Rhenium on:
[Wikipedia]
[Google]
[Amazon]
Rhenium is a
 The most common oxide is the volatile yellow Re2O7. The red
The most common oxide is the volatile yellow Re2O7. The red
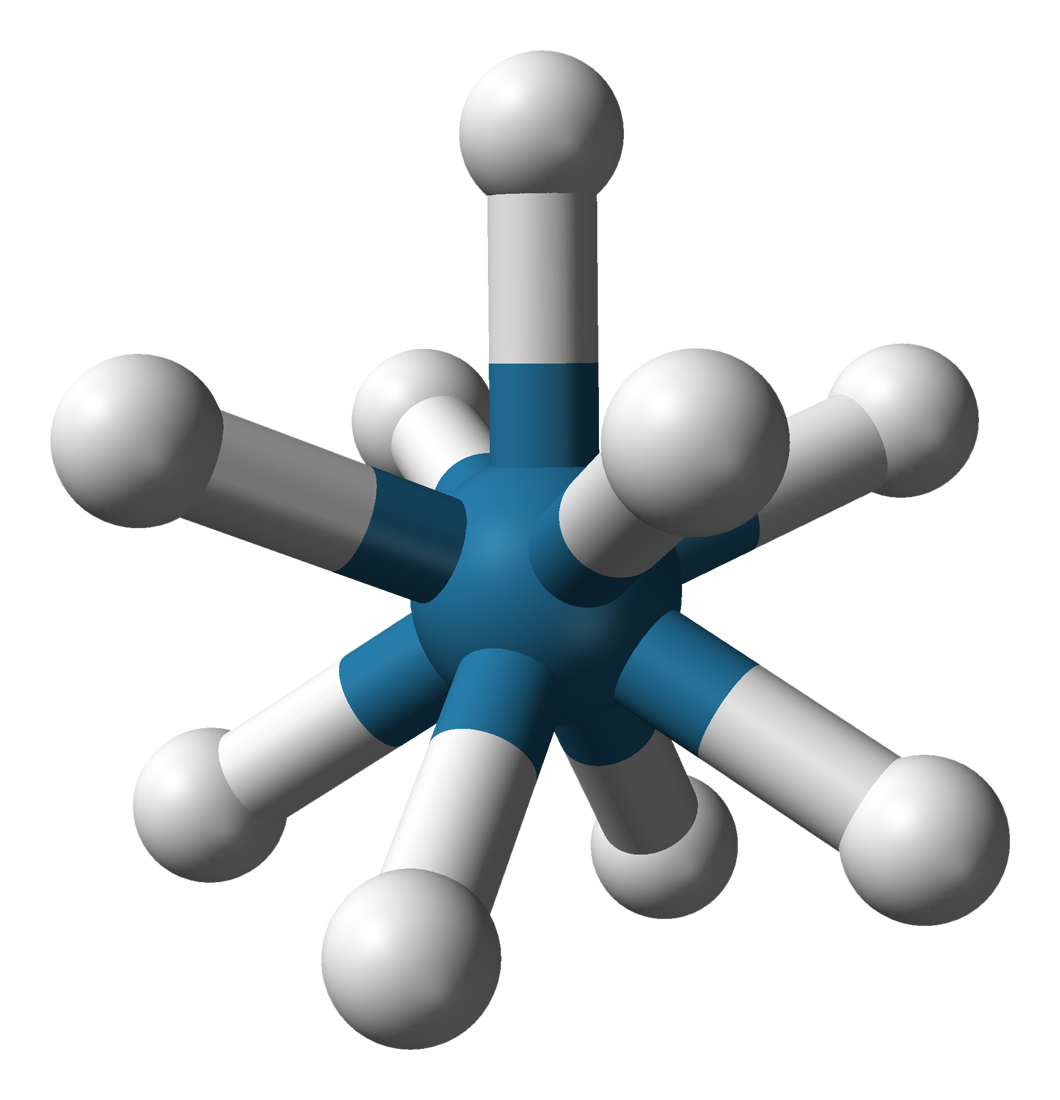 A distinctive derivative of rhenium is nonahydridorhenate, originally thought to be the ''rhenide'' anion, Re−, but actually containing the anion in which the oxidation state of rhenium is +7.
A distinctive derivative of rhenium is nonahydridorhenate, originally thought to be the ''rhenide'' anion, Re−, but actually containing the anion in which the oxidation state of rhenium is +7.
 For 2006, the consumption is given as 28% for
For 2006, the consumption is given as 28% for
Rhenium
at ''
chemical element
A chemical element is a species of atoms that have a given number of protons in their nuclei, including the pure substance consisting only of that species. Unlike chemical compounds, chemical elements cannot be broken down into simpler sub ...
with the symbol
A symbol is a mark, sign, or word that indicates, signifies, or is understood as representing an idea, object, or relationship. Symbols allow people to go beyond what is known or seen by creating linkages between otherwise very different conc ...
Re and atomic number
The atomic number or nuclear charge number (symbol ''Z'') of a chemical element is the charge number of an atomic nucleus. For ordinary nuclei, this is equal to the proton number (''n''p) or the number of protons found in the nucleus of every ...
75. It is a silvery-gray, heavy, third-row transition metal
In chemistry, a transition metal (or transition element) is a chemical element in the d-block of the periodic table (groups 3 to 12), though the elements of group 12 (and less often group 3) are sometimes excluded. They are the elements that can ...
in group 7 Group 7 may refer to:
* G7, an international group of finance minister
*Group 7 element, chemical element classification
*Halogens (alternative name)
*Group 7 Rugby League, rugby league competition in New South Wales, Australia
*Group 7 (racing)
G ...
of the periodic table
The periodic table, also known as the periodic table of the (chemical) elements, is a rows and columns arrangement of the chemical elements. It is widely used in chemistry, physics, and other sciences, and is generally seen as an icon of ch ...
. With an estimated average concentration of 1 part per billion
In science and engineering, the parts-per notation is a set of pseudo-units to describe small values of miscellaneous dimensionless quantities, e.g. mole fraction or mass fraction. Since these fractions are quantity-per-quantity measures, they ...
(ppb), rhenium is one of the rarest elements in the Earth's crust
Earth's crust is Earth's thin outer shell of rock, referring to less than 1% of Earth's radius and volume. It is the top component of the lithosphere, a division of Earth's layers that includes the crust and the upper part of the mantle. The ...
. Rhenium has the third-highest melting point
The melting point (or, rarely, liquefaction point) of a substance is the temperature at which it changes state from solid to liquid. At the melting point the solid and liquid phase exist in equilibrium. The melting point of a substance depends ...
and highest boiling point of any stable element at 5869 K. Rhenium resembles manganese
Manganese is a chemical element with the symbol Mn and atomic number 25. It is a hard, brittle, silvery metal, often found in minerals in combination with iron. Manganese is a transition metal with a multifaceted array of industrial alloy use ...
and technetium
Technetium is a chemical element with the symbol Tc and atomic number 43. It is the lightest element whose isotopes are all radioactive. All available technetium is produced as a synthetic element. Naturally occurring technetium is a spontaneous ...
chemically and is mainly obtained as a by-product
A by-product or byproduct is a secondary product derived from a production process, manufacturing process or chemical reaction; it is not the primary product or service being produced.
A by-product can be useful and marketable or it can be consid ...
of the extraction and refinement of molybdenum
Molybdenum is a chemical element with the symbol Mo and atomic number 42 which is located in period 5 and group 6. The name is from Neo-Latin ''molybdaenum'', which is based on Ancient Greek ', meaning lead, since its ores were confused with lea ...
and copper
Copper is a chemical element with the symbol Cu (from la, cuprum) and atomic number 29. It is a soft, malleable, and ductile metal with very high thermal and electrical conductivity. A freshly exposed surface of pure copper has a pinkis ...
ores. Rhenium shows in its compounds a wide variety of oxidation state
In chemistry, the oxidation state, or oxidation number, is the hypothetical charge of an atom if all of its bonds to different atoms were fully ionic. It describes the degree of oxidation (loss of electrons) of an atom in a chemical compound. C ...
s ranging from −1 to +7.
Discovered by Walter Noddack
Walter Noddack (17 August 1893 – 7 December 1960) was a German chemist. He, Ida Tacke (who later married Noddack), and Otto Berg reported the discovery of element 43 and element 75 in 1925.
Rhenium
They named element 75 rhenium (Latin ''Rh ...
, Ida Tacke
Ida Noddack (25 February 1896 – 24 September 1978), ''née'' Tacke, was a German chemist and physicist. In 1934 she was the first to mention the idea later named nuclear fission. With her husband - Walter Noddack - and Otto Berg (scientist), ...
and Otto Berg in 1925, rhenium was the last stable element to be discovered. It was named after the river Rhine
), Surselva, Graubünden, Switzerland
, source1_coordinates=
, source1_elevation =
, source2 = Rein Posteriur/Hinterrhein
, source2_location = Paradies Glacier, Graubünden, Switzerland
, source2_coordinates=
, so ...
in Europe, from which the earliest samples had been obtained and worked commercially.
Nickel
Nickel is a chemical element with symbol Ni and atomic number 28. It is a silvery-white lustrous metal with a slight golden tinge. Nickel is a hard and ductile transition metal. Pure nickel is chemically reactive but large pieces are slow to ...
-based superalloys of rhenium are used in combustion chambers, turbine blades, and exhaust nozzles of jet engine
A jet engine is a type of reaction engine discharging a fast-moving jet of heated gas (usually air) that generates thrust by jet propulsion. While this broad definition can include rocket, Pump-jet, water jet, and hybrid propulsion, the term ...
s. These alloys contain up to 6% rhenium, making jet engine construction the largest single use for the element. The second-most important use is as a catalyst
Catalysis () is the process of increasing the rate of a chemical reaction by adding a substance known as a catalyst (). Catalysts are not consumed in the reaction and remain unchanged after it. If the reaction is rapid and the catalyst recyc ...
: rhenium is an excellent catalyst for hydrogenation
Hydrogenation is a chemical reaction between molecular hydrogen (H2) and another compound or element, usually in the presence of a Catalysis, catalyst such as nickel, palladium or platinum. The process is commonly employed to redox, reduce or S ...
and isomerization, and is used for example in catalytic reforming
Catalytic reforming is a chemical process used to convert petroleum refinery naphthas distilled from crude oil (typically having low octane ratings) into high-octane liquid products called reformates, which are premium blending stocks for high-oc ...
of naphtha for use in gasoline (rheniforming process). Because of the low availability relative to demand, rhenium is expensive, with price reaching an all-time high in 2008/2009 of US$10,600 per kilogram
The kilogram (also kilogramme) is the unit of mass in the International System of Units (SI), having the unit symbol kg. It is a widely used measure in science, engineering and commerce worldwide, and is often simply called a kilo colloquially ...
(US$4,800 per pound). Due to increases in rhenium recycling and a drop in demand for rhenium in catalysts, the price of rhenium had dropped to US$2,844 per kilogram
The kilogram (also kilogramme) is the unit of mass in the International System of Units (SI), having the unit symbol kg. It is a widely used measure in science, engineering and commerce worldwide, and is often simply called a kilo colloquially ...
(US$1,290 per pound) as of July 2018.
History
Rhenium ( la, Rhenus meaning: "Rhine
), Surselva, Graubünden, Switzerland
, source1_coordinates=
, source1_elevation =
, source2 = Rein Posteriur/Hinterrhein
, source2_location = Paradies Glacier, Graubünden, Switzerland
, source2_coordinates=
, so ...
") was the last-discovered of the elements that have a stable isotope (other new elements discovered in nature since then, such as francium
Francium is a chemical element with the symbol Fr and atomic number 87. It is extremely radioactive; its most stable isotope, francium-223 (originally called actinium K after the natural decay chain it appears in), has a half-life of only 22&nb ...
, are radioactive). The existence of a yet-undiscovered element at this position in the periodic table
The periodic table, also known as the periodic table of the (chemical) elements, is a rows and columns arrangement of the chemical elements. It is widely used in chemistry, physics, and other sciences, and is generally seen as an icon of ch ...
had been first predicted by Dmitri Mendeleev
Dmitri Ivanovich Mendeleev (sometimes transliterated as Mendeleyev or Mendeleef) ( ; russian: links=no, Дмитрий Иванович Менделеев, tr. , ; 8 February Old_Style_and_New_Style_dates">O.S._27_January.html" ;"title="O ...
. Other calculated information was obtained by Henry Moseley
Henry Gwyn Jeffreys Moseley (; 23 November 1887 – 10 August 1915) was an English physicist, whose contribution to the science of physics was the justification from physical laws of the previous empirical and chemical concept of the atomic num ...
in 1914. In 1908, Japan
Japan ( ja, 日本, or , and formally , ''Nihonkoku'') is an island country in East Asia. It is situated in the northwest Pacific Ocean, and is bordered on the west by the Sea of Japan, while extending from the Sea of Okhotsk in the north ...
ese chemist Masataka Ogawa
was a Japanese chemist mainly known for the claimed discovery of element 43 (later known as technetium), which he named nipponium. In fact, he might have discovered, but misidentified, element 75 (later called rhenium).
After graduating from th ...
announced that he had discovered the 43rd element and named it ''nipponium'' (Np) after Japan
Japan ( ja, 日本, or , and formally , ''Nihonkoku'') is an island country in East Asia. It is situated in the northwest Pacific Ocean, and is bordered on the west by the Sea of Japan, while extending from the Sea of Okhotsk in the north ...
(''Nippon'' in Japanese). However, recent analysis indicated the presence of rhenium (element 75), not element 43, although this reinterpretation has been questioned by Eric Scerri
Eric R. Scerri is a chemist, writer and philosopher of science of Maltese origin. He is a lecturer at the University of California, Los Angeles; and the founder and editor-in-chief of '' Foundations of Chemistry'', an international peer reviewed ...
. The symbol Np was later used for the element neptunium
Neptunium is a chemical element with the Symbol (chemistry), symbol Np and atomic number 93. A radioactivity, radioactive actinide metal, neptunium is the first transuranic element. Its position in the periodic table just after uranium, named after ...
, and the name "nihonium", also named after Japan, along with symbol Nh, was later used for element 113. Element 113 was also discovered by a team of Japanese scientists and was named in respectful homage to Ogawa's work.
Rhenium is generally considered to have been discovered by Walter Noddack
Walter Noddack (17 August 1893 – 7 December 1960) was a German chemist. He, Ida Tacke (who later married Noddack), and Otto Berg reported the discovery of element 43 and element 75 in 1925.
Rhenium
They named element 75 rhenium (Latin ''Rh ...
, Ida Noddack
Ida Noddack (25 February 1896 – 24 September 1978), '' née'' Tacke, was a German chemist and physicist. In 1934 she was the first to mention the idea later named nuclear fission. With her husband - Walter Noddack - and Otto Berg she discov ...
, and Otto Berg in Germany
Germany,, officially the Federal Republic of Germany, is a country in Central Europe. It is the second most populous country in Europe after Russia, and the most populous member state of the European Union. Germany is situated betwe ...
. In 1925 they reported that they had detected the element in platinum ore and in the mineral columbite
Columbite, also called niobite, niobite-tantalite and columbate [], is a black mineral group that is an ore of niobium. It has a submetallic Lustre (mineralogy), luster and a high density and is a niobate of iron and manganese. This mineral group ...
. They also found rhenium in gadolinite and molybdenite
Molybdenite is a mineral of molybdenum disulfide, Mo S2. Similar in appearance and feel to graphite, molybdenite has a lubricating effect that is a consequence of its layered structure. The atomic structure consists of a sheet of molybdenum ato ...
. In 1928 they were able to extract 1 g of the element by processing 660 kg of molybdenite. It was estimated in 1968 that 75% of the rhenium metal in the United States
The United States of America (U.S.A. or USA), commonly known as the United States (U.S. or US) or America, is a country primarily located in North America. It consists of 50 states, a federal district, five major unincorporated territorie ...
was used for research and the development of refractory metal
Refractory metals are a class of metals that are extraordinarily resistant to heat and wear. The expression is mostly used in the context of materials science, metallurgy and engineering. The definition of which elements belong to this group diff ...
alloys. It took several years from that point before the superalloys became widely used.
Characteristics
Rhenium is a silvery-white metal with one of the highestmelting point
The melting point (or, rarely, liquefaction point) of a substance is the temperature at which it changes state from solid to liquid. At the melting point the solid and liquid phase exist in equilibrium. The melting point of a substance depends ...
s of all elements, exceeded by only tungsten
Tungsten, or wolfram, is a chemical element with the symbol W and atomic number 74. Tungsten is a rare metal found naturally on Earth almost exclusively as compounds with other elements. It was identified as a new element in 1781 and first isolat ...
and carbon
Carbon () is a chemical element with the symbol C and atomic number 6. It is nonmetallic and tetravalent
In chemistry, the valence (US spelling) or valency (British spelling) of an element is the measure of its combining capacity with o ...
. It also has one of the highest boiling points
The boiling point of a substance is the temperature at which the vapor pressure of a liquid equals the pressure surrounding the liquid and the liquid changes into a vapor.
The boiling point of a liquid varies depending upon the surrounding envir ...
of all elements, and the highest among stable elements. It is also one of the densest, exceeded only by platinum
Platinum is a chemical element with the symbol Pt and atomic number 78. It is a dense, malleable, ductile, highly unreactive, precious, silverish-white transition metal. Its name originates from Spanish , a diminutive of "silver".
Platinu ...
, iridium
Iridium is a chemical element with the symbol Ir and atomic number 77. A very hard, brittle, silvery-white transition metal of the platinum group, it is considered the second-densest naturally occurring metal (after osmium) with a density of ...
and osmium. Rhenium has a hexagonal close-packed crystal structure, with lattice parameters ''a'' = 276.1 pm and ''c'' = 445.6 pm.
Its usual commercial form is a powder, but this element can be consolidated by pressing and sintering
Clinker nodules produced by sintering
Sintering or frittage is the process of compacting and forming a solid mass of material by pressure or heat without melting it to the point of liquefaction.
Sintering happens as part of a manufacturing ...
in a vacuum or hydrogen
Hydrogen is the chemical element with the symbol H and atomic number 1. Hydrogen is the lightest element. At standard conditions hydrogen is a gas of diatomic molecules having the formula . It is colorless, odorless, tasteless, non-toxic, an ...
atmosphere. This procedure yields a compact solid having a density above 90% of the density of the metal. When annealed this metal is very ductile and can be bent, coiled, or rolled. Rhenium-molybdenum alloy
An alloy is a mixture of chemical elements of which at least one is a metal. Unlike chemical compounds with metallic bases, an alloy will retain all the properties of a metal in the resulting material, such as electrical conductivity, ductility, ...
s are superconductive
Superconductivity is a set of physical properties observed in certain materials where electrical resistance vanishes and magnetic flux fields are expelled from the material. Any material exhibiting these properties is a superconductor. Unlike ...
at 10 K; tungsten-rhenium alloys are also superconductive around 4–8 K, depending on the alloy. Rhenium metal superconducts at .
In bulk form and at room temperature and atmospheric pressure, the element resists alkalis, sulfuric acid
Sulfuric acid (American spelling and the preferred IUPAC name) or sulphuric acid ( Commonwealth spelling), known in antiquity as oil of vitriol, is a mineral acid composed of the elements sulfur, oxygen and hydrogen, with the molecular formu ...
, hydrochloric acid
Hydrochloric acid, also known as muriatic acid, is an aqueous solution of hydrogen chloride. It is a colorless solution with a distinctive pungent smell. It is classified as a strong acid
Acid strength is the tendency of an acid, symbol ...
, nitric acid
Nitric acid is the inorganic compound with the formula . It is a highly corrosive mineral acid. The compound is colorless, but older samples tend to be yellow cast due to decomposition into oxides of nitrogen. Most commercially available nitri ...
, and aqua regia. It will however, react with nitric acid upon heating.
Isotopes
Rhenium has onestable
A stable is a building in which livestock, especially horses, are kept. It most commonly means a building that is divided into separate stalls for individual animals and livestock. There are many different types of stables in use today; the ...
isotope, rhenium-185, which nevertheless occurs in minority abundance, a situation found only in two other elements (indium
Indium is a chemical element with the symbol In and atomic number 49. Indium is the softest metal that is not an alkali metal. It is a silvery-white metal that resembles tin in appearance. It is a post-transition metal that makes up 0.21 parts p ...
and tellurium
Tellurium is a chemical element with the symbol Te and atomic number 52. It is a brittle, mildly toxic, rare, silver-white metalloid. Tellurium is chemically related to selenium and sulfur, all three of which are chalcogens. It is occasionally fou ...
). Naturally occurring rhenium is only 37.4% 185Re, and 62.6% 187Re, which is unstable
In numerous fields of study, the component of instability within a system is generally characterized by some of the outputs or internal states growing without bounds. Not all systems that are not stable are unstable; systems can also be mar ...
but has a very long half-life
Half-life (symbol ) is the time required for a quantity (of substance) to reduce to half of its initial value. The term is commonly used in nuclear physics to describe how quickly unstable atoms undergo radioactive decay or how long stable ato ...
(≈1010 years). A kilogram of natural rhenium emits 1.07 MBq of radiation due to the presence of this isotope. This lifetime can be greatly affected by the charge state of the rhenium atom. The beta decay
In nuclear physics, beta decay (β-decay) is a type of radioactive decay in which a beta particle (fast energetic electron or positron) is emitted from an atomic nucleus, transforming the original nuclide to an isobar of that nuclide. For ...
of 187Re is used for rhenium–osmium dating
Rhenium–osmium dating is a form of radiometric dating based on the beta decay of the isotope 187 Re to 187 Os. This normally occurs with a half-life of 41.6 × 109 y, but studies using fully ionised 187 Re atoms have found that this can decrease ...
of ores. The available energy for this beta decay (2.6 keV Kev can refer to:
Given name
* Kev Adams, French comedian, actor, screenwriter and film producer born Kevin Smadja in 1991
* Kevin Kev Carmody (born 1946), Indigenous Australian singer-songwriter
* Kev Coghlan (born 1988), Scottish Grand Prix moto ...
) is one of the lowest known among all radionuclide
A radionuclide (radioactive nuclide, radioisotope or radioactive isotope) is a nuclide that has excess nuclear energy, making it unstable. This excess energy can be used in one of three ways: emitted from the nucleus as gamma radiation; transfer ...
s. The isotope rhenium-186m is notable as being one of the longest lived metastable isotope
A nuclear isomer is a metastable state of an atomic nucleus, in which one or more nucleons (protons or neutrons) occupy higher energy levels than in the ground state of the same nucleus. "Metastable" describes nuclei whose excited states have ha ...
s with a half-life of around 200,000 years. There are 33 other unstable isotopes that have been recognized, ranging from 160Re to 194Re, the longest-lived of which is 183Re with a half-life of 70 days.
Compounds
Rhenium compounds are known for all theoxidation states
In chemistry, the oxidation state, or oxidation number, is the hypothetical charge of an atom if all of its bonds to different atoms were fully ionic. It describes the degree of oxidation (loss of electrons) of an atom in a chemical compound. C ...
between −3 and +7 except −2. The oxidation states +7, +6, +4, and +2 are the most common. Rhenium is most available commercially as salts of perrhenate The perrhenate ion is the anion with the formula , or a compound containing this ion. The perrhenate anion is tetrahedral, being similar in size and shape to perchlorate and the valence isoelectronicity, isoelectronic permanganate. The perrhenate a ...
, including sodium
Sodium is a chemical element with the symbol Na (from Latin ''natrium'') and atomic number 11. It is a soft, silvery-white, highly reactive metal. Sodium is an alkali metal, being in group 1 of the periodic table. Its only stable iso ...
and ammonium perrhenate
Ammonium perrhenate (APR) is the ammonium salt of perrhenic acid, NH4ReO4. It is the most common form in which rhenium is traded. It is a white salt; soluble in ethanol and water, and mildly soluble in NH4Cl. It was first described soon after the ...
s. These are white, water-soluble compounds.Glemser, O. (1963) "Ammonium Perrhenate" in ''Handbook of Preparative Inorganic Chemistry'', 2nd ed., G. Brauer (ed.), Academic Press, NY., Vol. 1, pp. 1476–85. Tetrathioperrhenate anion eS4sup>− is possible.
Halides and oxyhalides
The most common rhenium chlorides are ReCl6, ReCl5, ReCl4, and ReCl3. The structures of these compounds often feature extensive Re-Re bonding, which is characteristic of this metal in oxidation states lower than VII. Salts of e2Cl8sup>2− feature aquadruple
Quadruple may refer to:
* 4-tuple, an ordered list of elements, with four elements
* Quad (figure skating), a figure skating jump
* Quadruple (computing), a term used as alternative for nibble in some contexts
* Quadruple-precision floating-point ...
metal-metal bond. Although the highest rhenium chloride features Re(VI), fluorine gives the d0 Re(VII) derivative rhenium heptafluoride
Rhenium heptafluoride is the compound with the formula ReF7. It is a yellow low melting solid and is the only thermally stable metal heptafluoride. It has a distorted pentagonal bipyramidal structure similar to IF7, which was confirmed by neutron ...
. Bromides and iodides of rhenium are also well known.
Like tungsten and molybdenum, with which it shares chemical similarities, rhenium forms a variety of oxyhalides
In chemistry, molecular oxohalides (oxyhalides) are a group of chemical compounds in which both oxygen and halogen atoms are attached to another chemical element A in a single molecule. They have the general formula , where X = fluorine (F), ch ...
. The oxychlorides are most common, and include ReOCl4, ReOCl3.
Oxides and sulfides
 The most common oxide is the volatile yellow Re2O7. The red
The most common oxide is the volatile yellow Re2O7. The red rhenium trioxide
Rhenium trioxide or rhenium(VI) oxide is an inorganic compound with the formula ReO3. It is a red solid with a metallic lustre that resembles copper in appearance. It is the only stable trioxide of the Group 7 elements ( Mn, Tc, Re).
Prepara ...
ReO3 adopts a perovskite
Perovskite (pronunciation: ) is a calcium titanium oxide mineral composed of calcium titanate (chemical formula ). Its name is also applied to the class of compounds which have the same type of crystal structure as (XIIA2+VIB4+X2−3), known as ...
-like structure. Other oxides include Re2O5, ReO2, and Re2O3. The sulfide
Sulfide (British English also sulphide) is an inorganic anion of sulfur with the chemical formula S2− or a compound containing one or more S2− ions. Solutions of sulfide salts are corrosive. ''Sulfide'' also refers to chemical compounds lar ...
s are ReS2 and Re2S7. Perrhenate salts can be converted to tetrathioperrhenate by the action of ammonium hydrosulfide
Ammonium hydrosulfide is the chemical compound with the formula .
Composition
It is the salt derived from the ammonium cation and the hydrosulfide anion. The salt exists as colourless, water-soluble, micaceous crystals. On Earth the compound ...
.
Other compounds
Rhenium diboride
Rhenium diboride (ReB2) is a synthetic superhard material. It was first synthesized in 1962 and re-emerged recently due to hopes of achieving high hardness comparable to that of diamond. The reported ultrahigh hardness has been questioned, althou ...
(ReB2) is a hard compound having a hardness similar to that of tungsten carbide
Tungsten carbide (chemical formula: WC) is a chemical compound (specifically, a carbide) containing equal parts of tungsten and carbon atoms. In its most basic form, tungsten carbide is a fine gray powder, but it can be pressed and formed into ...
, silicon carbide
Silicon carbide (SiC), also known as carborundum (), is a hard chemical compound containing silicon and carbon. A semiconductor, it occurs in nature as the extremely rare mineral moissanite, but has been mass-produced as a powder and crystal sin ...
, titanium diboride
Titanium diboride (TiB2) is an extremely hard ceramic which has excellent heat conductivity, oxidation stability and wear resistance. TiB2 is also a reasonable electrical conductor,J. Schmidt et al. "Preparation of titanium diboride TiB2 by spark ...
or zirconium diboride
Zirconium diboride (ZrB2) is a highly covalent refractory ceramic material with a hexagonal crystal structure. ZrB2 is an ultra high temperature ceramic (UHTC) with a melting point of 3246 °C. This along with its relatively low density of ...
.
Organorhenium compounds
Dirhenium decacarbonyl is the most common entry to organorhenium chemistry. Its reduction with sodiumamalgam
Amalgam most commonly refers to:
* Amalgam (chemistry), mercury alloy
* Amalgam (dentistry), material of silver tooth fillings
** Bonded amalgam, used in dentistry
Amalgam may also refer to:
* Amalgam Comics, a publisher
* Amalgam Digital
...
gives Na e(CO)5with rhenium in the formal oxidation state −1. Dirhenium decacarbonyl can be oxidised with bromine
Bromine is a chemical element with the symbol Br and atomic number 35. It is the third-lightest element in group 17 of the periodic table (halogens) and is a volatile red-brown liquid at room temperature that evaporates readily to form a simila ...
to bromopentacarbonylrhenium(I):
:Re2(CO)10 + Br2 → 2 Re(CO)5Br
Reduction of this pentacarbonyl with zinc
Zinc is a chemical element with the symbol Zn and atomic number 30. Zinc is a slightly brittle metal at room temperature and has a shiny-greyish appearance when oxidation is removed. It is the first element in group 12 (IIB) of the periodi ...
and acetic acid
Acetic acid , systematically named ethanoic acid , is an acidic, colourless liquid and organic compound with the chemical formula (also written as , , or ). Vinegar is at least 4% acetic acid by volume, making acetic acid the main component ...
gives pentacarbonylhydridorhenium:
:Re(CO)5Br + Zn + HOAc → Re(CO)5H + ZnBr(OAc)
Methylrhenium trioxide
Methylrhenium trioxide, also known as methyltrioxorhenium(VII), is an organometallic compound with the formula CH3ReO3. It is a volatile, colourless solid that has been used as a catalyst in some laboratory experiments. In this compound, rhenium h ...
("MTO"), CH3ReO3 is a volatile, colourless solid has been used as a catalyst
Catalysis () is the process of increasing the rate of a chemical reaction by adding a substance known as a catalyst (). Catalysts are not consumed in the reaction and remain unchanged after it. If the reaction is rapid and the catalyst recyc ...
in some laboratory experiments. It can be prepared by many routes, a typical method is the reaction of Re2O7 and tetramethyltin
Tetramethyltin is an organometallic compound with the formula (CH3)4Sn. This liquid, one of the simplest organotin compounds, is useful for transition-metal mediated conversion of acid chlorides to methyl ketones and aryl halides to aryl methyl ke ...
:
:Re2O7 + (CH3)4Sn → CH3ReO3 + (CH3)3SnOReO3
Analogous alkyl and aryl derivatives are known. MTO catalyses for the oxidations with hydrogen peroxide
Hydrogen peroxide is a chemical compound with the formula . In its pure form, it is a very pale blue liquid that is slightly more viscous than water. It is used as an oxidizer, bleaching agent, and antiseptic, usually as a dilute solution (3%� ...
. Terminal alkyne
\ce
\ce
Acetylene
\ce
\ce
\ce
Propyne
\ce
\ce
\ce
\ce
1-Butyne
In organic chemistry, an alkyne is an unsaturated hydrocarbon containing at least one carbon—carbon triple bond. The simplest acyclic alkynes with only one triple bond and n ...
s yield the corresponding acid or ester, internal alkynes yield diketones, and alkene
In organic chemistry, an alkene is a hydrocarbon containing a carbon–carbon double bond.
Alkene is often used as synonym of olefin, that is, any hydrocarbon containing one or more double bonds.H. Stephen Stoker (2015): General, Organic, an ...
s give epoxides. MTO also catalyses the conversion of aldehyde
In organic chemistry, an aldehyde () is an organic compound containing a functional group with the structure . The functional group itself (without the "R" side chain) can be referred to as an aldehyde but can also be classified as a formyl grou ...
s and diazoalkane
The diazo group is an organic moiety consisting of two linked nitrogen atoms ( azo) at the terminal position. Overall charge neutral organic compounds containing the diazo group bound to a carbon atom are called diazo compounds or diazoalkanes ...
s into an alkene.
Nonahydridorhenate
 A distinctive derivative of rhenium is nonahydridorhenate, originally thought to be the ''rhenide'' anion, Re−, but actually containing the anion in which the oxidation state of rhenium is +7.
A distinctive derivative of rhenium is nonahydridorhenate, originally thought to be the ''rhenide'' anion, Re−, but actually containing the anion in which the oxidation state of rhenium is +7.
Occurrence
Rhenium is one of the rarest elements inEarth's crust
Earth's crust is Earth's thin outer shell of rock, referring to less than 1% of Earth's radius and volume. It is the top component of the lithosphere, a division of Earth's layers that includes the crust and the upper part of the mantle. The ...
with an average concentration of 1 ppb; other sources quote the number of 0.5 ppb making it the 77th most abundant element in Earth's crust. Rhenium is probably not found free in nature (its possible natural occurrence is uncertain), but occurs in amounts up to 0.2% in the mineral molybdenite
Molybdenite is a mineral of molybdenum disulfide, Mo S2. Similar in appearance and feel to graphite, molybdenite has a lubricating effect that is a consequence of its layered structure. The atomic structure consists of a sheet of molybdenum ato ...
(which is primarily molybdenum disulfide), the major commercial source, although single molybdenite samples with up to 1.88% have been found. Chile
Chile, officially the Republic of Chile, is a country in the western part of South America. It is the southernmost country in the world, and the closest to Antarctica, occupying a long and narrow strip of land between the Andes to the east a ...
has the world's largest rhenium reserves, part of the copper ore deposits, and was the leading producer as of 2005. It was only recently that the first rhenium mineral
In geology and mineralogy, a mineral or mineral species is, broadly speaking, a solid chemical compound with a fairly well-defined chemical composition and a specific crystal structure that occurs naturally in pure form.John P. Rafferty, ed. ( ...
was found and described (in 1994), a rhenium sulfide mineral
The sulfide minerals are a class of minerals containing sulfide (S2−) or disulfide (S22−) as the major anion. Some sulfide minerals are economically important as metal ores. The sulfide class also includes the selenides, the tellurides, the ...
(ReS2) condensing from a fumarole
A fumarole (or fumerole) is a vent in the surface of the Earth or other rocky planet from which hot volcanic gases and vapors are emitted, without any accompanying liquids or solids. Fumaroles are characteristic of the late stages of volcani ...
on Kudriavy volcano, Iturup
, other_names = russian: Итуру́п; ja, 択捉島
, location = Sea of Okhotsk
, coordinates =
, archipelago = Kuril Islands
, total_islands =
, major_islands =
, area_km2 = 3139
, length_km = 200
, width_km = 27
, coastline =
, highest_moun ...
island, in the Kuril Islands
The Kuril Islands or Kurile Islands (; rus, Кури́льские острова́, r=Kuril'skiye ostrova, p=kʊˈrʲilʲskʲɪjə ɐstrɐˈva; Japanese: or ) are a volcanic archipelago currently administered as part of Sakhalin Oblast in the ...
. Kudriavy discharges up to 20–60 kg rhenium per year mostly in the form of rhenium disulfide. Named rheniite
Rheniite is a very rare rhenium sulfide mineral with the chemical formula ( ). It forms metallic, silver grey platey crystals in the triclinic - pinacoidal class. It has a specific gravity of 7.5.
It was discovered at the Kudriavy Volcano, Iturup ...
, this rare mineral commands high prices among collectors.
Production
Approximately 80% of rhenium is extracted from porphyry molybdenum deposits. Some ores contain 0.001% to 0.2% rhenium. Roasting the ore volatilizes rhenium oxides.Rhenium(VII) oxide
Rhenium(VII) oxide is the inorganic compound with the formula Re2 O7. This yellowish solid is the anhydride of HOReO3. Perrhenic acid, Re2O7·2H2O, is closely related to Re2O7. Re2O7 is the raw material for all rhenium compounds, being the volat ...
and perrhenic acid
Perrhenic acid is the chemical compound with the formula . It is obtained by evaporating aqueous solutions of . Conventionally, perrhenic acid is considered to have the formula , and a species of this formula forms when rhenium(VII) oxide sublime ...
readily dissolve in water; they are leached from flue dusts and gasses and extracted by precipitating with potassium
Potassium is the chemical element with the symbol K (from Neo-Latin ''kalium'') and atomic number19. Potassium is a silvery-white metal that is soft enough to be cut with a knife with little force. Potassium metal reacts rapidly with atmosphe ...
or ammonium chloride as the perrhenate The perrhenate ion is the anion with the formula , or a compound containing this ion. The perrhenate anion is tetrahedral, being similar in size and shape to perchlorate and the valence isoelectronicity, isoelectronic permanganate. The perrhenate a ...
salts, and purified by recrystallization. Total world production is between 40 and 50 tons/year; the main producers are in Chile, the United States, Peru, and Poland. Recycling of used Pt-Re catalyst and special alloys allow the recovery of another 10 tons per year. Prices for the metal rose rapidly in early 2008, from $1000–$2000 per kg in 2003–2006 to over $10,000 in February 2008. The metal form is prepared by reducing ammonium perrhenate
Ammonium perrhenate (APR) is the ammonium salt of perrhenic acid, NH4ReO4. It is the most common form in which rhenium is traded. It is a white salt; soluble in ethanol and water, and mildly soluble in NH4Cl. It was first described soon after the ...
with hydrogen
Hydrogen is the chemical element with the symbol H and atomic number 1. Hydrogen is the lightest element. At standard conditions hydrogen is a gas of diatomic molecules having the formula . It is colorless, odorless, tasteless, non-toxic, an ...
at high temperatures:
:2 NH4ReO4 + 7 H2 → 2 Re + 8 H2O + 2 NH3
There are technologies for the associated extraction of rhenium from productive solutions of underground leaching of uranium ores.
Applications
Rhenium is added to high-temperature superalloys that are used to makejet engine
A jet engine is a type of reaction engine discharging a fast-moving jet of heated gas (usually air) that generates thrust by jet propulsion. While this broad definition can include rocket, Pump-jet, water jet, and hybrid propulsion, the term ...
parts, using 70% of the worldwide rhenium production. Another major application is in platinum–rhenium catalyst
Catalysis () is the process of increasing the rate of a chemical reaction by adding a substance known as a catalyst (). Catalysts are not consumed in the reaction and remain unchanged after it. If the reaction is rapid and the catalyst recyc ...
s, which are primarily used in making lead
Lead is a chemical element with the symbol Pb (from the Latin ) and atomic number 82. It is a heavy metal that is denser than most common materials. Lead is soft and malleable, and also has a relatively low melting point. When freshly cu ...
-free, high-octane gasoline
Gasoline (; ) or petrol (; ) (see ) is a transparent, petroleum-derived flammable liquid that is used primarily as a fuel in most spark-ignited internal combustion engines (also known as petrol engines). It consists mostly of organic co ...
.
Alloys
The nickel-based superalloys have improved creep strength with the addition of rhenium. The alloys normally contain 3% or 6% of rhenium. Second-generation alloys contain 3%; these alloys were used in the engines for the F-15 and F-16, whereas the newer single-crystal third-generation alloys contain 6% of rhenium; they are used in theF-22
The Lockheed Martin F-22 Raptor is an American single-seat, twin-engine, all-weather stealth tactical fighter aircraft developed for the United States Air Force (USAF). As the result of the USAF's Advanced Tactical Fighter (ATF) program, th ...
and F-35
The Lockheed Martin F-35 Lightning II is an American family of single-seat, single-engine, all-weather stealth multirole combat aircraft that is intended to perform both air superiority and strike missions. It is also able to provide elect ...
engines. Rhenium is also used in the superalloys, such as CMSX-4 (2nd gen) and CMSX-10 (3rd gen) that are used in industrial gas turbine
A gas turbine, also called a combustion turbine, is a type of continuous flow internal combustion engine. The main parts common to all gas turbine engines form the power-producing part (known as the gas generator or core) and are, in the directi ...
engines like the GE 7FA. Rhenium can cause superalloys to become microstructurally unstable, forming undesirable topologically close packed (TCP) phases. In 4th- and 5th-generation superalloys, ruthenium
Ruthenium is a chemical element with the Symbol (chemistry), symbol Ru and atomic number 44. It is a rare transition metal belonging to the platinum group of the periodic table. Like the other metals of the platinum group, ruthenium is inert to ...
is used to avoid this effect. Among others the new superalloys are EPM-102 (with 3% Ru) and TMS-162 (with 6% Ru), as well as TMS-138 and TMS-174.
 For 2006, the consumption is given as 28% for
For 2006, the consumption is given as 28% for General Electric
General Electric Company (GE) is an American multinational conglomerate founded in 1892, and incorporated in New York state and headquartered in Boston. The company operated in sectors including healthcare, aviation, power, renewable energ ...
, 28% Rolls-Royce plc
Rolls-Royce Holdings plc is a British multinational aerospace and defence company incorporated in February 2011. The company owns Rolls-Royce, a business established in 1904 which today designs, manufactures and distributes power systems for ...
and 12% Pratt & Whitney
Pratt & Whitney is an American aerospace manufacturer with global service operations. It is a subsidiary of Raytheon Technologies. Pratt & Whitney's aircraft engines are widely used in both civil aviation (especially airlines) and military aviat ...
, all for superalloys, whereas the use for catalysts only accounts for 14% and the remaining applications use 18%. In 2006, 77% of rhenium consumption in the United States was in alloys. The rising demand for military jet engines and the constant supply made it necessary to develop superalloys with a lower rhenium content. For example, the newer CFM International CFM56
The CFM International CFM56 (U.S. military designation F108) series is a Franco-American family of high-bypass turbofan aircraft engines made by CFM International (CFMI), with a thrust range of . CFMI is a 50–50 joint-owned company of Safran ...
high-pressure turbine (HPT) blades will use Rene N515 with a rhenium content of 1.5% instead of Rene N5 with 3%.
Rhenium improves the properties of tungsten
Tungsten, or wolfram, is a chemical element with the symbol W and atomic number 74. Tungsten is a rare metal found naturally on Earth almost exclusively as compounds with other elements. It was identified as a new element in 1781 and first isolat ...
. Tungsten-rhenium alloys are more ductile at low temperature, allowing them to be more easily machined. The high-temperature stability is also improved. The effect increases with the rhenium concentration, and therefore tungsten alloys are produced with up to 27% of Re, which is the solubility limit. Tungsten-rhenium wire was originally created in efforts to develop a wire that was more ductile after recrystallization. This allows the wire to meet specific performance objectives, including superior vibration resistance, improved ductility, and higher resistivity. One application for the tungsten-rhenium alloys is X-ray
An X-ray, or, much less commonly, X-radiation, is a penetrating form of high-energy electromagnetic radiation. Most X-rays have a wavelength ranging from 10 picometers to 10 nanometers, corresponding to frequencies in the range 30&nb ...
sources. The high melting point of both elements, together with their high atomic mass, makes them stable against the prolonged electron impact. Rhenium tungsten alloys are also applied as thermocouple
A thermocouple, also known as a "thermoelectrical thermometer", is an electrical device consisting of two dissimilar electrical conductors forming an electrical junction. A thermocouple produces a temperature-dependent voltage as a result of the ...
s to measure temperatures up to 2200 ° C.
The high temperature stability, low vapor pressure, good wear resistance
Wear is the damaging, gradual removal or deformation of material at solid surfaces. Causes of wear can be mechanical (e.g., erosion) or chemical (e.g., corrosion). The study of wear and related processes is referred to as tribology.
Wear in m ...
and ability to withstand arc corrosion of rhenium are useful in self-cleaning electrical contacts
An electrical contact is an electrical circuit component found in electrical switches, relays, connectors and circuit breakers. Each contact is a piece of electrically conductive material, typically metal. When a pair of contacts touch, they ca ...
. In particular, the discharge that occurs during electrical switching oxidizes the contacts. However, rhenium oxide Re2O7 is volatile (sublimes at ~360 °C) and therefore is removed during the discharge.
Rhenium has a high melting point and a low vapor pressure similar to tantalum
Tantalum is a chemical element with the symbol Ta and atomic number 73. Previously known as ''tantalium'', it is named after Tantalus, a villain in Greek mythology. Tantalum is a very hard, ductile, lustrous, blue-gray transition metal that is ...
and tungsten. Therefore, rhenium filaments exhibit a higher stability if the filament is operated not in vacuum, but in oxygen-containing atmosphere. Those filaments are widely used in mass spectrometer
Mass spectrometry (MS) is an analytical technique that is used to measure the mass-to-charge ratio of ions. The results are presented as a '' mass spectrum'', a plot of intensity as a function of the mass-to-charge ratio. Mass spectrometry is us ...
s, ion gauge
Pressure measurement is the measurement of an applied force by a fluid (liquid or gas) on a surface. Pressure is typically measured in units of force per unit of surface area. Many techniques have been developed for the measurement of pressur ...
s and photoflash
A flash is a device used in photography that produces a brief burst of light (typically lasting 1/1000 to 1/200 of a second) at a color temperature of about 5500 K to help illuminate a scene. A major purpose of a flash is to illuminate a ...
lamps in photography
Photography is the art, application, and practice of creating durable images by recording light, either electronically by means of an image sensor, or chemically by means of a light-sensitive material such as photographic film. It is employed ...
.
Catalysts
Rhenium in the form of rhenium-platinum alloy is used as catalyst forcatalytic reforming
Catalytic reforming is a chemical process used to convert petroleum refinery naphthas distilled from crude oil (typically having low octane ratings) into high-octane liquid products called reformates, which are premium blending stocks for high-oc ...
, which is a chemical process to convert petroleum refinery naphthas with low octane rating
An octane rating, or octane number, is a standard measure of a fuel's ability to withstand compression in an internal combustion engine without detonating. The higher the octane number, the more compression the fuel can withstand before detonating ...
s into high-octane liquid products. Worldwide, 30% of catalysts used for this process contain rhenium. The olefin metathesis
Olefin metathesis is an organic reaction that entails the redistribution of fragments of alkenes (olefins) by the scission and regeneration of carbon-carbon double bonds. Because of the relative simplicity of olefin metathesis, it often create ...
is the other reaction for which rhenium is used as catalyst. Normally Re2O7 on alumina is used for this process. Rhenium catalysts are very resistant to chemical poisoning from nitrogen, sulfur and phosphorus, and so are used in certain kinds of hydrogenation reactions.
Other uses
The isotopes 188Re and 186Re are radioactive and are used for treatment ofliver cancer
Liver cancer (also known as hepatic cancer, primary hepatic cancer, or primary hepatic malignancy) is cancer that starts in the liver. Liver cancer can be primary (starts in liver) or secondary (meaning cancer which has spread from elsewhere to th ...
. They both have similar penetration depth in tissue (5 mm for 186Re and 11 mm for 188Re), but 186Re has advantage of longer lifetime (90 hours vs. 17 hours).
188Re is also being used experimentally in a novel treatment of pancreatic cancer where it is delivered by means of the bacterium ''Listeria monocytogenes''. The 188Re isotope is also used for the rhenium-SCT (skin cancer
Skin cancers are cancers that arise from the skin. They are due to the development of abnormal cells that have the ability to invade or spread to other parts of the body. There are three main types of skin cancers: basal-cell skin cancer (BCC) ...
therapy). The treatment uses the isotope's properties as a beta emitter
In nuclear physics, beta decay (β-decay) is a type of radioactive decay in which a beta particle (fast energetic electron or positron) is emitted from an atomic nucleus, transforming the original nuclide to an isobar of that nuclide. For exam ...
for brachytherapy
Brachytherapy is a form of radiation therapy where a sealed radiation source is placed inside or next to the area requiring treatment. ''Brachy'' is Greek for short. Brachytherapy is commonly used as an effective treatment for cervical, prost ...
in the treatment of basal cell carcinoma
Basal-cell carcinoma (BCC), also known as basal-cell cancer, is the most common type of skin cancer. It often appears as a painless raised area of skin, which may be shiny with small blood vessels running over it. It may also present as a raise ...
and squamous cell carcinoma of the skin.
Related by periodic trends
Periodic trends are specific patterns that are present in the periodic table that illustrate different aspects of a certain element. They were discovered by the Russian chemist Dmitri Mendeleev in the year 1863. Major periodic trends include atom ...
, rhenium has a similar chemistry to that of technetium
Technetium is a chemical element with the symbol Tc and atomic number 43. It is the lightest element whose isotopes are all radioactive. All available technetium is produced as a synthetic element. Naturally occurring technetium is a spontaneous ...
; work done to label rhenium onto target compounds can often be translated to technetium. This is useful for radiopharmacy, where it is difficult to work with technetium – especially the technetium-99m
Technetium-99m (99mTc) is a metastable nuclear isomer of technetium-99 (itself an isotope of technetium), symbolized as 99mTc, that is used in tens of millions of medical diagnostic procedures annually, making it the most commonly used medical ra ...
isotope used in medicine – due to its expense and short half-life.
Precautions
Very little is known about the toxicity of rhenium and its compounds because they are used in very small amounts. Soluble salts, such as the rhenium halides or perrhenates, could be hazardous due to elements other than rhenium or due to rhenium itself. Only a few compounds of rhenium have been tested for their acute toxicity; two examples are potassium perrhenate and rhenium trichloride, which were injected as a solution into rats. The perrhenate had an LD50 value of 2800 mg/kg after seven days (this is very low toxicity, similar to that of table salt) and the rhenium trichloride showed LD50 of 280 mg/kg.References
External links
Rhenium
at ''
The Periodic Table of Videos
''Periodic Videos'' (also known as ''The Periodic Table of Videos'') is a video project and YouTube channel on chemistry. It consists of a series of videos about chemical elements and the periodic table, with additional videos on other topics i ...
'' (University of Nottingham)
{{Good article
Chemical elements
Transition metals
Noble metals
Refractory metals
Chemical elements predicted by Dmitri Mendeleev
Chemical elements with hexagonal close-packed structure
Native element minerals