Ferredoxin-thioredoxin Reductase on:
[Wikipedia]
[Google]
[Amazon]
Ferredoxin-thioredoxin reductase , systematic name ''ferredoxin:thioredoxin disulfide oxidoreductase,'' is a Fe-4S protein that plays an important role in the
 The variable α subunit has an open
The variable α subunit has an open
ferredoxin
Ferredoxins (from Latin ''ferrum'': iron + redox, often abbreviated "fd") are iron–sulfur proteins that mediate electron transfer in a range of metabolic reactions. The term "ferredoxin" was coined by D.C. Wharton of the DuPont Co. and applied t ...
/ thioredoxin regulatory chain. It catalyzes the following reaction:
::: 2 reduced ferredoxin
Ferredoxins (from Latin ''ferrum'': iron + redox, often abbreviated "fd") are iron–sulfur proteins that mediate electron transfer in a range of metabolic reactions. The term "ferredoxin" was coined by D.C. Wharton of the DuPont Co. and applied t ...
+ thioredoxin disulfide 2 oxidized ferredoxin + thioredoxin thiols + 2 H+
Ferredoxin-Thioredoxin reductase (FTR) converts an electron signal (photoreduced ferredoxin) to a thiol signal (reduced thioredoxin), regulating enzymes by reduction of specific disulfide groups. It catalyses the light-dependent activation of several photosynthesis enzymes
Enzymes () are proteins that act as biological catalysts by accelerating chemical reactions. The molecules upon which enzymes may act are called substrate (chemistry), substrates, and the enzyme converts the substrates into different molecule ...
and constitutes the first historical example of a thiol/disulfide exchange cascade for enzyme regulation. It is a heterodimer
In biochemistry, a protein dimer is a macromolecular complex formed by two protein monomers, or single proteins, which are usually non-covalently bound. Many macromolecules, such as proteins or nucleic acids, form dimers. The word ''dimer'' has ...
of subunit alpha and subunit beta. Subunit alpha is the variable subunit, and beta is the catalytic chain. The structure
A structure is an arrangement and organization of interrelated elements in a material object or system, or the object or system so organized. Material structures include man-made objects such as buildings and machines and natural objects such as ...
of the beta subunit has been determined and found to fold around the FeS
Fez or Fes (; ar, فاس, fās; zgh, ⴼⵉⵣⴰⵣ, fizaz; french: Fès) is a city in northern inland Morocco and the capital of the Fès-Meknès administrative region. It is the second largest city in Morocco, with a population of 1.11 mi ...
cluster.
Biological Function
Major groups of oxygen-producing,photosynthetic
Photosynthesis is a process used by plants and other organisms to convert light energy into chemical energy that, through cellular respiration, can later be released to fuel the organism's activities. Some of this chemical energy is stored in c ...
organisms such as cyanobacteria
Cyanobacteria (), also known as Cyanophyta, are a phylum of gram-negative bacteria that obtain energy via photosynthesis. The name ''cyanobacteria'' refers to their color (), which similarly forms the basis of cyanobacteria's common name, blu ...
, algae
Algae (; singular alga ) is an informal term for a large and diverse group of photosynthetic eukaryotic organisms. It is a polyphyletic grouping that includes species from multiple distinct clades. Included organisms range from unicellular mic ...
, C4, C3, and crassulacean acid metabolism (CAM) plants use Ferredoxin-thioredoxin reductase for carbon fixation
Biological carbon fixation or сarbon assimilation is the process by which inorganic carbon (particularly in the form of carbon dioxide) is converted to organic compounds by living organisms. The compounds are then used to store energy and as ...
regulation. FTR, as part of a greater Ferredoxin-Thioredoxin system, allows plants to change their metabolism based on light intensity. Specifically, the Ferredoxin-Thioredoxin system controls enzymes in the Calvin Cycle
The Calvin cycle, light-independent reactions, bio synthetic phase, dark reactions, or photosynthetic carbon reduction (PCR) cycle of photosynthesis is a series of chemical reactions that convert carbon dioxide and hydrogen-carrier compounds into ...
and Pentose phosphate pathway - allowing plants to balance carbohydrate synthesis and degradation based on the availability of light. In the light, photosynthesis harnesses light energy and reduces Ferredoxin
Ferredoxins (from Latin ''ferrum'': iron + redox, often abbreviated "fd") are iron–sulfur proteins that mediate electron transfer in a range of metabolic reactions. The term "ferredoxin" was coined by D.C. Wharton of the DuPont Co. and applied t ...
. Using FTR, reduced Ferredoxin then reduces Thioredoxin. Thioredoxin, through thiol/disulfide exchange, then activates carbohydrate synthesis enzymes such as chloroplast fructose-1,6-bisphosphatase
The enzyme fructose bisphosphatase (EC 3.1.3.11; systematic name D-fructose-1,6-bisphosphate 1-phosphohydrolase) catalyses the conversion of fructose-1,6-bisphosphate to fructose 6-phosphate in gluconeogenesis and the Calvin cycle, which ar ...
, Sedoheptulose-bisphosphatase
Sedoheptulose-bisphosphatase (also sedoheptulose-1,7-bisphosphatase or SBPase, EC number 3.1.3.37; systematic name sedoheptulose-1,7-bisphosphate 1-phosphohydrolase) is an enzyme that catalyzes the removal of a phosphate group from sedoheptulose 1, ...
, and phosphoribulokinase. As a result, light uses FTR to activate carbohydrate biosynthesis. In the dark, Ferredoxin remains oxidized. This leaves Thioredoxin inactive and allows carbohydrate breakdown to dominate metabolism.
Structure
Ferredoxin-Thioredoxin Reductase is an α-βheterodimer
In biochemistry, a protein dimer is a macromolecular complex formed by two protein monomers, or single proteins, which are usually non-covalently bound. Many macromolecules, such as proteins or nucleic acids, form dimers. The word ''dimer'' has ...
of approximately 30 kDa. FTR structure across different plant species include a conserved catalytic β subunit and a variable α subunit. The structure of FTR from '' Synechocystis'' sp. PCC6803 has been studied in detail and resolved at 1.6 Å. FTR resembles a thin concave disc, 10 Å across the center where a Fe-4S clusterresides. One side of the cluster center contains redox-active disulfide bonds that reduce Thioredoxin while the opposite docks with reduced Ferredoxin. This two sided disc structure allows FTR to simultaneously interact with Thioredoxin and Ferredoxin.
 The variable α subunit has an open
The variable α subunit has an open β barrel
In protein structures, a beta barrel is a beta sheet composed of tandem repeats that twists and coils to form a closed toroidal structure in which the first strand is bonded to the last strand (hydrogen bond). Beta-strands in many beta-barrels are ...
structure made of five antiparallel β strands. Its interaction with the catalytic subunit occurs mainly with two loops between β strands. The residues in these two loops are mostly conserved and are thought to stabilize the 4Fe-4S cluster in the catalytic subunit. Structurally, the α subunit is very similar to the PsaE protein, a subunit of Photosystem I, though the similarity is not seen in their sequences or functions.
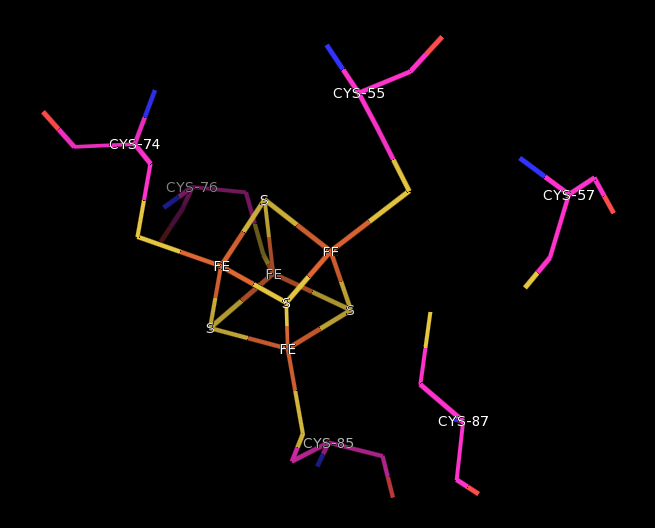
The catalytic β subunit has a general α-helical structure with an Fe-4S center The FeS center and redox-active Cysteine
Cysteine (symbol Cys or C; ) is a semiessential proteinogenic amino acid with the formula . The thiol side chain in cysteine often participates in enzymatic reactions as a nucleophile.
When present as a deprotonated catalytic residue, sometime ...
residues are located within the loops of these helices. Cysteine-55, 74, 76, and 85 are coordinated to the iron atoms of the cubane-type cluster.
Enzymatic Mechanism
FTR is ''unique'' among thioredoxin reductases because it uses an Fe-S cluster cofactor rather than flavoproteins to reduce disulfide bonds. FTR catalysis begins with its interaction with reduced Ferredoxin. This proceeds with the attraction between FTR Lys-47 and Ferredoxin Glu-92. One electron from Ferredoxin and one electron from the Fe-S center is abstracted to break FTR's Cys-87 and Cys-57 disulfide bond, create a nucleophilic Cys-57, and oxidize the Fe-S center from Fe-4Ssup>2+ to Fe-4Ssup>3+. The structure of this one-electron (from Ferredoxin) intermediate is contested: Staples et al. suggest Cys-87 is coordinated to a Sulfur in the Fe-S center while Dai et al. argue Cys-87 is coordinated to an Iron. Next, the nucleophilic Cys-57, encouraged by an adjacent Histidine residue, attacks a disulfide bridge on Thioredoxin, creating a hetero-disulfide Thioredoxin intermediate. Lastly, a newly docked Ferredoxin molecule delivers the final electron to the FeS center, reducing it to its original 2+ state, reforming the Cys-87, Cys-57 disulfide, and fully reducing thioredoxin to two thiols.References
External links
* {{Portal bar, Biology, border=no Protein domains EC 1.8.7