|
Ununennium
Ununennium, also known as eka-francium or element 119, is the hypothetical chemical element with symbol Uue and atomic number 119. ''Ununennium'' and ''Uue'' are the temporary systematic IUPAC name and symbol respectively, which are used until the element is discovered, confirmed, and a permanent name is decided upon. In the periodic table of the elements, it is expected to be an s-block element, an alkali metal, and the first element in the eighth period. It is the lightest element that has not yet been synthesized. An attempt to synthesize the element has been ongoing since 2018 in RIKEN in Japan. The Joint Institute for Nuclear Research in Dubna, Russia, plans to make an attempt at some point in the future, but a precise date has not been released to the public. Theoretical and experimental evidence has shown that the synthesis of ununennium will likely be far more difficult than that of the previous elements, and it may even be the penultimate element that can be synthesized ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Alkali Metal
The alkali metals consist of the chemical elements lithium (Li), sodium (Na), potassium (K),The symbols Na and K for sodium and potassium are derived from their Latin names, ''natrium'' and ''kalium''; these are still the origins of the names for the elements in some languages, such as German and Russian. rubidium (Rb), caesium (Cs), and francium (Fr). Together with hydrogen they constitute Group (periodic table)#Group names, group 1, which lies in the s-block of the periodic table. All alkali metals have their outermost electron in an atomic orbital, s-orbital: this shared electron configuration results in their having very similar characteristic properties. Indeed, the alkali metals provide the best example of periodic trends, group trends in properties in the periodic table, with elements exhibiting well-characterised homology (chemistry), homologous behaviour. This family of elements is also known as the lithium family after its leading element. The alkali metals are all sh ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Unbinilium
Unbinilium, also known as eka-radium or simply element 120, is the hypothetical chemical element in the periodic table with symbol Ubn and atomic number 120. ''Unbinilium'' and ''Ubn'' are the temporary systematic IUPAC name and symbol, which are used until the element is discovered, confirmed, and a permanent name is decided upon. In the periodic table of the elements, it is expected to be an s-block element, an alkaline earth metal, and the second element in the eighth period. It has attracted attention because of some predictions that it may be in the island of stability. Unbinilium has not yet been synthesized, despite multiple attempts from German and Russian teams. Another attempt by the Russian team is planned, but the precise timeframe has not been publicly released. Experimental evidence from these attempts shows that the period 8 elements would likely be far more difficult to synthesise than the previous known elements, and that unbinilium may even be the last element ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Einsteinium
Einsteinium is a synthetic element with the symbol Es and atomic number 99. Einsteinium is a member of the actinide series and it is the seventh transuranium element. It was named in honor of Albert Einstein. Einsteinium was discovered as a component of the debris of the first hydrogen bomb explosion in 1952. Its most common isotope, einsteinium-253 (half-life 20.47 days), is produced artificially from decay of californium-253 in a few dedicated high-power nuclear reactors with a total yield on the order of one milligram per year. The reactor synthesis is followed by a complex process of separating einsteinium-253 from other actinides and products of their decay. Other isotopes are synthesized in various laboratories, but in much smaller amounts, by bombarding heavy actinide elements with light ions. Owing to the small amounts of produced einsteinium and the short half-life of its most easily produced isotope, there are currently almost no practical applications for it outside bas ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Joint Institute For Nuclear Research
The Joint Institute for Nuclear Research (JINR, russian: Объединённый институт ядерных исследований, ОИЯИ), in Dubna, Moscow Oblast (110 km north of Moscow), Russia, is an international research center for nuclear sciences, with 5500 staff members including 1200 researchers holding over 1000 Ph.Ds from eighteen countries. Most scientists, however, are eminent Russian scientists. The institute has seven laboratories, each with its own specialisation: theoretical physics, high energy physics (particle physics), heavy ion physics, condensed matter physics, nuclear reactions, neutron physics, and information technology. The institute has a division to study radiation and radiobiological research and other ad hoc experimental physics experiments. Principal research instruments include a nuclotron superconductive particle accelerator (particle energy: 7 GeV), three isochronous cyclotrons (120, 145, 650 MeV), a phasitron (680 MeV) and a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Periodic Table
The periodic table, also known as the periodic table of the (chemical) elements, is a rows and columns arrangement of the chemical elements. It is widely used in chemistry, physics, and other sciences, and is generally seen as an icon of chemistry. It is a graphic formulation of the periodic law, which states that the properties of the chemical elements exhibit an approximate periodic dependence on their atomic numbers. The table is divided into four roughly rectangular areas called blocks. The rows of the table are called periods, and the columns are called groups. Elements from the same group of the periodic table show similar chemical characteristics. Trends run through the periodic table, with nonmetallic character (keeping their own electrons) increasing from left to right across a period, and from down to up across a group, and metallic character (surrendering electrons to other atoms) increasing in the opposite direction. The underlying reason for these trends is ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Darmstadt
Darmstadt () is a city in the States of Germany, state of Hesse in Germany, located in the southern part of the Frankfurt Rhine Main Area, Rhine-Main-Area (Frankfurt Metropolitan Region). Darmstadt has around 160,000 inhabitants, making it the fourth largest city in the state of Hesse after Frankfurt am Main, Wiesbaden, and Kassel. Darmstadt holds the official title "City of Science" (german: link=no, Wissenschaftsstadt) as it is a major centre of scientific institutions, universities, and high-technology companies. The European Organisation for the Exploitation of Meteorological Satellites (EUMETSAT) and the European Space Operations Centre (ESOC) are located in Darmstadt, as well as Gesellschaft für Schwerionenforschung, GSI Centre for Heavy Ion Research, where several chemical elements such as bohrium (1981), meitnerium (1982), hassium (1984), darmstadtium (1994), roentgenium (1994), and copernicium (1996) were discovered. The existence of the following elements were also ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
GSI Helmholtz Centre For Heavy Ion Research
The GSI Helmholtz Centre for Heavy Ion Research (german: GSI Helmholtzzentrum für Schwerionenforschung) is a federally and state co-funded heavy ion () research center in the Wixhausen suburb of Darmstadt, Germany. It was founded in 1969 as the Society for Heavy Ion Research (german: Gesellschaft für Schwerionenforschung), abbreviated GSI, to conduct research on and with heavy-ion accelerators. It is the only major user research center in the State of Hesse. The laboratory performs basic and applied research in physics and related natural science disciplines. Main fields of study include plasma physics, atomic physics, nuclear structure and reactions research, biophysics and medical research. The lab is a member of the Helmholtz Association of German Research Centres. Shareholders are the German Federal Government (90%) and the State of Hesse, Thuringia and Rhineland-Palatinate. As a member of the Helmholtz Association, the current name was given to the facility on 7 October ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
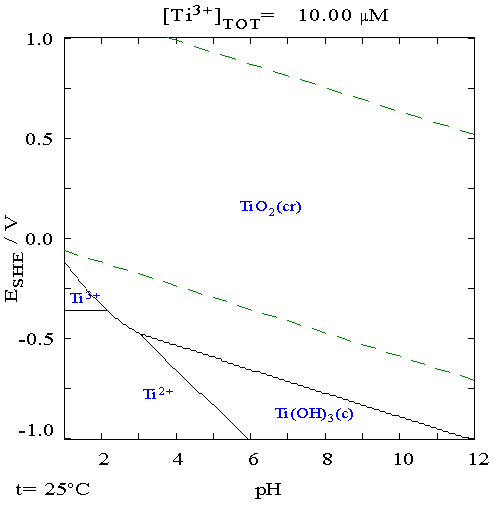
Titanium
Titanium is a chemical element with the symbol Ti and atomic number 22. Found in nature only as an oxide, it can be reduced to produce a lustrous transition metal with a silver color, low density, and high strength, resistant to corrosion in sea water, aqua regia, and chlorine. Titanium was discovered in Cornwall, Great Britain, by William Gregor in 1791 and was named by Martin Heinrich Klaproth after the Titans of Greek mythology. The element occurs within a number of minerals, principally rutile and ilmenite, which are widely distributed in the Earth's crust and lithosphere; it is found in almost all living things, as well as bodies of water, rocks, and soils. The metal is extracted from its principal mineral ores by the Kroll and Hunter processes. The most common compound, titanium dioxide, is a popular photocatalyst and is used in the manufacture of white pigments. Other compounds include titanium tetrachloride (TiCl4), a component of smoke screens and catalysts; and ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Flerovium
Flerovium is a superheavy chemical element with symbol Fl and atomic number 114. It is an extremely radioactive synthetic element. It is named after the Flerov Laboratory of Nuclear Reactions of the Joint Institute for Nuclear Research in Dubna, Russia, where the element was discovered in 1998. The lab's name, in turn, honours Russian physicist Georgy Flyorov ( in Cyrillic, hence the transliteration of " yo" to "e"). IUPAC adopted the name on 30 May 2012. The name and symbol had previously been proposed for element 102 (nobelium), but was not accepted by IUPAC at that time. It is a transactinide in the p-block of the periodic table. It is in period 7; the heaviest known member of the carbon group, and the last element whose chemistry has been investigated. Initial chemical studies in 2007–2008 indicated that flerovium was unexpectedly volatile for a group 14 element; in preliminary results it even seemed to exhibit properties similar to noble gases. More recent results show ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Berkelium
Berkelium is a transuranic radioactive chemical element with the symbol Bk and atomic number 97. It is a member of the actinide and transuranium element series. It is named after the city of Berkeley, California, the location of the Lawrence Berkeley National Laboratory (then the University of California Radiation Laboratory) where it was discovered in December 1949. Berkelium was the fifth transuranium element discovered after neptunium, plutonium, curium and americium. The major isotope of berkelium, 249Bk, is synthesized in minute quantities in dedicated high-flux nuclear reactors, mainly at the Oak Ridge National Laboratory in Tennessee, United States, and at the Research Institute of Atomic Reactors in Dimitrovgrad, Russia. The production of the second-most important isotope, 247Bk, involves the irradiation of the rare isotope 244Cm with high-energy alpha particles. Just over one gram of berkelium has been produced in the United States since 1967. There is no practical appl ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Half-life
Half-life (symbol ) is the time required for a quantity (of substance) to reduce to half of its initial value. The term is commonly used in nuclear physics to describe how quickly unstable atoms undergo radioactive decay or how long stable atoms survive. The term is also used more generally to characterize any type of exponential (or, rarely, non-exponential) decay. For example, the medical sciences refer to the biological half-life of drugs and other chemicals in the human body. The converse of half-life (in exponential growth) is doubling time. The original term, ''half-life period'', dating to Ernest Rutherford's discovery of the principle in 1907, was shortened to ''half-life'' in the early 1950s. Rutherford applied the principle of a radioactive element's half-life in studies of age determination of rocks by measuring the decay period of radium to lead-206. Half-life is constant over the lifetime of an exponentially decaying quantity, and it is a characteristic unit for ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cross Section (physics)
In physics, the cross section is a measure of the probability that a specific process will take place when some kind of radiant excitation (e.g. a particle beam, sound wave, light, or an X-ray) intersects a localized phenomenon (e.g. a particle or density fluctuation). For example, the Rutherford cross-section is a measure of probability that an alpha particle will be deflected by a given angle during an interaction with an atomic nucleus. Cross section is typically denoted ( sigma) and is expressed in units of area, more specifically in barns. In a way, it can be thought of as the size of the object that the excitation must hit in order for the process to occur, but more exactly, it is a parameter of a stochastic process. In classical physics, this probability often converges to a deterministic proportion of excitation energy involved in the process, so that, for example, with light scattering off of a particle, the cross section specifies the amount of optical power scattere ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |