|
Dimethocaine
Dimethocaine, also known as DMC or larocaine, is a compound with a stimulatory effect. This effect resembles that of cocaine, although dimethocaine appears to be less potent. Just like cocaine, dimethocaine is addictive due to its stimulation of the reward pathway in the brain. However, dimethocaine is a legal cocaine replacement in some countries and is even listed by the European Monitoring Centre for Drugs and Drug Addiction (EMCDDA) under the category “synthetic cocaine derivatives”. The structure of dimethocaine, being a 4-aminobenzoic acid ester, resembles that of procaine. It is found as a white powder at room temperature. When a product sold online in the UK in June 2010, advertised as dimethocaine was tested, it was found to be a mixture of caffeine and lidocaine, and the lack of any dopaminergic stimulant ingredient in such mixes may explain the limited recreational effects reported by many users. Other samples tested have however been shown to contain genuine dime ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrochloride
In chemistry, a hydrochloride is an acid salt resulting, or regarded as resulting, from the reaction of hydrochloric acid with an organic base (e.g. an amine). An alternative name is chlorhydrate, which comes from French. An archaic alternative name is muriate, derived from hydrochloric acid's ancient name: muriatic acid. Uses Converting amines into their hydrochlorides is a common way to improve their water solubility, which can be desirable for substances used in medications. The European Pharmacopoeia lists more than 200 hydrochlorides as active ingredients in medications. These hydrochlorides, compared to free bases, may more readily dissolve in the gastrointestinal tract and be absorbed into the bloodstream more quickly. Additionally, many hydrochlorides of amines have a longer shelf-life than their respective free bases. Amine hydrochlorides represent latent forms of a more reactive free base. In this regard, formation of an amine hydrochloride confers protection. This eff ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
In Vivo
Studies that are ''in vivo'' (Latin for "within the living"; often not italicized in English) are those in which the effects of various biological entities are tested on whole, living organisms or cells, usually animals, including humans, and plants, as opposed to a tissue extract or dead organism. This is not to be confused with experiments done ''in vitro'' ("within the glass"), i.e., in a laboratory environment using test tubes, Petri dishes, etc. Examples of investigations ''in vivo'' include: the pathogenesis of disease by comparing the effects of bacterial infection with the effects of purified bacterial toxins; the development of non-antibiotics, antiviral drugs, and new drugs generally; and new surgical procedures. Consequently, animal testing and clinical trials are major elements of ''in vivo'' research. ''In vivo'' testing is often employed over ''in vitro'' because it is better suited for observing the overall effects of an experiment on a living subject. In dr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
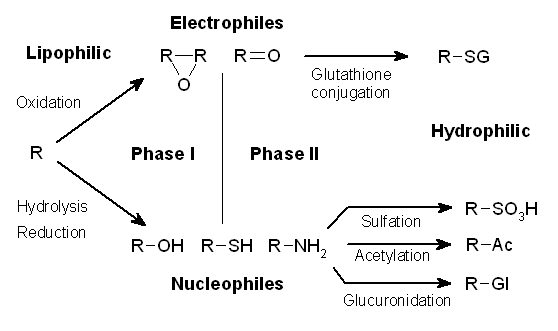
Glucuronidation
Glucuronidation is often involved in drug metabolism of substances such as drugs, pollutants, bilirubin, androgens, estrogens, mineralocorticoids, glucocorticoids, fatty acid derivatives, retinoids, and bile acids. These linkages involve glycosidic bonds. Mechanism Glucuronidation consists of transfer of the glucuronic acid component of uridine diphosphate glucuronic acid to a substrate by any of several types of UDP-glucuronosyltransferase. UDP-glucuronic acid (glucuronic acid linked via a glycosidic bond to uridine diphosphate) is an intermediate in the process and is formed in the liver. One example is the N-glucuronidation of an aromatic amine, 4-aminobiphenyl, by UGT1A4 or UGT1A9 from human, rat, or mouse liver. : The substances resulting from glucuronidation are known as glucuronides (or glucuronosides) and are typically much more water-soluble than the non-glucuronic acid-containing substances from which they were originally synthesised. The human body uses glucuronidat ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Acetylation
: In organic chemistry, acetylation is an organic esterification reaction with acetic acid. It introduces an acetyl group into a chemical compound. Such compounds are termed ''acetate esters'' or simply '' acetates''. Deacetylation is the opposite reaction, the removal of an acetyl group from a chemical compound. Organic synthesis Acetate esters and acetamides are generally prepared by acetylations. Acetylations are often used in making C-acetyl bonds in Friedel-Crafts reactions. Carbanions and their equivalents are susceptible to acetylations. Acetylation reagents Many acetylations are achieved using these three reagents: * Acetic anhydride. This reagent is common in the laboratory; its use cogenerates acetic acid. *Acetyl chloride. This reagent is also common in the laboratory, but its use cogenerates hydrogen chloride, which can be undesirable. *Ketene. At one time acetic anhydride was prepared by the reaction of ketene with acetic acid: :H2C=C=O + CH3COOH -> (CH3CO)2 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydroxylation
In chemistry, hydroxylation can refer to: *(i) most commonly, hydroxylation describes a chemical process that introduces a hydroxyl group () into an organic compound. *(ii) the ''degree of hydroxylation'' refers to the number of OH groups in a molecule. The ''pattern of hydroxylation'' refers to the location of hydroxy groups on a molecule or material. Hydroxylation reactions Synthetic hydroxylations Installing hydroxyl groups into organic compounds can be effected by various metal catalysts. Many such catalysts are biomimetic, i.e. they are inspired by or intended to mimic enzymes such as cytochrome P450. Whereas many hydroxylations insert O atoms into bonds, some reactions ''add'' OH groups to unsaturated substrates. The Sharpless dihydroxylation is such a reaction: it converts alkenes into diols. The hydroxy groups are provided by hydrogen peroxide, which adds across the double bond of alkenes. Biological hydroxylation In biochemistry, hydroxylation reactions are often ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Demethylation
Demethylation is the chemical process resulting in the removal of a methyl group (CH3) from a molecule. A common way of demethylation is the replacement of a methyl group by a hydrogen atom, resulting in a net loss of one carbon and two hydrogen atoms. The counterpart of demethylation is methylation. In biochemistry In biochemical systems, the process of demethylation is catalyzed by demethylases. These enzymes oxidize N-methyl groups, which occur in histones and some forms of DNA: :R2N-CH3 + O → R2N-H + CH2O One such oxidative enzyme family is the cytochrome P450. Alpha-ketoglutarate-dependent hydroxylases are active for demethylation of DNA, operating by a similar pathway. These reactions exploit the weak C-H bond adjacent to amines. In particular, 5-methylcytosines in DNA can be demethylated by TET enzymes as illustrated in the figure. TET enzymes are dioxygenases in the family of alpha-ketoglutarate-dependent hydroxylases. A TET enzyme is an alpha-ketoglut ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ester Hydrolysis
In chemistry, an ester is a compound derived from an oxoacid (organic or inorganic) in which at least one hydroxyl group () is replaced by an alkoxy group (), as in the substitution reaction of a carboxylic acid and an alcohol. Glycerides are fatty acid esters of glycerol; they are important in biology, being one of the main classes of lipids and comprising the bulk of animal fats and vegetable oils. Esters typically have a pleasant smell; those of low molecular weight are commonly used as fragrances and are found in essential oils and pheromones. They perform as high-grade solvents for a broad array of plastics, plasticizers, resins, and lacquers, and are one of the largest classes of synthetic lubricants on the commercial market. Polyesters are important plastics, with monomers linked by ester moieties. Phosphoesters form the backbone of DNA molecules. Nitrate esters, such as nitroglycerin, are known for their explosive properties. '' Nomenclature Etymology The word '' ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Drug Metabolism
Drug metabolism is the metabolic breakdown of drugs by living organisms, usually through specialized enzymatic systems. More generally, xenobiotic metabolism (from the Greek xenos "stranger" and biotic "related to living beings") is the set of metabolic pathways that modify the chemical structure of xenobiotics, which are compounds foreign to an organism's normal biochemistry, such as any drug or poison. These pathways are a form of biotransformation present in all major groups of organisms and are considered to be of ancient origin. These reactions often act to detoxify poisonous compounds (although in some cases the intermediates in xenobiotic metabolism can themselves cause toxic effects). The study of drug metabolism is called pharmacokinetics. The metabolism of pharmaceutical drugs is an important aspect of pharmacology and medicine. For example, the rate of metabolism determines the duration and intensity of a drug's pharmacologic action. Drug metabolism also affects mu ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Laboratory Rat
A laboratory rat or lab rat is a brown rat of the subspecies '' Rattus norvegicus domestica'' which is bred and kept for scientific research. While less commonly used for research than mice (see laboratory mouse), rats have served as an important animal model for research in psychology and biomedical science. Origins In 18th century Europe, wild brown rats ran rampant and this infestation fueled the industry of rat-catching. Rat-catchers would not only make money by trapping the rodents, but also by selling them for food or, more commonly, for rat-baiting. Rat-baiting was a popular sport, which involved filling a pit with rats and timing how long it took for a terrier to kill them all. Over time, breeding the rats for these contests may have produced variations in color, notably the albino and hooded varieties. The first time one of these albino mutants was brought into a laboratory for a study was in 1828 for an experiment on fasting. Over the next 30 years, rats were u ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nociception
Nociception (also nocioception, from Latin ''nocere'' 'to harm or hurt') is the sensory nervous system's process of encoding noxious stimuli. It deals with a series of events and processes required for an organism to receive a painful stimulus, convert it to a molecular signal, and recognize and characterize the signal in order to trigger an appropriate defense response. In nociception, intense chemical (e.g., capsaicin present in Chili pepper or Cayenne pepper), mechanical (e.g., cutting, crushing), or thermal (heat and cold) stimulation of sensory neurons called nociceptors produces a signal that travels along a chain of nerve fibers via the spinal cord to the brain. Nociception triggers a variety of physiological and behavioral responses to protect the organism against an aggression and usually results in a subjective experience, or perception, of pain in sentient beings. Detection of noxious stimuli Potentially damaging mechanical, thermal, and chemical stimuli are detected ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chloroprocaine
Chloroprocaine (trade name Nesacaine, Nesacaine-MPF) (often in the hydrochloride salt form as the aforementioned trade names) is a local anesthetic given by injection during surgical procedures and labor and delivery. Chloroprocaine vasodilates; this is in contrast to cocaine which vasoconstricts. Chloroprocaine is an ester anesthetic. Medical uses Chloroprocaine is used for regional anaesthesia including spinal anaesthesia, caudal anaesthesia and epidural anesthesia It is also indicated for local anaesthesia including brachial plexus block, cervical nerve block, occipital nerve block. mandibular nerve block or maxillary nerve block for dental anesthesia, ophthalmic anesthesia via infraorbital nerve block, ulnar nerve block, paravertebral block, intercostal nerve block, sciatic nerve block, stellate ganglion block, lumbar sympathetic block and interdigital block. It is also used for obstetric anesthesia including pudendal nerve block and paracervical block. Chloroprocaine i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tetracaine
Tetracaine, also known as amethocaine, is an ester local anesthetic used to numb the eyes, nose, or throat. It may also be applied to the skin before starting an intravenous (injection) to decrease pain from the procedure. Typically it is applied as a liquid to the area. Onset of effects when used in the eyes is within 30 seconds and last for less than 15 minutes. Common side effects include a brief period of burning at the site of use. Allergic reactions may uncommonly occur. Long-term use is generally not recommended as it may slow healing of the eye. It is unclear if use during pregnancy is safe for the baby. Tetracaine is in the ester-type local anesthetic family of medications. It works by blocking the sending of nerve impulses. Tetracaine was patented in 1930 and came into medical use in 1941. It is on the World Health Organization's List of Essential Medicines Medical uses A systematic review investigated tetracaine for use in emergency departments, especially for star ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |