Keratin on:
[Wikipedia]
[Google]
[Amazon]
 Keratin () is one of a family of structural
Keratin () is one of a family of structural
 Keratin () is one of a family of structural
Keratin () is one of a family of structural fibrous protein
In molecular biology, fibrous proteins or scleroproteins are one of the three main classifications of protein structure (alongside globular and membrane proteins). Fibrous proteins are made up of elongated or fibrous polypeptide chains which fo ...
s also known as ''scleroproteins''. Alpha-keratin
Alpha-keratin, or α-keratin, is a type of keratin found in vertebrates. This protein is the primary component in hairs, horns, mammalian claws, nails and the epidermis layer of the skin. α-keratin is a fibrous structural protein, meaning it i ...
(α-keratin) is a type of keratin found in vertebrates
Vertebrates () comprise all animal taxa within the subphylum Vertebrata () (chordates with backbones), including all mammals, birds, reptiles, amphibians, and fish. Vertebrates represent the overwhelming majority of the phylum Chordata, with c ...
. It is the key structural material making up scales
Scale or scales may refer to:
Mathematics
* Scale (descriptive set theory), an object defined on a set of points
* Scale (ratio), the ratio of a linear dimension of a model to the corresponding dimension of the original
* Scale factor, a number w ...
, hair, nails, feathers, horns Horns or The Horns may refer to:
* Plural of Horn (instrument), a group of musical instruments all with a horn-shaped bells
* The Horns (Colorado), a summit on Cheyenne Mountain
* ''Horns'' (novel), a dark fantasy novel written in 2010 by Joe Hill ...
, claw
A claw is a curved, pointed appendage found at the end of a toe or finger in most amniotes (mammals, reptiles, birds). Some invertebrates such as beetles and spiders have somewhat similar fine, hooked structures at the end of the leg or tarsus ...
s, hooves
The hoof (plural: hooves) is the tip of a toe of an ungulate mammal, which is covered and strengthened with a thick and horny keratin covering. Artiodactyls are even-toed ungulates, species whose feet have an even number of digits, yet the rumi ...
, and the outer layer of skin
Skin is the layer of usually soft, flexible outer tissue covering the body of a vertebrate animal, with three main functions: protection, regulation, and sensation.
Other animal coverings, such as the arthropod exoskeleton, have different de ...
among vertebrates. Keratin also protects epithelial
Epithelium or epithelial tissue is one of the four basic types of animal tissue, along with connective tissue, muscle tissue and nervous tissue. It is a thin, continuous, protective layer of compactly packed cells with a little intercell ...
cells from damage or stress. Keratin is extremely insoluble in water and organic solvents. Keratin monomer
In chemistry, a monomer ( ; '' mono-'', "one" + ''-mer'', "part") is a molecule that can react together with other monomer molecules to form a larger polymer chain or three-dimensional network in a process called polymerization.
Classification
...
s assemble into bundles to form intermediate filaments, which are tough and form strong unmineralized epidermal appendages found in reptiles, bird
Birds are a group of warm-blooded vertebrates constituting the class Aves (), characterised by feathers, toothless beaked jaws, the laying of hard-shelled eggs, a high metabolic rate, a four-chambered heart, and a strong yet lightweig ...
s, amphibians, and mammals. Excessive keratinization participate in fortification of certain tissues such as in horns of cattle and rhinos, and armadillos' osteoderm. The only other biological matter known to approximate the toughness
In materials science and metallurgy, toughness is the ability of a material to absorb energy and plastically deform without fracturing.chitin.
Keratin comes in two types, the primitive, softer forms found in all vertebrates and harder, derived forms found only among


 The
The
 Fibrous keratin molecules supercoil to form a very stable, left-handed superhelical motif to multimerise, forming filaments consisting of multiple copies of the keratin
Fibrous keratin molecules supercoil to form a very stable, left-handed superhelical motif to multimerise, forming filaments consisting of multiple copies of the keratin
Composition and β-sheet structure of silkHair-Science.com's entry on the microscopic elements of hair
Proteopedia page on keratins
{{Authority control Keratins Cytoskeleton Skin anatomy
sauropsids
Sauropsida ("lizard faces") is a clade of amniotes, broadly equivalent to the class Reptilia. Sauropsida is the sister taxon to Synapsida, the other clade of amniotes which includes mammals as its only modern representatives. Although early sy ...
(reptiles and birds).
Spider silk
Spider silk is a protein fibre spun by spiders. Spiders use their silk to make webs or other structures, which function as sticky nets to catch other animals, or as nests or cocoons to protect their offspring, or to wrap up prey. They can ...
is classified as keratin, although production of the protein may have evolved independently of the process in vertebrates.
Examples of occurrence

Alpha-keratin
Alpha-keratin, or α-keratin, is a type of keratin found in vertebrates. This protein is the primary component in hairs, horns, mammalian claws, nails and the epidermis layer of the skin. α-keratin is a fibrous structural protein, meaning it i ...
s (α-keratins) are found in all vertebrates. They form the hair (including wool
Wool is the textile fibre obtained from sheep and other mammals, especially goats, rabbits, and camelids. The term may also refer to inorganic materials, such as mineral wool and glass wool, that have properties similar to animal wool. ...
), the outer layer of skin, horns Horns or The Horns may refer to:
* Plural of Horn (instrument), a group of musical instruments all with a horn-shaped bells
* The Horns (Colorado), a summit on Cheyenne Mountain
* ''Horns'' (novel), a dark fantasy novel written in 2010 by Joe Hill ...
, nails, claws
A claw is a curved, pointed appendage found at the end of a toe or finger in most amniotes (mammals, reptiles, birds). Some invertebrates such as beetles and spiders have somewhat similar fine, hooked structures at the end of the leg or tarsus ...
and hooves
The hoof (plural: hooves) is the tip of a toe of an ungulate mammal, which is covered and strengthened with a thick and horny keratin covering. Artiodactyls are even-toed ungulates, species whose feet have an even number of digits, yet the rumi ...
of mammals, and the slime threads of hagfish
Hagfish, of the class Myxini (also known as Hyperotreti) and order Myxiniformes , are eel-shaped, slime-producing marine fish (occasionally called slime eels). They are the only known living animals that have a skull but no vertebral column, ...
. Keratin filaments are abundant in keratinocyte
Keratinocytes are the primary type of cell found in the epidermis, the outermost layer of the skin. In humans, they constitute 90% of epidermal skin cells.
Basal cells in the basal layer (''stratum basale'') of the skin are sometimes referre ...
s in the hornified layer of the epidermis; these are proteins which have undergone keratinization
Keratin () is one of a family of structural fibrous proteins also known as ''scleroproteins''. Alpha-keratin (α-keratin) is a type of keratin found in vertebrates. It is the key structural material making up scales, hair, nails, feathers, h ...
. They are also present in epithelial cells in general. For example, mouse thymic epithelial cells react with antibodies for keratin 5, keratin 8, and keratin 14. These antibodies are used as fluorescent markers to distinguish subsets of mouse thymic
The thymus is a specialized primary Lymphatic system#Structure, lymphoid organ of the immune system. Within the thymus, T cell, thymus cell lymphocytes or ''T cells'' mature. T cells are critical to the adaptive immune system, where the body ada ...
epithelial cells in genetic studies of the thymus
The thymus is a specialized primary lymphoid organ of the immune system. Within the thymus, thymus cell lymphocytes or ''T cells'' mature. T cells are critical to the adaptive immune system, where the body adapts to specific foreign invaders. ...
.
The harder beta-keratin
Beta-keratin (β-keratin), is a member of a structural protein family found in the epidermis of reptiles and birds. Beta-keratins were named so because they are components of epidermal stratum corneum rich in stacked beta sheets, in contrast to a ...
s (β-keratin)s are found only in the sauropsids, that is all living reptiles and bird
Birds are a group of warm-blooded vertebrates constituting the class Aves (), characterised by feathers, toothless beaked jaws, the laying of hard-shelled eggs, a high metabolic rate, a four-chambered heart, and a strong yet lightweig ...
s. They are found in the nails, scales
Scale or scales may refer to:
Mathematics
* Scale (descriptive set theory), an object defined on a set of points
* Scale (ratio), the ratio of a linear dimension of a model to the corresponding dimension of the original
* Scale factor, a number w ...
, and claws of reptiles, in some reptile shells (testudines
Turtles are an order of reptiles known as Testudines, characterized by a special shell developed mainly from their ribs. Modern turtles are divided into two major groups, the Pleurodira (side necked turtles) and Cryptodira (hidden necked t ...
, such as tortoise
Tortoises () are reptiles of the family Testudinidae of the order Testudines (Latin: ''tortoise''). Like other turtles, tortoises have a shell to protect from predation and other threats. The shell in tortoises is generally hard, and like oth ...
, turtle
Turtles are an order of reptiles known as Testudines, characterized by a special shell developed mainly from their ribs. Modern turtles are divided into two major groups, the Pleurodira (side necked turtles) and Cryptodira (hidden necked t ...
, terrapin
Terrapins are one of several small species of turtle (order Testudines) living in fresh or brackish water. Terrapins do not form a taxonomic unit and may not be closely related. Many belong to the families Geoemydidae and Emydidae.
The name ...
), and in the feathers, beak
The beak, bill, or rostrum is an external anatomical structure found mostly in birds, but also in turtles, non-avian dinosaurs and a few mammals. A beak is used for eating, preening, manipulating objects, killing prey, fighting, probing for foo ...
s, and claws of bird
Birds are a group of warm-blooded vertebrates constituting the class Aves (), characterised by feathers, toothless beaked jaws, the laying of hard-shelled eggs, a high metabolic rate, a four-chambered heart, and a strong yet lightweig ...
s. These keratins are formed primarily in beta sheets. However, beta sheets are also found in α-keratins.
The baleen plates of filter-feeding whale
Whales are a widely distributed and diverse group of fully aquatic placental marine mammals. As an informal and colloquial grouping, they correspond to large members of the infraorder Cetacea, i.e. all cetaceans apart from dolphins and ...
s are made of keratin. Recent scholarship has shown that sauropsid β-keratins are fundamentally different from α-keratins at a genetic and structural level. The new term ''corneous beta protein'' (CBP) has been proposed to avoid confusion with α-keratins.
Keratins (also described as cytokeratins
Cytokeratins are keratin proteins found in the intracytoplasmic cytoskeleton of epithelial tissue. They are an important component of intermediate filaments, which help cells resist mechanical stress. Expression of these cytokeratins within epi ...
) are polymer
A polymer (; Greek '' poly-'', "many" + ''-mer'', "part")
is a substance or material consisting of very large molecules called macromolecules, composed of many repeating subunits. Due to their broad spectrum of properties, both synthetic a ...
s of type I and type II intermediate filaments
Intermediate filaments (IFs) are cytoskeletal structural components found in the cells of vertebrates, and many invertebrates. Homologues of the IF protein have been noted in an invertebrate, the cephalochordate '' Branchiostoma''.
Intermedi ...
that have been found only in chordate
A chordate () is an animal of the phylum Chordata (). All chordates possess, at some point during their larval or adult stages, five synapomorphies, or primary physical characteristics, that distinguish them from all the other taxa. These fi ...
s (vertebrate
Vertebrates () comprise all animal taxa within the subphylum Vertebrata () (chordates with backbones), including all mammals, birds, reptiles, amphibians, and fish. Vertebrates represent the overwhelming majority of the phylum Chordata, with c ...
s, amphioxus
The lancelets ( or ), also known as amphioxi (singular: amphioxus ), consist of some 30 to 35 species of "fish-like" benthic filter feeding chordates in the order Amphioxiformes. They are the modern representatives of the subphylum Cephalochorda ...
, urochordates
A tunicate is a marine invertebrate animal, a member of the subphylum Tunicata (). It is part of the Chordata, a phylum which includes all animals with dorsal nerve cords and notochords (including vertebrates). The subphylum was at one time ...
). Nematodes and many other non-chordate animals seem to have only type VI intermediate filaments
Intermediate filaments (IFs) are cytoskeletal structural components found in the cells of vertebrates, and many invertebrates. Homologues of the IF protein have been noted in an invertebrate, the cephalochordate '' Branchiostoma''.
Intermedi ...
, fibers that structure the nucleus.
Genes
human genome
The human genome is a complete set of nucleic acid sequences for humans, encoded as DNA within the 23 chromosome pairs in cell nuclei and in a small DNA molecule found within individual mitochondria. These are usually treated separately as the ...
encodes 54 functional keratin gene
In biology, the word gene (from , ; "... Wilhelm Johannsen coined the word gene to describe the Mendelian units of heredity..." meaning ''generation'' or ''birth'' or ''gender'') can have several different meanings. The Mendelian gene is a b ...
s, located in two clusters on chromosome
A chromosome is a long DNA molecule with part or all of the genetic material of an organism. In most chromosomes the very long thin DNA fibers are coated with packaging proteins; in eukaryotic cells the most important of these proteins are ...
s 12 and 17. This suggests that they originated from a series of gene duplications on these chromosomes.
The keratins include the following proteins of which KRT23
Keratin, type I cytoskeletal 23 is a protein that in humans is encoded by the ''KRT23'' gene.
The protein encoded by this gene is a member of the keratin family. The keratins are intermediate filament proteins responsible for the structural integ ...
, KRT24
KRT24 is a keratin
Keratin () is one of a family of structural fibrous proteins also known as ''scleroproteins''. Alpha-keratin (α-keratin) is a type of keratin found in vertebrates. It is the key structural material making up scales, hair, ...
, KRT25
KRT25 is a keratin
Keratin () is one of a family of structural fibrous proteins also known as ''scleroproteins''. Alpha-keratin (α-keratin) is a type of keratin found in vertebrates. It is the key structural material making up Scale (anatomy ...
, KRT26
KRT26 is a keratin
Keratin () is one of a family of structural fibrous proteins also known as ''scleroproteins''. Alpha-keratin (α-keratin) is a type of keratin found in vertebrates. It is the key structural material making up Scale (anatomy ...
, KRT27
KRT27 is a keratin
Keratin () is one of a family of structural fibrous proteins also known as ''scleroproteins''. Alpha-keratin (α-keratin) is a type of keratin found in vertebrates. It is the key structural material making up scales, hair, ...
, KRT28
KRT28 is a keratin
Keratin () is one of a family of structural fibrous proteins also known as ''scleroproteins''. Alpha-keratin (α-keratin) is a type of keratin found in vertebrates. It is the key structural material making up scales, hair, ...
, KRT31
Keratin, type I cuticular Ha1 is a protein that in humans is encoded by the ''KRT31'' gene.
Function
The protein encoded by this gene is a member of the keratin gene family. As a type I hair keratin, it is an acidic protein which heterodimeri ...
, KRT32
Keratin, type I cuticular Ha2 is a protein that in humans is encoded by the ''KRT32'' gene.
The protein encoded by this gene is a member of the keratin gene family. As a type I hair keratin, it is an acidic protein which heterodimerizes with typ ...
, KRT33A
Keratin, type I cuticular Ha3-I is a protein that in humans is encoded by the ''KRT33A'' gene
In biology, the word gene (from , ; "... Wilhelm Johannsen coined the word gene to describe the Mendelian units of heredity..." meaning ''generat ...
, KRT33B
Keratin, type I cuticular Ha3-II is a protein that in humans is encoded by the ''KRT33B'' gene.
The protein encoded by this gene is a member of the keratin gene family. It is one of the type I hair keratin genes which are clustered in a region o ...
, KRT34
Keratin, type I cuticular Ha4 is a protein that in humans is encoded by the ''KRT34'' gene.
The protein encoded by this gene is a member of the keratin gene family. As a type I hair keratin, it is an acidic protein which heterodimerizes with type ...
, KRT35
Keratin, type I cuticular Ha5 is a keratin
Keratin () is one of a family of structural fibrous proteins also known as ''scleroproteins''. Alpha-keratin (α-keratin) is a type of keratin found in vertebrates. It is the key structural material ...
, KRT36
Keratin, type I cuticular Ha6 is a protein that in humans is encoded by the ''KRT36'' gene
In biology, the word gene (from , ; "...Wilhelm Johannsen coined the word gene to describe the Mendelian units of heredity..." meaning ''generation ...
, KRT37
Keratin 37 is a protein that in humans is encoded by the KRT37 gene. KRT37 is a member of the keratin gene family.
Clinical significance
KRT37 is the only keratin that is regulated by androgens. This sensitivity to androgens was acquired by ...
, KRT38
KRT38 is a keratin
Keratin () is one of a family of structural fibrous proteins also known as ''scleroproteins''. Alpha-keratin (α-keratin) is a type of keratin found in vertebrates. It is the key structural material making up scales, hair, ...
, KRT39
KRT39 is a keratin
Keratin () is one of a family of structural fibrous proteins also known as ''scleroproteins''. Alpha-keratin (α-keratin) is a type of keratin found in vertebrates. It is the key structural material making up Scale (anatomy ...
, KRT40
KRT40 is a keratin
Keratin () is one of a family of structural fibrous proteins also known as ''scleroproteins''. Alpha-keratin (α-keratin) is a type of keratin found in vertebrates. It is the key structural material making up Scale (anatomy ...
, KRT71
KRT71 is a keratin
Keratin () is one of a family of structural fibrous proteins also known as ''scleroproteins''. Alpha-keratin (α-keratin) is a type of keratin found in vertebrates. It is the key structural material making up scales, hair, ...
, KRT72
KRT72 is a keratin gene. It is responsible for hair formation, and it encodes a protein present in the inner root sheath The inner root sheath or internal root sheath of the hair follicle is located between the outer root sheath The outer root shea ...
, KRT73
KRT73 is a keratin gene. It is responsible for hair formation, along with other genes, and it encodes a protein present in the inner root sheath of hair follicles.
References
{{Cytoskeletal proteins ...
, KRT74
KRT74 is a keratin gene.
Mutations
In biology, a mutation is an alteration in the nucleic acid sequence of the genome of an organism, virus, or extrachromosomal DNA. Viral genomes contain either DNA or RNA. Mutations result from errors ...
, KRT75, KRT76, KRT77
''KRT77'' encodes keratin 77, a member of the type II keratin
Keratin () is one of a family of structural fibrous proteins also known as ''scleroproteins''. Alpha-keratin (α-keratin) is a type of keratin found in vertebrates. It is the key s ...
, KRT78
Keratin, type II cytoskeletal 78 is a protein that in humans is encoded by the ''KRT78'' gene.
This gene is a member of the type II keratin gene family and encodes a protein with an intermediate filament domain. Keratins are the major structural ...
, KRT79
Keratin 79 also known as KRT79 is a protein which humans is encoded by the ''KRT79'' gene.
Function
Keratins, such as KRT79, are filament proteins that make up one of the major structural fibers of epithelial cells
Epithelium or epith ...
, KRT8
Keratin, type II cytoskeletal 8 also known as cytokeratin-8 (CK-8) or keratin-8 (K8) is a keratin protein that is encoded in humans by the ''KRT8'' gene. It is often paired with keratin 18.
Utility as an immunohistochemical stain
Antibodies to CK ...
, KRT80
Keratin 80, also known as KRT80, is a protein which humans is encoded by the ''KRT80'' gene.
Function
Keratin
Keratin () is one of a family of structural fibrous proteins also known as ''scleroproteins''. Alpha-keratin (α-keratin) is a typ ...
, KRT81
Keratin, type II cuticular Hb1 is a protein that in humans is encoded by the ''KRT81'' gene
In biology, the word gene (from , ; "...Wilhelm Johannsen coined the word gene to describe the Mendelian units of heredity..." meaning ''generation ...
, KRT82
KRT82 is a keratin
Keratin () is one of a family of structural fibrous proteins also known as ''scleroproteins''. Alpha-keratin (α-keratin) is a type of keratin found in vertebrates. It is the key structural material making up Scale (anatomy) ...
, KRT83
Keratin 83, also known as KRT83, is a protein which humans is encoded by the ''KRT83'' gene.
Function
The protein encoded by this gene is a member of the keratin gene family. As a type II hair keratin, it is a basic protein which heterodimeri ...
, KRT84
KRT84 is a keratin gene, for a type II hair keratin Hair keratin is a type of keratin found in hair and nails. There are two types of hair keratin:
* the acidic ''type I hair keratin''
** type I hair keratin 1,
** type I hair keratin 2,
** ty ...
, KRT85
Keratin, type II cuticular Hb5 is a protein that in humans is encoded by the ''KRT85'' gene
In biology, the word gene (from , ; "...Wilhelm Johannsen coined the word gene to describe the Mendelian units of heredity..." meaning ''generation ...
and KRT86
Keratin, type II cuticular Hb6 is a protein that in humans is encoded by the ''KRT86'' gene
In biology, the word gene (from , ; "... Wilhelm Johannsen coined the word gene to describe the Mendelian units of heredity..." meaning ''generation ...
have been used to describe keratins past 20.
Protein structure
The first sequences of keratins were determined byIsrael Hanukoglu
Israel Hanukoglu ( tr, İsrael Hanukoğlu) is a Turkish-born Israeli scientist. He is a full professor of biochemistry and molecular biology at Ariel University and former science and technology adviser to the prime minister of Israel (1996–199 ...
and Elaine Fuchs
Elaine V. Fuchs is an American cell biologist famous for her work on the biology and molecular mechanisms of mammalian skin and skin diseases, who helped lead the modernization of dermatology. Fuchs pioneered reverse genetics approaches, which ...
(1982, 1983). These sequences revealed that there are two distinct but homologous keratin families, which were named type I and type II keratins. By analysis of the primary structures of these keratins and other intermediate filament proteins, Hanukoglu and Fuchs suggested a model in which keratins and intermediate filament proteins contain a central ~310 residue domain with four segments in α-helical conformation that are separated by three short linker segments predicted to be in beta-turn conformation. This model has been confirmed by the determination of the crystal structure of a helical domain of keratins.
 Fibrous keratin molecules supercoil to form a very stable, left-handed superhelical motif to multimerise, forming filaments consisting of multiple copies of the keratin
Fibrous keratin molecules supercoil to form a very stable, left-handed superhelical motif to multimerise, forming filaments consisting of multiple copies of the keratin monomer
In chemistry, a monomer ( ; '' mono-'', "one" + ''-mer'', "part") is a molecule that can react together with other monomer molecules to form a larger polymer chain or three-dimensional network in a process called polymerization.
Classification
...
.
The major force that keeps the coiled-coil structure is hydrophobic interactions
The hydrophobic effect is the observed tendency of nonpolar substances to aggregate in an aqueous solution and exclude water#Properties, water molecules. The word hydrophobic literally means "water-fearing", and it describes the Segregation in m ...
between apolar
In chemistry, polarity is a separation of electric charge leading to a molecule or its chemical groups having an electric dipole moment, with a negatively charged end and a positively charged end.
Polar molecules must contain one or more po ...
residues along the keratins helical segments.
Limited interior space is the reason why the triple helix
In the fields of geometry and biochemistry, a triple helix (plural triple helices) is a set of three congruent geometrical helices with the same axis, differing by a translation along the axis. This means that each of the helices keeps the same ...
of the (unrelated) structural protein collagen, found in skin
Skin is the layer of usually soft, flexible outer tissue covering the body of a vertebrate animal, with three main functions: protection, regulation, and sensation.
Other animal coverings, such as the arthropod exoskeleton, have different de ...
, cartilage and bone
A bone is a rigid organ that constitutes part of the skeleton in most vertebrate animals. Bones protect the various other organs of the body, produce red and white blood cells, store minerals, provide structure and support for the body, ...
, likewise has a high percentage of glycine
Glycine (symbol Gly or G; ) is an amino acid that has a single hydrogen atom as its side chain. It is the simplest stable amino acid ( carbamic acid is unstable), with the chemical formula NH2‐ CH2‐ COOH. Glycine is one of the proteinog ...
. The connective tissue protein elastin also has a high percentage of both glycine and alanine
Alanine (symbol Ala or A), or α-alanine, is an α-amino acid that is used in the biosynthesis of proteins. It contains an amine group and a carboxylic acid group, both attached to the central carbon atom which also carries a methyl group side ...
. Silk
Silk is a natural protein fiber, some forms of which can be woven into textiles. The protein fiber of silk is composed mainly of fibroin and is produced by certain insect larvae to form cocoons. The best-known silk is obtained from the ...
fibroin
Fibroin is an insoluble protein present in silk produced by numerous insects, such as the larvae of ''Bombyx mori'', and other moth genera such as '' Antheraea'', '' Cricula'', '' Samia'' and '' Gonometa''. Silk in its raw state consists of tw ...
, considered a β-keratin, can have these two as 75–80% of the total, with 10–15% serine, with the rest having bulky side groups. The chains are antiparallel, with an alternating C → N orientation. A preponderance of amino acid
Amino acids are organic compounds that contain both amino and carboxylic acid functional groups. Although hundreds of amino acids exist in nature, by far the most important are the alpha-amino acids, which comprise proteins. Only 22 alpha a ...
s with small, nonreactive side groups is characteristic of structural proteins, for which H-bonded close packing is more important than chemical specificity
Chemical specificity is the ability of binding site of a macromolecule (such as a protein) to bind specific ligands. The fewer ligands a protein can bind, the greater its specificity.
Specificity describes the strength of binding between a giv ...
.
Disulfide bridges
In addition to intra- and intermolecular hydrogen bonds, the distinguishing feature of keratins is the presence of large amounts of the sulfur-containing amino acid cysteine, required for thedisulfide bridges
In biochemistry, a disulfide (or disulphide in British English) refers to a functional group with the structure . The linkage is also called an SS-bond or sometimes a disulfide bridge and is usually derived by the coupling of two thiol groups. In ...
that confer additional strength and rigidity by permanent, thermally stable crosslinking Cross-linking may refer to
*Cross-link
In chemistry and biology a cross-link is a bond or a short sequence of bonds that links one polymer chain to another. These links may take the form of covalent bonds or ionic bonds and the polymers ca ...
—in much the same way that non-protein sulfur bridges stabilize vulcanized
Vulcanization (British: Vulcanisation) is a range of processes for hardening rubbers. The term originally referred exclusively to the treatment of natural rubber with sulfur, which remains the most common practice. It has also grown to inclu ...
rubber
Rubber, also called India rubber, latex, Amazonian rubber, ''caucho'', or ''caoutchouc'', as initially produced, consists of polymers of the organic compound isoprene, with minor impurities of other organic compounds. Thailand, Malaysia, an ...
. Human hair is approximately 14% cysteine. The pungent
Pungency () refers to the taste of food commonly referred to as spiciness, hotness or heat, found in foods such as chili peppers. Highly pungent tastes may be experienced as unpleasant. The term piquancy () is sometimes applied to foods with a l ...
smells of burning hair and skin are due to the volatile sulfur compounds formed. Extensive disulfide bonding contributes to the insolubility
In chemistry, solubility is the ability of a substance, the solute, to form a solution with another substance, the solvent. Insolubility is the opposite property, the inability of the solute to form such a solution.
The extent of the solubi ...
of keratins, except in a small number of solvents such as dissociating or reducing agents.
The more flexible and elastic keratins of hair have fewer interchain disulfide bridges than the keratins in mammalian
Mammals () are a group of vertebrate animals constituting the class Mammalia (), characterized by the presence of mammary glands which in females produce milk for feeding (nursing) their young, a neocortex (a region of the brain), fur o ...
fingernail
A nail is a claw-like plate found at the tip of the fingers and toes on most primates. Nails correspond to the claws found in other animals. Fingernails and toenails are made of a tough protective protein called alpha-keratin, which is a polymer ...
s, hooves and claws (homologous structures), which are harder and more like their analogs in other vertebrate classes. Hair and other α-keratins consist of α-helically coiled single protein strands (with regular intra-chain H-bonding
In chemistry, a hydrogen bond (or H-bond) is a primarily electrostatic force of attraction between a hydrogen (H) atom which is covalently bound to a more electronegative "donor" atom or group (Dn), and another electronegative atom bearing a ...
), which are then further twisted into superhelical rope
A rope is a group of yarns, plies, fibres, or strands that are twisted or braided together into a larger and stronger form. Ropes have tensile strength and so can be used for dragging and lifting. Rope is thicker and stronger than similar ...
s that may be further coiled. The β-keratins of reptiles and birds have β-pleated sheets twisted together, then stabilized and hardened by disulfide bridges.
Filament formation
It has been proposed that keratins can be divided into 'hard' and 'soft' forms, or 'cytokeratin
Cytokeratins are keratin proteins found in the intracytoplasmic cytoskeleton of epithelial tissue. They are an important component of intermediate filaments, which help cells resist mechanical stress. Expression of these cytokeratins within epi ...
s' and 'other keratins'. That model is now understood to be correct. A new nuclear addition in 2006 to describe keratins takes this into account.
Keratin filaments are intermediate filaments. Like all intermediate filaments, keratin proteins form filamentous polymers in a series of assembly steps beginning with dimerization; dimers assemble into tetramers and octamers and eventually, if the current hypothesis holds, into unit-length-filaments (ULF) capable of annealing end-to-end into long filaments.
Pairing
Cornification
Cornification is the process of forming an epidermal barrier in stratified squamous epithelial tissue. At the cellular level, cornification is characterised by: * production of keratin * production of small proline-rich (SPRR) proteins and transglutaminase which eventually form a cornified cell envelope beneath the plasma membrane *terminal differentiation
Cellular differentiation is the process in which a stem cell alters from one type to a differentiated one. Usually, the cell changes to a more specialized type. Differentiation happens multiple times during the development of a multicellular ...
* loss of nuclei and organelles, in the final stages of cornification
Metabolism ceases, and the cells are almost completely filled by keratin. During the process of epithelial differentiation, cells become cornified as keratin protein is incorporated into longer keratin intermediate filaments. Eventually the nucleus and cytoplasmic organelles disappear, metabolism ceases and cells undergo a programmed death as they become fully keratinized. In many other cell types, such as cells of the dermis, keratin filaments and other intermediate filaments function as part of the cytoskeleton to mechanically stabilize the cell against physical stress. It does this through connections to desmosomes, cell–cell junctional plaques, and hemidesmosomes, cell-basement membrane adhesive structures.
Cells in the epidermis contain a structural matrix of keratin, which makes this outermost layer of the skin almost waterproof, and along with collagen and elastin gives skin its strength. Rubbing and pressure cause thickening of the outer, cornified layer of the epidermis and form protective calluses, which are useful for athletes and on the fingertips of musicians who play stringed instruments. Keratinized epidermal cells are constantly shed and replaced.
These hard, integumentary structures are formed by intercellular cementing of fibers formed from the dead, cornified cells generated by specialized beds deep within the skin. Hair grows continuously and feathers molt and regenerate. The constituent proteins may be phylogenetically homologous but differ somewhat in chemical structure and supermolecular organization. The evolutionary relationships are complex and only partially known. Multiple genes have been identified for the β-keratins in feathers, and this is probably characteristic of all keratins.
Silk
Thesilk
Silk is a natural protein fiber, some forms of which can be woven into textiles. The protein fiber of silk is composed mainly of fibroin and is produced by certain insect larvae to form cocoons. The best-known silk is obtained from the ...
fibroin
Fibroin is an insoluble protein present in silk produced by numerous insects, such as the larvae of ''Bombyx mori'', and other moth genera such as '' Antheraea'', '' Cricula'', '' Samia'' and '' Gonometa''. Silk in its raw state consists of tw ...
s produced by insect
Insects (from Latin ') are pancrustacean hexapod invertebrates of the class Insecta. They are the largest group within the arthropod phylum. Insects have a chitinous exoskeleton, a three-part body ( head, thorax and abdomen), three ...
s and spider
Spiders ( order Araneae) are air-breathing arthropods that have eight legs, chelicerae with fangs generally able to inject venom, and spinnerets that extrude silk. They are the largest order of arachnids and rank seventh in total species ...
s are often classified as keratins, though it is unclear whether they are phylogenetically related to vertebrate keratins.
Silk found in insect pupa
A pupa ( la, pupa, "doll"; plural: ''pupae'') is the life stage of some insects undergoing transformation between immature and mature stages. Insects that go through a pupal stage are holometabolous: they go through four distinct stages in thei ...
e, and in spider web
A spider web, spiderweb, spider's web, or cobweb (from the archaic word '' coppe'', meaning "spider") is a structure created by a spider out of proteinaceous spider silk extruded from its spinnerets, generally meant to catch its prey.
Spi ...
s and egg casings, also has twisted β-pleated sheets incorporated into fibers wound into larger supermolecular aggregates. The structure of the spinneret
A spinneret is a silk-spinning organ of a spider or the larva of an insect. Some adult insects also have spinnerets, such as those borne on the forelegs of Embioptera. Spinnerets are usually on the underside of a spider's opisthosoma, and a ...
s on spiders’ tails, and the contributions of their interior glands, provide remarkable control of fast extrusion
Extrusion is a process used to create objects of a fixed cross-sectional profile by pushing material through a die of the desired cross-section. Its two main advantages over other manufacturing processes are its ability to create very complex ...
. Spider silk is typically about 1 to 2 micrometers (µm) thick, compared with about 60 µm for human hair, and more for some mammals. The biologically
Biology is the scientific study of life. It is a natural science with a broad scope but has several unifying themes that tie it together as a single, coherent field. For instance, all organisms are made up of cells that process hereditary i ...
and commercially
Commerce is the large-scale organized system of activities, functions, procedures and institutions directly and indirectly related to the exchange (buying and selling) of goods and services among two or more parties within local, regional, nation ...
useful properties of silk fibers depend on the organization of multiple adjacent protein chains into hard, crystal
A crystal or crystalline solid is a solid material whose constituents (such as atoms, molecules, or ions) are arranged in a highly ordered microscopic structure, forming a crystal lattice that extends in all directions. In addition, macro ...
line regions of varying size, alternating with flexible, amorphous regions where the chains are randomly coiled. A somewhat analogous situation occurs with synthetic polymer
A polymer (; Greek '' poly-'', "many" + ''-mer'', "part")
is a substance or material consisting of very large molecules called macromolecules, composed of many repeating subunits. Due to their broad spectrum of properties, both synthetic a ...
s such as nylon
Nylon is a generic designation for a family of synthetic polymers composed of polyamides ( repeating units linked by amide links).The polyamides may be aliphatic or semi-aromatic.
Nylon is a silk-like thermoplastic, generally made from pe ...
, developed as a silk substitute. Silk from the hornet
Hornets (insects in the genus ''Vespa'') are the largest of the eusocial wasps, and are similar in appearance to their close relatives yellowjackets. Some species can reach up to in length. They are distinguished from other vespine wasps by th ...
cocoon contains doublets about 10 µm across, with cores and coating, and may be arranged in up to 10 layers, also in plaques of variable shape. Adult hornets also use silk as a glue, as do spiders.
Glue
Glues made from partially-hydrolysed keratin includehoof glue Hoof glue is a form of animal glue made by boiling down the hooves of ungulates into partially hydrolyzed keratin. It is not to be confused with hide glue.
History
Hoof glue applications include stiffening bow strings, adhering fabric to wood, ...
and horn glue.
Clinical significance
Abnormal growth of keratin can occur in a variety of conditions includingkeratosis
Keratosis (from '' kerat-'' + '' -osis'') is a growth of keratin on the skin or on mucous membranes stemming from keratinocytes, the prominent cell type in the epidermis. More specifically, it can refer to:
* actinic keratosis (also known as solar ...
, hyperkeratosis
Hyperkeratosis is thickening of the stratum corneum (the outermost layer of the epidermis, or skin), often associated with the presence of an abnormal quantity of keratin,Kumar, Vinay; Fausto, Nelso; Abbas, Abul (2004) ''Robbins & Cotran Pathologi ...
and keratoderma
Keratoderma is a hornlike skin condition.
Classification
The keratodermas are classified into the following subgroups:Freedberg, et al. (2003). ''Fitzpatrick's Dermatology in General Medicine''. (6th ed.). McGraw-Hill. .
Congenital
* Simple ker ...
.
Mutations in keratin gene expression can lead to, among others:
* Alopecia Areata
Alopecia areata, also known as spot baldness, is a condition in which hair is lost from some or all areas of the body. Often, it results in a few bald spots on the scalp, each about the size of a coin. Psychological stress and illness are pos ...
* Epidermolysis bullosa simplex
Epidermolysis bullosa simplex (EBS) is a disorder resulting from mutations in the genes encoding keratin 5 or keratin 14.Freedberg, et al. (2003). ''Fitzpatrick's Dermatology in General Medicine''. (6th ed.). McGraw-Hill. .
Epidermolysis bullosa ...
* Ichthyosis bullosa of Siemens
Ichthyosis bullosa of Siemens is a type of familial, autosomal dominant ichthyosis, a rare skin disorder.Freedberg, et al. (2003). ''Fitzpatrick's Dermatology in General Medicine''. (6th ed.). McGraw-Hill. . It is also known as bullous congenital ...
* Epidermolytic hyperkeratosis
Epidermolytic ichthyosis (EI), also known as bullous epidermis ichthyosis (BEI), epidermolytic hyperkeratosis (EHK), bullous congenital ichthyosiform erythroderma (BCIE), bullous ichthyosiform erythrodermaFreedberg, et al. (2003). ''Fitzpatrick' ...
* Steatocystoma multiplex
Steatocystoma multiplex, is a benign, autosomal dominant congenital condition resulting in multiple cysts on a person's body. Steatocystoma simplex is the solitary counterpart to steatocystoma multiplex.
In steatocystoma multiplex, the tendency to ...
* Keratosis pharyngis
Keratosis Pharyngis is a medical condition where keratin grows on the surface of the pharynx, that is the part of the throat at the back of the mouth. Keratin is a protein that normally occurs as the main component of hair and nails.
It is charact ...
* Rhabdoid cell formation in Large cell lung carcinoma with rhabdoid phenotype
Several diseases, such as athlete's foot
Athlete's foot, known medically as ''tinea pedis'', is a common skin infection of the feet caused by a fungus. Signs and symptoms often include itching, scaling, cracking and redness. In rare cases the skin may blister. Athlete's foot fungus ...
and ringworm
Dermatophytosis, also known as ringworm, is a fungal infection of the skin. Typically it results in a red, itchy, scaly, circular rash. Hair loss may occur in the area affected. Symptoms begin four to fourteen days after exposure. Multiple ar ...
, are caused by infectious fungi that feed on keratin.
Keratin is highly resistant to digestive acids if ingested. Cat
The cat (''Felis catus'') is a domestic species of small carnivorous mammal. It is the only domesticated species in the family Felidae and is commonly referred to as the domestic cat or house cat to distinguish it from the wild members of ...
s regularly ingest hair as part of their grooming behavior, leading to the gradual formation of hairball
A hairball is a small collection of hair or fur formed in the stomach of animals, and uncommonly in humans, that is occasionally vomited up when it becomes too big. Hairballs are primarily a tight elongated cylinder of packed fur, but may inclu ...
s that may be expelled orally or excreted. In humans, trichophagia
Trichophagia is the compulsive eating of hair associated with trichotillomania (hair pulling). In trichophagia, people with trichotillomania also ingest the hair that they pull; in extreme cases this can lead to a hair ball ( trichobezoar). The te ...
may lead to Rapunzel syndrome
Rapunzel syndrome is an extremely rare intestinal condition in humans resulting from ingesting hair ( trichophagia). The syndrome is named after the long-haired girl Rapunzel in the fairy tale by the Brothers Grimm. Trichophagia is sometimes asso ...
, an extremely rare but potentially fatal intestinal condition.
Diagnostic use
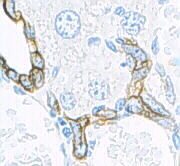
Keratin expression is helpful in determining epithelial origin inanaplastic
Anaplasia (from grc, ἀνά ''ana'', "backward" + πλάσις ''plasis'', "formation") is a condition of cells with poor cellular differentiation, losing the morphological characteristics of mature cells and their orientation with respect to ...
cancers. Tumors that express keratin include carcinomas, thymoma
A thymoma is a tumor originating from the epithelial cells of the thymus that is considered a rare malignancy. Thymomas are frequently associated with neuromuscular disorders such as myasthenia gravis; thymoma is found in 20% of patients with m ...
s, sarcoma
A sarcoma is a malignant tumor, a type of cancer that arises from transformed cells of mesenchymal ( connective tissue) origin. Connective tissue is a broad term that includes bone, cartilage, fat, vascular, or hematopoietic tissues, and sar ...
s and trophoblastic neoplasm
Gestational trophoblastic neoplasia (GTN) is group of rare diseases related to pregnancy and included in gestational trophoblastic disease (GTD) in which abnormal trophoblast cells grow in the uterus. GTN can be classified into benign and maligna ...
s. Furthermore, the precise expression-pattern of keratin subtypes allows prediction of the origin of the primary tumor when assessing metastases
Metastasis is a pathogenic agent's spread from an initial or primary site to a different or secondary site within the host's body; the term is typically used when referring to metastasis by a cancerous tumor. The newly pathological sites, then ...
. For example, hepatocellular carcinomas typically express CK8 and CK18, and cholangiocarcinomas express CK7, CK8 and CK18, while metastases of colorectal carcinoma
Colorectal cancer (CRC), also known as bowel cancer, colon cancer, or rectal cancer, is the development of cancer from the colon or rectum (parts of the large intestine). Signs and symptoms may include blood in the stool, a change in bowel ...
s express CK20, but not CK7.
See also
*List of cutaneous conditions caused by mutations in keratins
There are many different keratin proteins normally expressed in the human integumentary system. Mutations in keratin proteins in the skin can cause disease.
Of note, other structural proteins in the epidermis of the skin that are closely rel ...
* List of keratins expressed in the human integumentary system
There are many different keratin proteins normally expressed in the human integumentary system.
See also
* List of cutaneous conditions caused by mutations in keratins
* List of target antigens in pemphigoid
* List of target antigens in p ...
* List of keratins The first sequences of keratins revealed that keratins could be grouped into two categories based on their sequence homologies. These two groups of keratins were named as type I and type II keratins. These two categories also represent the first two ...
References
External links
Composition and β-sheet structure of silk
Proteopedia page on keratins
{{Authority control Keratins Cytoskeleton Skin anatomy