|
Thiocarbonyl Compounds
In organic chemistry, thioketones (; also known as thiones or thiocarbonyls) are organosulfur compounds related to conventional ketones in which the oxygen has been replaced by a sulfur. Instead of a structure of , thioketones have the structure , which is reflected by the prefix "thio-" in the name of the functional group. Thus the simplest thioketone is thioacetone, the sulfur analog of acetone. Unhindered alkylthioketones typically tend to form polymers or rings. Structure and bonding The C=S bond length of thiobenzophenone is 1.63 Å, which is comparable to 1.64 Å, the C=S bond length of thioformaldehyde, measured in the gas phase. Due to steric interactions, the phenyl groups are not coplanar and the dihedral angle SC-CC is 36°. Unhindered dialkylthiones polymerize or oligomerize but thiocamphor is well characterized red solid. Consistent with the double bond rule, most alkyl thioketones are unstable with respect to dimerization.Organosulfur Chemistry I: Topics in Curre ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Lawesson's Reagent
Lawesson's reagent (LR) is a chemical compound used in organic synthesis as a thiation agent. Lawesson's reagent was first made popular by Sven-Olov Lawesson, who did not, however, invent it. Lawesson's reagent was first made in 1956 during a systematic study of the reactions of arenes with P4S10. Preparation Lawesson's reagent is commercially available. It can also be conveniently prepared in the laboratory by heating a mixture of anisole with phosphorus pentasulfide until the mixture is clear and no more hydrogen sulfide is formed, then recrystallized from toluene or xylene. Samples give a strong odor of hydrogen sulfide owing to partial hydrolysis. One common and effective method of destroying the foul smelling residues is to use an excess of sodium hypochlorite ( chlorine bleach). Mechanism of action Lawesson's reagent has a four membered ring of alternating sulfur and phosphorus atoms. The central phosphorus/sulfur four-membered ring dissociates to form two reactive di ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dithiirane
In organic chemistry, an episulfide is an organic compound that contain a saturated, heterocyclic ring consisting of two carbon atoms and one sulfur atom. It is the sulfur analogue of an epoxide or aziridine. They are also known as thiiranes, olefin sulfides, thioalkylene oxides, and thiacyclopropanes. Episulfides are less common and generally less stable than epoxides. The most common derivative is ethylene sulfide (). Structure According to electron diffraction, the and distances in ethylene sulfide are respectively 1.473 and 1.811 Å. The and angles are respectively 66.0 and 48.0°. Preparation History A number of chemists in the early 1900s, including Staudinger and Pfenninger (1916), as well as Delepine (1920) studied episulfides.Sander, M. Thiiranes. Chem. Rev. 1966, 66(3), 297-339. In 1934 Dachlauer and Jackel devised a general synthesis of episulfides from epoxides using alkali thiocyanates and thiourea. Contemporary methods Following the lead of Dachlaue ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
1,3-dipole
In organic chemistry, a 1,3-dipolar compound or 1,3-dipole is a dipolar compound with delocalized electrons and a separation of charge over three atoms. They are reactants in 1,3-dipolar cycloadditions. The dipole has at least one resonance structure with positive and negative charges having a 1,3 relationship which can generally be denoted as , where a may be a carbon, oxygen or nitrogen, b may be nitrogen or oxygen, and c may be a carbon, oxygen or nitrogen. Known 1,3-dipoles are: * Azides () * Ozone () * Nitro compounds () * Diazo compounds () * Some oxides ** Azoxide compounds (RN(O)NR) ** Carbonyl oxides ( Criegee zwitterions)Li, Jie Jack''Criegee mechanism of ozonolysis''Book: Name Reactions. 2006, 173-174, ** Nitrile oxides () ** Nitrous oxide () ** Nitrones () * Some imines: ** Azomethine imine ** Nitrilimines (, analogous to nitrile oxide) ** Carbonyl imines * Some ylide An ylide () or ylid () is a neutral dipolar molecule containing a formally negatively c ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
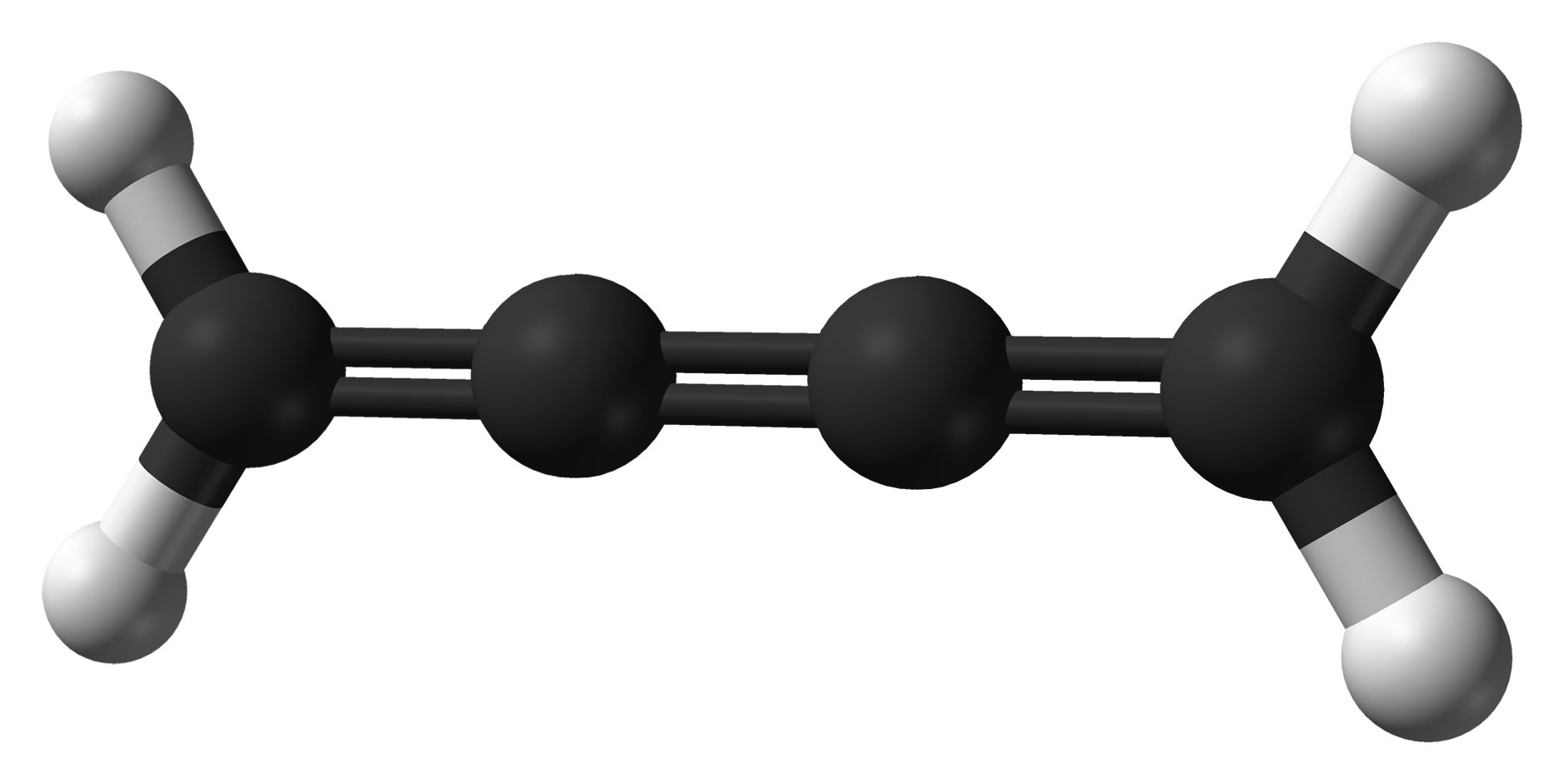
Cumulene
A cumulene is a compound having three or more ''cumulative'' (consecutive) double bonds. They are analogous to allenes, only having a more extensive chain. The simplest molecule in this class is butatriene (), which is also called simply ''cumulene''. Unlike most alkanes and alkenes, cumulenes tend to be rigid, comparable to polyynes. Cumulene carbenes for ''n'' from 3 to 6 have been observed in interstellar molecular cloud A molecular cloud—sometimes called a stellar nursery if star formation is occurring within—is a type of interstellar cloud of which the density and size permit absorption nebulae, the formation of molecules (most commonly molecular hydrogen, ...s and in laboratory experiments by using microwave and infrared spectroscopy. (The more stable cumulenes are difficult to detect optically because they lack an electric dipole moment.) Cumulenes containing heteroatoms are called heterocumulenes; an example is carbon suboxide. Synthesis The first reporte ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Benzophenone
Benzophenone is a naturally occurring organic compound with the formula (C6H5)2CO, generally abbreviated Ph2CO. Benzophenone has been found in some fungi, fruits and plants, including grapes. It is a white solid with a low melting point and rose-like odor that is soluble in organic solvents. Benzophenone is the simplest diaromatic ketone. It is a widely used building block in organic chemistry, being the parent diarylketone. History Carl Graebe of the University of Königsberg, in an early literature report from 1874, described working with benzophenone. Uses Benzophenone can be used as a photo initiator in ultraviolet (UV)-curing applications such as inks, imaging, and clear coatings in the printing industry. Benzophenone prevents UV light from damaging scents and colors in products such as perfumes and soaps. Benzophenone can also be added to plastic packaging as a UV blocker to prevent photo-degradation of the packaging polymers or its contents. Its use allows manufactur ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Blue
Blue is one of the three primary colours in the RYB color model, RYB colour model (traditional colour theory), as well as in the RGB color model, RGB (additive) colour model. It lies between Violet (color), violet and cyan on the optical spectrum, spectrum of visible light. The term ''blue'' generally describes colours perceived by humans observing light with a dominant wavelength that's between approximately 450 and 495 nanometres. Most blues contain a slight mixture of other colours; Azure (color), azure contains some green, while ultramarine contains some violet. The clear daytime sky and the deep sea appear blue because of an optical effect known as Rayleigh scattering#Cause of the blue colour of the sky, Rayleigh scattering. An optical effect called the Tyndall effect explains Eye color#Blue, blue eyes. Distant objects appear more blue because of another optical effect called aerial perspective. Blue has been an important colour in art and decoration since ancient t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Thiobenzophenone
Thiobenzophenone is an organosulfur compound with the formula (C6H5)2CS. It is the prototypical thioketone. Unlike other thioketones that tend to dimerize to form rings and polymers, thiobenzophenone is quite stable, although it photoxidizes in air back to benzophenone and sulfur. Thiobenzophenone is deep blue and dissolves readily in many organic solvents. Structure The C=S bond length of thiobenzophenone is 1.63 Å, which is comparable to 1.64 Å, the C=S bond length of thioformaldehyde, measured in the gas phase. Due to steric interactions, the phenyl groups are not coplanar and the dihedral angle SC-CC is 36°.Sustmann, R.; Sicking, W.; Huisgen, R. "A Computational Study of the Cycloaddition of Thiobenzophenone S-Methylide to Thiobenzophenone". J. Am. Chem. Soc. 2003, 125, 14425-14434. A variety of thiones with structures and stability related to thiobenzophenone have also been prepared. Synthesis One of the first reported syntheses of thiobenzophenone involves the reaction ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carbon
Carbon () is a chemical element; it has chemical symbol, symbol C and atomic number 6. It is nonmetallic and tetravalence, tetravalent—meaning that its atoms are able to form up to four covalent bonds due to its valence shell exhibiting 4 electrons. It belongs to group 14 of the periodic table. Carbon makes up about 0.025 percent of Earth's crust. Three Isotopes of carbon, isotopes occur naturally, carbon-12, C and carbon-13, C being stable, while carbon-14, C is a radionuclide, decaying with a half-life of 5,700 years. Carbon is one of the timeline of chemical element discoveries#Pre-modern and early modern discoveries, few elements known since antiquity. Carbon is the 15th abundance of elements in Earth's crust, most abundant element in the Earth's crust, and the abundance of the chemical elements, fourth most abundant element in the universe by mass after hydrogen, helium, and oxygen. Carbon's abundance, its unique diversity of organic compounds, and its unusual abi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electronegativity
Electronegativity, symbolized as , is the tendency for an atom of a given chemical element to attract shared electrons (or electron density) when forming a chemical bond. An atom's electronegativity is affected by both its atomic number and the distance at which its valence electrons reside from the charged nucleus. The higher the associated electronegativity, the more an atom or a substituent group attracts electrons. Electronegativity serves as a simple way to quantitatively estimate the bond energy, and the sign and magnitude of a bond's chemical polarity, which characterizes a bond along the continuous scale from covalent to ionic bonding. The loosely defined term electropositivity is the opposite of electronegativity: it characterizes an element's tendency to donate valence electrons. On the most basic level, electronegativity is determined by factors like the nuclear charge (the more protons an atom has, the more "pull" it will have on electrons) and the number and lo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Methylene Group
A methylene group is any part of a molecule that consists of two hydrogen atoms bound to a carbon Carbon () is a chemical element; it has chemical symbol, symbol C and atomic number 6. It is nonmetallic and tetravalence, tetravalent—meaning that its atoms are able to form up to four covalent bonds due to its valence shell exhibiting 4 ... atom, which is connected to the remainder of the molecule by two single bonds. The group may be represented as or , where the '>' denotes the two bonds. This stands in contrast to a situation where the carbon atom is bound to the rest of the molecule by a double bond, which is preferably called a methylidene group, represented . Formerly the methylene name was used for both isomers. The name “ methylene bridge“ can be used for the single-bonded isomer, to emphatically exclude methylidene. The distinction is often important, because the double bond is chemically different from two single bonds. The methylene group should be d ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |