making TiO2 and is also used to produce the Ziegler–Natta catalyst. Titanium tetrachloride is also used to iridize glass and, because it fumes strongly in moist air, it is used to make smoke screens.
Consumer and architectural
Titanium metal is used in automotive applications, particularly in automobile and motorcycle racing where low weight and high strength and rigidity are critical. The metal is generally too expensive for the general consumer market, though some late model Corvettes have been manufactured with titanium exhausts, and a Corvette Z06's LT4 supercharged engine uses lightweight, solid titanium intake valves for greater strength and resistance to heat. Titanium is used in many sporting goods: tennis rackets, golf clubs, lacrosse stick shafts; cricket, hockey, lacrosse, and football helmet grills, and bicycle frames and components. Although not a mainstream material for bicycle production, titanium bikes have been used by racing teams and adventure cyclists. Titanium alloys are used in spectacle frames that are rather expensive but highly durable, long lasting, light weight, and cause no skin allergies. Titanium is a common material for backpacking cookware and eating utensils. Though more expensive than traditional steel or aluminium alternatives, titanium products can be significantly lighter without compromising strength. Titanium horseshoes are preferred to steel by farriers because they are lighter and more durable. Titanium has occasionally been used in architecture. The Monument to Yuri Gagarin, the first man to travel in space (), as well as theMonument to the Conquerors of Space
A monument is a type of structure that was explicitly created to commemorate a person or event, or which has become relevant to a social group as a part of their remembrance of historic times or cultural heritage, due to its artistic, historical ...
on top of the Cosmonaut Museum in Moscow are made of titanium for the metal's attractive color and association with rocketry. The Guggenheim Museum Bilbao and the Cerritos Millennium Library were the first buildings in Europe and North America, respectively, to be sheathed in titanium panels. Titanium sheathing was used in the Frederic C. Hamilton Building in Denver, Colorado.
Because of titanium's superior strength and light weight relative to other metals (steel, stainless steel, and aluminium), and because of recent advances in metalworking techniques, its use has become more widespread in the manufacture of firearms. Primary uses include pistol frames and revolver cylinders. For the same reasons, it is used in the body of some laptop computers (for example, in Apple
An apple is a round, edible fruit produced by an apple tree (''Malus'' spp.). Fruit trees of the orchard or domestic apple (''Malus domestica''), the most widely grown in the genus, are agriculture, cultivated worldwide. The tree originated ...
's PowerBook G4).
In 2023, Apple launched the iPhone 15 Pro, which uses a titanium enclosure.
Some upmarket lightweight and corrosion-resistant tools, such as shovels, knife handles and flashlights, are made of titanium or titanium alloys.
Jewelry
 Because of its durability, titanium has become more popular for designer jewelry (particularly, titanium rings). Its inertness makes it a good choice for those with allergies or those who will be wearing the jewelry in environments such as swimming pools. Titanium is also alloyed with gold to produce an alloy that can be marketed as 24-karat gold because the 1% of alloyed Ti is insufficient to require a lesser mark. The resulting alloy is roughly the hardness of 14-karat gold and is more durable than pure 24-karat gold.
Titanium's durability, light weight, and dent and corrosion resistance make it useful for
Because of its durability, titanium has become more popular for designer jewelry (particularly, titanium rings). Its inertness makes it a good choice for those with allergies or those who will be wearing the jewelry in environments such as swimming pools. Titanium is also alloyed with gold to produce an alloy that can be marketed as 24-karat gold because the 1% of alloyed Ti is insufficient to require a lesser mark. The resulting alloy is roughly the hardness of 14-karat gold and is more durable than pure 24-karat gold.
Titanium's durability, light weight, and dent and corrosion resistance make it useful for watch
A watch is a timepiece carried or worn by a person. It is designed to maintain a consistent movement despite the motions caused by the person's activities. A wristwatch is worn around the wrist, attached by a watch strap or another type of ...
cases. Some artists work with titanium to produce sculptures, decorative objects and furniture.
Titanium may be anodized to vary the thickness of the surface oxide layer, causing optical interference fringes and a variety of bright colors. With this coloration and chemical inertness, titanium is a popular metal for body piercing.
Titanium has a minor use in dedicated non-circulating coins and medals. In 1999, Gibraltar released the world's first titanium coin for the millennium celebration. The Gold Coast Titans, an Australian rugby league team, award a medal of pure titanium to their player of the year.
Medical
Because titanium is biocompatible (non-toxic and not rejected by the body), it has many medical uses, including surgical implements and implants, such as hip balls and sockets ( joint replacement) and dental implants that can stay in place for up to 20 years. The titanium is often alloyed with about 4% aluminium or 6% Al and 4% vanadium. Titanium has the inherent ability to osseointegrate, enabling use in dental implants that can last for over 30 years. This property is also useful for orthopedic implant applications. These benefit from titanium's lower modulus of elasticity (
Titanium has the inherent ability to osseointegrate, enabling use in dental implants that can last for over 30 years. This property is also useful for orthopedic implant applications. These benefit from titanium's lower modulus of elasticity (Young's modulus
Young's modulus (or the Young modulus) is a mechanical property of solid materials that measures the tensile or compressive stiffness when the force is applied lengthwise. It is the modulus of elasticity for tension or axial compression. Youn ...
) to more closely match that of the bone that such devices are intended to repair. As a result, skeletal loads are more evenly shared between bone and implant, leading to a lower incidence of bone degradation due to stress shielding and periprosthetic bone fractures, which occur at the boundaries of orthopedic implants. However, titanium alloys' stiffness is still more than twice that of bone, so adjacent bone bears a greatly reduced load and may deteriorate.
Because titanium is non-ferromagnetic
Ferromagnetism is a property of certain materials (such as iron) that results in a significant, observable magnetic permeability, and in many cases, a significant magnetic coercivity, allowing the material to form a permanent magnet. Ferromagne ...
, patients with titanium implants can be safely examined with magnetic resonance imaging
Magnetic resonance imaging (MRI) is a medical imaging technique used in radiology to generate pictures of the anatomy and the physiological processes inside the body. MRI scanners use strong magnetic fields, magnetic field gradients, and ...
(convenient for long-term implants). Preparing titanium for implantation in the body involves subjecting it to a high-temperature plasma arc which removes the surface atoms, exposing fresh titanium that is instantly oxidized.
Modern advancements in additive manufacturing techniques have increased potential for titanium use in orthopedic implant applications. Complex implant scaffold designs can be 3D-printed using titanium alloys, which allows for more patient-specific applications and increased implant osseointegration.
Titanium is used for the surgical instrument
A surgical instrument is a medical device for performing specific actions or carrying out desired effects during a surgery or operation, such as modifying biological tissue, or to provide access for viewing it. Over time, many different kinds of ...
s used in image-guided surgery, as well as wheelchairs, crutches, and any other products where high strength and low weight are desirable.
Titanium dioxide nanoparticle
A nanoparticle or ultrafine particle is a particle of matter 1 to 100 nanometres (nm) in diameter. The term is sometimes used for larger particles, up to 500 nm, or fibers and tubes that are less than 100 nm in only two directions. At ...
s are widely used in electronics and the delivery of pharmaceuticals and cosmetics.
Nuclear waste storage
Because of its corrosion resistance, containers made of titanium have been studied for the long-term storage of nuclear waste. Containers lasting more than 100,000 years are thought possible with manufacturing conditions that minimize material defects. A titanium "drip shield" could also be installed over containers of other types to enhance their longevity.Precautions
Titanium is non-toxic even in large doses and does not play any natural role inside thehuman body
The human body is the entire structure of a Human, human being. It is composed of many different types of Cell (biology), cells that together create Tissue (biology), tissues and subsequently Organ (biology), organs and then Organ system, org ...
. An estimated quantity of 0.8 milligrams of titanium is ingested by humans each day, but most passes through without being absorbed in the tissues. It does, however, sometimes bio-accumulate in tissues that contain silica
Silicon dioxide, also known as silica, is an oxide of silicon with the chemical formula , commonly found in nature as quartz. In many parts of the world, silica is the major constituent of sand. Silica is one of the most complex and abundant f ...
. One study indicates a possible connection between titanium and yellow nail syndrome.
As a powder or in the form of metal shavings, titanium metal poses a significant fire hazard and, when heated in air, an explosion hazard. Water and carbon dioxide
Carbon dioxide is a chemical compound with the chemical formula . It is made up of molecules that each have one carbon atom covalent bond, covalently double bonded to two oxygen atoms. It is found in a gas state at room temperature and at norma ...
are ineffective for extinguishing a titanium fire; Class D dry powder agents must be used instead.
When used in the production or handling of chlorine, titanium should not be exposed to dry chlorine gas because it may result in a titanium–chlorine fire.
Titanium can catch fire when a fresh, non-oxidized surface comes in contact with liquid oxygen.
Function in plants
plant
Plants are the eukaryotes that form the Kingdom (biology), kingdom Plantae; they are predominantly Photosynthesis, photosynthetic. This means that they obtain their energy from sunlight, using chloroplasts derived from endosymbiosis with c ...
s may use titanium to stimulate the production of carbohydrate
A carbohydrate () is a biomolecule composed of carbon (C), hydrogen (H), and oxygen (O) atoms. The typical hydrogen-to-oxygen atomic ratio is 2:1, analogous to that of water, and is represented by the empirical formula (where ''m'' and ''n'' ...
s and encourage growth. This may explain why most plants contain about 1 part per million (ppm) of titanium, food plants have about 2 ppm, and horsetail and nettle
Nettle refers to plants with stinging hairs, particularly those of the genus '' Urtica''. It can also refer to plants which resemble ''Urtica'' species in appearance but do not have stinging hairs. Plants called "nettle" include:
* ball nettle ...
contain up to 80 ppm.
See also
* Titanium alloys * Suboxide * Titanium in zircon geothermometry * Titanium ManFootnotes
References
Bibliography
* * * * * *External links
"Titanium: Our Next Major Metal"
in ''
Popular Science
Popular science (also called pop-science or popsci) is an interpretation of science intended for a general audience. While science journalism focuses on recent scientific developments, popular science is more broad ranging. It may be written ...
'' (October 1950), one of first general public detailed articles on Titanium
Titanium
at '' Periodic Videos'' (University of Nottingham)
Titanium.org
official website of the International Titanium Association, an industry association
Metallurgy of Titanium and its Alloys
- slide presentations, movies, and other material from Harshad Bhadeshia and other
Cambridge University
The University of Cambridge is a Public university, public collegiate university, collegiate research university in Cambridge, England. Founded in 1209, the University of Cambridge is the List of oldest universities in continuous operation, wo ...
metallurgists
{{Authority control
Aerospace materials
Biomaterials
Chemical elements with hexagonal close-packed structure
Chemical elements
Native element minerals
Pyrotechnic fuels
Transition metals