PET scans on:
[Wikipedia]
[Google]
[Amazon]
Positron emission tomography (PET) is a functional imaging technique that uses radioactive substances known as
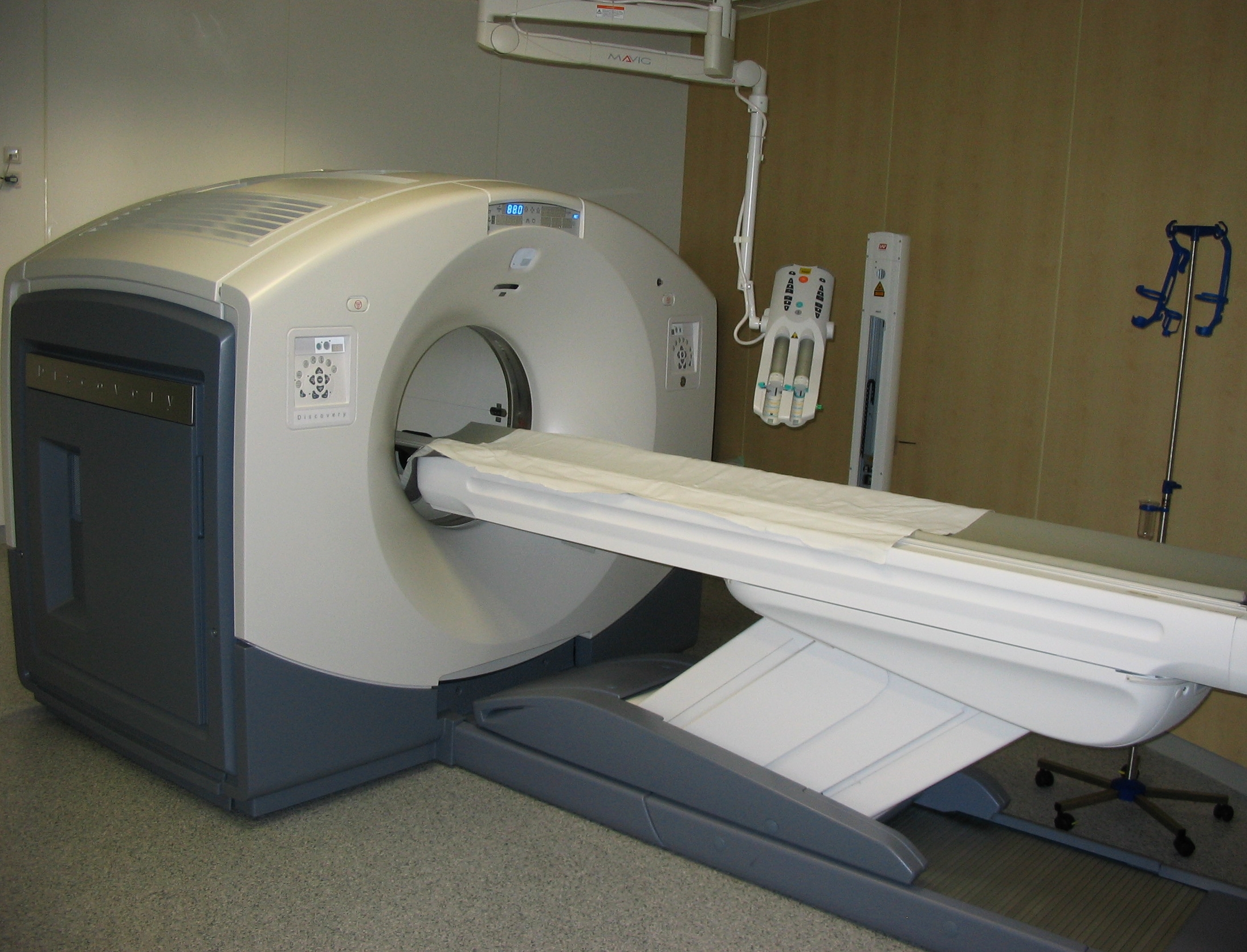 PET is both a medical and research tool used in pre-clinical and clinical settings. It is used heavily in the imaging of
PET is both a medical and research tool used in pre-clinical and clinical settings. It is used heavily in the imaging of
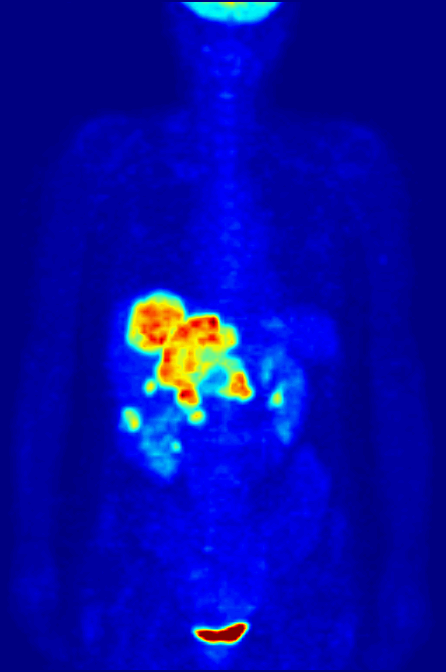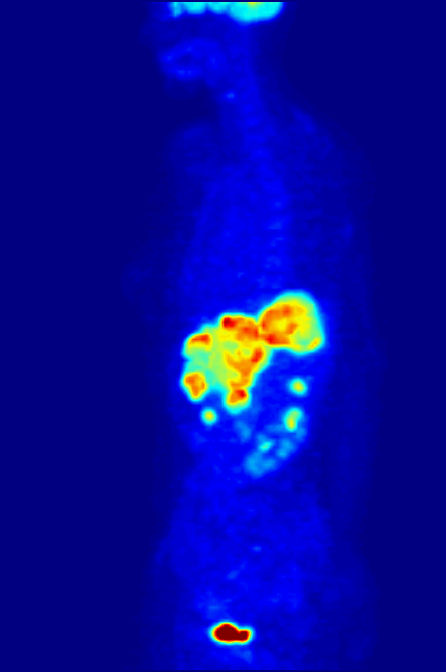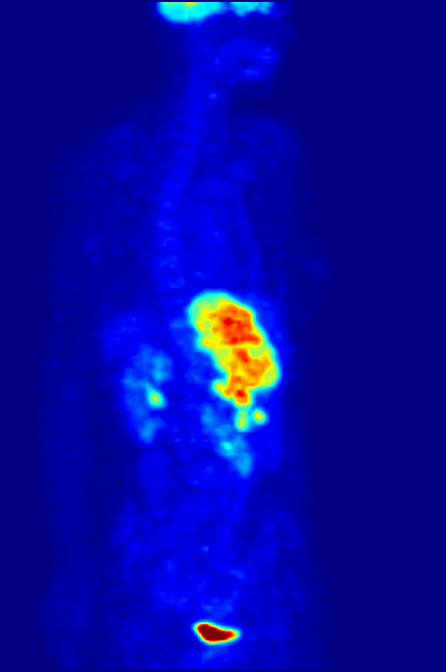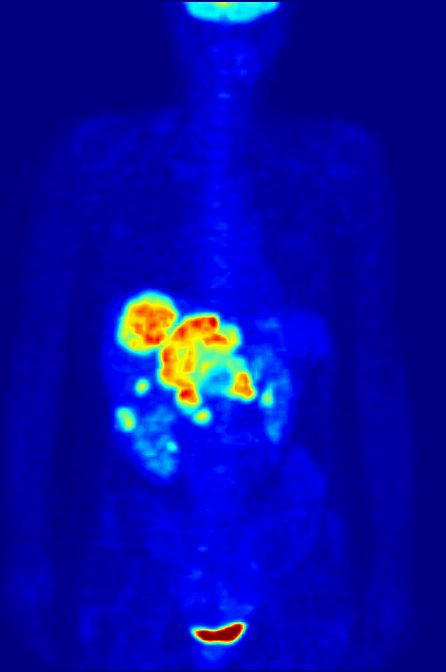 PET scanning with the tracer 18F-FDG is widely used in clinical
PET scanning with the tracer 18F-FDG is widely used in clinical
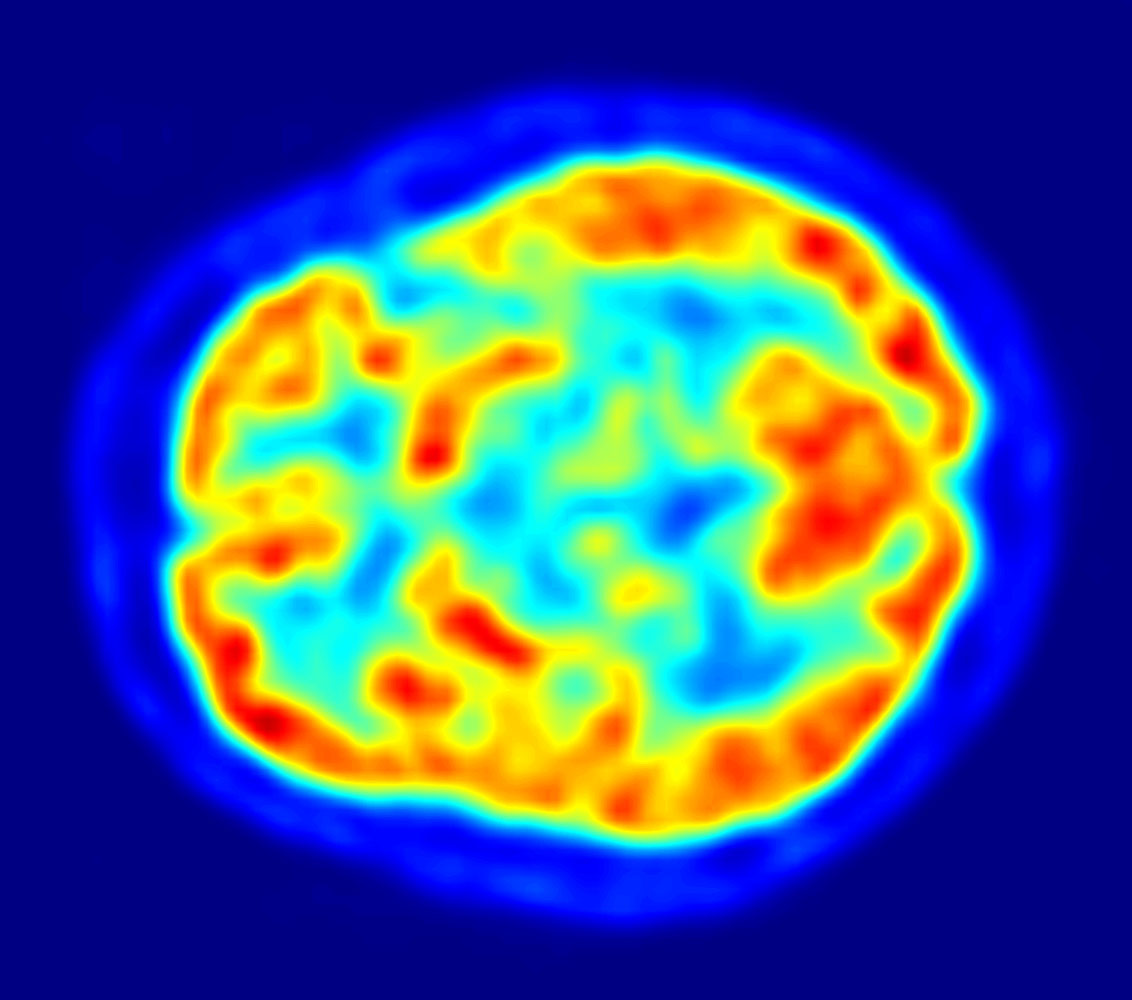 PET imaging with oxygen-15 indirectly measures blood flow to the brain. In this method, increased radioactivity signal indicates increased blood flow which is assumed to correlate with increased brain activity. Because of its 2-minute half-life, 15O must be piped directly from a medical
PET imaging with oxygen-15 indirectly measures blood flow to the brain. In this method, increased radioactivity signal indicates increased blood flow which is assumed to correlate with increased brain activity. Because of its 2-minute half-life, 15O must be piped directly from a medical
 Radionuclides are incorporated either into compounds normally used by the body such as
Radionuclides are incorporated either into compounds normally used by the body such as
 To conduct the scan, a short-lived radioactive tracer
To conduct the scan, a short-lived radioactive tracer
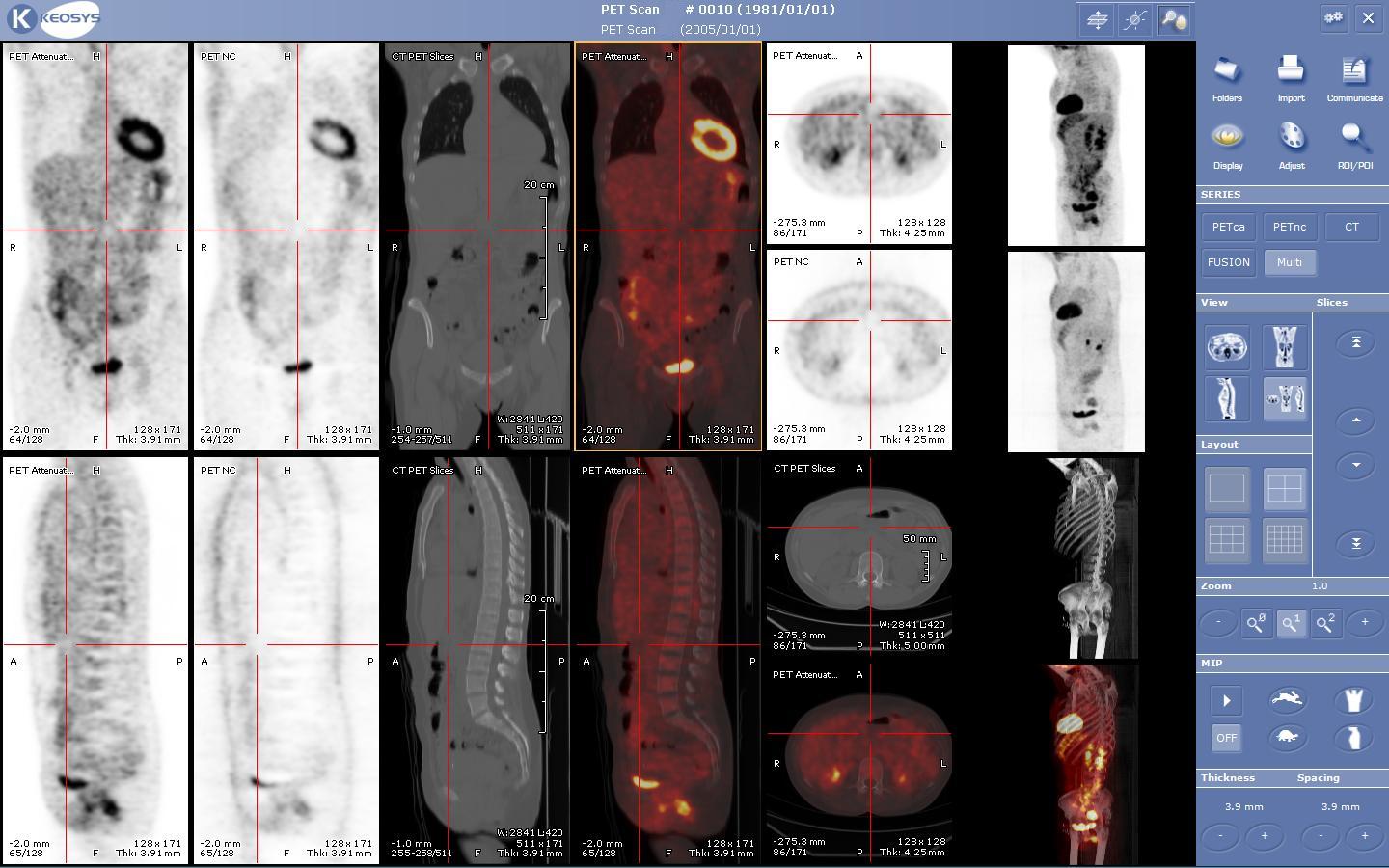
 PET scans are increasingly read alongside CT or magnetic resonance imaging (MRI) scans, with the combination (called "co-registration") giving both anatomic and metabolic information (i.e., what the structure is, and what it is doing biochemically). Because PET imaging is most useful in combination with anatomical imaging, such as CT, modern PET scanners are now available with integrated high-end multi-detector-row CT scanners (so-called "PET-CT"). Because the two scans can be performed in immediate sequence during the same session, with the patient not changing position between the two types of scans, the two sets of images are more precisely registered, so that areas of abnormality on the PET imaging can be more perfectly correlated with anatomy on the CT images. This is very useful in showing detailed views of moving organs or structures with higher anatomical variation, which is more common outside the brain.
At the Jülich Institute of Neurosciences and Biophysics, the world's largest PET-MRI device began operation in April 2009: a 9.4- tesla magnetic resonance tomograph (MRT) combined with a positron emission tomograph (PET). Presently, only the head and brain can be imaged at these high magnetic field strengths.
For brain imaging, registration of CT, MRI and PET scans may be accomplished without the need for an integrated PET-CT or PET-MRI scanner by using a device known as the
PET scans are increasingly read alongside CT or magnetic resonance imaging (MRI) scans, with the combination (called "co-registration") giving both anatomic and metabolic information (i.e., what the structure is, and what it is doing biochemically). Because PET imaging is most useful in combination with anatomical imaging, such as CT, modern PET scanners are now available with integrated high-end multi-detector-row CT scanners (so-called "PET-CT"). Because the two scans can be performed in immediate sequence during the same session, with the patient not changing position between the two types of scans, the two sets of images are more precisely registered, so that areas of abnormality on the PET imaging can be more perfectly correlated with anatomy on the CT images. This is very useful in showing detailed views of moving organs or structures with higher anatomical variation, which is more common outside the brain.
At the Jülich Institute of Neurosciences and Biophysics, the world's largest PET-MRI device began operation in April 2009: a 9.4- tesla magnetic resonance tomograph (MRT) combined with a positron emission tomograph (PET). Presently, only the head and brain can be imaged at these high magnetic field strengths.
For brain imaging, registration of CT, MRI and PET scans may be accomplished without the need for an integrated PET-CT or PET-MRI scanner by using a device known as the
PET-CT atlas Harvard Medical School
National Isotope Development Center
€”U.S. government source of radionuclides including those for PET—production, research, development, distribution, and information {{Authority control 3D nuclear medical imaging American inventions Antimatter Medical physics Neuroimaging Radiation therapy Medicinal radiochemistry Armenian inventions
radiotracer
A radioactive tracer, radiotracer, or radioactive label is a chemical compound in which one or more atoms have been replaced by a radionuclide so by virtue of its radioactive decay it can be used to explore the mechanism of chemical reactions by tr ...
s to visualize and measure changes in metabolic processes, and in other physiological activities including blood flow
Hemodynamics or haemodynamics are the dynamics of blood flow. The circulatory system is controlled by homeostatic mechanisms of autoregulation, just as hydraulic circuits are controlled by control systems. The hemodynamic response continuously m ...
, regional chemical composition, and absorption. Different tracers are used for various imaging purposes, depending on the target process within the body. For example, -FDG is commonly used to detect cancer
Cancer is a group of diseases involving abnormal cell growth with the potential to invade or spread to other parts of the body. These contrast with benign tumors, which do not spread. Possible signs and symptoms include a lump, abnormal b ...
, NaF is widely used for detecting bone formation, and oxygen-15 is sometimes used to measure blood flow.
PET is a common imaging technique, a medical scintillography technique used in nuclear medicine
Nuclear medicine or nucleology is a medical specialty involving the application of radioactive substances in the diagnosis and treatment of disease. Nuclear imaging, in a sense, is " radiology done inside out" because it records radiation emi ...
. A radiopharmaceutical — radioisotope
A radionuclide (radioactive nuclide, radioisotope or radioactive isotope) is a nuclide that has excess nuclear energy, making it unstable. This excess energy can be used in one of three ways: emitted from the nucleus as gamma radiation; transferr ...
attached to a drug — is injected into the body as a tracer. When the radiopharmaceutical undergoes beta plus decay
Positron emission, beta plus decay, or β+ decay is a subtype of radioactive decay called beta decay, in which a proton inside a radionuclide nucleus is converted into a neutron while releasing a positron and an electron neutrino (). Positron ...
, a positron is emitted, and when the positron collides with an ordinary electron, the two particles annihilate and gamma rays
A gamma ray, also known as gamma radiation (symbol γ or \gamma), is a penetrating form of electromagnetic radiation arising from the radioactive decay of atomic nuclei. It consists of the shortest wavelength electromagnetic waves, typically ...
are emitted. These gamma rays are detected by gamma camera
A gamma camera (γ-camera), also called a scintillation camera or Anger camera, is a device used to image gamma radiation emitting radioisotopes, a technique known as scintigraphy. The applications of scintigraphy include early drug development ...
s to form a three-dimensional image, in a similar way that an X-ray image is captured.
PET scanners can incorporate a CT scanner and are known as PET-CT scanners. PET scan images can be reconstructed using a CT scan performed using one scanner during the same session.
One of the disadvantages of a PET scanner is its high initial cost and ongoing operating costs.
Uses
 PET is both a medical and research tool used in pre-clinical and clinical settings. It is used heavily in the imaging of
PET is both a medical and research tool used in pre-clinical and clinical settings. It is used heavily in the imaging of tumor
A neoplasm () is a type of abnormal and excessive growth of tissue. The process that occurs to form or produce a neoplasm is called neoplasia. The growth of a neoplasm is uncoordinated with that of the normal surrounding tissue, and persists ...
s and the search for metastases
Metastasis is a pathogenic agent's spread from an initial or primary site to a different or secondary site within the host's body; the term is typically used when referring to metastasis by a cancerous tumor. The newly pathological sites, then ...
within the field of clinical oncology
Oncology is a branch of medicine that deals with the study, treatment, diagnosis and prevention of cancer. A medical professional who practices oncology is an ''oncologist''. The name's etymological origin is the Greek word ὄγκος (''à ...
, and for the clinical diagnosis of certain diffuse brain diseases such as those causing various types of dementias. PET is a valuable research tool to learn and enhance our knowledge of the normal human brain, heart function, and support drug development. PET is also used in pre-clinical studies using animals. It allows repeated investigations into the same subjects over time, where subjects can act as their own control and substantially reduces the numbers of animals required for a given study. This approach allows research studies to reduce the sample size needed while increasing the statistical quality of its results.
Physiological processes lead to anatomical changes in the body. Since PET is capable of detecting biochemical processes as well as expression of some proteins, PET can provide molecular-level information much before any anatomic changes are visible. PET scanning does this by using radiolabelled molecular probes that have different rates of uptake depending on the type and function of tissue involved. Regional tracer uptake in various anatomic structures can be visualized and relatively quantified in terms of injected positron emitter within a PET scan.
PET imaging is best performed using a dedicated PET scanner. It is also possible to acquire PET images using a conventional dual-head gamma camera
A gamma camera (γ-camera), also called a scintillation camera or Anger camera, is a device used to image gamma radiation emitting radioisotopes, a technique known as scintigraphy. The applications of scintigraphy include early drug development ...
fitted with a coincidence detector. The quality of gamma-camera PET imaging is lower, and the scans take longer to acquire. However, this method allows a low-cost on-site solution to institutions with low PET scanning demand. An alternative would be to refer these patients to another center or relying on a visit by a mobile scanner.
Alternative methods of medical imaging include single-photon emission computed tomography (SPECT), x-ray
An X-ray, or, much less commonly, X-radiation, is a penetrating form of high-energy electromagnetic radiation. Most X-rays have a wavelength ranging from 10 picometers to 10 nanometers, corresponding to frequencies in the range 30&nb ...
computed tomography (CT), magnetic resonance imaging (MRI) and functional magnetic resonance imaging (fMRI), and ultrasound
Ultrasound is sound waves with frequencies higher than the upper audible limit of human hearing. Ultrasound is not different from "normal" (audible) sound in its physical properties, except that humans cannot hear it. This limit varies ...
. SPECT is an imaging technique similar to PET that uses radioligand
A radioligand is a radioactive biochemical substance (in particular, a ligand (biochemistry), ligand that is radioactive tracer, radiolabeled) that is used for diagnosis or for research-oriented study of the receptor (biochemistry), receptor system ...
s to detect molecules in the body. SPECT is less expensive and provides inferior image quality than PET.
Oncology
 PET scanning with the tracer 18F-FDG is widely used in clinical
PET scanning with the tracer 18F-FDG is widely used in clinical oncology
Oncology is a branch of medicine that deals with the study, treatment, diagnosis and prevention of cancer. A medical professional who practices oncology is an ''oncologist''. The name's etymological origin is the Greek word ὄγκος (''à ...
. FDG is a glucose
Glucose is a simple sugar with the molecular formula . Glucose is overall the most abundant monosaccharide, a subcategory of carbohydrates. Glucose is mainly made by plants and most algae during photosynthesis from water and carbon dioxide, u ...
analog
Analog or analogue may refer to:
Computing and electronics
* Analog signal, in which information is encoded in a continuous variable
** Analog device, an apparatus that operates on analog signals
*** Analog electronics, circuits which use analog ...
that is taken up by glucose-using cells and phosphorylated by hexokinase
A hexokinase is an enzyme that phosphorylates hexoses (six-carbon sugars), forming hexose phosphate. In most organisms, glucose is the most important substrate for hexokinases, and glucose-6-phosphate is the most important product. Hexok ...
(whose mitochondrial form is significantly elevated in rapidly growing malignant
Malignancy () is the tendency of a medical condition to become progressively worse.
Malignancy is most familiar as a characterization of cancer. A ''malignant'' tumor contrasts with a non-cancerous ''benign'' tumor in that a malignancy is not s ...
tumors). Metabolic trapping
Metabolic trapping refers to a localization mechanism of synthesized radiocompounds in the human body. It can be defined as the intracellular accumulation of a radioactive tracer based on the relative metabolic activity of the body's tissues. It ...
of the radioactive glucose molecule allows the PET scan to be utilized. The concentrations of imaged FDG tracer indicate tissue metabolic activity as it corresponds to the regional glucose uptake. 18F-FDG is used to explore the possibility of cancer spreading to other body sites (cancer
Cancer is a group of diseases involving abnormal cell growth with the potential to invade or spread to other parts of the body. These contrast with benign tumors, which do not spread. Possible signs and symptoms include a lump, abnormal b ...
metastasis
Metastasis is a pathogenic agent's spread from an initial or primary site to a different or secondary site within the host's body; the term is typically used when referring to metastasis by a cancerous tumor. The newly pathological sites, then ...
). These 18F-FDG PET scans for detecting cancer metastasis are the most common in standard medical care (representing 90% of current scans). The same tracer may also be used for the diagnosis of types of dementia
Dementia is a disorder which manifests as a set of related symptoms, which usually surfaces when the brain is damaged by injury or disease. The symptoms involve progressive impairments in memory, thinking, and behavior, which negatively affe ...
. Less often, other radioactive tracers
A radioactive tracer, radiotracer, or radioactive label is a chemical compound in which one or more atoms have been replaced by a radionuclide so by virtue of its radioactive decay it can be used to explore the mechanism of chemical reactions by ...
, usually but not always labelled with fluorine-18
Fluorine-18 (18F) is a fluorine radioisotope which is an important source of positrons. It has a mass of 18.0009380(6) u and its half-life is 109.771(20) minutes. It decays by positron emission 96% of the time and electron capture 4% of the time ...
, are used to image the tissue concentration of different kinds of molecules of interest inside the body.
A typical dose of FDG used in an oncological scan has an effective radiation dose of 7.6 mSv.ARSAC - Notes for Guidance on the Clinical Administration of Radiopharmaceuticals and use of Sealed Sources (March 2018 p.35) Because the hydroxy group that is replaced by fluorine-18 to generate FDG is required for the next step in glucose metabolism
Carbohydrate metabolism is the whole of the biochemical processes responsible for the metabolic formation, breakdown, and interconversion of carbohydrates in living organisms.
Carbohydrates are central to many essential metabolic pathways. Pla ...
in all cells, no further reactions occur in FDG. Furthermore, most tissues (with the notable exception of liver and kidneys) cannot remove the phosphate
In chemistry, a phosphate is an anion, salt, functional group or ester derived from a phosphoric acid. It most commonly means orthophosphate, a derivative of orthophosphoric acid .
The phosphate or orthophosphate ion is derived from phosph ...
added by hexokinase
A hexokinase is an enzyme that phosphorylates hexoses (six-carbon sugars), forming hexose phosphate. In most organisms, glucose is the most important substrate for hexokinases, and glucose-6-phosphate is the most important product. Hexok ...
. This means that FDG is trapped in any cell that takes it up until it decays, since phosphorylated
In chemistry, phosphorylation is the attachment of a phosphate group to a molecule or an ion. This process and its inverse, dephosphorylation, are common in biology and could be driven by natural selection. Text was copied from this source, wh ...
sugars, due to their ionic charge, cannot exit from the cell. This results in intense radiolabeling of tissues with high glucose uptake, such as the normal brain, liver, kidneys, and most cancers, which have a higher glucose uptake than most normal tissue due to the Warburg effect. As a result, FDG-PET can be used for diagnosis, staging, and monitoring treatment of cancers, particularly in Hodgkin lymphoma
Hodgkin lymphoma (HL) is a type of lymphoma, in which cancer originates from a specific type of white blood cell called lymphocytes, where multinucleated Reed–Sternberg cells (RS cells) are present in the patient's lymph nodes. The condition w ...
, non-Hodgkin lymphoma
Non-Hodgkin lymphoma (NHL), also known as non-Hodgkin's lymphoma, is a group of blood cancers that includes all types of lymphomas except Hodgkin lymphomas. Symptoms include enlarged lymph nodes, fever, night sweats, weight loss, and tirednes ...
, and lung cancer
Lung cancer, also known as lung carcinoma (since about 98–99% of all lung cancers are carcinomas), is a malignant lung tumor characterized by uncontrolled cell growth in tissues of the lung. Lung carcinomas derive from transformed, malign ...
.
A 2020 review of research on the use of PET for Hodgkin lymphoma found evidence that negative findings in interim PET scans are linked to higher overall survival
Survival rate is a part of survival analysis. It is the proportion of people in a study or treatment group still alive at a given period of time after diagnosis. It is a method of describing prognosis in certain disease conditions, and can be use ...
and progression-free survival Progression-free survival (PFS) is "the length of time during and after the treatment of a disease, such as cancer, that a patient lives with the disease but it does not get worse". In oncology, PFS usually refers to situations in which a tumor is p ...
; however, the certainty of the available evidence was moderate for survival, and very low for progression-free survival.
A few other isotopes and radiotracers are slowly being introduced into oncology for specific purposes. For example, 11C-labelled metomidate
Metomidate is a non-barbiturate imidazole that was discovered by Janssen Pharmaceutica in 1965 and under the names (Hypnodil, Nokemyl) is sold as a sedative-hypnotic drug used in Europe to treat humans and for veterinary purposes.
11C-labelled ...
(11C-metomidate) has been used to detect tumors of adrenocortical
The adrenal cortex is the outer region and also the largest part of an adrenal gland. It is divided into three separate zones: zona glomerulosa, zona fasciculata and zona reticularis. Each zone is responsible for producing specific hormones. It is ...
origin. Also, FDOPA PET/CT (or F-18-DOPA PET/CT) has proven to be a more sensitive alternative to finding and also localizing pheochromocytoma
Pheochromocytoma (PHEO or PCC) is a rare tumor of the adrenal medulla composed of chromaffin cells, also known as pheochromocytes. When a tumor composed of the same cells as a pheochromocytoma develops outside the adrenal gland, it is referred t ...
than the MIBG scan.
Neuroimaging
Neurology
 PET imaging with oxygen-15 indirectly measures blood flow to the brain. In this method, increased radioactivity signal indicates increased blood flow which is assumed to correlate with increased brain activity. Because of its 2-minute half-life, 15O must be piped directly from a medical
PET imaging with oxygen-15 indirectly measures blood flow to the brain. In this method, increased radioactivity signal indicates increased blood flow which is assumed to correlate with increased brain activity. Because of its 2-minute half-life, 15O must be piped directly from a medical cyclotron
A cyclotron is a type of particle accelerator invented by Ernest O. Lawrence in 1929–1930 at the University of California, Berkeley, and patented in 1932. Lawrence, Ernest O. ''Method and apparatus for the acceleration of ions'', filed: Jan ...
for such uses, which is difficult.
PET imaging with 18F-FDG takes advantage of the fact that the brain is normally a rapid user of glucose. Standard 18F-FDG PET of the brain measures regional glucose use and can be used in neuropathological diagnosis.
Brain pathologies such as Alzheimer's disease greatly decrease brain metabolism of both glucose and oxygen in tandem. Therefore 18F-FDG PET of the brain may also be used to successfully differentiate Alzheimer's disease from other dementing processes, and also to make early diagnoses of Alzheimer's disease. The advantage of 18F-FDG PET for these uses is its much wider availability. Some 18F based radioactive tracers used for Alzheimer's include florbetapir
Florbetapir (18F), sold under the brand name Amyvid, is a PET scanning radiopharmaceutical compound containing the radionuclide fluorine-18 that was approved for use in the United States in 2012, as a diagnostic tool for Alzheimer's disease.
* ...
, flutemetamol
Flutemetamol (18F) (trade name Vizamyl, by GE Healthcare) is a PET scanning radiopharmaceutical containing the radionuclide fluorine-18, used as a diagnostic tool for Alzheimer's disease.
Adverse effects
Adverse effects of flutemetamol include ...
, PiB and florbetaben
Florbetaben, a fluorine-18 (18F)-labeled stilbene derivative (formerly known as BAY-949172), trade name NeuraCeq, is a diagnostic radiotracer developed for routine clinical application to visualize β-amyloid plaques in the brain. It is indicat ...
, which are all used to detect amyloid-beta
Amyloid beta (Aβ or Abeta) denotes peptides of 36–43 amino acids that are the main component of the amyloid plaques found in the brains of people with Alzheimer's disease. The peptides derive from the amyloid precursor protein (APP), which is ...
plaques (a potential biomarker for Alzheimer's) in the brain.
PET imaging with FDG can also be used for localization of ''seizure focus''. A seizure focus will appear as hypometabolic during an interictal scan. Several radiotracer
A radioactive tracer, radiotracer, or radioactive label is a chemical compound in which one or more atoms have been replaced by a radionuclide so by virtue of its radioactive decay it can be used to explore the mechanism of chemical reactions by tr ...
s (i.e. radioligand
A radioligand is a radioactive biochemical substance (in particular, a ligand (biochemistry), ligand that is radioactive tracer, radiolabeled) that is used for diagnosis or for research-oriented study of the receptor (biochemistry), receptor system ...
s) have been developed for PET that are ligands for specific neuroreceptor subtypes such as sup>11Craclopride
Raclopride is a typical antipsychotic. It acts as a selective antagonist on D2 dopamine receptors. It has been used in trials studying Parkinson Disease.
Its selectivity to the cerebral D2 receptors is characterized by its respective Ki-valu ...
, sup>18F fallypride and sup>18F desmethoxyfallypride for dopamine D2/D3 receptors, sup>11CMcN 5652 and sup>11CDASB
DASB, also known as 3-amino-4-(2-dimethylaminomethylphenylsulfanyl)-benzonitrile, is a compound that binds to the serotonin transporter.
Labeled with carbon-11 — a radioactive isotope — it has been used as a radioligand in neuroimagin ...
for serotonin transporter
The serotonin transporter (SERT or 5-HTT) also known as the sodium-dependent serotonin transporter and solute carrier family 6 member 4 is a protein that in humans is encoded by the SLC6A4 gene. SERT is a type of monoamine transporter protein tha ...
s, sup>18F Mefway for serotonin 5HT1A receptors, sup>18F Nifene for nicotinic acetylcholine receptor
Nicotinic acetylcholine receptors, or nAChRs, are receptor polypeptides that respond to the neurotransmitter acetylcholine. Nicotinic receptors also respond to drugs such as the agonist nicotine. They are found in the central and peripheral ner ...
s or enzyme substrates (e.g. 6- FDOPA for the AADC enzyme). These agents permit the visualization of neuroreceptor pools in the context of a plurality of neuropsychiatric and neurologic illnesses.
PET may also be used for the diagnosis of hippocampal sclerosis
Hippocampal sclerosis (HS) or mesial temporal sclerosis (MTS) is a neuropathological condition with severe neuronal cell loss and gliosis in the hippocampus, specifically in the CA-1 (Cornu Ammonis area 1) and subiculum of the hippocampus. It w ...
, which causes epilepsy. FDG, and the less common tracers flumazenil
Flumazenil (also known as flumazepil, code name Ro 15-1788) is a selective GABAA receptor antagonist administered via injection, otic insertion, or intranasally. Therapeutically, it acts as both an antagonist and antidote to benzodiazepines ( ...
and MPPF have been explored for this purpose. If the sclerosis is unilateral (right hippocampus or left hippocampus), 18F-FDG uptake can be compared with the healthy side. Even if the diagnosis is difficult with MRI, it may be diagnosed with PET.
The development of a number of novel probes for noninvasive, in vivo PET imaging of neuroaggregate in human brain has brought amyloid imaging close to clinical use. The earliest amyloid imaging probes included 2-(1-ethylidene)malononitrile ( sup>18FDDNP) developed at the University of California, Los Angeles and N-methyl- sup>11C-(4'-methylaminophenyl)-6-hydroxybenzothiazole (termed Pittsburgh compound B
Pittsburgh compound B (PiB) is a radioactive analog of thioflavin T, which can be used in positron emission tomography scans to image beta-amyloid plaques in neuronal tissue. Due to this property, Pittsburgh compound B may be used in investiga ...
) developed at the University of Pittsburgh. These amyloid imaging probes permit the visualization of amyloid
Amyloids are aggregates of proteins characterised by a fibrillar morphology of 7–13 nm in diameter, a beta sheet (β-sheet) secondary structure (known as cross-β) and ability to be stained by particular dyes, such as Congo red. In the huma ...
plaques in the brains of Alzheimer's patients and could assist clinicians in making a positive clinical diagnosis of AD pre-mortem and aid in the development of novel anti-amyloid therapies. sup>11CMP (N- sup>11Cethylpiperidin-4-yl propionate) is a novel radiopharmaceutical used in PET imaging to determine the activity of the acetylcholinergic neurotransmitter system by acting as a substrate for acetylcholinesterase. Post-mortem examination of AD patients have shown decreased levels of acetylcholinesterase. sup>11CMP is used to map the acetylcholinesterase activity in the brain, which could allow for pre-mortem diagnoses of AD and help to monitor AD treatments. Avid Radiopharmaceuticals has developed and commercialized a compound called florbetapir
Florbetapir (18F), sold under the brand name Amyvid, is a PET scanning radiopharmaceutical compound containing the radionuclide fluorine-18 that was approved for use in the United States in 2012, as a diagnostic tool for Alzheimer's disease.
* ...
that uses the longer-lasting radionuclide fluorine-18
Fluorine-18 (18F) is a fluorine radioisotope which is an important source of positrons. It has a mass of 18.0009380(6) u and its half-life is 109.771(20) minutes. It decays by positron emission 96% of the time and electron capture 4% of the time ...
to detect amyloid plaques using PET scans.
Neuropsychology or cognitive neuroscience
To examine links between specific psychological processes or disorders and brain activity.Psychiatry
Numerous compounds that bind selectively to neuroreceptors of interest in biological psychiatry have been radiolabeled with C-11 or F-18.Radioligand
A radioligand is a radioactive biochemical substance (in particular, a ligand (biochemistry), ligand that is radioactive tracer, radiolabeled) that is used for diagnosis or for research-oriented study of the receptor (biochemistry), receptor system ...
s that bind to dopamine receptor
Dopamine receptors are a class of G protein-coupled receptors that are prominent in the vertebrate central nervous system (CNS). Dopamine receptors activate different effectors through not only G-protein coupling, but also signaling through diffe ...
s (D1, D2 receptor, reuptake transporter), serotonin receptors (5HT1A, 5HT2A, reuptake transporter) opioid receptor
Opioid receptors are a group of inhibitory G protein-coupled receptors with opioids as ligands. The endogenous opioids are dynorphins, enkephalins, endorphins, endomorphins and nociceptin. The opioid receptors are ~40% identical to somatostatin ...
s (mu and kappa) cholinergic receptors (nicotinic and muscarinic) and other sites have been used successfully in studies with human subjects. Studies have been performed examining the state of these receptors in patients compared to healthy controls in schizophrenia
Schizophrenia is a mental disorder characterized by continuous or relapsing episodes of psychosis. Major symptoms include hallucinations (typically hearing voices), delusions, and disorganized thinking. Other symptoms include social wit ...
, substance abuse, mood disorder
A mood disorder, also known as an affective disorder, is any of a group of conditions of mental and behavioral disorder where a disturbance in the person's mood is the main underlying feature. The classification is in the '' Diagnostic and St ...
s and other psychiatric conditions.
Stereotactic surgery and radiosurgery
PET-image guided surgery Image-guided surgery (IGS) is any surgical procedure where the surgeon uses tracked surgical instruments in conjunction with preoperative or intraoperative images in order to directly or indirectly guide the procedure. Image guided surgery systems ...
facilitates treatment of intracranial tumors, arteriovenous malformations and other surgically treatable conditions.
Cardiology
Cardiology
Cardiology () is a branch of medicine that deals with disorders of the heart and the cardiovascular system. The field includes medical diagnosis and treatment of congenital heart defects, coronary artery disease, heart failure, valvular h ...
, atherosclerosis
Atherosclerosis is a pattern of the disease arteriosclerosis in which the wall of the artery develops abnormalities, called lesions. These lesions may lead to narrowing due to the buildup of atheromatous plaque. At onset there are usually no s ...
and vascular disease study: 18F-FDG PET can help in identifying hibernating myocardium. However, the cost-effectiveness of PET for this role versus SPECT
Single-photon emission computed tomography (SPECT, or less commonly, SPET) is a nuclear medicine tomographic imaging technique using gamma rays. It is very similar to conventional nuclear medicine planar imaging using a gamma camera (that is, ...
is unclear. 18F-FDG PET imaging of atherosclerosis
Atherosclerosis is a pattern of the disease arteriosclerosis in which the wall of the artery develops abnormalities, called lesions. These lesions may lead to narrowing due to the buildup of atheromatous plaque. At onset there are usually no s ...
to detect patients at risk of stroke is also feasible. Also, it can help test the efficacy of novel anti-atherosclerosis therapies.
Infectious diseases
Imaging infections withmolecular imaging
Molecular imaging is a field of medical imaging that focuses on imaging molecules of medical interest within living patients. This is in contrast to conventional methods for obtaining molecular information from preserved tissue samples, such as h ...
technologies can improve diagnosis and treatment follow-up. Clinically, PET has been widely used to image bacterial infections using fluorodeoxyglucose
18F.html" ;"title="sup>18F">sup>18Fluorodeoxyglucose ( INN), or fluorodeoxyglucose F 18 (USAN and USP), also commonly called fluorodeoxyglucose and abbreviated 18F.html" ;"title="sup>18F">sup>18FDG, 2- 18F.html" ;"title="sup>18F">sup>18FDG or ...
(FDG) to identify the infection-associated inflammatory response. Three different PET contrast agents have been developed to image bacterial infections in vivo are sup>18F maltose, sup>18Faltohexaose, and sup>18F-fluorodeoxy sorbitol (FDS). FDS has the added benefit of being able to target only Enterobacteriaceae.
Bio-distribution studies
In pre-clinical trials, a new drug can beradiolabeled
A radioactive tracer, radiotracer, or radioactive label is a chemical compound in which one or more atoms have been replaced by a radionuclide so by virtue of its radioactive decay it can be used to explore the mechanism of chemical reactions by tr ...
and injected into animals. Such scans are referred to as biodistribution studies. The information regarding drug uptake, retention and elimination over time can be obtained quickly and cost-effectively compare to the older technique of killing and dissecting the animals. Commonly, drug occupancy at a purported site of action can be inferred indirectly by competition studies between unlabeled drug and radiolabeled compounds known apriori to bind with specificity to the site. A single radioligand can be used this way to test many potential drug candidates for the same target. A related technique involves scanning with radioligands that compete with an endogenous (naturally occurring) substance at a given receptor to demonstrate that a drug causes the release of the natural substance.
Small animal imaging
A miniature animal PET has been constructed that is small enough for a fully conscious rat to be scanned. This RatCAP (Rat Conscious Animal PET) allows animals to be scanned without the confounding effects of anesthesia. PET scanners designed specifically for imaging rodents, often referred to as microPET, as well as scanners for small primates, are marketed for academic and pharmaceutical research. The scanners are based on microminiature scintillators and amplified avalanche photodiodes (APDs) through a system that uses single-chip silicon photomultipliers. In 2018 theUC Davis School of Veterinary Medicine The University of California, Davis, School of Veterinary Medicine is the largest veterinary school in the United States. Established in 1948, the school is the primary health resource for California's various animal populations. In 2020, the schoo ...
became the first veterinary center to employ a small clinical PET-scanner as a pet-PET scan for clinical (rather than research) animal diagnosis. Because of cost as well as the marginal utility of detecting cancer metastases in companion animals (the primary use of this modality), veterinary PET scanning is expected to be rarely available in the immediate future.
Musculo-skeletal imaging
PET imaging has been used for imaging muscles and bones. 18F-FDG is the most commonly used tracer for imaging muscles, andNaF-F18
Sodium fluoride (NaF) is an inorganic compound with the formula . It is used in trace amounts in the fluoridation of drinking water, in toothpaste, in metallurgy, and as a flux. It is a colorless or white solid that is readily soluble in water. I ...
is the most widely used tracer for imaging bones.
Muscles
PET is a feasible technique for studying skeletal muscles during exercises like walking. Also, PET can provide muscle activation data about deep-lying muscles (such as the vastus intermedialis and thegluteus minimus
The gluteus minimus, or glutæus minimus, the smallest of the three gluteal muscles, is situated immediately beneath the gluteus medius.
Structure
It is fan-shaped, arising from the outer surface of the ilium, between the anterior and infer ...
) compared to techniques like electromyography, which can be used only on superficial muscles directly under the skin. However, a disadvantage is that PET provides no timing information about muscle activation because it has to be measured after the exercise is completed. This is due to the time it takes for FDG to accumulate in the activated muscles.
Bones
Together with NaF-F18, PET for bone imaging has been in use for 60 years for measuring regional bone metabolism and blood flow using static and dynamic scans. Researchers have recently started using NaF-18 to study bone metastasis as well.Safety
PET scanning is non-invasive, but it does involve exposure to ionizing radiation. 18 F-FDG, which is now the standard radiotracer used for PET neuroimaging and cancer patient management, has an effective radiation dose of 14 mSv. The amount of radiation in 18F-FDG is similar to the effective dose of spending one year in the American city ofDenver, Colorado
Denver () is a consolidated city and county, the capital, and most populous city of the U.S. state of Colorado. Its population was 715,522 at the 2020 census, a 19.22% increase since 2010. It is the 19th-most populous city in the Unit ...
(12.4 mSv/year). For comparison, radiation dosage for other medical procedures range from 0.02 mSv for a chest x-ray and 6.5–8 mSv for a CT scan of the chest. Average civil aircrews are exposed to 3 mSv/year, and the whole body occupational dose limit for nuclear energy workers in the USA is 50mSv/year. For scale, see Orders of magnitude (radiation)
Recognized effects of higher acute radiation doses are described in more detail in the article on radiation poisoning. Although the International System of Units (SI) defines the sievert (Sv) as the unit of radiation dose equivalent, chronic radi ...
.
For PET-CT
Positron emission tomography–computed tomography (better known as PET-CT or PET/CT) is a nuclear medicine technique which combines, in a single gantry, a positron emission tomography (PET) scanner and an x-ray computed tomography (CT) scan ...
scanning, the radiation exposure may be substantial—around 23–26 mSv (for a 70 kg person—dose is likely to be higher for higher body weights).
Operation
Radionuclides and radiotracers
 Radionuclides are incorporated either into compounds normally used by the body such as
Radionuclides are incorporated either into compounds normally used by the body such as glucose
Glucose is a simple sugar with the molecular formula . Glucose is overall the most abundant monosaccharide, a subcategory of carbohydrates. Glucose is mainly made by plants and most algae during photosynthesis from water and carbon dioxide, u ...
(or glucose analogues), water
Water (chemical formula ) is an Inorganic compound, inorganic, transparent, tasteless, odorless, and Color of water, nearly colorless chemical substance, which is the main constituent of Earth's hydrosphere and the fluids of all known living ...
, or ammonia
Ammonia is an inorganic compound of nitrogen and hydrogen with the formula . A stable binary hydride, and the simplest pnictogen hydride, ammonia is a colourless gas with a distinct pungent smell. Biologically, it is a common nitrogenous wa ...
, or into molecules that bind to receptors or other sites of drug action. Such labelled compounds are known as radiotracer
A radioactive tracer, radiotracer, or radioactive label is a chemical compound in which one or more atoms have been replaced by a radionuclide so by virtue of its radioactive decay it can be used to explore the mechanism of chemical reactions by tr ...
s. PET technology can be used to trace the biologic pathway of any compound in living humans (and many other species as well), provided it can be radiolabeled with a PET isotope. Thus, the specific processes that can be probed with PET are virtually limitless, and radiotracers for new target molecules and processes are continuing to be synthesized; as of this writing there are already dozens in clinical use and hundreds applied in research. In 2020 by far the most commonly used radiotracer in clinical PET scanning is the carbohydrate derivative fluorodeoxyglucose (18F) (18F-FDG). This radiotracer is used in essentially all scans for oncology and most scans in neurology, and thus makes up the large majority of radiotracer (>95%) used in PET and PET-CT scanning.
Due to the short half-lives of most positron-emitting radioisotopes, the radiotracers have traditionally been produced using a cyclotron
A cyclotron is a type of particle accelerator invented by Ernest O. Lawrence in 1929–1930 at the University of California, Berkeley, and patented in 1932. Lawrence, Ernest O. ''Method and apparatus for the acceleration of ions'', filed: Jan ...
in close proximity to the PET imaging facility. The half-life of fluorine-18 is long enough that radiotracers labeled with fluorine-18 can be manufactured commercially at offsite locations and shipped to imaging centers. Recently rubidium-82 generators have become commercially available. These contain strontium-82, which decays by electron capture
Electron capture (K-electron capture, also K-capture, or L-electron capture, L-capture) is a process in which the proton-rich nucleus of an electrically neutral atom absorbs an inner atomic electron, usually from the K or L electron shells. Thi ...
to produce positron-emitting rubidium-82.
The use of positron-emitting isotopes of metals in PET scans has been reviewed, including elements not listed above, such as lanthanides.
Immuno-PET
The isotope 89Zr has been applied to the tracking and quantification of molecular antibodies with positron emission tomography (PET) cameras (a method called "immuno-PET"). Thebiological half-life
Biological half-life (also known as elimination half-life, pharmacologic half-life) is the time taken for concentration of a biological substance (such as a medication) to decrease from its maximum concentration ( Cmax) to half of Cmax in the bl ...
of antibodies is typically on the order of days, see daclizumab
Daclizumab (trade name Zinbryta) is a therapeutic humanized monoclonal antibody which was used for the treatment of adults with relapsing forms of multiple sclerosis (MS). Daclizumab works by binding to CD25, the alpha subunit of the IL-2 receptor ...
and erenumab
Erenumab, sold under the brand name Aimovig, is a medication which targets the calcitonin gene-related peptide receptor (CGRPR) for the prevention of migraine. It is administered by subcutaneous injection.
Erenumab, which was developed by Amg ...
by way of example. To visualize and quantify the distribution of such antibodies in the body, the PET isotope 89Zr is well suited because its physical half-life matches the typical biological half-life of antibodies, see table above.
Emission
 To conduct the scan, a short-lived radioactive tracer
To conduct the scan, a short-lived radioactive tracer isotope
Isotopes are two or more types of atoms that have the same atomic number (number of protons in their nuclei) and position in the periodic table (and hence belong to the same chemical element), and that differ in nucleon numbers (mass numb ...
is injected into the living subject (usually into blood circulation). Each tracer atom has been chemically incorporated into a biologically active molecule. There is a waiting period while the active molecule becomes concentrated in tissues of interest; then the subject is placed in the imaging scanner. The molecule most commonly used for this purpose is F-18 labeled fluorodeoxyglucose
18F.html" ;"title="sup>18F">sup>18Fluorodeoxyglucose ( INN), or fluorodeoxyglucose F 18 (USAN and USP), also commonly called fluorodeoxyglucose and abbreviated 18F.html" ;"title="sup>18F">sup>18FDG, 2- 18F.html" ;"title="sup>18F">sup>18FDG or ...
(FDG), a sugar, for which the waiting period is typically an hour. During the scan, a record of tissue concentration is made as the tracer decays.
As the radioisotope undergoes positron emission
Positron emission, beta plus decay, or β+ decay is a subtype of radioactive decay called beta decay, in which a proton inside a radionuclide nucleus is converted into a neutron while releasing a positron and an electron neutrino (). Positron ...
decay (also known as positive beta decay
In nuclear physics, beta decay (β-decay) is a type of radioactive decay in which a beta particle (fast energetic electron or positron) is emitted from an atomic nucleus, transforming the original nuclide to an isobar of that nuclide. For ...
), it emits a positron, an antiparticle
In particle physics, every type of particle is associated with an antiparticle with the same mass but with opposite physical charges (such as electric charge). For example, the antiparticle of the electron is the positron (also known as an antie ...
of the electron
The electron ( or ) is a subatomic particle with a negative one elementary electric charge. Electrons belong to the first generation of the lepton particle family,
and are generally thought to be elementary particles because they have no ...
with opposite charge. The emitted positron travels in tissue for a short distance (typically less than 1 mm, but dependent on the isotope), during which time it loses kinetic energy, until it decelerates to a point where it can interact with an electron. The encounter annihilates both electron and positron, producing a pair of annihilation
In particle physics, annihilation is the process that occurs when a subatomic particle collides with its respective antiparticle to produce other particles, such as an electron colliding with a positron to produce two photons. The total energy ...
( gamma) photon
A photon () is an elementary particle that is a quantum of the electromagnetic field, including electromagnetic radiation such as light and radio waves, and the force carrier for the electromagnetic force. Photons are massless, so they a ...
s moving in approximately opposite directions. These are detected when they reach a scintillator in the scanning device, creating a burst of light which is detected by photomultiplier A photomultiplier is a device that converts incident photons into an electrical signal.
Kinds of photomultiplier include:
* Photomultiplier tube, a vacuum tube converting incident photons into an electric signal. Photomultiplier tubes (PMTs for sh ...
tubes or silicon avalanche photodiode
An avalanche photodiode (APD) is a highly sensitive semiconductor photodiode detector that exploits the photoelectric effect to convert light into electricity. From a functional standpoint, they can be regarded as the semiconductor analog of phot ...
s (Si APD). The technique depends on simultaneous or coincident detection of the pair of photons moving in approximately opposite directions (they would be exactly opposite in their center of mass frame, but the scanner has no way to know this, and so has a built-in slight direction-error tolerance). Photons that do not arrive in temporal "pairs" (i.e. within a timing-window of a few nanoseconds) are ignored.
Localization of the positron annihilation event
The most significant fraction of electron–positron annihilations results in two 511 keV gamma photons being emitted at almost 180 degrees to each other; hence, it is possible to localize their source along a straight line of coincidence (also called the line of response, or LOR). In practice, the LOR has a non-zero width as the emitted photons are not exactly 180 degrees apart. If the resolving time of the detectors is less than 500picoseconds
A picosecond (abbreviated as ps) is a unit of time in the International System of Units (SI) equal to 10−12 or (one trillionth) of a second. That is one trillionth, or one millionth of one millionth of a second, or 0.000 000 000&nbs ...
rather than about 10 nanoseconds
A nanosecond (ns) is a unit of time in the International System of Units (SI) equal to one billionth of a second, that is, of a second, or 10 seconds.
The term combines the SI prefix ''nano-'' indicating a 1 billionth submultiple of an SI unit ( ...
, it is possible to localize the event to a segment of a chord, whose length is determined by the detector timing resolution. As the timing resolution improves, the signal-to-noise ratio (SNR) of the image will improve, requiring fewer events to achieve the same image quality. This technology is not yet common, but it is available on some new systems.
Image reconstruction
The raw data collected by a PET scanner are a list of 'coincidence events' representing near-simultaneous detection (typically, within a window of 6 to 12 nanoseconds of each other) of annihilation photons by a pair of detectors. Each coincidence event represents a line in space connecting the two detectors along which the positron emission occurred (i.e., the line of response (LOR)). Analytical techniques, much like the reconstruction of computed tomography (CT) and single-photon emission computed tomography (SPECT) data, are commonly used, although thedata set A data set (or dataset) is a collection of data. In the case of tabular data, a data set corresponds to one or more database tables, where every column of a table represents a particular variable, and each row corresponds to a given record of the ...
collected in PET is much poorer than CT, so reconstruction techniques are more difficult. Coincidence events can be grouped into projection images, called sinograms. The sinograms are sorted by the angle of each view and tilt (for 3D images). The sinogram images are analogous to the projections captured by computed tomography (CT) scanners, and can be reconstructed in a similar way. The statistics of data thereby obtained are much worse than those obtained through transmission tomography. A normal PET data set has millions of counts for the whole acquisition, while the CT can reach a few billion counts. This contributes to PET images appearing "noisier" than CT. Two major sources of noise in PET are scatter (a detected pair of photons, at least one of which was deflected from its original path by interaction with matter in the field of view, leading to the pair being assigned to an incorrect LOR) and random events (photons originating from two different annihilation events but incorrectly recorded as a coincidence pair because their arrival at their respective detectors occurred within a coincidence timing window).
In practice, considerable pre-processing of the data is required—correction for random coincidences, estimation and subtraction of scattered photons, detector dead-time correction (after the detection of a photon, the detector must "cool down" again) and detector-sensitivity correction (for both inherent detector sensitivity and changes in sensitivity due to angle of incidence).
Filtered back projection (FBP) has been frequently used to reconstruct images from the projections. This algorithm has the advantage of being simple while having a low requirement for computing resources. Disadvantages are that shot noise in the raw data is prominent in the reconstructed images, and areas of high tracer uptake tend to form streaks across the image. Also, FBP treats the data deterministically—it does not account for the inherent randomness associated with PET data, thus requiring all the pre-reconstruction corrections described above.
Statistical, likelihood-based approaches:
Statistical, likelihood-based
iterative expectation-maximization algorithms such as the Shepp–Vardi algorithm
are now the preferred method of reconstruction. These algorithms compute an estimate of the likely distribution of annihilation events that led to the measured data, based on statistical principles. The advantage is a better noise profile and resistance to the streak artifacts common with FBP, but the disadvantage is higher computer resource requirements. A further advantage of statistical image reconstruction techniques is that the physical effects that would need to be pre-corrected for when using an analytical reconstruction algorithm, such as scattered photons, random coincidences, attenuation and detector dead-time, can be incorporated into the likelihood model being used in the reconstruction, allowing for additional noise reduction. Iterative reconstruction has also been shown to result in improvements in the resolution of the reconstructed images, since more sophisticated models of the scanner physics can be incorporated into the likelihood model than those used by analytical reconstruction methods, allowing for improved quantification of the radioactivity distribution.
Research has shown that Bayesian
Thomas Bayes (/beɪz/; c. 1701 – 1761) was an English statistician, philosopher, and Presbyterian minister.
Bayesian () refers either to a range of concepts and approaches that relate to statistical methods based on Bayes' theorem, or a followe ...
methods that involve a Poisson likelihood function and an appropriate prior probability
In Bayesian statistical inference, a prior probability distribution, often simply called the prior, of an uncertain quantity is the probability distribution that would express one's beliefs about this quantity before some evidence is taken into ...
(e.g., a smoothing prior leading to total variation regularization
In signal processing, particularly image processing, total variation denoising, also known as total variation regularization or total variation filtering, is a noise removal process (filter). It is based on the principle that signals with excessi ...
or a Laplacian distribution leading to -based regularization in a wavelet
A wavelet is a wave-like oscillation with an amplitude that begins at zero, increases or decreases, and then returns to zero one or more times. Wavelets are termed a "brief oscillation". A taxonomy of wavelets has been established, based on the num ...
or other domain), such as via Ulf Grenander
Ulf Grenander (23 July 1923 – 12 May 2016) was a Swedish statistician and professor of applied mathematics at Brown University.
His early research was in probability theory, stochastic processes, time series analysis, and statistical theory (p ...
's Sieve estimator In statistics, sieve estimators are a class of non-parametric estimators which use progressively more complex models to estimate an unknown high-dimensional function as more data becomes available, with the aim of asymptotically reducing error tow ...
or via Bayes penalty methods
or via I.J. Good's roughness method may yield superior performance to expectation-maximization-based methods which involve a Poisson likelihood function but do not involve such a prior.
Attenuation correction: Quantitative PET Imaging requires attenuation correction. In these systems attenuation correction is based on a transmission scan using 68Ge rotating rod source.
Transmission scans directly measure attenuation values at 511keV. Attenuation occurs when photon
A photon () is an elementary particle that is a quantum of the electromagnetic field, including electromagnetic radiation such as light and radio waves, and the force carrier for the electromagnetic force. Photons are massless, so they a ...
s emitted by the radiotracer inside the body are absorbed by intervening tissue between the detector and the emission of the photon. As different LORs must traverse different thicknesses of tissue, the photons are attenuated differentially. The result is that structures deep in the body are reconstructed as having falsely low tracer uptake. Contemporary scanners can estimate attenuation using integrated x-ray
An X-ray, or, much less commonly, X-radiation, is a penetrating form of high-energy electromagnetic radiation. Most X-rays have a wavelength ranging from 10 picometers to 10 nanometers, corresponding to frequencies in the range 30&nb ...
CT equipment, in place of earlier equipment that offered a crude form of CT using a gamma ray
A gamma ray, also known as gamma radiation (symbol γ or \gamma), is a penetrating form of electromagnetic radiation arising from the radioactive decay of atomic nuclei. It consists of the shortest wavelength electromagnetic waves, typically ...
( positron emitting) source and the PET detectors.
While attenuation-corrected images are generally more faithful representations, the correction process is itself susceptible to significant artifacts. As a result, both corrected and uncorrected images are always reconstructed and read together.
2D/3D reconstruction: Early PET scanners had only a single ring of detectors, hence the acquisition of data and subsequent reconstruction was restricted to a single transverse plane. More modern scanners now include multiple rings, essentially forming a cylinder of detectors.
There are two approaches to reconstructing data from such a scanner: 1) treat each ring as a separate entity, so that only coincidences within a ring are detected, the image from each ring can then be reconstructed individually (2D reconstruction), or 2) allow coincidences to be detected between rings as well as within rings, then reconstruct the entire volume together (3D).
3D techniques have better sensitivity (because more coincidences are detected and used) and therefore less noise, but are more sensitive to the effects of scatter and random coincidences, as well as requiring correspondingly greater computer resources. The advent of sub-nanosecond timing resolution detectors affords better random coincidence rejection, thus favoring 3D image reconstruction.
Time-of-flight (TOF) PET: For modern systems with a higher time resolution (roughly 3 nanoseconds) a technique called "time-of-flight" is used to improve the overall performance. Time-of-flight PET makes use of very fast gamma-ray detectors and data processing system which can more precisely decide the difference in time between the detection of the two photons. It is impossible to localize the point of origin of the annihilation event exactly (currently within 10 cm). Therefore image reconstruction is still needed. TOF technique gives a remarkable improvement in image quality, especially signal-to-noise ratio.
Combination of PET with CT or MRI
N-localizer
The N-localizer is a device that enables guidance of stereotactic surgery or radiosurgery using tomographic images that are obtained via computed tomography (CT), magnetic resonance imaging (MRI), or positron emission tomography (PET). The N-local ...
.
Limitations
The minimization of radiation dose to the subject is an attractive feature of the use of short-lived radionuclides. Besides its established role as a diagnostic technique, PET has an expanding role as a method to assess the response to therapy, in particular, cancer therapy, where the risk to the patient from lack of knowledge about disease progress is much greater than the risk from the test radiation. Since the tracers are radioactive, the elderly and pregnant are unable to use it due to risks posed by radiation. Limitations to the widespread use of PET arise from the high costs ofcyclotrons
A cyclotron is a type of particle accelerator invented by Ernest O. Lawrence in 1929–1930 at the University of California, Berkeley, and patented in 1932. Lawrence, Ernest O. ''Method and apparatus for the acceleration of ions'', filed: Janu ...
needed to produce the short-lived radionuclides for PET scanning and the need for specially adapted on-site chemical synthesis apparatus to produce the radiopharmaceuticals after radioisotope preparation. Organic radiotracer molecules that will contain a positron-emitting radioisotope cannot be synthesized first and then the radioisotope prepared within them, because bombardment with a cyclotron to prepare the radioisotope destroys any organic carrier for it. Instead, the isotope must be prepared first, then afterward, the chemistry to prepare any organic radiotracer (such as FDG) accomplished very quickly, in the short time before the isotope decays. Few hospitals and universities are capable of maintaining such systems, and most clinical PET is supported by third-party suppliers of radiotracers that can supply many sites simultaneously. This limitation restricts clinical PET primarily to the use of tracers labelled with fluorine-18, which has a half-life of 110 minutes and can be transported a reasonable distance before use, or to rubidium-82 (used as rubidium-82 chloride) with a half-life of 1.27 minutes, which is created in a portable generator and is used for myocardial
Cardiac muscle (also called heart muscle, myocardium, cardiomyocytes and cardiac myocytes) is one of three types of vertebrate muscle tissues, with the other two being skeletal muscle and smooth muscle. It is an involuntary, striated muscle that ...
perfusion studies. Nevertheless, in recent years a few on-site cyclotrons with integrated shielding and "hot labs" (automated chemistry labs that are able to work with radioisotopes) have begun to accompany PET units to remote hospitals. The presence of the small on-site cyclotron promises to expand in the future as the cyclotrons shrink in response to the high cost of isotope transportation to remote PET machines. In recent years the shortage of PET scans has been alleviated in the US, as rollout of radiopharmacies to supply radioisotopes has grown 30%/year.
Because the half-life of fluorine-18 is about two hours, the prepared dose of a radiopharmaceutical bearing this radionuclide will undergo multiple half-lives of decay during the working day. This necessitates frequent recalibration of the remaining dose (determination of activity per unit volume) and careful planning with respect to patient scheduling.
History
The concept of emission and transmissiontomography
Tomography is imaging by sections or sectioning that uses any kind of penetrating wave. The method is used in radiology, archaeology, biology, atmospheric science, geophysics, oceanography, plasma physics, materials science, astrophysics, ...
was introduced by David E. Kuhl, Luke Chapman and Roy Edwards in the late 1950s. Their work later led to the design and construction of several tomographic instruments at the University of Pennsylvania
The University of Pennsylvania (also known as Penn or UPenn) is a private research university in Philadelphia. It is the fourth-oldest institution of higher education in the United States and is ranked among the highest-regarded universitie ...
. In 1975 tomographic imaging techniques were further developed by Michel Ter-Pogossian
Michel Matthew Ter-Pogossian (April 21, 1925June 19, 1996) was an American medical physicist. He was professor of radiology at the Washington University School of Medicine for over 30 years. A pioneer in nuclear medicine, he is best known for his ...
, Michael E. Phelps, Edward J. Hoffman
Edward Joseph Hoffman (January 1, 1942 – July 1, 2004) helped invent the first human PET scanner, a commonly used whole-body scanning procedure for detecting diseases like cancer. Hoffman, with Michel Ter-Pogossian and Michael E. Phelps, dev ...
and others at Washington University School of Medicine
Washington University School of Medicine (WUSM) is the medical school of Washington University in St. Louis in St. Louis, Missouri. Founded in 1891, the School of Medicine has 1,260 students, 604 of which are pursuing a medical degree with ...
.
Work by Gordon Brownell, Charles Burnham and their associates at the Massachusetts General Hospital beginning in the 1950s contributed significantly to the development of PET technology and included the first demonstration of annihilation radiation for medical imaging. Their innovations, including the use of light pipes and volumetric analysis, have been important in the deployment of PET imaging. In 1961, James Robertson and his associates at Brookhaven National Laboratory built the first single-plane PET scan, nicknamed the "head-shrinker."
One of the factors most responsible for the acceptance of positron imaging was the development of radiopharmaceuticals. In particular, the development of labeled 2-fluorodeoxy-D-glucose (2FDG) by the Brookhaven group under the direction of Al Wolf and Joanna Fowler was a major factor in expanding the scope of PET imaging. The compound was first administered to two normal human volunteers by Abass Alavi
, image =
, image_size =
, birth_place = Tabriz, Iran
, nationality = American
, field = Molecular imaging
, work_institutions = University of PennsylvaniaDepartment of Radiology
, alma_mater ...
in August 1976 at the University of Pennsylvania. Brain images obtained with an ordinary (non-PET) nuclear scanner demonstrated the concentration of FDG in that organ. Later, the substance was used in dedicated positron tomographic scanners, to yield the modern procedure.
The logical extension of positron instrumentation was a design using two 2-dimensional arrays. PC-I was the first instrument using this concept and was designed in 1968, completed in 1969 and reported in 1972. The first applications of PC-I in tomographic mode as distinguished from the computed tomographic mode were reported in 1970. It soon became clear to many of those involved in PET development that a circular or cylindrical array of detectors was the logical next step in PET instrumentation. Although many investigators took this approach, James Robertson and Zang-Hee Cho were the first to propose a ring system that has become the prototype of the current shape of PET.
The PET-CT scanner, attributed to David Townsend and Ronald Nutt, was named by ''Time
Time is the continued sequence of existence and events that occurs in an apparently irreversible succession from the past, through the present, into the future. It is a component quantity of various measurements used to sequence events, ...
'' as the medical invention of the year in 2000.
Cost
As of August 2008,Cancer Care Ontario
Cancer Care Ontario was an agency of the provincial Government of Ontario that was responsible for improving cancer services. It was created by the government of Bob Rae in April 1995, and was formally launched in 1997. The agency was governed u ...
reports that the current average incremental cost to perform a PET scan in the province is Can$1,000–1,200 per scan. This includes the cost of the radiopharmaceutical and a stipend for the physician reading the scan.
In the United States, a PET scan is estimated to be ~$5,000, and most insurance companies don't pay for routine PET scans after cancer treatment due to the fact that these scans are often unnecessary and present potentially more risks than benefits.
In England, the National Health Service
The National Health Service (NHS) is the umbrella term for the publicly funded healthcare systems of the United Kingdom (UK). Since 1948, they have been funded out of general taxation. There are three systems which are referred to using the " ...
reference cost (2015–2016) for an adult outpatient PET scan is £798.
In Australia, as of July 2018, the Medicare Benefits Schedule Fee for whole body FDG PET ranges from A$953 to A$999, depending on the indication for the scan.
Quality control
The overall performance of PET systems can be evaluated by quality control tools such as theJaszczak phantom A Jaszczak phantom (pronounced "JAY-zak") aka Data Spectrum ECT phantom is an imaging phantom used for validating scanner geometry, 3D contrast, uniformity, resolution, attenuation and scatter correction or alignment tasks in nuclear medicine. It is ...
.Prekeges, Jennifer (2012). ''Nuclear Medicine Instrumentation''. Jones & Bartlett Publishers. . p. 189.
See also
*Diffuse optical imaging
Diffuse optical imaging (DOI) is a method of imaging using near-infrared spectroscopy (NIRS) or fluorescence-based methods.
When used to create 3D volumetric models of the imaged material DOI is referred to as diffuse optical tomography, whereas ...
* Hot cell
Shielded nuclear radiation containment chambers are commonly referred to as hot cells. The word "hot" refers to radioactivity.
Hot cells are used in both the nuclear-energy and the nuclear-medicines industries.
They are required to protect in ...
(equipment used to produce the radiopharmaceuticals used in PET)
* Molecular imaging
Molecular imaging is a field of medical imaging that focuses on imaging molecules of medical interest within living patients. This is in contrast to conventional methods for obtaining molecular information from preserved tissue samples, such as h ...
References
External links
*PET-CT atlas Harvard Medical School
National Isotope Development Center
€”U.S. government source of radionuclides including those for PET—production, research, development, distribution, and information {{Authority control 3D nuclear medical imaging American inventions Antimatter Medical physics Neuroimaging Radiation therapy Medicinal radiochemistry Armenian inventions