|
1,2,4-Trihydroxyanthraquinone
1,2,4-Trihydroxyanthraquinone, commonly called purpurin, is an anthraquinone. It is a naturally occurring red/yellow dye. It is formally derived from 9,10-anthraquinone by replacement of three hydrogen atoms by hydroxyl (OH) groups. Purpurin is also called verantin, smoke Brown G, hydroxylizaric acid, and C.I. 58205. It is a minor component of the classical lake pigment "madder lake" or Rose Madder. History Madder root has been used for dying cloth at least since 1500 BC.Madder Root'' catalog entry at Natural Pigments website. Accessed on 2010-01-22. Purpurin and alizarin were isolated from the root by Pierre Robiquet and Colin, two French chemists, in 1826. They were identified as anthracene derivatives by Gräbe and Liebermann in 1868. They also synthesized alizarin from bromoanthraquinone, which, together with the conversion of alizarin into purpurin published previously by M. F. De Lalande, provided the first synthetic route to purpurin. The positions of t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Alizarin
Alizarin (also known as 1,2-dihydroxyanthraquinone, Mordant Red 11, C.I. 58000, and Turkey Red) is an organic compound with formula that has been used throughout history as a prominent red dye, principally for dyeing textile fabrics. Historically it was derived from the roots of plants of the madder genus.The primary madder species from which alizarin historically has been obtained is ''Rubia tinctorum''. See also In 1869, it became the first natural dye to be produced synthetically. Alizarin is the main ingredient for the manufacture of the madder lake pigments known to painters as rose madder and alizarin crimson. Alizarin in the most common usage of the term has a deep red color, but the term is also part of the name for several related non-red dyes, such as Alizarine Cyanine Green and Alizarine Brilliant Blue. A notable use of alizarin in modern times is as a staining agent in biological research because it stains free calcium and certain calcium compounds a red or ligh ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
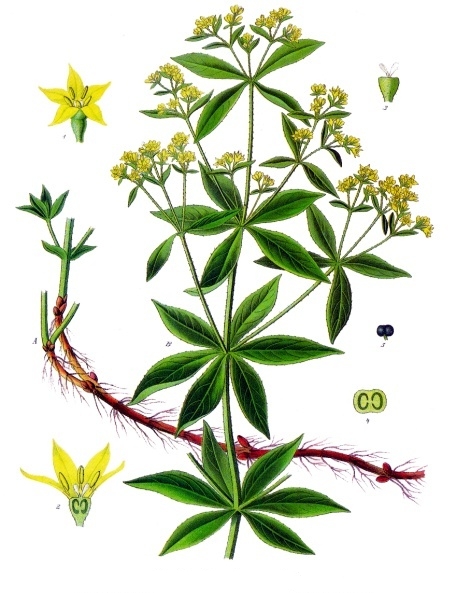
Rubia Tinctorum
''Rubia tinctorum'', the rose madder or common madder or dyer's madder, is a herbaceous perennial plant species belonging to the Galium, bedstraw and Coffea, coffee family Rubiaceae. Description The common madder can grow up to 1.5 m in height. The evergreen leaf, leaves are approximately 5–10 cm long and 2–3 cm broad, produced in whorls of 4–7 starlike around the central stem. It climbs with tiny hooks at the leaves and stems. The flowers are small (3–5 mm across), with five pale yellow petals, in dense racemes, and appear from June to August, followed by small (4–6 mm diameter) red to black berry (botany), berries. The roots can be over a metre long, up to 12 mm thick and the source of red dyes known as rose madder and Turkey red. It prefers loamy soils (sand and clay soil) with a constant level of moisture. Madder is used as a food plant by the larvae of some Lepidoptera species including the Macroglossum stellatarum, hummingbird hawk moth. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pierre Robiquet
Pierre Jean Robiquet (13 January 1780 – 29 April 1840) was a French chemist. He laid founding work in identifying amino acids, the fundamental building blocks of proteins. He did this through recognizing the first of them, asparagine, in 1806, in the industry's adoption of industrial dyes, with the identification of alizarin in 1826, and in the emergence of modern medications, through the identification of codeine in 1832, a opiate alkaloid substance of widespread use with analgesic and antidiarrheal properties. Robiquet was born in Rennes. He was at first a pharmacist in the French armies during the French Revolution years and became a professor at the École de pharmacie in Paris, where he died. Notable scientific achievements were among other things his isolation and characterization of properties of asparagine (the first amino acid to be identified, from asparagus, achieved. In 1806, with Louis Nicolas Vauquelin), cantharidin (1810), the sigma-1 receptor agonist noscapin ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Trihydroxyanthraquinone
A trihydroxyanthraquinone or trihydroxyanthracenedione is any of several isomeric organic compounds with formula , formally derived from anthraquinone by replacing three hydrogen atoms by hydroxyl groups. They include several historically important dyes. Wahl, Andre; Atack, F. W (1919) ''The Manufacture Of Organic Dyestuffs''. G. Bell And Sons, LimitedOnline versionaccessed on 2010-01-22. Hugh Alister McGuigan (1921), ''An introduction to chemical pharmacology; pharmacodynamics in relation to chemistry''. P. Blakiston's son, PhiladelphiaOnline versionat archive.org, accessed on 2010-01-30. The isomers may differ in the parent anthraquinone isomer and/or of the three hydroxyl groups. In general there are 56 ways of choosing three out of the 8 hydrogens. However, if the underlying core is symmetrical, some of these choices will give identical molecules. Isomers From 9,10-anthraquinone Due to the symmetry of the 9,10-anthraquinone core, there are only 14 isomers. CRC (1996) ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Natural Anthraquinone
''For the parent molecule 9,10-anthraquinone, see anthraquinone'' Anthraquinones (also known as anthraquinonoids) are a class of naturally occurring phenolic compounds based on the 9,10-anthraquinone skeleton. They are widely used industrially and occur naturally. Occurrence in plants : Natural pigments that are derivatives of anthraquinone are found, inter alia, in aloe latex, senna (plant), senna, rhubarb, and rhamnus purshiana, cascara buckthorn, fungi, lichens, and some insects. A type II polyketide synthase is responsible for anthraquinone biosynthesis in the bacterium ''Photorhabdus luminescens''. Chorismate, formed by isochorismate synthase in the shikimate pathway, is a precursor of anthraquinones in ''Morinda citrifolia''. Tests for anthraquinones in natural extracts have been established. *Senna glycosides from the Senna (plant), senna. *Frangulin in Frangula alnus. *Aloe-emodin in aloe resin. *Carmine, a bright-red pigment derived from insects. *Hypericin and fagopyrin ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Precipitate
In an aqueous solution, precipitation is the process of transforming a dissolved substance into an insoluble solid from a super-saturated solution. The solid formed is called the precipitate. In case of an inorganic chemical reaction leading to precipitation, the chemical reagent causing the solid to form is called the ''precipitant''. The clear liquid remaining above the precipitated or the centrifuged solid phase is also called the 'supernate' or 'supernatant'. The notion of precipitation can also be extended to other domains of chemistry (organic chemistry and biochemistry) and even be applied to the solid phases (''e.g.'', metallurgy and alloys) when solid impurities segregate from a solid phase. Supersaturation The precipitation of a compound may occur when its concentration exceeds its solubility. This can be due to temperature changes, solvent evaporation, or by mixing solvents. Precipitation occurs more rapidly from a strongly supersaturated solution. The formati ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hexane
Hexane () is an organic compound, a straight-chain alkane with six carbon atoms and has the molecular formula C6H14. It is a colorless liquid, odorless when pure, and with boiling points approximately . It is widely used as a cheap, relatively safe, largely unreactive, and easily evaporated non-polar solvent, and modern gasoline blends contain about 3% hexane. The term ''hexanes'' refers to a mixture, composed largely (>60%) of hexane, with varying amounts of the isomeric compounds 2-methylpentane and 3-methylpentane, and, possibly, smaller amounts of nonisomeric C5, C6, and C7 (cyclo)alkanes. These ''hexanes'' are cheaper than pure hexane and are often used in large-scale operations not requiring a single isomer (e.g., as cleaning solvent or for chromatography). Isomers Uses In industry, hexanes are used in the formulation of adhesive, glues for shoes, leather products, and roofing. They are also used to extract cooking oils (such as canola oil or soy oil) from seeds, for c ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chloroform
Chloroform, or trichloromethane, is an organic compound with chemical formula, formula Carbon, CHydrogen, HChlorine, Cl3 and a common organic solvent. It is a colorless, strong-smelling, dense liquid produced on a large scale as a precursor to PTFE. It is also a precursor to various refrigerants. It is trihalomethane. It is a powerful anesthetic, euphoriant, anxiolytic, and sedative when inhaled or ingested. Structure The molecule adopts a tetrahedral molecular geometry with C3v symmetry group, symmetry. Natural occurrence The total global flux of chloroform through the environment is approximately tonnes per year, and about 90% of emissions are natural in origin. Many kinds of seaweed produce chloroform, and fungi are believed to produce chloroform in soil. Abiotic processes are also believed to contribute to natural chloroform productions in soils although the mechanism is still unclear. Chloroform volatilizes readily from soil and surface water and undergoes degradation in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Aluminum Sulfate
Aluminium sulfate is a salt with the formula Al2 (SO4)3. It is soluble in water and is mainly used as a coagulating agent (promoting particle collision by neutralizing charge) in the purification of drinking water and wastewater treatment plants, and also in paper manufacturing. The anhydrous form occurs naturally as a rare mineral millosevichite, found for example in volcanic environments and on burning coal-mining waste dumps. Aluminium sulfate is rarely, if ever, encountered as the anhydrous salt. It forms a number of different hydrates, of which the hexadecahydrate Al2(SO4)3·16H2O and octadecahydrate Al2(SO4)3·18H2O are the most common. The heptadecahydrate, whose formula can be written as l(H2O)6sub>2(SO4)3·5H2O, occurs naturally as the mineral alunogen. Aluminium sulfate is sometimes called alum or papermaker's alum in certain industries. However, the name "alum" is more commonly and properly used for any double sulfate salt with the generic formula , where ''X'' is a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
1,2,7-trihydroxyanthraquinone
Anthrapurpurin, or 1,2,7-trihydroxyanthraquinone, is a purple dye used in histology for the detection of calcium. See also * * Purpurin * * * |
Purgative
Laxatives, purgatives, or aperients are substances that loosen stools and increase bowel movements. They are used to treat and prevent constipation. Laxatives vary as to how they work and the side effects they may have. Certain stimulant, lubricant and saline laxatives are used to evacuate the colon for rectal and bowel examinations, and may be supplemented by enemas under certain circumstances. Sufficiently high doses of laxatives may cause diarrhea. Some laxatives combine more than one active ingredient. Laxatives may be administered orally or rectally. Types Bulk-forming agents Bulk-forming laxatives, also known as roughage, are substances, such as fiber in food and hydrophilic agents in over-the-counter drugs, that add bulk and water to stools so that they can pass more easily through the intestines (lower part of the digestive tract). Properties * Site of action: small and large intestines * Onset of action: 12–72 hours * Examples: dietary fiber, Metamucil, Citru ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ethanol
Ethanol (abbr. EtOH; also called ethyl alcohol, grain alcohol, drinking alcohol, or simply alcohol) is an organic compound. It is an Alcohol (chemistry), alcohol with the chemical formula . Its formula can be also written as or (an ethyl group linked to a hydroxyl group). Ethanol is a Volatility (chemistry), volatile, Combustibility and flammability, flammable, colorless liquid with a characteristic wine-like odor and pungent taste. It is a psychoactive recreational drug, the active ingredient in alcoholic drinks. Ethanol is naturally produced by the fermentation process of Carbohydrate, sugars by yeasts or via Petrochemistry, petrochemical processes such as ethylene hydration. It has medical applications as an antiseptic and disinfectant. It is used as a chemical solvent and in the Chemical synthesis, synthesis of organic compounds, and as a Alcohol fuel, fuel source. Ethanol also can be dehydrated to make ethylene, an important chemical feedstock. As of 2006, world produ ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |