|
1,3-dipole
In organic chemistry, a 1,3-dipolar compound or 1,3-dipole is a dipolar compound with delocalized electrons and a separation of charge over three atoms. They are reactants in 1,3-dipolar cycloadditions. The dipole has at least one resonance structure with positive and negative charges having a 1,3 relationship which can generally be denoted as , where a may be a carbon, oxygen or nitrogen, b may be nitrogen or oxygen, and c may be a carbon, oxygen or nitrogen. Known 1,3-dipoles are: * Azides () * Ozone () * Nitro compounds () * Diazo compounds () * Some oxides ** Azoxide compounds (RN(O)NR) ** Carbonyl oxides (Criegee zwitterions)Li, Jie Jack''Criegee mechanism of ozonolysis''Book: Name Reactions. 2006, 173-174, ** Nitrile oxides () ** Nitrous oxide () ** Nitrones () * Some imines: ** Azomethine imine ** Nitrilimines (, analogous to nitrile oxide) ** Carbonyl imines * Some ylides ** Azomethine ylide ** Nitrile ylide () ** Carbonyl ylide In organic chemistry, a carbo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Azomethine Ylide
Azomethine ylides are nitrogen-based 1,3-dipoles, consisting of an iminium ion next to a carbanion. They are used in 1,3-dipolar cycloaddition reactions to form five-membered heterocycles, including pyrrolidines and pyrrolines. These reactions are highly stereo- and regioselective, and have the potential to form four new contiguous stereocenters. Azomethine ylides thus have high utility in total synthesis, and formation of chiral ligands and pharmaceuticals. Azomethine ylides can be generated from many sources, including aziridines, imines, and iminiums. They are often generated ''in situ'', and immediately reacted with dipolarophiles. Structure The resonance structures below show the 1,3-dipole contribution, in which the two carbon atoms adjacent to the nitrogen have a negative or positive charge. The most common representation of azomethine ylides is that in which the nitrogen is positively charged, and the negative charge is shared between the two carbon atoms. The r ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dipolar Compound
In organic chemistry, a dipolar compound or simply dipole is an electrically neutral molecule carrying a positive and a negative charge in at least one canonical description. In most dipolar compounds the charges are delocalized. Unlike salts, dipolar compounds have charges on separate atoms, not on positive and negative ions that make up the compound. Dipolar compounds exhibit a dipole moment. Dipolar compounds can be represented by a resonance structure. Contributing structures containing charged atoms are denoted as zwitterions. {{cite journal, last1=MacHiguchi, first1=Takahisa, last2=Okamoto, first2=Junko, last3=Takachi, first3=Junpei, last4=Hasegawa, first4=Toshio, last5=Yamabe, first5=Shinichi, last6=Minato, first6=Tsutomu, title=Exclusive Formation of α-Methyleneoxetanes in Ketene−Alkene Cycloadditions. Evidence for Intervention of Both an α-Methyleneoxetane and the Subsequent 1,4-Zwitterion, journal=Journal of the American Chemical Society, volume=125, issue=4 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nitrone
In organic chemistry, a nitrone is a functional group consisting of an ''N''-oxide of an imine. The general structure is , where R’ is not a hydrogen. A nitrone is a 1,3-dipole, and is used in 1,3-dipolar cycloadditions. Other reactions of nitrones are known, including formal +3cycloadditions to form 6-membered rings, as well as formal +2cycloadditions to form 7-membered rings. Generation of nitrones Nitrones are generated most often either by the oxidation of hydroxylamines or condensation of monosubstituted hydroxylamines with carbonyl compounds (ketones or aldehydes). The most general reagent used for the oxidation of hydroxylamines is mercury(II) oxide. Carbonyl condensation methods avoid issues of site selectivity associated with the oxidation of hydroxylamines with two sets of (alpha) hydrogens. A significant problem associated with many reactive nitrones is dimerization. This issue is alleviated experimentally by employing an excess of the nitrone or increasing ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
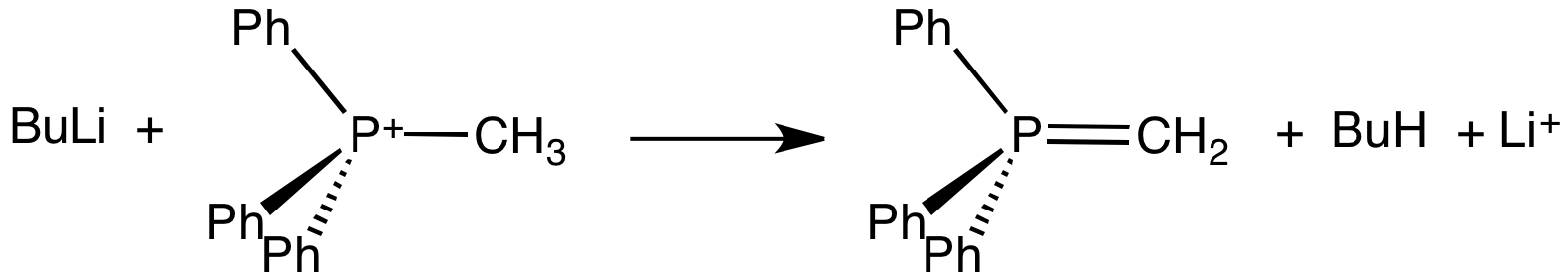
Ylide
An ylide or ylid () is a neutral dipolar molecule containing a formally negatively charged atom (usually a carbanion) directly attached to a heteroatom with a formal positive charge (usually nitrogen, phosphorus or sulfur), and in which both atoms have full octets of electrons. The result can be viewed as a structure in which two adjacent atoms are connected by both a covalent and an ionic bond; normally written X+–Y−. Ylides are thus 1,2- dipolar compounds, and a subclass of zwitterions. They appear in organic chemistry as reagents or reactive intermediates. The class name "ylide" for the compound should not be confused with the suffix "-ylide". Resonance structures Many ylides may be depicted by a multiple bond form in a resonance structure, known as the ylene form, while the actual structure lies in between both forms: : The actual bonding picture of these types of ylides is strictly zwitterionic (the structure on the right) with the strong Coulombic attraction betw ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Diazo
The diazo group is an organic moiety consisting of two linked nitrogen atoms ( azo) at the terminal position. Overall charge neutral organic compounds containing the diazo group bound to a carbon atom are called diazo compounds or diazoalkanes and are described by the general structural formula R2C=N+=N–. The simplest example of a diazo compound is diazomethane, CH2N2. Diazo compounds (R2C=N2) should not be confused with azo compounds of the type R-N=N-R or with diazonium compounds of the type R-N2+. Structure The electronic structure of diazo compounds is characterized by π electron density delocalized over the α-carbon and two nitrogen atoms, along with an orthogonal π system with electron density delocalized over only the terminal nitrogen atoms. Because all octet rule-satisfying resonance forms of diazo compounds have formal charges, they are members of a class of compounds known as 1,3-dipoles. Some of the most stable diazo compounds are α-diazo-β-diketones ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nitrile Ylide
Nitrile ylides also known as ''nitrilium ylides'', or ''nitrilium methylides'' are generally reactive intermediates. With a few exceptions, they cannot be isolated. However, a structure has been determined on particularly stable nitrile ylideby X-ray crystallography. Another nitrile ylide has been captured under cryogenic conditions. As ylides, they possess a negative charge and a positive charge on adjacent atoms. As a 1,3-dipole, several resonance structures can be drawn (1a–d). The most appropriate resonance structure is dependent upon the substituent pattern (the identity of the R and R′ groups in 1 and 2). The 3-dimensional structure of the nitrilium ylide itself may also provide a clue as to the most appropriate resonance structure, with a linear R–C≡N–C unit supportive of the charge distribution indicated for resonance structures 1a & 1b and also consistent with the nomenclature ''nitrilium ylide''. As resonance structures 1c and 1d become more important the nitri ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ozone
Ozone (), or trioxygen, is an inorganic molecule with the chemical formula . It is a pale blue gas with a distinctively pungent smell. It is an allotrope of oxygen that is much less stable than the diatomic allotrope , breaking down in the lower atmosphere to ( dioxygen). Ozone is formed from dioxygen by the action of ultraviolet (UV) light and electrical discharges within the Earth's atmosphere. It is present in very low concentrations throughout the latter, with its highest concentration high in the ozone layer of the stratosphere, which absorbs most of the Sun's ultraviolet (UV) radiation. Ozone's odour is reminiscent of chlorine, and detectable by many people at concentrations of as little as in air. Ozone's O3 structure was determined in 1865. The molecule was later proven to have a bent structure and to be weakly diamagnetic. In standard conditions, ozone is a pale blue gas that condenses at cryogenic temperatures to a dark blue liquid and finally a violet-black ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nitrilimine
Nitrilimines or nitrile amides are a class of organic compounds sharing a common functional group with the general structure R-CN-NR corresponding to the conjugate base of an amine bonded to the N-terminus of a nitrile. The dominant structure for the parent compound nitrilimine is that of the propargyl-like in ''scheme 1'' with a C-N triple bond and with a formal positive charge on nitrogen and two lone pairs and a formal negative charge on the terminal nitrogen. Other structures such as hypervalent , allene-like , allylic and carbene are of lesser relevance. Nitrilimines were first observed in the thermal decomposition of 2-tetrazoles releasing nitrogen:Communications: ''The Formation of Nitrile Imines in the Thermal Breakdown of 2,5-Disubstituted Tetrazoles Rolf Huisgen'' Michael Seidel, Juergen Sauer, James McFarland, Guenter WallbillichJ. Org. Chem.; 1959; 24(6); 892-89Abstract/ref> Nitrilimines are linear 1,3-dipoles represented by structures and . A major use is in hete ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nitrile Oxide
In organic chemistry, a nitrile is any organic compound that has a functional group. The prefix ''cyano-'' is used interchangeably with the term ''nitrile'' in industrial literature. Nitriles are found in many useful compounds, including methyl cyanoacrylate, used in super glue, and nitrile rubber, a nitrile-containing polymer used in latex-free laboratory and medical gloves. Nitrile rubber is also widely used as automotive and other seals since it is resistant to fuels and oils. Organic compounds containing multiple nitrile groups are known as cyanocarbons. Inorganic compounds containing the group are not called nitriles, but cyanides instead. Though both nitriles and cyanides can be derived from cyanide salts, most nitriles are not nearly as toxic. Structure and basic properties The N−C−C geometry is linear in nitriles, reflecting the sp hybridization of the triply bonded carbon. The C−N distance is short at 1.16 Å, consistent with a triple bond. Nitriles ar ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Criegee Zwitterion
A Criegee intermediate (also called a Criegee zwitterion or Criegee biradical) is a carbonyl oxide with two charge centres. These chemicals may react with sulfur dioxide and nitrogen oxides in the earth's atmosphere, and are implicated in the formation of aerosols, which are an important factor in controlling global climate. Criegee intermediates are also an important source of OH (hydroxyl radicals). OH radicals are the most important oxidant in the troposphere, and are important in controlling air quality and pollution. The formation of this sort of structure was first postulated in the 1950s by Rudolf Criegee, for whom it is named. It was not until 2012 that direct detection of such chemicals was reported. Infrared spectroscopy suggests the electronic structure has a substantially zwitterionic character rather than the biradical character that had previously been proposed. Formation Criegee intermediates are formed by the gas-phase reactions of alkenes and ozone in th ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |