Rodent on:
[Wikipedia]
[Google]
[Amazon]
Rodents (from
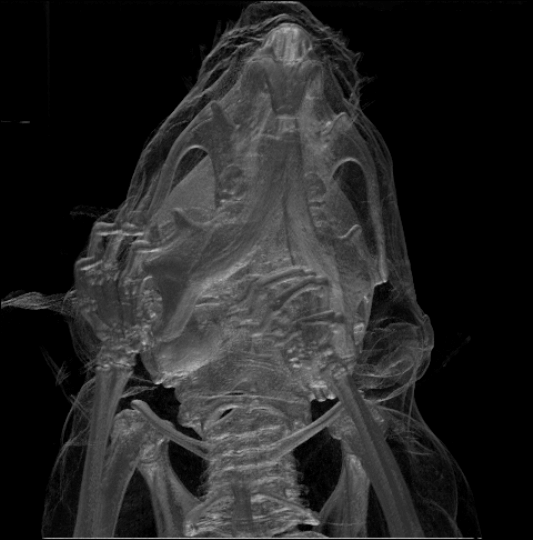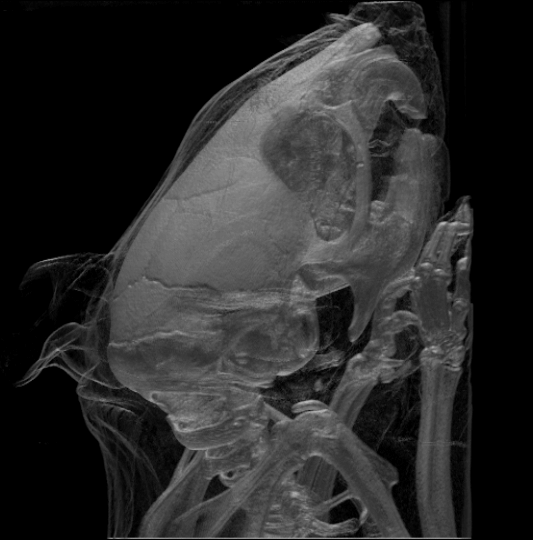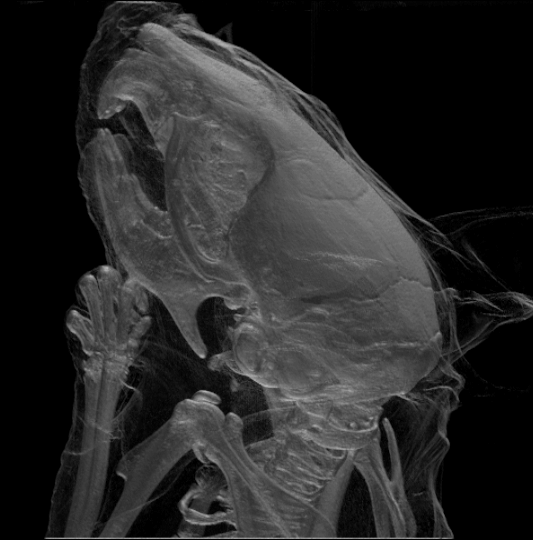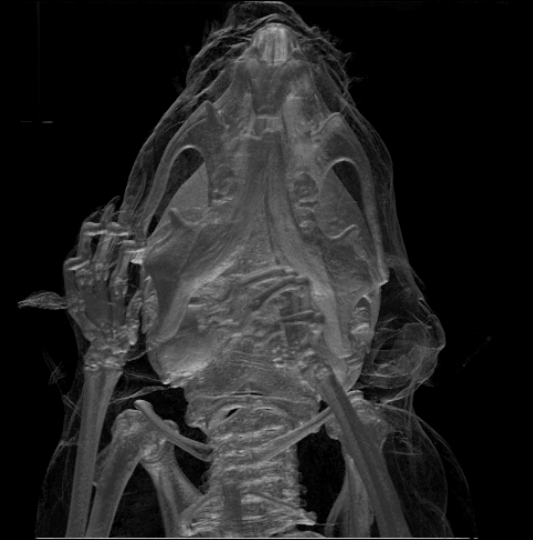 While the largest species, the
While the largest species, the  Rodents generally have well-developed senses of smell, hearing, and vision. Nocturnal species often have enlarged eyes and some are sensitive to
Rodents generally have well-developed senses of smell, hearing, and vision. Nocturnal species often have enlarged eyes and some are sensitive to
 One of the most widespread groups of mammals, rodents can be found on every continent except Antarctica. They are the only terrestrial placental mammals to have colonized Australia and
One of the most widespread groups of mammals, rodents can be found on every continent except Antarctica. They are the only terrestrial placental mammals to have colonized Australia and  Though some species are common pests for humans, rodents also play important ecological roles. Some rodents are considered keystone species and ecosystem engineers in their respective habitats. In the
Though some species are common pests for humans, rodents also play important ecological roles. Some rodents are considered keystone species and ecosystem engineers in their respective habitats. In the
 Most rodents are herbivorous, feeding exclusively on plant material such as seeds, stems, leaves, flowers, and roots. Some are
Most rodents are herbivorous, feeding exclusively on plant material such as seeds, stems, leaves, flowers, and roots. Some are  A strategy for dealing with seasonal plenty is to eat as much as possible and store the surplus nutrients as fat. Marmots do this, and may be 50% heavier in the autumn than in the spring. They rely on their fat reserves during their long winter
A strategy for dealing with seasonal plenty is to eat as much as possible and store the surplus nutrients as fat. Marmots do this, and may be 50% heavier in the autumn than in the spring. They rely on their fat reserves during their long winter
 Among the most social of rodents are the ground squirrels, which typically form colonies based on female kinship, with males dispersing after weaning and becoming nomadic as adults. Cooperation in ground squirrels varies between species and typically includes making alarm calls, defending territories, sharing food, protecting nesting areas, and preventing infanticide. The black-tailed prairie dog forms large towns that may cover many hectares. The burrows do not interconnect, but are excavated and occupied by territorial family groups known as coteries. A coterie often consists of an adult male, three or four adult females, several nonbreeding yearlings, and the current year's offspring. Individuals within coteries are friendly with each other, but hostile towards outsiders.
Perhaps the most extreme examples of colonial behavior in rodents are the eusocial naked mole rat and Damaraland mole rat. The naked mole rat lives completely underground and can form colonies of up to 80 individuals. Only one female and up to three males in the colony reproduce, while the rest of the members are smaller and sterile, and function as workers. Some individuals are of intermediate size. They help with the rearing of the young and can take the place of a reproductive if one dies. The Damaraland mole rat is characterized by having a single reproductively active male and female in a colony where the remaining animals are not truly sterile, but become fertile only if they establish a colony of their own. The naked mole-rat has a particularly long life-span for a small rodent, about 30 years, and the basis for this longevity has been investigated. Naked mole-rats express DNA repair genes, including core genes in several DNA repair pathways, at a higher level than shorter-lived mice, and thus it was suggested that DNA repair acts as a longevity assurance system.
Among the most social of rodents are the ground squirrels, which typically form colonies based on female kinship, with males dispersing after weaning and becoming nomadic as adults. Cooperation in ground squirrels varies between species and typically includes making alarm calls, defending territories, sharing food, protecting nesting areas, and preventing infanticide. The black-tailed prairie dog forms large towns that may cover many hectares. The burrows do not interconnect, but are excavated and occupied by territorial family groups known as coteries. A coterie often consists of an adult male, three or four adult females, several nonbreeding yearlings, and the current year's offspring. Individuals within coteries are friendly with each other, but hostile towards outsiders.
Perhaps the most extreme examples of colonial behavior in rodents are the eusocial naked mole rat and Damaraland mole rat. The naked mole rat lives completely underground and can form colonies of up to 80 individuals. Only one female and up to three males in the colony reproduce, while the rest of the members are smaller and sterile, and function as workers. Some individuals are of intermediate size. They help with the rearing of the young and can take the place of a reproductive if one dies. The Damaraland mole rat is characterized by having a single reproductively active male and female in a colony where the remaining animals are not truly sterile, but become fertile only if they establish a colony of their own. The naked mole-rat has a particularly long life-span for a small rodent, about 30 years, and the basis for this longevity has been investigated. Naked mole-rats express DNA repair genes, including core genes in several DNA repair pathways, at a higher level than shorter-lived mice, and thus it was suggested that DNA repair acts as a longevity assurance system.
 Vibrations can provide cues to conspecifics about specific behaviors being performed, predator warning and avoidance, herd or group maintenance, and courtship. The Middle East blind mole rat was the first mammal for which
Vibrations can provide cues to conspecifics about specific behaviors being performed, predator warning and avoidance, herd or group maintenance, and courtship. The Middle East blind mole rat was the first mammal for which
 Some species of rodent are monogamous, with an adult male and female forming a lasting
Some species of rodent are monogamous, with an adult male and female forming a lasting
 Rodents may be born either
Rodents may be born either  Mother rodents provide both direct parental care, such as nursing, grooming, retrieving and huddling, and indirect parenting, such as food caching, nest building and protection to their offspring. In many social species, young may be cared for by individuals other than their parents, a practice known as alloparenting or cooperative breeding. This is known to occur in black-tailed prairie dogs and Belding's ground squirrels, where mothers have communal nests and nurse unrelated young along with their own. There is some question as to whether these mothers can distinguish which young are theirs. In the Patagonian mara, young are also placed in communal warrens, but mothers do not permit youngsters other than their own to nurse.
Infanticide exists in numerous rodent species and may be practiced by adult conspecifics of either sex. Several reasons have been proposed for this behavior, including nutritional stress, resource competition, avoiding misdirecting parental care and, in the case of males, attempting to make the mother sexually receptive. The latter reason is well supported in
Mother rodents provide both direct parental care, such as nursing, grooming, retrieving and huddling, and indirect parenting, such as food caching, nest building and protection to their offspring. In many social species, young may be cared for by individuals other than their parents, a practice known as alloparenting or cooperative breeding. This is known to occur in black-tailed prairie dogs and Belding's ground squirrels, where mothers have communal nests and nurse unrelated young along with their own. There is some question as to whether these mothers can distinguish which young are theirs. In the Patagonian mara, young are also placed in communal warrens, but mothers do not permit youngsters other than their own to nurse.
Infanticide exists in numerous rodent species and may be practiced by adult conspecifics of either sex. Several reasons have been proposed for this behavior, including nutritional stress, resource competition, avoiding misdirecting parental care and, in the case of males, attempting to make the mother sexually receptive. The latter reason is well supported in
 Rodents have advanced
Rodents have advanced

 During the
During the
 Order Rodentia (from Latin, ''rodere'', to gnaw)
Order Rodentia (from Latin, ''rodere'', to gnaw)


 *Suborder Anomaluromorpha
*::*Family Anomaluridae: scaly-tailed squirrels
*::*Family Pedetidae: springhares
*::*Family Zenkerellidae: Cameroon scaly-tail
*Suborder Castorimorpha
*:*Superfamily Castoroidea
*:** Family
*Suborder Anomaluromorpha
*::*Family Anomaluridae: scaly-tailed squirrels
*::*Family Pedetidae: springhares
*::*Family Zenkerellidae: Cameroon scaly-tail
*Suborder Castorimorpha
*:*Superfamily Castoroidea
*:** Family
 Humanity has long used animal skins for clothing, as the leather is durable and the fur provides extra insulation. The native people of North America made much use of beaver pelts, tanning and sewing them together to make robes. Europeans appreciated the quality of these and the North American fur trade developed and became of prime importance to early settlers. In Europe, the soft underfur known as "beaver wool" was found to be ideal for felting and was made into beaver hats and trimming for clothing. Later, the coypu took over as a cheaper source of fur for felting and was farmed extensively in America and Europe; however, fashions changed, new materials became available and this area of the animal fur industry declined. The chinchilla has a soft and silky coat and the demand for its fur was so high that it was nearly wiped out in the wild before farming took over as the main source of pelts. The quills and guardhairs of porcupines are used for traditional decorative clothing. For example, their guardhairs are used in the creation of the Native American "porky roach" headdress. The main quills may be dyed, and then applied in combination with thread to embellish leather accessories such as knife sheaths and leather bags.
Humanity has long used animal skins for clothing, as the leather is durable and the fur provides extra insulation. The native people of North America made much use of beaver pelts, tanning and sewing them together to make robes. Europeans appreciated the quality of these and the North American fur trade developed and became of prime importance to early settlers. In Europe, the soft underfur known as "beaver wool" was found to be ideal for felting and was made into beaver hats and trimming for clothing. Later, the coypu took over as a cheaper source of fur for felting and was farmed extensively in America and Europe; however, fashions changed, new materials became available and this area of the animal fur industry declined. The chinchilla has a soft and silky coat and the demand for its fur was so high that it was nearly wiped out in the wild before farming took over as the main source of pelts. The quills and guardhairs of porcupines are used for traditional decorative clothing. For example, their guardhairs are used in the creation of the Native American "porky roach" headdress. The main quills may be dyed, and then applied in combination with thread to embellish leather accessories such as knife sheaths and leather bags.
 Rodents are used widely as
Rodents are used widely as
 Rodents including guinea pigs, mice, rats, hamsters, gerbils, chinchillas, degus and chipmunks make convenient pets able to live in small spaces, each species with its own qualities. Most are normally kept in cages of suitable sizes and have varied requirements for space and social interaction. If handled from a young age, they are usually docile and do not bite. Guinea pigs have a long lifespan and need a large cage. Rats also need plenty of space and can become very tame, can learn tricks and seem to enjoy human companionship. Mice are short-lived but take up very little space. Hamsters are solitary but tend to be nocturnal. They have interesting behaviors, but unless handled regularly they may be defensive. Gerbils are not usually aggressive, rarely bite and are sociable animals that enjoy the company of humans and their own kind.
Rodents including guinea pigs, mice, rats, hamsters, gerbils, chinchillas, degus and chipmunks make convenient pets able to live in small spaces, each species with its own qualities. Most are normally kept in cages of suitable sizes and have varied requirements for space and social interaction. If handled from a young age, they are usually docile and do not bite. Guinea pigs have a long lifespan and need a large cage. Rats also need plenty of space and can become very tame, can learn tricks and seem to enjoy human companionship. Mice are short-lived but take up very little space. Hamsters are solitary but tend to be nocturnal. They have interesting behaviors, but unless handled regularly they may be defensive. Gerbils are not usually aggressive, rarely bite and are sociable animals that enjoy the company of humans and their own kind.
 Some rodent species are serious agricultural
Some rodent species are serious agricultural
ArchéoZooThèque : Rodent osteology
(photos)
ArchéoZooThèque : Rodent skeleton drawings
African rodentia
Rodent photos
on
Rodent Species Fact Sheets
from the National Pest Management Association on Deer Mice, Norway Rats, and other rodent species {{Authority control Articles containing video clips Extant Thanetian first appearances Taxa named by Thomas Edward Bowdich
Latin
Latin ( or ) is a classical language belonging to the Italic languages, Italic branch of the Indo-European languages. Latin was originally spoken by the Latins (Italic tribe), Latins in Latium (now known as Lazio), the lower Tiber area aroun ...
, 'to gnaw') are mammal
A mammal () is a vertebrate animal of the Class (biology), class Mammalia (). Mammals are characterised by the presence of milk-producing mammary glands for feeding their young, a broad neocortex region of the brain, fur or hair, and three ...
s of the order Rodentia ( ), which are characterized by a single pair of continuously growing incisors
Incisors (from Latin ''incidere'', "to cut") are the front teeth present in most mammals. They are located in the premaxilla above and on the mandible below. Humans have a total of eight (two on each side, top and bottom). Opossums have 18, wher ...
in each of the upper and lower jaw
In jawed vertebrates, the mandible (from the Latin ''mandibula'', 'for chewing'), lower jaw, or jawbone is a bone that makes up the lowerand typically more mobilecomponent of the mouth (the upper jaw being known as the maxilla).
The jawbone i ...
s. About 40% of all mammal species are rodents. They are native to all major land masses except for Antarctica
Antarctica () is Earth's southernmost and least-populated continent. Situated almost entirely south of the Antarctic Circle and surrounded by the Southern Ocean (also known as the Antarctic Ocean), it contains the geographic South Pole. ...
, and several oceanic islands, though they have subsequently been introduced to most of these land masses by human activity.
Rodents are extremely diverse in their ecology and lifestyles and can be found in almost every terrestrial habitat, including human-made environments. Species can be arboreal
Arboreal locomotion is the locomotion of animals in trees. In habitats in which trees are present, animals have evolved to move in them. Some animals may scale trees only occasionally (scansorial), but others are exclusively arboreal. The hab ...
, fossorial
A fossorial animal () is one that is adapted to digging and which lives primarily (but not solely) underground. Examples of fossorial vertebrates are Mole (animal), moles, badgers, naked mole-rats, meerkats, armadillos, wombats, and mole salamand ...
(burrowing), saltatorial/ricochetal (leaping on their hind legs), or semiaquatic. However, all rodents share several morphological features, including having only a single upper and lower pair of ever-growing incisors. Well-known rodents include mice
A mouse (: mice) is a small rodent. Characteristically, mice are known to have a pointed snout, small rounded ears, a body-length scaly tail, and a high breeding rate. The best known mouse species is the common house mouse (''Mus musculus' ...
, rats, squirrel
Squirrels are members of the family Sciuridae (), a family that includes small or medium-sized rodents. The squirrel family includes tree squirrels, ground squirrels (including chipmunks and prairie dogs, among others), and flying squirrel ...
s, prairie dogs, porcupine
Porcupines are large rodents with coats of sharp Spine (zoology), spines, or quills, that protect them against predation. The term covers two Family (biology), families of animals: the Old World porcupines of the family Hystricidae, and the New ...
s, beaver
Beavers (genus ''Castor'') are large, semiaquatic rodents of the Northern Hemisphere. There are two existing species: the North American beaver (''Castor canadensis'') and the Eurasian beaver (''C. fiber''). Beavers are the second-large ...
s, guinea pig
The guinea pig or domestic guinea pig (''Cavia porcellus''), also known as the cavy or domestic cavy ( ), is a species of rodent belonging to the genus ''Cavia'', family Caviidae. Animal fancy, Breeders tend to use the name "cavy" for the ani ...
s, and hamsters. Once included with rodents, rabbit
Rabbits are small mammals in the family Leporidae (which also includes the hares), which is in the order Lagomorpha (which also includes pikas). They are familiar throughout the world as a small herbivore, a prey animal, a domesticated ...
s, hare
Hares and jackrabbits are mammals belonging to the genus ''Lepus''. They are herbivores and live Solitary animal, solitarily or in pairs. They nest in slight depressions called forms, and their young are precociality, able to fend for themselves ...
s, and pikas, which also have incisors that grow continuously (but have ''two'' pairs of upper incisors instead of one), are now considered to be in a separate order, the Lagomorpha. Nonetheless, Rodentia and Lagomorpha are sister group
In phylogenetics, a sister group or sister taxon, also called an adelphotaxon, comprises the closest relative(s) of another given unit in an evolutionary tree.
Definition
The expression is most easily illustrated by a cladogram:
Taxon A and ...
s, sharing a single common ancestor and forming the clade
In biology, a clade (), also known as a Monophyly, monophyletic group or natural group, is a group of organisms that is composed of a common ancestor and all of its descendants. Clades are the fundamental unit of cladistics, a modern approach t ...
of Glires.
Most rodents are small animals with robust bodies, short limbs, and long tails. They use their sharp incisors to gnaw food, excavate burrows, and defend themselves. Most eat seeds or other plant material, but some have more varied diets. They tend to be social animals and many species live in societies with complex ways of communicating with each other. Mating among rodents can vary from monogamy
Monogamy ( ) is a social relation, relationship of Dyad (sociology), two individuals in which they form a mutual and exclusive intimate Significant other, partnership. Having only one partner at any one time, whether for life or #Serial monogamy ...
, to polygyny
Polygyny () is a form of polygamy entailing the marriage of a man to several women. The term polygyny is from Neoclassical Greek πολυγυνία (); .
Incidence
Polygyny is more widespread in Africa than in any other continent. Some scholar ...
, to promiscuity
Promiscuity is the practice of engaging in sexual activity frequently with different partners or being indiscriminate in the choice of sexual partners. The term can carry a moral judgment. A common example of behavior viewed as promiscuous by man ...
. Many have litters of underdeveloped, altricial
Precocial species in birds and mammals are those in which the young are relatively mature and mobile from the moment of birth or hatching. They are normally nidifugous, meaning that they leave the nest shortly after birth or hatching. Altricial ...
young, while others are precocial
Precocial species in birds and mammals are those in which the young are relatively mature and mobile from the moment of birth or hatching. They are normally nidifugous, meaning that they leave the nest shortly after birth or hatching. Altricial ...
(relatively well developed) at birth.
The rodent fossil
A fossil (from Classical Latin , ) is any preserved remains, impression, or trace of any once-living thing from a past geological age. Examples include bones, shells, exoskeletons, stone imprints of animals or microbes, objects preserve ...
record dates back to the Paleocene
The Paleocene ( ), or Palaeocene, is a geological epoch (geology), epoch that lasted from about 66 to 56 mya (unit), million years ago (mya). It is the first epoch of the Paleogene Period (geology), Period in the modern Cenozoic Era (geology), ...
on the supercontinent of Laurasia
Laurasia () was the more northern of two large landmasses that formed part of the Pangaea supercontinent from around ( Mya), the other being Gondwana. It separated from Gondwana (beginning in the late Triassic period) during the breakup of Pa ...
. Rodents greatly diversified in the Eocene
The Eocene ( ) is a geological epoch (geology), epoch that lasted from about 56 to 33.9 million years ago (Ma). It is the second epoch of the Paleogene Period (geology), Period in the modern Cenozoic Era (geology), Era. The name ''Eocene'' comes ...
, as they spread across continents, sometimes even crossing oceans. Rodents reached both South America and Madagascar from Africa and, until the arrival of ''Homo sapiens'', were the only terrestrial placental mammals to reach and colonize Australia.
Rodents have been used as food, for clothing, as pets, and as laboratory animals in research. Some species, in particular, the brown rat
The brown rat (''Rattus norvegicus''), also known as the common rat, street rat, sewer rat, wharf rat, Hanover rat, Norway rat and Norwegian rat, is a widespread species of common rat. One of the largest Muroidea, muroids, it is a brown or grey ...
, the black rat
The black rat (''Rattus rattus''), also known as the roof rat, ship rat, or house rat, is a common long-tailed rodent of the stereotypical rat genus ''Rattus'', in the subfamily Murinae. It likely originated in the Indian subcontinent, but is n ...
, and the house mouse
The house mouse (''Mus musculus'') is a small mammal of the rodent family Muridae, characteristically having a pointed snout, large rounded ears, and a long and almost hairless tail. It is one of the most abundant species of the genus '' Mus''. A ...
, are serious pests
PESTS was an anonymous American activist group formed in 1986 to critique racism, tokenism, and exclusion in the art world. PESTS produced newsletters, posters, and other print material highlighting examples of discrimination in gallery represent ...
, eating and spoiling food stored by humans and spreading diseases. Accidentally introduced species
An introduced species, alien species, exotic species, adventive species, immigrant species, foreign species, non-indigenous species, or non-native species is a species living outside its native distributional range, but which has arrived ther ...
of rodents are often considered to be invasive and have caused the extinction of numerous species, such as island birds, the dodo
The dodo (''Raphus cucullatus'') is an extinction, extinct flightless bird that was endemism, endemic to the island of Mauritius, which is east of Madagascar in the Indian Ocean. The dodo's closest relative was the also-extinct and flightles ...
being an example, previously isolated from land-based predators.
Characteristics
The distinguishing feature of the rodents is their pairs of continuously growing, razor-sharp, open-rootedincisors
Incisors (from Latin ''incidere'', "to cut") are the front teeth present in most mammals. They are located in the premaxilla above and on the mandible below. Humans have a total of eight (two on each side, top and bottom). Opossums have 18, wher ...
. These incisors have thick layers of enamel on the front and little enamel on the back. Because they do not stop growing, the animal must continue to wear them down so that they do not reach and pierce the skull. As the incisors grind against each other, the softer dentine on the rear of the teeth wears away, leaving the sharp enamel edge shaped like the blade of a chisel
A chisel is a hand tool with a characteristic Wedge, wedge-shaped cutting edge on the end of its blade. A chisel is useful for carving or cutting a hard material such as woodworking, wood, lapidary, stone, or metalworking, metal.
Using a chi ...
. Most species have up to 22 teeth with no canines or anterior premolar
The premolars, also called premolar Tooth (human), teeth, or bicuspids, are transitional teeth located between the Canine tooth, canine and Molar (tooth), molar teeth. In humans, there are two premolars per dental terminology#Quadrant, quadrant in ...
s. A gap, or diastema, occurs between the incisors and the cheek teeth in most species. This allows rodents to suck in their cheeks or lips to shield their mouth and throat from wood shavings and other inedible material, discarding this waste from the sides of their mouths. Chinchillas and guinea pigs have a high-fiber diet; their molars have no roots and grow continuously like their incisors.
In many species, the molars are relatively large, intricately structured, and highly cusped or ridged. Rodent molars are well equipped to grind food into small particles. The jaw musculature is strong. The lower jaw is thrust forward while gnawing and is pulled backwards during chewing. Gnawing uses incisors and chewing uses molars, however, due to the cranial anatomy of rodents these feeding methods cannot be used at the same time and are considered to be mutually exclusive. Among rodents, the masseter
In anatomy, the masseter is one of the muscles of mastication. Found only in mammals, it is particularly powerful in herbivores to facilitate chewing of plant matter. The most obvious muscle of mastication is the masseter muscle, since it is the ...
muscle plays a key role in chewing, making up 60% – 80% of the total muscle mass among masticatory muscles and reflects rodents' herbivorous diet. Rodent groups differ in the arrangement of the jaw muscles and associated skull structures, both from other mammals and amongst themselves.
The Sciuromorpha
Sciuromorpha ( 'squirrel-like') is a rodent Order (biology), suborder that includes several rodent Family (biology), families. It includes all members of the Sciuridae (the squirrel family) as well as the mountain beaver species.
Traditionally, ...
, such as the eastern grey squirrel, have a large deep masseter
In anatomy, the masseter is one of the muscles of mastication. Found only in mammals, it is particularly powerful in herbivores to facilitate chewing of plant matter. The most obvious muscle of mastication is the masseter muscle, since it is the ...
, making them efficient at biting with the incisors. The Myomorpha, such as the brown rat, have enlarged temporalis and masseter muscles, making them able to chew powerfully with their molars. In rodents, masseter muscles insert behind the eyes and contribute to eye boggling that occurs during gnawing where the quick contraction and relaxation of the muscle causes the eyeballs to move up and down. The Hystricomorpha, such as the guinea pig, have larger superficial masseter muscles and smaller deep masseter muscles than rats or squirrels, possibly making them less efficient at biting with the incisors, but their enlarged internal pterygoid muscles may allow them to move the jaw further sideways when chewing. The cheek pouch is a specific morphological feature used for storing food and is evident in particular subgroups of rodents like kangaroo rats, hamsters, chipmunks and gophers which have two bags that may range from the mouth to the front of the shoulders. True mice and rats do not contain this structure but their cheeks are elastic due to a high degree of musculature and innervation in the region.
 While the largest species, the
While the largest species, the capybara
The capybara or greater capybara (''Hydrochoerus hydrochaeris'') is the largest living rodent, native to South America. It is a member of the genus '' Hydrochoerus''. The only other extant member is the lesser capybara (''Hydrochoerus isthmi ...
, can weigh as much as , most rodents weigh less than . Rodents have wide-ranging morphologies, but typically have squat bodies and short limbs. The fore limbs usually have five digits, including an opposable thumb, while the hind limbs have three to five digits. The elbow gives the forearms great flexibility. The majority of species are plantigrade, walking on both the palms and soles of their feet, and have claw-like nails. The nails of burrowing species tend to be long and strong, while arboreal rodents have shorter, sharper nails. Rodent species use a wide variety of methods of locomotion including quadrupedal walking, running, burrowing, climbing, bipedal hopping ( kangaroo rats and hopping mice
A hopping mouse is any of about ten different Australian native mice in the genus ''Notomys''. They are rodents, not marsupials, and their ancestors are thought to have arrived from Asia about 5 million years ago.
All are brown or fawn, fading t ...
), swimming and even gliding.
Scaly-tailed squirrels and flying squirrel
Flying squirrels (scientifically known as Pteromyini or Petauristini) are a tribe (biology), tribe of 50 species of squirrels in the family (biology), family Squirrel, Sciuridae. Despite their name, they are not in fact capable of full flight i ...
s, although not closely related, can both glide from tree to tree using parachute-like membranes that stretch from the fore to the hind limbs. The agouti is fleet-footed and antelope
The term antelope refers to numerous extant or recently extinct species of the ruminant artiodactyl family Bovidae that are indigenous to most of Africa, India, the Middle East, Central Asia, and a small area of Eastern Europe. Antelopes do ...
-like, being digitigrade and having hoof-like nails. The majority of rodents have tails, which can be of many shapes and sizes. Some tails are prehensile, as in the Eurasian harvest mouse, and the fur on the tails can vary from bushy to completely bald. The tail is sometimes used for communication, as when beavers slap their tails on the water surface or house mice rattle their tails to indicate alarm. Some species have vestigial tails or no tails at all. In some species, the tail is capable of regeneration if a part is broken off.
 Rodents generally have well-developed senses of smell, hearing, and vision. Nocturnal species often have enlarged eyes and some are sensitive to
Rodents generally have well-developed senses of smell, hearing, and vision. Nocturnal species often have enlarged eyes and some are sensitive to ultraviolet
Ultraviolet radiation, also known as simply UV, is electromagnetic radiation of wavelengths of 10–400 nanometers, shorter than that of visible light, but longer than X-rays. UV radiation is present in sunlight and constitutes about 10% of ...
light. Many species have long, sensitive whiskers or vibrissae for touch or "whisking". Whisker action is mostly driven by the brain stem, which is itself provoked by the cortex. However Legg et al. 1989 find an alternate circuit between the cortex and whiskers through the cerebellar circuits, and Hemelt & Keller 2008 the superior colliculus. Some rodents have cheek pouches, which may be lined with fur. These can be turned inside out for cleaning. In many species, the tongue cannot reach past the incisors. Rodents have efficient digestive systems, absorbing nearly 80% of ingested energy. When eating cellulose
Cellulose is an organic compound with the chemical formula, formula , a polysaccharide consisting of a linear chain of several hundred to many thousands of glycosidic bond, β(1→4) linked glucose, D-glucose units. Cellulose is an important s ...
, the food is softened in the stomach and passed to the cecum
The cecum ( caecum, ; plural ceca or caeca, ) is a pouch within the peritoneum that is considered to be the beginning of the large intestine. It is typically located on the right side of the body (the same side of the body as the appendix (a ...
, where bacteria reduce it to its carbohydrate
A carbohydrate () is a biomolecule composed of carbon (C), hydrogen (H), and oxygen (O) atoms. The typical hydrogen-to-oxygen atomic ratio is 2:1, analogous to that of water, and is represented by the empirical formula (where ''m'' and ''n'' ...
elements. The rodent then practices coprophagy, eating its own fecal pellets, so the nutrients can be absorbed by the gut. Rodents therefore often produce a hard and dry fecal pellet. Horn et al. 2013 makes the finding that rodents entirely lack the ability to vomit. In many species, the penis contains a bone, the baculum
The baculum (: bacula), also known as the penis bone, penile bone, ''os penis'', ''os genitale'', or ''os priapi'', is a bone in the penis of many placental mammals. It is not present in humans, but is present in the penises of some primates, ...
; the testes can be located either abdominally or at the groin.
Sexual dimorphism
Sexual dimorphism is the condition where sexes of the same species exhibit different Morphology (biology), morphological characteristics, including characteristics not directly involved in reproduction. The condition occurs in most dioecy, di ...
occurs in many rodent species. In some rodents, males are larger than females, while in others the reverse is true. Male-bias sexual dimorphism is typical for ground squirrels, kangaroo rats, solitary mole rat Mole-rat or mole rat can refer to several groups of burrowing Old World rodents:
* Bathyergidae, a family of about 20 hystricognath species in six genera from Africa also called blesmols.
*''Heterocephalus glaber'', the naked mole-rat.
* Spalacidae ...
s and pocket gopher
Pocket gophers, commonly referred to simply as gophers, are burrowing rodents of the family Geomyidae. The roughly 41 speciesSearch results for "Geomyidae" on thASM Mammal Diversity Database are all endemic to North and Central America. They ar ...
s; it likely developed due to sexual selection
Sexual selection is a mechanism of evolution in which members of one sex mate choice, choose mates of the other sex to mating, mate with (intersexual selection), and compete with members of the same sex for access to members of the opposite sex ...
and greater male–male combat. Female-bias sexual dimorphism exists among chipmunks and jumping mice. It is not understood why this pattern occurs, but in the case of yellow-pine chipmunks, males may have selected larger females due to their greater reproductive success. In some species, such as vole
Voles are small rodents that are relatives of lemmings and hamsters, but with a stouter body; a longer, hairy tail; a slightly rounder head; smaller eyes and ears; and differently formed molars (high-crowned with angular cusps instead of lo ...
s, sexual dimorphism can vary from population to population. In bank vole
The bank vole (''Clethrionomys glareolus'') is a small vole with red-brown fur and some grey patches, with a tail about half as long as its body. A rodent, it lives in woodland areas and is around in length. The bank vole is found in much of Eu ...
s, females are typically larger than males, but male-bias sexual dimorphism occurs in alpine populations, possibly because of the lack of predators and greater competition between males.
Distribution and habitat
 One of the most widespread groups of mammals, rodents can be found on every continent except Antarctica. They are the only terrestrial placental mammals to have colonized Australia and
One of the most widespread groups of mammals, rodents can be found on every continent except Antarctica. They are the only terrestrial placental mammals to have colonized Australia and New Guinea
New Guinea (; Hiri Motu: ''Niu Gini''; , fossilized , also known as Papua or historically ) is the List of islands by area, world's second-largest island, with an area of . Located in Melanesia in the southwestern Pacific Ocean, the island is ...
without human intervention. Humans have also allowed the animals to spread to many remote oceanic islands (e.g., the Polynesian rat). Rodents have adapted to almost every terrestrial habitat, from cold tundra
In physical geography, a tundra () is a type of biome where tree growth is hindered by frigid temperatures and short growing seasons. There are three regions and associated types of tundra: #Arctic, Arctic, Alpine tundra, Alpine, and #Antarctic ...
(where they can live under snow) to hot deserts.
Some species such as tree squirrel
Tree squirrels are the members of the squirrel Family (biology), family (Sciuridae) commonly just referred to as "squirrels". They include more than 100 arboreal species native to all continents except Antarctica and Oceania.
They do not form a ...
s and New World porcupines are arboreal
Arboreal locomotion is the locomotion of animals in trees. In habitats in which trees are present, animals have evolved to move in them. Some animals may scale trees only occasionally (scansorial), but others are exclusively arboreal. The hab ...
, while some, such as gopher
Pocket gophers, commonly referred to simply as gophers, are burrowing rodents of the family Geomyidae. The roughly 41 speciesSearch results for "Geomyidae" on thASM Mammal Diversity Database are all endemic to North and Central America. They ar ...
s, tuco-tucos, and mole rats, live almost completely underground, where they build complex burrow systems. Others dwell on the surface of the ground, but may have a burrow into which they can retreat. Beavers and muskrats are known for being semiaquatic, but the rodent best adapted for aquatic life is probably the earless water rat from New Guinea. Rodents have also thrived in human-created environments such as agricultural
Agriculture encompasses crop and livestock production, aquaculture, and forestry for food and non-food products. Agriculture was a key factor in the rise of sedentary human civilization, whereby farming of domesticated species created f ...
and urban area
An urban area is a human settlement with a high population density and an infrastructure of built environment. Urban areas originate through urbanization, and researchers categorize them as cities, towns, conurbations or suburbs. In urbani ...
s.
Great Plains
The Great Plains is a broad expanse of plain, flatland in North America. The region stretches east of the Rocky Mountains, much of it covered in prairie, steppe, and grassland. They are the western part of the Interior Plains, which include th ...
of North America, the burrowing activities of prairie dogs play important roles in soil aeration
Aeration (also called aerification or aeriation) is the process by which air is circulated through, mixed with or dissolved in a liquid or other substances that act as a fluid (such as soil). Aeration processes create additional surface area in t ...
and nutrient redistribution, raising the organic content of the soil and increasing the absorption of water. They maintain these grassland habitats, and some large herbivores such as bison
A bison (: bison) is a large bovine in the genus ''Bison'' (from Greek, meaning 'wild ox') within the tribe Bovini. Two extant taxon, extant and numerous extinction, extinct species are recognised.
Of the two surviving species, the American ...
and pronghorn prefer to graze near prairie dog colonies due to the increased nutritional quality of forage.
Extirpation of prairie dogs can also contribute to regional and local biodiversity loss
Biodiversity loss happens when plant or animal species disappear completely from Earth (extinction) or when there is a decrease or disappearance of species in a specific area. Biodiversity loss means that there is a reduction in Biodiversity, b ...
, increased seed depredation, and the establishment and spread of invasive shrubs. Burrowing rodents may eat the fruiting bodies of fungi
A fungus (: fungi , , , or ; or funguses) is any member of the group of eukaryotic organisms that includes microorganisms such as yeasts and mold (fungus), molds, as well as the more familiar mushrooms. These organisms are classified as one ...
and spread spore
In biology, a spore is a unit of sexual reproduction, sexual (in fungi) or asexual reproduction that may be adapted for biological dispersal, dispersal and for survival, often for extended periods of time, in unfavourable conditions. Spores fo ...
s through their feces, thereby allowing the fungi to disperse and form symbiotic relationship
Symbiosis (Ancient Greek : living with, companionship < : together; and ''bíōsis'': living) is any type of a close and long-term biolo ...
s with the roots of plants (which usually cannot thrive without them). As such, these rodents may play a role in maintaining healthy forests.
In many temperate regions, beavers play an essential hydrological
Hydrology () is the scientific study of the movement, distribution, and management of water on Earth and other planets, including the water cycle, water resources, and drainage basin sustainability. A practitioner of hydrology is called a hydro ...
role. When building their dams and lodges, beavers alter the paths of streams and rivers and allow for the creation of extensive wetland habitats. One study found that engineering by beavers leads to a 33 percent increase in the number of herbaceous plant
Herbaceous plants are vascular plants that have no persistent woody stems above ground. This broad category of plants includes many perennials, and nearly all annuals and biennials.
Definitions of "herb" and "herbaceous"
The fourth edition o ...
species in riparian area
A riparian zone or riparian area is the interface between land and a river or stream. In some regions, the terms riparian woodland, riparian forest, riparian buffer zone, riparian corridor, and riparian strip are used to characterize a ripar ...
s. Another study found that beavers increase wild salmon populations. Meanwhile, some rodents are seen as pests
PESTS was an anonymous American activist group formed in 1986 to critique racism, tokenism, and exclusion in the art world. PESTS produced newsletters, posters, and other print material highlighting examples of discrimination in gallery represent ...
, due to their wide range.
Behavior and life history
Feeding
 Most rodents are herbivorous, feeding exclusively on plant material such as seeds, stems, leaves, flowers, and roots. Some are
Most rodents are herbivorous, feeding exclusively on plant material such as seeds, stems, leaves, flowers, and roots. Some are omnivorous
An omnivore () is an animal that regularly consumes significant quantities of both plant and animal matter. Obtaining energy and nutrients from plant and animal matter, omnivores digest carbohydrates, protein, fat, and fiber, and metabolize ...
and a few are predators. The field vole is a typical herbivorous rodent and feeds on grasses, herbs, root tubers, moss, and other vegetation, and gnaws on bark during the winter. It occasionally eats invertebrates such as insect larvae. The plains pocket gopher eats plant material found underground during tunneling, and also collects grasses, roots, and tubers in its cheek pouches and caches them in underground larder chambers.
The Texas pocket gopher avoids emerging onto the surface to feed by seizing the roots of plants with its jaws and pulling them downwards into its burrow. It also practices coprophagy. The African pouched rat forages on the surface, gathering anything that might be edible into its capacious cheek pouches until its face bulges out sideways. It then returns to its burrow to sort through the material it has gathered and eats the nutritious items.
Agouti species are one of the few animal groups that can break open the large capsules of the Brazil nut fruit. Too many seeds are inside to be consumed in one meal, so the agouti carries some off and caches them. This helps dispersal of the seeds as any that the agouti fails to retrieve are distant from the parent tree when they germinate. Other nut-bearing trees tend to bear a glut of fruits in the autumn. These are too numerous to be eaten in one meal and squirrels gather and store the surplus in crevices and hollow trees. In desert regions, seeds are often available only for short periods. The kangaroo rat collects all it can find and stores them in larder chambers in its burrow.
 A strategy for dealing with seasonal plenty is to eat as much as possible and store the surplus nutrients as fat. Marmots do this, and may be 50% heavier in the autumn than in the spring. They rely on their fat reserves during their long winter
A strategy for dealing with seasonal plenty is to eat as much as possible and store the surplus nutrients as fat. Marmots do this, and may be 50% heavier in the autumn than in the spring. They rely on their fat reserves during their long winter hibernation
Hibernation is a state of minimal activity and metabolic reduction entered by some animal species. Hibernation is a seasonal heterothermy characterized by low body-temperature, slow breathing and heart-rate, and low metabolic rate. It is mos ...
. Beavers feed on the leaves, buds, and inner bark of growing trees, as well as aquatic plants. They store food for winter use by felling small trees and leafy branches in the autumn and immersing them in their pond, sticking the ends into the mud to anchor them. Here, they can access their food supply underwater even when their pond is frozen over.
Although rodents have been regarded traditionally as herbivores, most small rodents opportunistically include insects, worms, fungi, fish, or meat in their diets and a few have become specialized to rely on a diet of animal matter. A functional-morphological study of the rodent tooth system supports the idea that primitive rodents were omnivores rather than herbivores. Studies of the literature show that numerous members of the Sciuromorpha and Myomorpha, and a few members of the Hystricomorpha, have either included animal matter in their diets or been prepared to eat such food when offered it in captivity. Examination of the stomach contents of the North American white-footed mouse, normally considered to be herbivorous, showed 34% animal matter.
More specialized carnivores include the shrewlike rats of the Philippines, which feed on insects and soft-bodied invertebrates, and the rakali or Australian water-rat, which devours aquatic insects, fish, crustaceans, mussels, snails, frogs, birds' eggs, and water birds. The grasshopper mouse from dry regions of North America feeds on insects, scorpions, and other small mice, and only a small part of its diet is plant material. It has a chunky body with short legs and tail, but is agile and can easily overpower prey as large as itself.
Social behavior
Rodents exhibit a wide range of types of social behavior ranging from the mammalian caste system of the naked mole-rat, the extensive "town" of the colonial prairie dog, through family groups to the independent, solitary life of theedible dormouse
''Glis'' is a genus of rodent that contains two extant species, both known as edible dormice or fat dormice: the European edible dormouse ''(Glis glis'') and the Iranian edible dormouse (''Glis persicus''). It also contains a number of fossil spe ...
. Adult dormice may have overlapping feeding ranges, but they live in individual nests and feed separately, coming together briefly in the breeding season to mate. The pocket gopher
Pocket gophers, commonly referred to simply as gophers, are burrowing rodents of the family Geomyidae. The roughly 41 speciesSearch results for "Geomyidae" on thASM Mammal Diversity Database are all endemic to North and Central America. They ar ...
is also a solitary animal outside the breeding season, each individual digging a complex tunnel system and maintaining a territory.
Larger rodents tend to live in family units where parents and their offspring live together until the young disperse. Beaver
Beavers (genus ''Castor'') are large, semiaquatic rodents of the Northern Hemisphere. There are two existing species: the North American beaver (''Castor canadensis'') and the Eurasian beaver (''C. fiber''). Beavers are the second-large ...
s live in extended family units typically with a pair of adults, this year's kits, the previous year's offspring, and sometimes older young. Brown rats usually live in small colonies with up to six females sharing a burrow and one male defending a territory
A territory is an area of land, sea, or space, belonging or connected to a particular country, person, or animal.
In international politics, a territory is usually a geographic area which has not been granted the powers of self-government, ...
around the burrow. At high population densities, this system breaks down and males show a hierarchical system of dominance with overlapping ranges. Female offspring remain in the colony while male young disperse. The prairie vole
The prairie vole (''Microtus ochrogaster'') is a small vole found in central North America.
Description
The vole has long, coarse grayish-brown fur on the upper portion of the body and yellowish fur on the lower portion of the body. It has sh ...
is monogamous and forms a lifelong pair bond. Outside the breeding season, prairie voles live with others in small colonies. A male is not aggressive towards other males until he has mated, after which time he defends a territory, a female, and a nest against other males. The pair huddles together, grooms one another, and shares nesting and pup-raising responsibilities.
 Among the most social of rodents are the ground squirrels, which typically form colonies based on female kinship, with males dispersing after weaning and becoming nomadic as adults. Cooperation in ground squirrels varies between species and typically includes making alarm calls, defending territories, sharing food, protecting nesting areas, and preventing infanticide. The black-tailed prairie dog forms large towns that may cover many hectares. The burrows do not interconnect, but are excavated and occupied by territorial family groups known as coteries. A coterie often consists of an adult male, three or four adult females, several nonbreeding yearlings, and the current year's offspring. Individuals within coteries are friendly with each other, but hostile towards outsiders.
Perhaps the most extreme examples of colonial behavior in rodents are the eusocial naked mole rat and Damaraland mole rat. The naked mole rat lives completely underground and can form colonies of up to 80 individuals. Only one female and up to three males in the colony reproduce, while the rest of the members are smaller and sterile, and function as workers. Some individuals are of intermediate size. They help with the rearing of the young and can take the place of a reproductive if one dies. The Damaraland mole rat is characterized by having a single reproductively active male and female in a colony where the remaining animals are not truly sterile, but become fertile only if they establish a colony of their own. The naked mole-rat has a particularly long life-span for a small rodent, about 30 years, and the basis for this longevity has been investigated. Naked mole-rats express DNA repair genes, including core genes in several DNA repair pathways, at a higher level than shorter-lived mice, and thus it was suggested that DNA repair acts as a longevity assurance system.
Among the most social of rodents are the ground squirrels, which typically form colonies based on female kinship, with males dispersing after weaning and becoming nomadic as adults. Cooperation in ground squirrels varies between species and typically includes making alarm calls, defending territories, sharing food, protecting nesting areas, and preventing infanticide. The black-tailed prairie dog forms large towns that may cover many hectares. The burrows do not interconnect, but are excavated and occupied by territorial family groups known as coteries. A coterie often consists of an adult male, three or four adult females, several nonbreeding yearlings, and the current year's offspring. Individuals within coteries are friendly with each other, but hostile towards outsiders.
Perhaps the most extreme examples of colonial behavior in rodents are the eusocial naked mole rat and Damaraland mole rat. The naked mole rat lives completely underground and can form colonies of up to 80 individuals. Only one female and up to three males in the colony reproduce, while the rest of the members are smaller and sterile, and function as workers. Some individuals are of intermediate size. They help with the rearing of the young and can take the place of a reproductive if one dies. The Damaraland mole rat is characterized by having a single reproductively active male and female in a colony where the remaining animals are not truly sterile, but become fertile only if they establish a colony of their own. The naked mole-rat has a particularly long life-span for a small rodent, about 30 years, and the basis for this longevity has been investigated. Naked mole-rats express DNA repair genes, including core genes in several DNA repair pathways, at a higher level than shorter-lived mice, and thus it was suggested that DNA repair acts as a longevity assurance system.
Communication
Olfactory
Rodents use scent marking in many social contexts including inter- and intra-species communication, the marking of trails and the establishment of territories. Their urine provides genetic information about individuals including the species, the sex and individual identity, and metabolic information on dominance, reproductive status and health. Compounds derived from themajor histocompatibility complex
The major histocompatibility complex (MHC) is a large Locus (genetics), locus on vertebrate DNA containing a set of closely linked polymorphic genes that code for Cell (biology), cell surface proteins essential for the adaptive immune system. The ...
(MHC) are bound to several urinary proteins. The odor of a predator depresses scent-marking behavior.
Rodents are able to recognize close relatives by smell and this allows them to show nepotism
Nepotism is the act of granting an In-group favoritism, advantage, privilege, or position to Kinship, relatives in an occupation or field. These fields can include business, politics, academia, entertainment, sports, religion or health care. In ...
(preferential behavior toward their kin) and also avoid inbreeding. This kin recognition is by olfactory cues from urine, feces and glandular secretions. The main assessment may involve the MHC, where the degree of relatedness of two individuals is correlated to the MHC genes they have in common. In non-kin communication, where more permanent odor markers are required, as at territorial borders, then non-volatile major urinary proteins (MUPs), which function as pheromone
A pheromone () is a secreted or excreted chemical factor that triggers a social response in members of the same species. Pheromones are chemicals capable of acting like hormones outside the body of the secreting individual, to affect the behavio ...
transporters, may also be used. MUPs may also signal individual identity, with each male house mouse
The house mouse (''Mus musculus'') is a small mammal of the rodent family Muridae, characteristically having a pointed snout, large rounded ears, and a long and almost hairless tail. It is one of the most abundant species of the genus '' Mus''. A ...
(''Mus musculus'') excreting urine containing about a dozen genetically encoded MUPs.
House mice deposit urine, which contains pheromones, for territorial marking, individual and group recognition, and social organization. Territorial beavers and red squirrels investigate and become familiar with the scents of their neighbors and respond less aggressively to intrusions by them than to those made by non-territorial "floaters" or strangers. This is known as the " dear enemy effect".
Auditory
Many rodent species, particularly those that are diurnal and social, have a wide range of alarm calls that are emitted when they perceive threats. There are both direct and indirect benefits of doing this. A potential predator may stop when it knows it has been detected, or an alarm call can allow conspecifics or related individuals to take evasive action. Several species, for example prairie dogs, have complex anti-predator alarm call systems. These species may have different calls for different predators (e.g. aerial predators or ground-based predators) and each call contains information about the nature of the precise threat. The urgency of the threat is also conveyed by the acoustic properties of the call. Social rodents have a wider range of vocalizations than do solitary species. Fifteen different call-types have been recognized in adult Kataba mole rats and four in juveniles. Similarly, the common degu, another social, burrowing rodent, exhibits a wide array of communication methods and has an elaborate vocal repertoire comprising fifteen different categories of sound. Ultrasonic calls play a part in social communication betweendormice
A dormouse is a rodent of the family (biology), family Gliridae (this family is also variously called Myoxidae or Muscardinidae by different taxonomists). Dormice are nocturnal animals found in Africa, Asia, and Europe. They are named for their ...
and are used when the individuals are out of sight of each other.
House mice use both audible and ultrasonic calls in a variety of contexts. Audible vocalizations can often be heard during agonistic or aggressive encounters, whereas ultrasound is used in sexual communication and also by pups when they have fallen out of the nest.
Laboratory rat
Laboratory rats or lab rats are strain (biology), strains of the rat subspecies ''Rattus norvegicus domestica'' (Domestic Norwegian rat) which are bred and kept for scientific research. While Animal testing on rodents, less commonly used for re ...
s (which are brown rats, ''Rattus norvegicus'') emit short, high frequency, ultrasonic vocalizations during purportedly pleasurable experiences such as rough-and-tumble play, when anticipating routine doses of morphine
Morphine, formerly also called morphia, is an opiate that is found naturally in opium, a dark brown resin produced by drying the latex of opium poppies (''Papaver somniferum''). It is mainly used as an analgesic (pain medication). There are ...
, during mating, and when tickled. The vocalization, described as a distinct "chirping", has been likened to laughter
Laughter is a pleasant physical reaction and emotion consisting usually of rhythmical, usually audible contractions of the diaphragm and other parts of the respiratory system. It is a response to certain external or internal stimuli. Laug ...
, and is interpreted as an expectation of something rewarding. In clinical studies, the chirping is associated with positive emotional feelings, and social bonding occurs with the tickler, resulting in the rats becoming conditioned to seek the tickling. However, as the rats age, the tendency to chirp declines. Like most rat vocalizations, the chirping is at frequencies too high for humans to hear without special equipment, so bat detectors have been used for this purpose.
Visual
Rodents, like all placental mammals except primates, have just two types of light receptive cones in their retina, a short wavelength "blue-UV" type and a middle wavelength "green" type. They are therefore classified as dichromats; however, they are visually sensitive into the ultraviolet (UV) spectrum and therefore can see light that humans cannot. The functions of this UV sensitivity are not always clear. In degus, for example, the belly reflects more UV light than the back. Therefore, when a degu stands up on its hind legs, which it does when alarmed, it exposes its belly to other degus and ultraviolet vision may serve a purpose in communicating the alarm. When it stands on all fours, its low UV-reflectance back could help make the degu less visible to predators. Ultraviolet light is abundant during the day but not at night. There is a large increase in the ratio of ultraviolet to visible light in the morning and evening twilight hours. Many rodents are active during twilight hours (crepuscular activity), and UV-sensitivity would be advantageous at these times. Ultraviolet reflectivity is of dubious value for nocturnal rodents. The urine of many rodents (e.g. voles, degus, mice, rats) strongly reflects UV light and this may be used in communication by leaving visible as well as olfactory markings. However, the amount of UV that is reflected decreases with time, which in some circumstances can be disadvantageous; thecommon kestrel
The common kestrel (''Falco tinnunculus''), also known as the European kestrel, Eurasian kestrel or Old World kestrel, is a species of bird of prey, predatory bird belonging to the kestrel group of the falcon family (biology), family Falconidae. ...
can distinguish between old and fresh rodent trails and has greater success hunting over more recently marked routes.
Tactile
 Vibrations can provide cues to conspecifics about specific behaviors being performed, predator warning and avoidance, herd or group maintenance, and courtship. The Middle East blind mole rat was the first mammal for which
Vibrations can provide cues to conspecifics about specific behaviors being performed, predator warning and avoidance, herd or group maintenance, and courtship. The Middle East blind mole rat was the first mammal for which seismic communication
Seismic or vibrational communication is a process of conveying information through mechanical (seismology, seismic) vibrations of the substrate. The substrate may be the earth, a plant stem or leaf, the surface of a body of water, a spider's web, ...
was documented. These fossorial
A fossorial animal () is one that is adapted to digging and which lives primarily (but not solely) underground. Examples of fossorial vertebrates are Mole (animal), moles, badgers, naked mole-rats, meerkats, armadillos, wombats, and mole salamand ...
rodents bang their head against the walls of their tunnels. This behavior was initially interpreted as part of their tunnel building behavior, but it was eventually realized that they generate temporally patterned seismic signals for long-distance communication with neighboring mole rats.
Footdrumming is used widely as a predator warning or defensive action. It is used primarily by fossorial or semi-fossorial rodents. The banner-tailed kangaroo rat produces several complex footdrumming patterns in a number of different contexts, one of which is when it encounters a snake. The footdrumming may alert nearby offspring but most likely conveys that the rat is too alert for a successful attack, thus preventing the snake's predatory pursuit. Several studies have indicated intentional use of ground vibrations as a means of intra-specific communication during courtship among the Cape mole rat. Footdrumming has been reported to be involved in male-male competition; the dominant male indicates its resource holding potential by drumming, thus minimizing physical contact with potential rivals.
Mating strategies
 Some species of rodent are monogamous, with an adult male and female forming a lasting
Some species of rodent are monogamous, with an adult male and female forming a lasting pair bond
In biology, a pair bond is the strong affinity that develops in some species between a mating pair, often leading to the production and rearing of young and potentially a lifelong bond. Pair-bonding is a term coined in the 1940s that is frequently ...
. Monogamy can come in two forms; obligate and facultative. In obligate monogamy, both parents care for the offspring and play an important part in their survival. This occurs in species such as California mice, oldfield mice, Malagasy giant rats and beavers. In these species, males usually mate only with their partners. In addition to increased care for young, obligate monogamy can also be beneficial to the adult male as it decreases the chances of never finding a mate or mating with an infertile female. In facultative monogamy, the males do not provide direct parental care and stay with one female because they cannot access others due to being spatially dispersed. Prairie voles appear to be an example of this form of monogamy, with males guarding and defending females within their vicinity.
In polygynous species, males will try to monopolize and mate with multiple females. As with monogamy, polygyny in rodents can come in two forms; defense and non-defense. Defense polygyny involves males controlling territories that contain resources that attract females. This occurs in ground squirrels like yellow-bellied marmots, California ground squirrels, Columbian ground squirrels and Richardson's ground squirrel
Richardson's ground squirrel (''Urocitellus richardsonii''), also known as the dakrat or flickertail, is a North American ground squirrel in the genus ''Urocitellus''. Like a number of other ground squirrels, they are sometimes called prairie dog ...
s. Males with territories are known as "resident" males and the females that live within the territories are known as "resident" females. In the case of marmots, resident males do not appear to ever lose their territories and always win encounters with invading males. Some species are also known to directly defend their resident females and the ensuing fights can lead to severe wounding. In species with non-defense polygyny, males are not territorial and wander widely in search of females to monopolize. These males establish dominance hierarchies, with the high-ranking males having access to the most females. This occurs in species like Belding's ground squirrels and some tree squirrel species.
thumbnail, A mating plug in a female Richardson's ground squirrel
Richardson's ground squirrel (''Urocitellus richardsonii''), also known as the dakrat or flickertail, is a North American ground squirrel in the genus ''Urocitellus''. Like a number of other ground squirrels, they are sometimes called prairie dog ...
Promiscuity#Other animals, Promiscuity, in which both males and females mate with multiple partners, also occurs in rodents. In species such as the white-footed mouse, females give birth to litters with multiple paternities. Promiscuity leads to increased sperm competition
Sperm competition is the competitive process between Spermatozoon, spermatozoa of two or more different males to fertility, fertilize the same Egg cell, egg during sexual reproduction. Competition can occur when females have multiple potential m ...
and males tend to have larger testicles. In the Cape ground squirrel, the male's testes can be 20 percent of its head-body length. Several rodent species have flexible mating systems that can vary between monogamy, polygyny and promiscuity.
Female rodents play an active role in choosing their mates. Factors that contribute to female preference may include the size, dominance and spatial ability of the male. In the eusocial naked mole rats, a single female monopolizes mating from at least three males. Reproductively active female naked mole-rats tend to associate with unfamiliar males (generally non-kin), whereas females that are reproductively inactive do not tend to discriminate. The preference of reproductively active females for unfamiliar males is thought to be an adaptation for inbreeding avoidance, since inbreeding ordinarily leads to the expression of recessive deleterious alleles.
In most rodent species, such as brown rats and house mice, ovulation
Ovulation is an important part of the menstrual cycle in female vertebrates where the egg cells are released from the ovaries as part of the ovarian cycle. In female humans ovulation typically occurs near the midpoint in the menstrual cycle and ...
occurs on a regular cycle while in others, such as voles, it is induced by mating. During copulation, males of some rodent species deposit a mating plug in the female's genital opening, both to prevent sperm leakage and to protect against other males inseminating the female. Females can remove the plug and may do so either immediately or after several hours.
Metabolism
Metabolism (, from ''metabolē'', "change") is the set of life-sustaining chemical reactions in organisms. The three main functions of metabolism are: the conversion of the energy in food to energy available to run cellular processes; the co ...
of thyroid hormones and iodine
Iodine is a chemical element; it has symbol I and atomic number 53. The heaviest of the stable halogens, it exists at standard conditions as a semi-lustrous, non-metallic solid that melts to form a deep violet liquid at , and boils to a vi ...
in the mediobasal hypothalamus changes in response to photoperiod. Thyroid hormones in turn induce reproductive changes. This is found by Watanabe et al. 2004 and 2007, Barrett et al. 2007, Freeman et al. 2007, and Herwig et al. 2009 in Siberian hamsters, Revel et al. 2006 and Yasuo et al. 2007 in Syrian hamsters, Yasuo et al. 2007 and Ross et al. 2011 in rats, and Ono et al. 2008 in mice.
Birth and parenting
 Rodents may be born either
Rodents may be born either altricial
Precocial species in birds and mammals are those in which the young are relatively mature and mobile from the moment of birth or hatching. They are normally nidifugous, meaning that they leave the nest shortly after birth or hatching. Altricial ...
(blind, hairless and relatively underdeveloped) or precocial
Precocial species in birds and mammals are those in which the young are relatively mature and mobile from the moment of birth or hatching. They are normally nidifugous, meaning that they leave the nest shortly after birth or hatching. Altricial ...
(mostly furred, eyes open and fairly developed) depending on the species. The altricial state is typical for squirrels and mice, while the precocial state usually occurs in species like guinea pigs and porcupines. Females with altricial young typically build elaborate nests before they give birth and maintain them until their offspring are weaned. The female gives birth sitting or lying down and the young emerge in the direction she is facing. The newborns first venture out of the nest a few days after they have opened their eyes and initially keep returning regularly. As they get older and more developed, they visit the nest less often and leave permanently when weaned.
In precocial species, the mothers invest little in nest building and some do not build nests at all. The female gives birth standing and the young emerge behind her. Mothers of these species maintain contact with their highly mobile young with maternal contact calls. Though relatively independent and weaned within days, precocial young may continue to nurse and be groomed by their mothers. Rodent litter sizes also vary and females with smaller litters spend more time in the nest than those with larger litters.
 Mother rodents provide both direct parental care, such as nursing, grooming, retrieving and huddling, and indirect parenting, such as food caching, nest building and protection to their offspring. In many social species, young may be cared for by individuals other than their parents, a practice known as alloparenting or cooperative breeding. This is known to occur in black-tailed prairie dogs and Belding's ground squirrels, where mothers have communal nests and nurse unrelated young along with their own. There is some question as to whether these mothers can distinguish which young are theirs. In the Patagonian mara, young are also placed in communal warrens, but mothers do not permit youngsters other than their own to nurse.
Infanticide exists in numerous rodent species and may be practiced by adult conspecifics of either sex. Several reasons have been proposed for this behavior, including nutritional stress, resource competition, avoiding misdirecting parental care and, in the case of males, attempting to make the mother sexually receptive. The latter reason is well supported in
Mother rodents provide both direct parental care, such as nursing, grooming, retrieving and huddling, and indirect parenting, such as food caching, nest building and protection to their offspring. In many social species, young may be cared for by individuals other than their parents, a practice known as alloparenting or cooperative breeding. This is known to occur in black-tailed prairie dogs and Belding's ground squirrels, where mothers have communal nests and nurse unrelated young along with their own. There is some question as to whether these mothers can distinguish which young are theirs. In the Patagonian mara, young are also placed in communal warrens, but mothers do not permit youngsters other than their own to nurse.
Infanticide exists in numerous rodent species and may be practiced by adult conspecifics of either sex. Several reasons have been proposed for this behavior, including nutritional stress, resource competition, avoiding misdirecting parental care and, in the case of males, attempting to make the mother sexually receptive. The latter reason is well supported in primate
Primates is an order (biology), order of mammals, which is further divided into the Strepsirrhini, strepsirrhines, which include lemurs, galagos, and Lorisidae, lorisids; and the Haplorhini, haplorhines, which include Tarsiiformes, tarsiers a ...
s and lion
The lion (''Panthera leo'') is a large Felidae, cat of the genus ''Panthera'', native to Sub-Saharan Africa and India. It has a muscular, broad-chested body (biology), body; a short, rounded head; round ears; and a dark, hairy tuft at the ...
s but less so in rodents. Infanticide appears to be widespread in black-tailed prairie dogs, including infanticide from invading males and immigrant females, as well as occasional cannibalism of an individual's own offspring. To protect against infanticide from other adults, female rodents may employ avoidance or direct aggression against potential perpetrators, multiple mating, territoriality or early termination of pregnancy. Feticide
Foeticide (British English), or feticide (North American English), is the act of killing a fetus, or causing a miscarriage. Definitions differ between legal and medical applications, whereas in law, feticide frequently refers to a criminal offe ...
can also occur among rodents; in alpine marmots, dominant females tend to suppress the reproduction of subordinates by being antagonistic towards them while they are pregnant. The resulting stress causes the fetuses to abort.
Intelligence
 Rodents have advanced
Rodents have advanced cognitive
Cognition is the "mental action or process of acquiring knowledge and understanding through thought, experience, and the senses". It encompasses all aspects of intellectual functions and processes such as: perception, attention, thought, ...
abilities. They can quickly learn to avoid poisoned baits, which makes them difficult pests to deal with. Guinea pigs can learn and remember complex pathways to food. Squirrels and kangaroo rats are able to locate caches of food by spatial memory
In cognitive psychology and neuroscience, spatial memory is a form of memory responsible for the recording and recovery of information needed to plan a course to a location and to recall the location of an object or the occurrence of an event. Sp ...
, rather than just by smell.
Because laboratory mice (house mice) and rats (brown rats) are widely used as scientific models to further our understanding of biology, a great deal has come to be known about their cognitive capacities. Brown rats exhibit cognitive bias
A cognitive bias is a systematic pattern of deviation from norm (philosophy), norm or rationality in judgment. Individuals create their own "subjective reality" from their perception of the input. An individual's construction of reality, not the ...
, where information processing is biased by whether they are in a positive or negative affective state. For example, laboratory rats trained to respond to a specific tone by pressing a lever to receive a reward, and to press another lever in response to a different tone so as to avoid receiving an electric shock, are more likely to respond to an intermediate tone by choosing the reward lever if they have just been tickled (something they enjoy), indicating "a link between the directly measured positive affective state and decision making under uncertainty in an animal model."
Laboratory (brown) rats may have the capacity for metacognition
Metacognition is an awareness of one's thought processes and an understanding of the patterns behind them. The term comes from the root word ''Meta (prefix), meta'', meaning "beyond", or "on top of".Metcalfe, J., & Shimamura, A. P. (1994). ''Metac ...
—to consider their own learning and then make decisions based on what they know, or do not know, as indicated by choices they make apparently trading off difficulty of tasks and expected rewards, making them the first animals other than primates known to have this capacity, but these findings are disputed, since the rats may have been following simple operant conditioning
Operant conditioning, also called instrumental conditioning, is a learning process in which voluntary behaviors are modified by association with the addition (or removal) of reward or aversive stimuli. The frequency or duration of the behavior ma ...
principles, or a behavioral economic model. Brown rats use social learning in a wide range of situations, but perhaps especially so in acquiring food preferences.
Evolutionary history
Dentition
Dentition pertains to the development of teeth and their arrangement in the mouth. In particular, it is the characteristic arrangement, kind, and number of teeth in a given species at a given age. That is, the number, type, and morpho-physiology ...
is the key feature by which fossil
A fossil (from Classical Latin , ) is any preserved remains, impression, or trace of any once-living thing from a past geological age. Examples include bones, shells, exoskeletons, stone imprints of animals or microbes, objects preserve ...
rodents are recognized and the earliest record of such mammals comes from the Paleocene
The Paleocene ( ), or Palaeocene, is a geological epoch (geology), epoch that lasted from about 66 to 56 mya (unit), million years ago (mya). It is the first epoch of the Paleogene Period (geology), Period in the modern Cenozoic Era (geology), ...
, shortly after the extinction of the non-avian dinosaur
Dinosaurs are a diverse group of reptiles of the clade Dinosauria. They first appeared during the Triassic Geological period, period, between 243 and 233.23 million years ago (mya), although the exact origin and timing of the #Evolutio ...
s some 66 million years ago. These fossils are found in Laurasia
Laurasia () was the more northern of two large landmasses that formed part of the Pangaea supercontinent from around ( Mya), the other being Gondwana. It separated from Gondwana (beginning in the late Triassic period) during the breakup of Pa ...
, the supercontinent composed of modern-day North America, Europe, and Asia. The divergence of Glires, a clade consisting of rodents and lagomorphs (rabbits, hares and pikas), from other placental mammals occurred within a few million years after the Cretaceous-Paleogene boundary; rodents and lagomorphs then radiated during the Cenozoic
The Cenozoic Era ( ; ) is Earth's current geological era, representing the last 66million years of Earth's history. It is characterized by the dominance of mammals, insects, birds and angiosperms (flowering plants). It is the latest of three g ...
. Some molecular clock data suggest modern rodents (members of the order Rodentia) had appeared by the late Cretaceous
The Cretaceous ( ) is a geological period that lasted from about 143.1 to 66 mya (unit), million years ago (Mya). It is the third and final period of the Mesozoic Era (geology), Era, as well as the longest. At around 77.1 million years, it is the ...
, although other molecular divergence estimations are in agreement with the fossil record.
Rodents are thought to have evolved in Asia, where local multituberculate faunas were severely affected by the Cretaceous–Paleogene extinction event
The Cretaceous–Paleogene (K–Pg) extinction event, also known as the K–T extinction, was the extinction event, mass extinction of three-quarters of the plant and animal species on Earth approximately 66 million years ago. The event cau ...
and never fully recovered, unlike their North American and European relatives. In the resulting ecological vacuum, rodents and other Glires were able to evolve and diversify, taking the niches left by extinct multituberculates. The correlation between the spread of rodents and the demise of multituberculates is a controversial topic, not fully resolved. American and European multituberculate assemblages do decline in diversity in correlation with the introduction of rodents in these areas, but the remaining Asian multituberculates co-existed with rodents with no observable replacement taking place, and ultimately both clades co-existed for at least 15 million years.
The history of the colonization of the world's continents by rodents is complex. The movements of the large superfamily Muroidea
The Muroidea are a large Taxonomic rank, superfamily of rodents, including mice, rats, voles, hamsters, lemmings, Gerbillinae, gerbils, and many other relatives. Although the Muroidea originated in Eurasia, they occupy a vast variety of habitat ...
(including hamsters, gerbils, true mice
A mouse (: mice) is a small rodent. Characteristically, mice are known to have a pointed snout, small rounded ears, a body-length scaly tail, and a high breeding rate. The best known mouse species is the common house mouse (''Mus musculus' ...
and rats) may have involved up to seven colonizations of Africa, five of North America, four of Southeast Asia, two of South America and up to ten of Eurasia.
 During the
During the Eocene
The Eocene ( ) is a geological epoch (geology), epoch that lasted from about 56 to 33.9 million years ago (Ma). It is the second epoch of the Paleogene Period (geology), Period in the modern Cenozoic Era (geology), Era. The name ''Eocene'' comes ...
, rodents began to diversify. Beavers appeared in Eurasia in the late Eocene before spreading to North America in the late Miocene. Late in the Eocene, hystricognaths invaded Africa, most probably having originated in Asia at least 39.5 million years ago. From Africa, fossil evidence shows that some hystricognaths ( caviomorphs) colonized South America, which was an isolated continent at the time, evidently making use of ocean currents to cross the Atlantic on floating debris. Caviomorphs had arrived in South America by 41 million years ago (implying a date at least as early as this for hystricognaths in Africa), and had reached the Greater Antilles by the early Oligocene
The Oligocene ( ) is a geologic epoch (geology), epoch of the Paleogene Geologic time scale, Period that extends from about 33.9 million to 23 million years before the present ( to ). As with other older geologic periods, the rock beds that defin ...
, suggesting that they must have dispersed rapidly across South America.
Nesomyid rodents are thought to have rafted from Africa to Madagascar 20–24 million years ago. All 27 species of native Malagasy rodents appear to be descendants of a single colonization event.
By 20 million years ago, fossils recognizably belonging to the current families such as Muridae had emerged. By the Miocene
The Miocene ( ) is the first epoch (geology), geological epoch of the Neogene Period and extends from about (Ma). The Miocene was named by Scottish geologist Charles Lyell; the name comes from the Greek words (', "less") and (', "new") and mea ...
, when Africa had collided with Asia, African rodents such as the porcupine
Porcupines are large rodents with coats of sharp Spine (zoology), spines, or quills, that protect them against predation. The term covers two Family (biology), families of animals: the Old World porcupines of the family Hystricidae, and the New ...
began to spread into Eurasia
Eurasia ( , ) is a continental area on Earth, comprising all of Europe and Asia. According to some geographers, Physical geography, physiographically, Eurasia is a single supercontinent. The concept of Europe and Asia as distinct continents d ...
. Some fossil species were very large in comparison to modern rodents and included the giant beaver, '' Castoroides ohioensis'', which grew to a length of and weight of . The largest known rodent was '' Josephoartigasia monesi'', a pacarana with an estimated body length of 3 m (10 ft).
The first rodents arrived in Australia via Indonesia around 5 million years ago. Although marsupial
Marsupials are a diverse group of mammals belonging to the infraclass Marsupialia. They are natively found in Australasia, Wallacea, and the Americas. One of marsupials' unique features is their reproductive strategy: the young are born in a r ...
s are the most prominent mammals in Australia, many rodents, all belonging to the subfamily Murinae, are among the continent's mammal species. There are about fifty species of 'old endemics', the first wave of rodents to colonize the country in the Miocene and early Pliocene
The Pliocene ( ; also Pleiocene) is the epoch (geology), epoch in the geologic time scale that extends from 5.33 to 2.58Rattus
''Rattus'' is a genus of muroid rodents, all typically called rats. However, the term rat can also be applied to rodent species outside of this genus.
Species and description
The best-known ''Rattus'' species are the black rat (''R. rattus'') ...
'') species of 'new endemics', arriving in a subsequent wave in the late Pliocene or early Pleistocene
The Pleistocene ( ; referred to colloquially as the ''ice age, Ice Age'') is the geological epoch (geology), epoch that lasted from to 11,700 years ago, spanning the Earth's most recent period of repeated glaciations. Before a change was fin ...
. The earliest fossil rodents in Australia have a maximum age of 4.5 million years, and molecular data is consistent with the colonization of New Guinea from the west during the late Miocene or early Pliocene followed by rapid diversification. A further wave of adaptive radiation occurred after one or more colonizations of Australia some 2 to 3 million years later.
Rodents participated in the Great American Interchange
The Great American Biotic Interchange (commonly abbreviated as GABI), also known as the Great American Interchange and the Great American Faunal Interchange, was an important late Cenozoic paleozoogeographic biotic interchange event in which land ...
that resulted from the joining of the Americas by formation of the Isthmus of Panama
The Isthmus of Panama, historically known as the Isthmus of Darien, is the narrow strip of land that lies between the Caribbean Sea and the Pacific Ocean, linking North America, North and South America. The country of Panama is located on the i ...
, around 3 million years ago in the Piacenzian
The Piacenzian is in the international geologic time scale the upper stage (stratigraphy), stage or latest age (geology), age of the Pliocene. It spans the time between 3.6 ± 0.005 year#SI prefix multipliers, Ma and 2.58 Ma (million years ago). T ...
age. In this exchange, a small number of species such as the New World porcupines (Erethizontidae) headed north. However, the main southward invasion of sigmodontines preceded formation of the land bridge by at least several million years, probably occurring via rafting. Sigmodontines diversified explosively once in South America, although some degree of diversification may have already occurred in Central America before the colonization.
Classification
The use of the order name "Rodentia" is attributed to the English traveler and naturalist Thomas Edward Bowdich (1821). The Modern Latin word is derived from , present participle of – "to gnaw", "eat away". Thehare
Hares and jackrabbits are mammals belonging to the genus ''Lepus''. They are herbivores and live Solitary animal, solitarily or in pairs. They nest in slight depressions called forms, and their young are precociality, able to fend for themselves ...
s, rabbit
Rabbits are small mammals in the family Leporidae (which also includes the hares), which is in the order Lagomorpha (which also includes pikas). They are familiar throughout the world as a small herbivore, a prey animal, a domesticated ...
s and pikas (order Lagomorpha) have continuously growing incisors, as do rodents, and were at one time included in the order. However, they have an additional pair of incisors in the upper jaw and the two orders have quite separate evolutionary histories. The phylogeny of the rodents places them in the clade
In biology, a clade (), also known as a Monophyly, monophyletic group or natural group, is a group of organisms that is composed of a common ancestor and all of its descendants. Clades are the fundamental unit of cladistics, a modern approach t ...
s Glires, Euarchontoglires and Boreoeutheria
Boreoeutheria (, "northern eutherians") is a magnorder of Placentalia, placental mammals that groups together superorders Euarchontoglires and Laurasiatheria. The clade includes groups as diverse as giraffes, Sus (genus), pigs, zebras, Rhinoceros ...
. The cladogram below shows the inner and outer relations of Rodentia based on a 2012 attempt by Wu et al. to align the molecular clock with paleontological data:
The living rodent families based on the study done by Fabre et al. 2012.
The order Rodentia may be divided into suborders, infraorder
Order () is one of the eight major hierarchical taxonomic ranks in Linnaean taxonomy. It is classified between Family_(biology), family and Class_(biology), class. In biological classification, the order is a taxonomic rank used in the classific ...
s, superfamilies and families. There is a great deal of parallelism and convergence
Convergence may refer to:
Arts and media Literature
*''Convergence'' (book series), edited by Ruth Nanda Anshen
*Convergence (comics), "Convergence" (comics), two separate story lines published by DC Comics:
**A four-part crossover storyline that ...
among rodents caused by the fact that they have tended to evolve to fill largely similar niches. This parallel evolution includes not only the structure of the teeth, but also the infraorbital region of the skull (below the eye socket) and makes classification difficult as similar traits may not be due to common ancestry. Brandt (1855) was the first to propose dividing Rodentia into three suborders, Sciuromorpha, Hystricomorpha and Myomorpha, based on the development of certain muscles in the jaw and this system was widely accepted. Schlosser (1884) performed a comprehensive review of rodent fossils, mainly using the cheek teeth, and found that they fitted into the classical system, but Tullborg (1899) proposed just two sub-orders, Sciurognathi and Hystricognathi. These were based on the degree of inflection of the lower jaw and were to be further subdivided into Sciuromorpha, Myomorpha, Hystricomorpha and Bathyergomorpha. Matthew (1910) created a phylogenetic tree
A phylogenetic tree or phylogeny is a graphical representation which shows the evolutionary history between a set of species or taxa during a specific time.Felsenstein J. (2004). ''Inferring Phylogenies'' Sinauer Associates: Sunderland, MA. In ...
of New World rodents but did not include the more problematic Old World species. Further attempts at classification continued without agreement, with some authors adopting the classical three suborder system and others Tullborg's two suborders.
These disagreements remain unresolved, nor have molecular studies fully resolved the situation though they have confirmed the monophyly
In biological cladistics for the classification of organisms, monophyly is the condition of a taxonomic grouping being a clade – that is, a grouping of organisms which meets these criteria:
# the grouping contains its own most recent comm ...
of the group and that the clade has descended from a common Paleocene ancestor. Carleton and Musser (2005) in ''Mammal Species of the World'' have provisionally adopted a five suborder system: Sciuromorpha, Castorimorpha, Myomorpha, Anomaluromorpha, and Hystricomorpha. As of 2021 the American Society of Mammalogists recognizes 34 recent families containing more than 481 genera and 2277 species.  Order Rodentia (from Latin, ''rodere'', to gnaw)
Order Rodentia (from Latin, ''rodere'', to gnaw)

 *Suborder Anomaluromorpha
*::*Family Anomaluridae: scaly-tailed squirrels
*::*Family Pedetidae: springhares
*::*Family Zenkerellidae: Cameroon scaly-tail
*Suborder Castorimorpha
*:*Superfamily Castoroidea
*:** Family
*Suborder Anomaluromorpha
*::*Family Anomaluridae: scaly-tailed squirrels
*::*Family Pedetidae: springhares
*::*Family Zenkerellidae: Cameroon scaly-tail
*Suborder Castorimorpha
*:*Superfamily Castoroidea
*:** Family Castoridae
Castoridae is a family of rodents that contains the two living species of beavers and their fossil relatives. A formerly diverse group, only a single genus is extant today, ''beaver, Castor.'' Two other genera of "giant beavers", ''Castoroides'' ...
: beaver
Beavers (genus ''Castor'') are large, semiaquatic rodents of the Northern Hemisphere. There are two existing species: the North American beaver (''Castor canadensis'') and the Eurasian beaver (''C. fiber''). Beavers are the second-large ...
s
*:*Superfamily Geomyoidea
*:**Family Geomyidae: pocket gophers (true gophers)
*:**Family Heteromyidae: kangaroo rats, kangaroo mice
*Suborder Hystricomorpha
**Infraorder Ctenodactylomorphi
**:*Family Ctenodactylidae: gundis
**:*Family Diatomyidae: Laotian rock rat
**Infraorder Hystricognathi
***Parvorder Phiomorpha
**:*Family Bathyergidae: African mole rats
**:*Family Heterocephalidae: naked mole-rat
**:*Family Hystricidae: Old World porcupines
**:*Family Petromuridae: dassie rat
**:*Family Thryonomyidae: cane rats
***Parvorder Caviomorpha
****Superfamily Erethizontoidea
*****Family Erethizontidae: New World porcupines
****Superfamily Chinchilloidea
*****Family Chinchillidae
The family Chinchillidae is in the order Rodentia and consists of the chinchillas, the viscachas, and their fossil relatives. This family is restricted to southern and western South America, mostly living in mountainous regions of the Andes, ex ...
: chinchillas, viscachas
*****Family Dinomyidae: pacaranas
****Superfamily Cavioidea
*****Family Caviidae: cavies, including guinea pig
The guinea pig or domestic guinea pig (''Cavia porcellus''), also known as the cavy or domestic cavy ( ), is a species of rodent belonging to the genus ''Cavia'', family Caviidae. Animal fancy, Breeders tend to use the name "cavy" for the ani ...
s and the capybara
The capybara or greater capybara (''Hydrochoerus hydrochaeris'') is the largest living rodent, native to South America. It is a member of the genus '' Hydrochoerus''. The only other extant member is the lesser capybara (''Hydrochoerus isthmi ...
*****Family Dasyproctidae: agoutis
*****Family Cuniculidae: pacas
****Superfamily Octodontoidea
Caviomorpha is the rodent parvorder that unites all New World hystricognaths. It is supported by both fossil and molecular phylogeny, molecular evidence. The Caviomorpha was for a time considered to be a separate order outside the Rodentia, but i ...
*****Family Abrocomidae: chinchilla rats
*****Family Ctenomyidae: tuco-tucos
*****Family Echimyidae: spiny rats, hutias, and nutria
The nutria () or coypu () (''Myocastor coypus'') is a herbivore, herbivorous, semiaquatic rodent from South America.
Classified for a long time as the only member of the family Myocastoridae, ''Myocastor'' has since been included within Echimy ...
*****Family Octodontidae: octodonts
*Suborder Myomorpha
*:*Superfamily Dipodoidea
Dipodoidea is a Superfamily (biology), superfamily of Rodent, rodents, also known as dipodoids, found across the Northern Hemisphere. This superfamily includes over 50 species among the 16 genera in 3 families. They include the Jerboa, jerboas (f ...
*:**Family Dipodidae
Jerboas () are the members of the family Dipodidae. They are hopping desert rodents found throughout North Africa and Asia. They tend to live in hot deserts.
When chased, jerboas can run at up to . Some species are preyed on by little owls ('' ...
: jerboas
*:**Family Sminthidae
Sminthidae is a family of mouse-like jumping rodents. They are represented by only one extant genus, '' Sicista'', represented by 19 species found throughout most of Eurasia, from central Europe east to Siberia, and south to southern China. How ...
: birch mice
*:**Family Zapodidae: jumping mice
*:*Superfamily Muroidea
The Muroidea are a large Taxonomic rank, superfamily of rodents, including mice, rats, voles, hamsters, lemmings, Gerbillinae, gerbils, and many other relatives. Although the Muroidea originated in Eurasia, they occupy a vast variety of habitat ...
*:**Family Calomyscidae: mouse-like hamsters
*:**Family Cricetidae
The Cricetidae are a family of rodents in the large and complex superfamily Muroidea. It includes true hamsters, voles, lemmings, muskrats, and New World rats and mice. At over 870 species, it is either the largest or second-largest family ...
: hamsters, New World rats and mice
The New World rats and mice are a group of related rodents found in North and South America. They are extremely diverse in appearance and ecology, ranging from the tiny '' Baiomys'' to the large '' Kunsia''. They represent one of the few example ...
, muskrats, vole
Voles are small rodents that are relatives of lemmings and hamsters, but with a stouter body; a longer, hairy tail; a slightly rounder head; smaller eyes and ears; and differently formed molars (high-crowned with angular cusps instead of lo ...
s, lemmings
*:**Family Muridae
The Muridae, or murids, are either the largest or second-largest family of rodents and of mammals, containing approximately 870 species, including many species of mice, rats, and gerbils found naturally throughout Eurasia, Africa, and Australia.
...
: true mice and rats, gerbils, spiny mice, crested rat
The maned rat or (African) crested rat (''Lophiomys imhausi'') is a nocturnal, long-haired and bushy-tailed East African rodent that superficially resembles a porcupine. The world's only known poisonous rodent, the maned rat sequesters toxins fro ...
*:**Family Nesomyidae: climbing mice, rock mice, white-tailed rat, Malagasy rats and mice
*:**Family Platacanthomyidae: spiny dormice
*:**Family Spalacidae: mole rats, bamboo rats, zokors
*Suborder Sciuromorpha
Sciuromorpha ( 'squirrel-like') is a rodent Order (biology), suborder that includes several rodent Family (biology), families. It includes all members of the Sciuridae (the squirrel family) as well as the mountain beaver species.
Traditionally, ...
*::*Family Aplodontiidae: mountain beaver
The mountain beaver (''Aplodontia rufa'')Other names include boomer, mountain boomer, ground bear, giant mole, gehalis, sewellel, suwellel, showhurll, showtl, and showte, as well as a number of other Native American terms. "Mountain beaver" is a ...
*::*Family Gliridae (also Myoxidae, Muscardinidae): dormice
*::*Family Sciuridae
Squirrels are members of the family Sciuridae (), a family that includes small or medium-sized rodents. The squirrel family includes tree squirrels, ground squirrels (including chipmunks and prairie dogs, among others), and flying squirrel ...
: squirrels, including chipmunks, prairie dogs, marmots
Interaction with humans
Conservation
While rodents are not the most seriously threatened order of mammals, there are 168 species in 126 genera that are said to warrant conservation attention in the face of limited appreciation by the public. Since 76 percent of rodent genera contain only one species, much phylogenetic diversity could be lost with a comparatively small number of extinctions. In the absence of more detailed knowledge of species at risk and accurate taxonomy, conservation must be based mainly on higher taxa (such as families rather than species) and geographical hot spots. Several species of rice rat have become extinct since the 19th century, probably through habitat loss and the introduction of alien species. In Colombia, the brown hairy dwarf porcupine was recorded from only two mountain localities in the 1920s, while the red crested soft-furred spiny rat is known only from its type locality on the Caribbean coast, so these species are considered vulnerable. The IUCN Species Survival Commission writes "We can safely conclude that many South American rodents are seriously threatened, mainly by environmental disturbance and intensive hunting". The "three now cosmopolitan commensal rodent pest species" 3–5 March 1992 (the brown rat, theblack rat
The black rat (''Rattus rattus''), also known as the roof rat, ship rat, or house rat, is a common long-tailed rodent of the stereotypical rat genus ''Rattus'', in the subfamily Murinae. It likely originated in the Indian subcontinent, but is n ...
and the house mouse) have been dispersed in association with humans, partly on sailing ships in the Age of Exploration
The Age of Discovery (), also known as the Age of Exploration, was part of the early modern period and overlapped with the Age of Sail. It was a period from approximately the 15th to the 17th century, during which Seamanship, seafarers fro ...
, and with a fourth species in the Pacific, the Polynesian rat (''Rattus exulans''), have severely damaged island biotas around the world. For example, when the black rat reached Lord Howe Island
Lord Howe Island (; formerly Lord Howe's Island) is an irregularly crescent-shaped volcanic remnant in the Tasman Sea between Australia and New Zealand, part of the Australian state of New South Wales. It lies directly east of mainland Port ...
in 1918, over 40 percent of the terrestrial bird species of the island, including the Lord Howe fantail, became extinct within ten years. Similar destruction has been seen on Midway Island (1943) and Big South Cape Island (1962). Conservation projects can with careful planning completely eradicate these pest rodents from islands using an anticoagulant
An anticoagulant, commonly known as a blood thinner, is a chemical substance that prevents or reduces the coagulation of blood, prolonging the clotting time. Some occur naturally in blood-eating animals, such as leeches and mosquitoes, which ...
rodenticide such as brodifacoum. This approach has been successful on the island of Lundy in the United Kingdom, where the eradication of an estimated 40,000 brown rats is giving populations of Manx shearwater and Atlantic puffin
The Atlantic puffin ('), also known as the common puffin, is a species of seabird in the auk family (biology), family. It is the only puffin native to the Atlantic Ocean; two related species, the tufted puffin and the horned puffin being found ...
a chance to recover from near-extinction.
Rodents have also been susceptible to climate change
Present-day climate change includes both global warming—the ongoing increase in Global surface temperature, global average temperature—and its wider effects on Earth's climate system. Climate variability and change, Climate change in ...
, especially species living on low-lying islands. The Bramble Cay melomys, which lived in the northernmost point of land of Australia
Australia, officially the Commonwealth of Australia, is a country comprising mainland Australia, the mainland of the Australia (continent), Australian continent, the island of Tasmania and list of islands of Australia, numerous smaller isl ...
, was the first mammal species to be declared extinct as a consequence of human-caused climate change.
Exploitation
Fur
 Humanity has long used animal skins for clothing, as the leather is durable and the fur provides extra insulation. The native people of North America made much use of beaver pelts, tanning and sewing them together to make robes. Europeans appreciated the quality of these and the North American fur trade developed and became of prime importance to early settlers. In Europe, the soft underfur known as "beaver wool" was found to be ideal for felting and was made into beaver hats and trimming for clothing. Later, the coypu took over as a cheaper source of fur for felting and was farmed extensively in America and Europe; however, fashions changed, new materials became available and this area of the animal fur industry declined. The chinchilla has a soft and silky coat and the demand for its fur was so high that it was nearly wiped out in the wild before farming took over as the main source of pelts. The quills and guardhairs of porcupines are used for traditional decorative clothing. For example, their guardhairs are used in the creation of the Native American "porky roach" headdress. The main quills may be dyed, and then applied in combination with thread to embellish leather accessories such as knife sheaths and leather bags.
Humanity has long used animal skins for clothing, as the leather is durable and the fur provides extra insulation. The native people of North America made much use of beaver pelts, tanning and sewing them together to make robes. Europeans appreciated the quality of these and the North American fur trade developed and became of prime importance to early settlers. In Europe, the soft underfur known as "beaver wool" was found to be ideal for felting and was made into beaver hats and trimming for clothing. Later, the coypu took over as a cheaper source of fur for felting and was farmed extensively in America and Europe; however, fashions changed, new materials became available and this area of the animal fur industry declined. The chinchilla has a soft and silky coat and the demand for its fur was so high that it was nearly wiped out in the wild before farming took over as the main source of pelts. The quills and guardhairs of porcupines are used for traditional decorative clothing. For example, their guardhairs are used in the creation of the Native American "porky roach" headdress. The main quills may be dyed, and then applied in combination with thread to embellish leather accessories such as knife sheaths and leather bags. Lakota
Lakota may refer to:
*Lakota people, a confederation of seven related Native American tribes
*Lakota language
Lakota ( ), also referred to as Lakhota, Teton or Teton Sioux, is a Siouan languages, Siouan language spoken by the Lakota people of ...
women would harvest the quills for quillwork
Quillwork is a form of textile embellishment traditionally practiced by Indigenous peoples of the Americas, Indigenous peoples of North America that employs the Spine (zoology), quills of porcupines as an aesthetic element. Quills from bird feathe ...
by throwing a blanket over a porcupine and retrieving the quills it left stuck in the blanket.
Consumption
At least 89 species of rodent, mostly Hystricomorpha such as guinea pigs, agoutis and capybaras, are eaten by humans; in 1985, there were at least 42 different societies in which people eat rats. Guinea pigs were first raised for food around 2500 B.C. and by 1500 B.C. had become the main source of meat for theInca Empire
The Inca Empire, officially known as the Realm of the Four Parts (, ), was the largest empire in pre-Columbian America. The administrative, political, and military center of the empire was in the city of Cusco. The History of the Incas, Inca ...
. Dormice were raised by the Romans in special pots called "gliraria", or in large outdoor enclosures, where they were fattened on walnuts, chestnuts, and acorns. The dormice were also caught from the wild in autumn when they were fattest, and either roasted and dipped into honey or baked while stuffed with a mixture of pork, pine nuts, and other flavorings. Researchers found that in Amazonia, where large mammals were scarce, pacas and common agoutis accounted for around 40 percent of the annual game taken by the indigenous people, but in forested areas where larger mammals were abundant, these rodents constituted only about 3 percent of the take.
Guinea pigs are used in the cuisine of Cuzco, Peru, in dishes such as ''cuy al horno'', baked guinea pig. The traditional Andean stove, known as a ''qoncha'' or a ''fogón'', is made from mud and clay reinforced with straw and hair from animals such as guinea pigs. In Peru, there are at any time 20 million domestic guinea pigs, which annually produce 64 million edible carcasses. This animal is an excellent food source since the flesh is 19% protein. In the United States, mostly squirrels, but also muskrats, porcupines, and groundhogs are eaten by humans. The Navajo people ate prairie dog baked in mud, while the Paiute ate gophers, squirrels, and rats.
Animal testing
 Rodents are used widely as
Rodents are used widely as model organism
A model organism is a non-human species that is extensively studied to understand particular biological phenomena, with the expectation that discoveries made in the model organism will provide insight into the workings of other organisms. Mo ...
s in animal testing. Albino mutant rats were first used for research in 1828 and later became the first animal domesticated for purely scientific purposes. Nowadays, the house mouse is the most commonly used laboratory rodent, and in 1979 it was estimated that fifty million were used annually worldwide. They are favored because of their small size, fertility, short gestation period and ease of handling and because they are susceptible to many of the conditions and infections that afflict humans. They are used in research into genetics
Genetics is the study of genes, genetic variation, and heredity in organisms.Hartl D, Jones E (2005) It is an important branch in biology because heredity is vital to organisms' evolution. Gregor Mendel, a Moravian Augustinians, Augustinian ...
, developmental biology
Developmental biology is the study of the process by which animals and plants grow and develop. Developmental biology also encompasses the biology of Regeneration (biology), regeneration, asexual reproduction, metamorphosis, and the growth and di ...
, cell biology
Cell biology (also cellular biology or cytology) is a branch of biology that studies the structure, function, and behavior of cells. All living organisms are made of cells. A cell is the basic unit of life that is responsible for the living an ...
, oncology
Oncology is a branch of medicine that deals with the study, treatment, diagnosis, and prevention of cancer. A medical professional who practices oncology is an ''oncologist''. The name's Etymology, etymological origin is the Greek word ὄγ ...
and immunology
Immunology is a branch of biology and medicine that covers the study of Immune system, immune systems in all Organism, organisms.
Immunology charts, measures, and contextualizes the Physiology, physiological functioning of the immune system in ...
. Guinea pigs were popular laboratory animals until the late 20th century; about 2.5 million guinea pigs were used annually in the United States for research in the 1960s, but that total decreased to about 375,000 by the mid-1990s. In 2007, they constituted about 2% of all laboratory animals. Guinea pigs played a major role in the establishment of germ theory in the late 19th century, through the experiments of Louis Pasteur
Louis Pasteur (, ; 27 December 1822 – 28 September 1895) was a French chemist, pharmacist, and microbiologist renowned for his discoveries of the principles of vaccination, Fermentation, microbial fermentation, and pasteurization, the la ...
, Émile Roux, and Robert Koch. They have been launched into orbital space flight several times—first by the USSR
The Union of Soviet Socialist Republics. (USSR), commonly known as the Soviet Union, was a List of former transcontinental countries#Since 1700, transcontinental country that spanned much of Eurasia from 1922 until Dissolution of the Soviet ...
on the Sputnik 9 biosatellite of 9 March 1961, with a successful recovery. The naked mole rat is the only known mammal that is poikilothermic; it is used in studies on thermoregulation
Thermoregulation is the ability of an organism to keep its body temperature within certain boundaries, even when the surrounding temperature is very different. A thermoconforming organism, by contrast, simply adopts the surrounding temperature ...
. It is also unusual in not producing the neurotransmitter substance P, a fact which researchers find useful in studies on pain
Pain is a distressing feeling often caused by intense or damaging Stimulus (physiology), stimuli. The International Association for the Study of Pain defines pain as "an unpleasant sense, sensory and emotional experience associated with, or res ...
.
Rodents have sensitive olfactory abilities, which have been used by humans to detect odors or chemicals of interest. The Gambian pouched rat is able to detect tuberculosis
Tuberculosis (TB), also known colloquially as the "white death", or historically as consumption, is a contagious disease usually caused by ''Mycobacterium tuberculosis'' (MTB) bacteria. Tuberculosis generally affects the lungs, but it can al ...
bacilli with a sensitivity of up to 86.6%, and specificity (detecting the absence of the bacilli) of over 93%; the same species has been trained to detect land mine
A land mine, or landmine, is an explosive weapon often concealed under or camouflaged on the ground, and designed to destroy or disable enemy targets as they pass over or near it. Land mines are divided into two types: anti-tank mines, wh ...
s. Rats have been studied for possible use in hazardous situations such as in disaster zones. They can be trained to respond to commands, which may be given remotely, and even persuaded to venture into brightly lit areas, which rats usually avoid.
As pets
 Rodents including guinea pigs, mice, rats, hamsters, gerbils, chinchillas, degus and chipmunks make convenient pets able to live in small spaces, each species with its own qualities. Most are normally kept in cages of suitable sizes and have varied requirements for space and social interaction. If handled from a young age, they are usually docile and do not bite. Guinea pigs have a long lifespan and need a large cage. Rats also need plenty of space and can become very tame, can learn tricks and seem to enjoy human companionship. Mice are short-lived but take up very little space. Hamsters are solitary but tend to be nocturnal. They have interesting behaviors, but unless handled regularly they may be defensive. Gerbils are not usually aggressive, rarely bite and are sociable animals that enjoy the company of humans and their own kind.
Rodents including guinea pigs, mice, rats, hamsters, gerbils, chinchillas, degus and chipmunks make convenient pets able to live in small spaces, each species with its own qualities. Most are normally kept in cages of suitable sizes and have varied requirements for space and social interaction. If handled from a young age, they are usually docile and do not bite. Guinea pigs have a long lifespan and need a large cage. Rats also need plenty of space and can become very tame, can learn tricks and seem to enjoy human companionship. Mice are short-lived but take up very little space. Hamsters are solitary but tend to be nocturnal. They have interesting behaviors, but unless handled regularly they may be defensive. Gerbils are not usually aggressive, rarely bite and are sociable animals that enjoy the company of humans and their own kind.
As pests and disease vectors
pests
PESTS was an anonymous American activist group formed in 1986 to critique racism, tokenism, and exclusion in the art world. PESTS produced newsletters, posters, and other print material highlighting examples of discrimination in gallery represent ...
, eating large quantities of food stored by humans. For example, in 2003, the amount of rice
Rice is a cereal grain and in its Domestication, domesticated form is the staple food of over half of the world's population, particularly in Asia and Africa. Rice is the seed of the grass species ''Oryza sativa'' (Asian rice)—or, much l ...
lost to mice and rats in Asia was estimated to be enough to feed 200 million people. Most of the damage worldwide is caused by a relatively small number of species, chiefly rats and mice. In Indonesia
Indonesia, officially the Republic of Indonesia, is a country in Southeast Asia and Oceania, between the Indian Ocean, Indian and Pacific Ocean, Pacific oceans. Comprising over List of islands of Indonesia, 17,000 islands, including Sumatra, ...
and Tanzania
Tanzania, officially the United Republic of Tanzania, is a country in East Africa within the African Great Lakes region. It is bordered by Uganda to the northwest; Kenya to the northeast; the Indian Ocean to the east; Mozambique and Malawi to t ...
, rodents reduce crop yields by around fifteen percent, while in some instances in South America losses have reached ninety percent. Across Africa, rodents including '' Mastomys'' and ''Arvicanthis
''Arvicanthis'' is a genus of rodent from Africa. They are commonly referred to as unstriped grass mice, unstriped grass rats, and kusu rats.
Species
Genus ''Arvicanthis'' - unstriped grass mice
*Abyssinian grass rat, ''Arvicanthis abyssinicus' ...
'' damage cereals, groundnuts, vegetables and cacao. In Asia, rats, mice and species such as '' Microtus brandti'', '' Meriones unguiculatus'' and '' Eospalax baileyi'' damage crops of rice, sorghum
''Sorghum bicolor'', commonly called sorghum () and also known as great millet, broomcorn, guinea corn, durra, imphee, jowar, or milo, is a species in the Poaceae, grass genus ''Sorghum (genus), Sorghum'' cultivated for its grain. The grain i ...
, tubers, vegetables and nuts. In Europe, as well as rats and mice, species of ''Apodemus
''Apodemus'' is a genus of Muridae, murid (true mice and rats) containing the field mice as well as other well-known species like the wood mouse and the yellow-necked mouse. The name is unrelated to that of the ''Mus (genus), Mus'' genus, instead ...
'', ''Microtus'' and in occasional outbreaks '' Arvicola terrestris'' cause damage to orchards, vegetables and pasture as well as cereals. In South America, a wider range of rodent species, such as '' Holochilus'', '' Akodon'', '' Calomys'', '' Oligoryzomys'', '' Phyllotis'', '' Sigmodon'' and '' Zygodontomys'', damage many crops including sugar cane, fruits, vegetables, and tubers.
Rodents are also significant vectors of disease. The black rat, with the fleas that it carries, plays a primary role in spreading the bacterium '' Yersinia pestis'' responsible for bubonic plague, and carries the organisms responsible for typhus
Typhus, also known as typhus fever, is a group of infectious diseases that include epidemic typhus, scrub typhus, and murine typhus. Common symptoms include fever, headache, and a rash. Typically these begin one to two weeks after exposu ...
, Weil's disease, toxoplasmosis and trichinosis. A number of rodents carry hantavirus
''Orthohantavirus'' is a genus of viruses that includes all hantaviruses (family ''Hantaviridae'') that cause disease in humans. Orthohantaviruses, hereafter referred to as hantaviruses, are naturally found primarily in rodents. In general, each ...
es, including the Puumala, Dobrava and Saaremaa viruses, which can infect humans. Rodents also help to transmit diseases including babesiosis, cutaneous leishmaniasis
Leishmaniasis is a wide array of clinical manifestations caused by protozoal parasites of the Trypanosomatida genus ''Leishmania''. It is generally spread through the bite of Phlebotominae, phlebotomine Sandfly, sandflies, ''Phlebotomus'' an ...
, human granulocytic anaplasmosis, Lyme disease
Lyme disease, also known as Lyme borreliosis, is a tick-borne disease caused by species of ''Borrelia'' bacteria, Disease vector, transmitted by blood-feeding ticks in the genus ''Ixodes''. It is the most common disease spread by ticks in th ...
, Omsk hemorrhagic fever, Powassan virus, rickettsialpox, relapsing fever, Rocky Mountain spotted fever, and West Nile virus.
Because rodents are a nuisance and endanger public health
Public health is "the science and art of preventing disease, prolonging life and promoting health through the organized efforts and informed choices of society, organizations, public and private, communities and individuals". Analyzing the de ...
, human societies often attempt to control them. Traditionally, this involved poisoning and trapping, methods that were not always safe or effective. More recently, integrated pest management
Integrated pest management (IPM), also known as integrated pest control (IPC) integrates both chemical and non-chemical practices for economic control of pests. The UN's Food and Agriculture Organization defines IPM as "the careful consideratio ...
attempts to improve control with a combination of surveys to determine the size and distribution of the pest population, the establishment of tolerance limits (levels of pest activity at which to intervene), interventions, and evaluation of effectiveness based on repeated surveys. Interventions may include education, making and applying laws and regulations, modifying the habitat, changing farming practices, and biological control
Biological control or biocontrol is a method of controlling pests, whether pest animals such as insects and mites, weeds, or pathogens affecting animals or plants by using other organisms. It relies on predation, parasitism, herbivory, or o ...
using pathogen
In biology, a pathogen (, "suffering", "passion" and , "producer of"), in the oldest and broadest sense, is any organism or agent that can produce disease. A pathogen may also be referred to as an infectious agent, or simply a Germ theory of d ...
s or predator
Predation is a biological interaction in which one organism, the predator, kills and eats another organism, its prey. It is one of a family of common List of feeding behaviours, feeding behaviours that includes parasitism and micropredation ...
s, as well as poisoning
Poisoning is the harmful effect which occurs when Toxicity, toxic substances are introduced into the body. The term "poisoning" is a derivative of poison, a term describing any chemical substance that may harm or kill a living organism upon ...
and trapping. The use of pathogens such as ''Salmonella
''Salmonella'' is a genus of bacillus (shape), rod-shaped, (bacillus) Gram-negative bacteria of the family Enterobacteriaceae. The two known species of ''Salmonella'' are ''Salmonella enterica'' and ''Salmonella bongori''. ''S. enterica'' ...
'' has the drawback that they can infect man and domestic animals, and rodents often become resistant. The use of predators including ferrets, mongooses and monitor lizards has been found unsatisfactory. Domestic and feral cats are able to control rodents effectively, provided the rodent population is not too large. In the UK, two species in particular, the house mouse and the brown rat, are actively controlled to limit damage in growing crops, loss and contamination of stored crops and structural damage to facilities, as well as to comply with the law.
See also
* RodentologyReferences
Further reading
* * ** Carleton, M. D.; Musser, G. G. "Order Rodentia", pages 745–752 in Wilson & Reeder (2005).External links
Zoology, osteology, comparative anatomy
ArchéoZooThèque : Rodent osteology
(photos)
ArchéoZooThèque : Rodent skeleton drawings
Various
African rodentia
Rodent photos
on
Flickr
Flickr ( ) is an image hosting service, image and Online video platform, video hosting service, as well as an online community, founded in Canada and headquartered in the United States. It was created by Ludicorp in 2004 and was previously a co ...
Rodent Species Fact Sheets
from the National Pest Management Association on Deer Mice, Norway Rats, and other rodent species {{Authority control Articles containing video clips Extant Thanetian first appearances Taxa named by Thomas Edward Bowdich