tertiary hydroxyl groups on:
[Wikipedia]
[Google]
[Amazon]
 In chemistry, an alcohol is a type of
In chemistry, an alcohol is a type of
 Alcohols have a long history of myriad uses. For simple mono-alcohols, which is the focus on this article, the following are most important industrial alcohols:.
*methanol, mainly for the production of
Alcohols have a long history of myriad uses. For simple mono-alcohols, which is the focus on this article, the following are most important industrial alcohols:.
*methanol, mainly for the production of
Al(C2H5)3 + 9 C2H4 -> Al(C8H17)3
:Al(C8H17)3 + 3O + 3 H2O -> 3 HOC8H17 + Al(OH)3
The process generates a range of alcohols that are separated by RCH=CH2 + H2 + CO -> RCH2CH2CHO
:RCH2CH2CHO + 3 H2 -> RCH2CH2CH2OH
Such processes give fatty alcohols, which are useful for detergents.
 The hydroboration-oxidation and
The hydroboration-oxidation and
2 R-OH + 2 NaH -> 2 R-O-Na+ + 2 H2
: 2 R-OH + 2 Na -> 2 R-O-Na+ + H2
The acidity of alcohols is strongly affected by
 Alcohols may, likewise, be converted to alkyl bromides using
Alcohols may, likewise, be converted to alkyl bromides using 3 R-OH + PBr3 -> 3 RBr + H3PO3
In the Barton-McCombie deoxygenation an alcohol is deoxygenated to an alkane with
 Upon treatment with strong acids, alcohols undergo the E1
Upon treatment with strong acids, alcohols undergo the E1 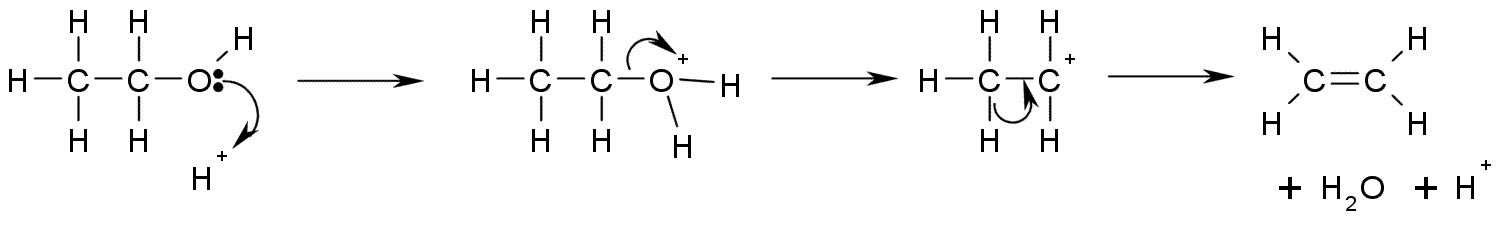 A more controlled elimination reaction requires the formation of the xanthate ester.
A more controlled elimination reaction requires the formation of the xanthate ester.
R-OH + R'-CO2H -> R'-CO2R + H2O
Other types of ester are prepared in a similar manner for example,
 Reagents useful for the transformation of primary alcohols to aldehydes are normally also suitable for the
Reagents useful for the transformation of primary alcohols to aldehydes are normally also suitable for the
 In chemistry, an alcohol is a type of
In chemistry, an alcohol is a type of organic compound
In chemistry, organic compounds are generally any chemical compounds that contain carbon-hydrogen or carbon-carbon bonds. Due to carbon's ability to catenate (form chains with other carbon atoms), millions of organic compounds are known. T ...
that carries at least one hydroxyl
In chemistry, a hydroxy or hydroxyl group is a functional group with the chemical formula and composed of one oxygen atom covalently bonded to one hydrogen atom. In organic chemistry, alcohols and carboxylic acids contain one or more hydro ...
() functional group
In organic chemistry, a functional group is a substituent or moiety in a molecule that causes the molecule's characteristic chemical reactions. The same functional group will undergo the same or similar chemical reactions regardless of the re ...
bound to a saturated
Saturation, saturated, unsaturation or unsaturated may refer to:
Chemistry
* Saturation, a property of organic compounds referring to carbon-carbon bonds
** Saturated and unsaturated compounds
**Degree of unsaturation
** Saturated fat or fatty ac ...
carbon atom. The term ''alcohol'' originally referred to the primary alcohol ethanol
Ethanol (abbr. EtOH; also called ethyl alcohol, grain alcohol, drinking alcohol, or simply alcohol) is an organic compound. It is an alcohol with the chemical formula . Its formula can be also written as or (an ethyl group linked to a ...
(ethyl alcohol), which is used as a drug and is the main alcohol present in alcoholic drink
An alcoholic beverage (also called an alcoholic drink, adult beverage, or a drink) is a drink that contains ethanol, a type of alcohol that acts as a drug and is produced by fermentation of grains, fruits, or other sources of sugar. The c ...
s. An important class of alcohols, of which methanol and ethanol are the simplest examples, includes all compounds which conform to the general formula . Simple monoalcohols that are the subject of this article include primary (), secondary () and tertiary () alcohols.
The suffix ''-ol'' appears in the IUPAC chemical name of all substances where the hydroxyl group is the functional group with the highest priority. When a higher priority group is present in the compound, the prefix ''hydroxy-'' is used in its IUPAC
The International Union of Pure and Applied Chemistry (IUPAC ) is an international federation of National Adhering Organizations working for the advancement of the chemical sciences, especially by developing nomenclature and terminology. It is ...
name. The suffix ''-ol'' in non-IUPAC names (such as paracetamol or cholesterol
Cholesterol is any of a class of certain organic molecules called lipids. It is a sterol (or modified steroid), a type of lipid. Cholesterol is biosynthesized by all animal cells and is an essential structural component of animal cell mem ...
) also typically indicates that the substance is an alcohol. However, some compounds that contain hydroxyl functional groups have ''trivial names'' which do not include the suffix ''-ol'' or the prefix ''hydroxy-'', e.g. the sugars glucose
Glucose is a simple sugar with the molecular formula . Glucose is overall the most abundant monosaccharide, a subcategory of carbohydrates. Glucose is mainly made by plants and most algae during photosynthesis from water and carbon dioxide, u ...
and sucrose.
History
The inflammable nature of the exhalations of wine was already known to ancient natural philosophers such asAristotle
Aristotle (; grc-gre, Ἀριστοτέλης ''Aristotélēs'', ; 384–322 BC) was a Greek philosopher and polymath during the Classical period in Ancient Greece. Taught by Plato, he was the founder of the Peripatetic school of ph ...
(384–322 BCE), Theophrastus
Theophrastus (; grc-gre, Θεόφραστος ; c. 371c. 287 BC), a Greek philosopher and the successor to Aristotle in the Peripatetic school. He was a native of Eresos in Lesbos.Gavin Hardy and Laurence Totelin, ''Ancient Botany'', Routle ...
(–287 BCE), and Pliny the Elder
Gaius Plinius Secundus (AD 23/2479), called Pliny the Elder (), was a Roman author, naturalist and natural philosopher, and naval and army commander of the early Roman Empire, and a friend of the emperor Vespasian. He wrote the encyclopedic ' ...
(23/24–79 CE). However, this did not immediately lead to the isolation of alcohol, even despite the development of more advanced distillation techniques in second- and third-century Roman Egypt. An important recognition, first found in one of the writings attributed to Jābir ibn Ḥayyān (ninth century CE), was that by adding salt to boiling wine, which increases the wine's relative volatility Relative volatility is a measure comparing the vapor pressures of the components in a liquid mixture of chemicals. This quantity is widely used in designing large industrial distillation processes. In effect, it indicates the ease or difficulty of u ...
, the flammability of the resulting vapors may be enhanced. The distillation of wine is attested in Arabic works attributed to al-Kindī
Abū Yūsuf Yaʻqūb ibn ʼIsḥāq aṣ-Ṣabbāḥ al-Kindī (; ar, أبو يوسف يعقوب بن إسحاق الصبّاح الكندي; la, Alkindus; c. 801–873 AD) was an Arab Muslim philosopher, polymath, mathematician, physician ...
(–873 CE) and to al-Fārābī (–950), and in the 28th book of al-Zahrāwī's (Latin: Abulcasis, 936–1013) ''Kitāb al-Taṣrīf'' (later translated into Latin as ''Liber servatoris''). In the twelfth century, recipes for the production of ''aqua ardens'' ("burning water", i.e., alcohol) by distilling wine with salt started to appear in a number of Latin works, and by the end of the thirteenth century it had become a widely known substance among Western European chemists.
The works of Taddeo Alderotti
Taddeo Alderotti (Latin: Thaddaeus Alderottus, French : Thaddée de Florence), born in Florence between 1206 and 1215, died in 1295, was an Italian doctor and professor of medicine at the University of Bologna, who made important contributions t ...
(1223–1296) describe a method for concentrating alcohol involving repeated fractional distillation through a water-cooled still, by which an alcohol purity of 90% could be obtained. The medicinal properties of ethanol were studied by Arnald of Villanova
Arnaldus de Villa Nova (also called Arnau de Vilanova in Catalan, his language, Arnaldus Villanovanus, Arnaud de Ville-Neuve or Arnaldo de Villanueva, c. 1240–1311) was a physician and a religious reformer. He was also thought to be an alchem ...
(1240–1311 CE) and John of Rupescissa
:''Johannes de Rupescissa may also refer to Cardinal Jean de La Rochetaillée''
Jean de Roquetaillade, also known as John of Rupescissa, (ca. 1310 – between 1366 and 1370) was a French Franciscan alchemist and eschatologist.
Biography
Aft ...
(–1366), the latter of whom regarded it as a life-preserving substance able to prevent all diseases (the '' aqua vitae'' or "water of life", also called by John the '' quintessence'' of wine).
Nomenclature
Etymology
The word "alcohol" derives from the Arabic '' kohl'' ( ar, الكحل, al-kuḥl), a powder used as an eyeliner. The first part of the word () is the Arabicdefinite article
An article is any member of a class of dedicated words that are used with noun phrases to mark the identifiability of the referents of the noun phrases. The category of articles constitutes a part of speech.
In English, both "the" and "a(n)" a ...
, equivalent to ''the'' in English. The second part of the word () has several antecedents in Semitic languages
The Semitic languages are a branch of the Afroasiatic language family. They are spoken by more than 330 million people across much of West Asia, the Horn of Africa, and latterly North Africa, Malta, West Africa, Chad, and in large immigra ...
, ultimately deriving from the Akkadian Akkadian or Accadian may refer to:
* Akkadians, inhabitants of the Akkadian Empire
* Akkadian language, an extinct Eastern Semitic language
* Akkadian literature, literature in this language
* Akkadian cuneiform
Cuneiform is a logo- syllabi ...
𒎎𒋆𒁉𒍣𒁕 (guḫlum), meaning stibnite or antimony
Antimony is a chemical element with the symbol Sb (from la, stibium) and atomic number 51. A lustrous gray metalloid, it is found in nature mainly as the sulfide mineral stibnite (Sb2S3). Antimony compounds have been known since ancient t ...
.
Like its antecedents in Arabic and older languages, the term ''alcohol'' was originally used for the very fine powder produced by the sublimation of the natural mineral stibnite
Stibnite, sometimes called antimonite, is a sulfide mineral with the formula Sb2 S3. This soft grey material crystallizes in an orthorhombic space group. It is the most important source for the metalloid antimony. The name is derived from the ...
to form antimony trisulfide
Antimony trisulfide (Sb2S3) is found in nature as the crystalline mineral stibnite and the amorphous red mineral (actually a mineraloid) metastibnite. It is manufactured for use in safety matches, military ammunition, explosives and fireworks. It ...
. It was considered to be the essence or "spirit" of this mineral. It was used as an antiseptic
An antiseptic (from Greek ἀντί ''anti'', "against" and σηπτικός ''sēptikos'', "putrefactive") is an antimicrobial substance or compound that is applied to living tissue/skin to reduce the possibility of infection, sepsis, or putre ...
, eyeliner, and cosmetic. Later the meaning of alcohol was extended to distilled substances in general, and then narrowed again to ethanol, when "spirits" was a synonym for hard liquor
Liquor (or a spirit) is an alcoholic drink produced by distillation of grains, fruits, vegetables, or sugar, that have already gone through alcoholic fermentation. Other terms for liquor include: spirit drink, distilled beverage or hard l ...
.
Bartholomew Traheron
Bartholomew Traheron (1510?–1558?) was an English Protestant writer and Marian exile.
Life
Born about 1510, he is said to have been a native of Cornwall. Bartholomew was early left an orphan, and was brought up under the care of Richard Tracy ...
, in his 1543 translation of John of Vigo
John is a common English name and surname:
* John (given name)
* John (surname)
John may also refer to:
New Testament
Works
* Gospel of John, a title often shortened to John
* First Epistle of John, often shortened to 1 John
* Second ...
, introduces the word as a term used by "barbarous" authors for "fine powder." Vigo wrote: "the barbarous auctours use alcohol, or (as I fynde it sometymes wryten) alcofoll, for moost fine poudre."
The 1657 ''Lexicon Chymicum'', by William Johnson glosses the word as "antimonium sive stibium." By extension, the word came to refer to any fluid obtained by distillation, including "alcohol of wine," the distilled essence of wine. Libavius in ''Alchymia'' (1594) refers to "vini alcohol vel vinum alcalisatum". Johnson (1657) glosses ''alcohol vini'' as "quando omnis superfluitas vini a vino separatur, ita ut accensum ardeat donec totum consumatur, nihilque fæcum aut phlegmatis in fundo remaneat." The word's meaning became restricted to "spirit of wine" (the chemical known today as ethanol
Ethanol (abbr. EtOH; also called ethyl alcohol, grain alcohol, drinking alcohol, or simply alcohol) is an organic compound. It is an alcohol with the chemical formula . Its formula can be also written as or (an ethyl group linked to a ...
) in the 18th century and was extended to the class of substances so-called as "alcohols" in modern chemistry after 1850.
The term ''ethanol'' was invented in 1892, blending "ethane
Ethane ( , ) is an organic chemical compound with chemical formula . At standard temperature and pressure, ethane is a colorless, odorless gas. Like many hydrocarbons, ethane is isolated on an industrial scale from natural gas and as a petroc ...
" with the "-ol" ending of "alcohol", which was generalized as a libfix
In linguistics, a libfix is a productive bound morpheme affix created by rebracketing and back-formation, often a generalization of a component of a blended or portmanteau word. For example, ''walkathon'' was coined in 1932 as a blend of ''walk'' ...
.
Systematic names
IUPAC nomenclature
A chemical nomenclature is a set of rules to generate systematic names for chemical compounds. The nomenclature used most frequently worldwide is the one created and developed by the International Union of Pure and Applied Chemistry (IUPAC).
T ...
is used in scientific publications and where precise identification of the substance is important. In naming simple alcohols, the name of the alkane chain loses the terminal ''e'' and adds the suffix ''-ol'', ''e.g.'', as in "ethanol" from the alkane chain name "ethane". When necessary, the position of the hydroxyl group is indicated by a number between the alkane name and the ''-ol'': propan-1-ol
Propan-1-ol (also propanol, n-propyl alcohol) is a primary alcohol with the formula and sometimes represented as PrOH or ''n''-PrOH. It is a colorless liquid and an isomer of 2-propanol. It is formed naturally in small amounts during many ferm ...
for , propan-2-ol
Isopropyl alcohol (IUPAC name propan-2-ol and also called isopropanol or 2-propanol) is a colorless, flammable organic compound with a pungent alcoholic odor. As an isopropyl group linked to a hydroxyl group (chemical formula ) it is the simpl ...
for . If a higher priority group is present (such as an aldehyde
In organic chemistry, an aldehyde () is an organic compound containing a functional group with the structure . The functional group itself (without the "R" side chain) can be referred to as an aldehyde but can also be classified as a formyl grou ...
, ketone, or carboxylic acid), then the prefix ''hydroxy-''is used, e.g., as in 1-hydroxy-2-propanone (). Compounds having more than one hydroxy group are called polyol
In organic chemistry, a polyol is an organic compound containing multiple hydroxyl groups (). The term "polyol" can have slightly different meanings depending on whether it is used in food science or polymer chemistry. Polyols containing two, thr ...
s. They are named using suffixes -diol, -triol, etc., following a list of the position numbers of the hydroxyl groups, as in propane-1,2-diol
Propylene glycol (IUPAC name: propane-1,2-diol) is a viscous, colorless liquid, which is nearly odorless but possesses a faintly sweet taste. Its chemical formula is CH3CH(OH)CH2OH.
Containing two alcohol groups, it is classed as a diol. It is ...
for CH3CH(OH)CH2OH (propylene glycol).
In cases where the hydroxy group is bonded to an sp2 carbon on an aromatic ring
In chemistry, aromaticity is a chemical property of cyclic ( ring-shaped), ''typically'' planar (flat) molecular structures with pi bonds in resonance (those containing delocalized electrons) that gives increased stability compared to satur ...
, the molecule is classified separately as a phenol
Phenol (also called carbolic acid) is an aromatic organic compound with the molecular formula . It is a white crystalline solid that is volatile. The molecule consists of a phenyl group () bonded to a hydroxy group (). Mildly acidic, it ...
and is named using the IUPAC rules for naming phenols. Phenols have distinct properties and are not classified as alcohols.
Common names
In other less formal contexts, an alcohol is often called with the name of the corresponding alkyl group followed by the word "alcohol", e.g., methyl alcohol, ethyl alcohol.Propyl
In organic chemistry, propyl is a three-carbon alkyl substituent with chemical formula for the linear form. This substituent form is obtained by removing one hydrogen atom attached to the terminal carbon of propane. A propyl substituent is often ...
alcohol may be ''n''-propyl alcohol or isopropyl alcohol
Isopropyl alcohol (IUPAC name propan-2-ol and also called isopropanol or 2-propanol) is a colorless, flammable organic compound with a pungent alcoholic odor. As an isopropyl group linked to a hydroxyl group ( chemical formula ) it is the s ...
, depending on whether the hydroxyl group is bonded to the end or middle carbon on the straight propane chain. As described under systematic naming, if another group on the molecule takes priority, the alcohol moiety is often indicated using the "hydroxy-" prefix.
In archaic nomenclature, alcohols can be named as derivatives of methanol using "-carbinol" as the ending. For instance, can be named trimethylcarbinol.
Primary, secondary, and tertiary
Alcohols are then classified into primary, secondary (''sec-'', ''s-''), and tertiary (''tert-'', ''t-''), based upon the number of carbon atoms connected to the carbon atom that bears thehydroxyl
In chemistry, a hydroxy or hydroxyl group is a functional group with the chemical formula and composed of one oxygen atom covalently bonded to one hydrogen atom. In organic chemistry, alcohols and carboxylic acids contain one or more hydro ...
functional group
In organic chemistry, a functional group is a substituent or moiety in a molecule that causes the molecule's characteristic chemical reactions. The same functional group will undergo the same or similar chemical reactions regardless of the re ...
. (The respective numeric shorthands 1°, 2°, and 3° are sometimes used in informal settings.) The primary alcohols have general formulas . The simplest primary alcohol is methanol (), for which R=H, and the next is ethanol, for which , the methyl group
In organic chemistry, a methyl group is an alkyl derived from methane, containing one carbon atom bonded to three hydrogen atoms, having chemical formula . In formulas, the group is often abbreviated as Me. This hydrocarbon group occurs in ma ...
. Secondary alcohols are those of the form RR'CHOH, the simplest of which is 2-propanol (). For the tertiary alcohols the general form is RR'R"COH. The simplest example is tert-butanol
''tert''-Butyl alcohol is the simplest tertiary alcohol, with a formula of (CH3)3COH (sometimes represented as ''t''-BuOH). Its isomers are 1-butanol, isobutanol, and butan-2-ol. ''tert''-Butyl alcohol is a colorless solid, which melts near r ...
(2-methylpropan-2-ol), for which each of R, R', and R" is . In these shorthands, R, R', and R" represent substituents
A substituent is one or a group of atoms that replaces (one or more) atoms, thereby becoming a moiety in the resultant (new) molecule. (In organic chemistry and biochemistry, the terms ''substituent'' and ''functional group'', as well as ''side ...
, alkyl or other attached, generally organic groups.
Examples
Applications
 Alcohols have a long history of myriad uses. For simple mono-alcohols, which is the focus on this article, the following are most important industrial alcohols:.
*methanol, mainly for the production of
Alcohols have a long history of myriad uses. For simple mono-alcohols, which is the focus on this article, the following are most important industrial alcohols:.
*methanol, mainly for the production of formaldehyde
Formaldehyde ( , ) (systematic name methanal) is a naturally occurring organic compound with the formula and structure . The pure compound is a pungent, colourless gas that polymerises spontaneously into paraformaldehyde (refer to section ...
and as a fuel additive
Petrol additives increase petrol's octane rating or act as corrosion inhibitors or lubricants, thus allowing the use of higher compression ratios for greater efficiency and power. Types of additives include metal deactivators, corrosion inhib ...
*ethanol, mainly for alcoholic beverages, fuel additive, solvent
*1-propanol, 1-butanol, and isobutyl alcohol for use as a solvent and precursor to solvents
*C6–C11 alcohols used for plasticizer
A plasticizer ( UK: plasticiser) is a substance that is added to a material to make it softer and more flexible, to increase its plasticity, to decrease its viscosity, and/or to decrease friction during its handling in manufacture.
Plasticiz ...
s, e.g. in polyvinylchloride
*fatty alcohol (C12–C18), precursors to detergents
Methanol is the most common industrial alcohol, with about 12 million tons/y produced in 1980. The combined capacity of the other alcohols is about the same, distributed roughly equally.
Toxicity
With respect to acute toxicity, simple alcohols have low acutetoxic
Toxicity is the degree to which a chemical substance or a particular mixture of substances can damage an organism. Toxicity can refer to the effect on a whole organism, such as an animal, bacterium, or plant, as well as the effect on a subs ...
ities. Doses of several milliliters are tolerated. For pentanols, hexanol
Hexanol may refer to any of the following isomeric organic compounds with the formula C6H13OH:
:
See also
* Cyclohexanol
* Amyl alcohol
An amyl alcohol is any of eight alcohols with the formula C5H12O. A mixture of amyl alcohols (also called a ...
s, octanol
Octanols are alcohols with the formula C8H17OH. A simple and important member is 1-octanol, with an unbranched chain of carbons. Other commercially important octanols are 2-octanol and 2-ethylhexanol.
There are 89 possible isomers
In che ...
s and longer alcohols, LD50
In toxicology, the median lethal dose, LD50 (abbreviation for "lethal dose, 50%"), LC50 (lethal concentration, 50%) or LCt50 is a toxic unit that measures the lethal dose of a toxin, radiation, or pathogen. The value of LD50 for a substance is the ...
range from 2–5 g/kg (rats, oral). Ethanol is less acutely toxic. All alcohols are mild skin irritants.
The metabolism of methanol (and ethylene glycol
Ethylene glycol (IUPAC name: ethane-1,2-diol) is an organic compound (a vicinal diol) with the formula . It is mainly used for two purposes, as a raw material in the manufacture of polyester fibers and for antifreeze formulations. It is an odo ...
) is affected by the presence of ethanol, which has a higher affinity for liver alcohol dehydrogenase. In this way methanol will be excreted intact in urine.
Physical properties
In general, the hydroxyl group makes alcohols polar. Those groups can form hydrogen bonds to one another and to most other compounds. Owing to the presence of the polar OH alcohols are more water-soluble than simple hydrocarbons. Methanol, ethanol, and propanol are miscible in water.Butanol
Butanol (also called butyl alcohol) is a four-carbon alcohol with a formula of C4 H9 O H, which occurs in five isomeric structures (four structural isomers), from a straight-chain primary alcohol to a branched-chain tertiary alcohol; all are a bu ...
, with a four-carbon chain, is moderately soluble.
Because of hydrogen bonding
In chemistry, a hydrogen bond (or H-bond) is a primarily electrostatic force of attraction between a hydrogen (H) atom which is covalently bound to a more electronegative "donor" atom or group (Dn), and another electronegative atom bearing a l ...
, alcohols tend to have higher boiling points than comparable hydrocarbon
In organic chemistry, a hydrocarbon is an organic compound consisting entirely of hydrogen and carbon. Hydrocarbons are examples of group 14 hydrides. Hydrocarbons are generally colourless and hydrophobic, and their odors are usually weak or ...
s and ether
In organic chemistry, ethers are a class of compounds that contain an ether group—an oxygen atom connected to two alkyl or aryl groups. They have the general formula , where R and R′ represent the alkyl or aryl groups. Ethers can again be ...
s. The boiling point of the alcohol ethanol is 78.29 °C, compared to 69 °C for the hydrocarbon hexane
Hexane () is an organic compound, a straight-chain alkane with six carbon atoms and has the molecular formula C6H14.
It is a colorless liquid, odorless when pure, and with boiling points approximately . It is widely used as a cheap, relatively ...
, and 34.6 °C for diethyl ether
Diethyl ether, or simply ether, is an organic compound in the ether class with the formula , sometimes abbreviated as (see Pseudoelement symbols). It is a colourless, highly volatile, sweet-smelling ("ethereal odour"), extremely flammable li ...
.
Occurrence in nature
Simple alcohols are found widely in nature. Ethanol is the most prominent because it is the product of fermentation, a major energy-producing pathway. Other simple alcohols, chiefly fusel alcohols, are formed in only trace amounts. More complex alcohols however are pervasive, as manifested in sugars, some amino acids, and fatty acids.Production
Ziegler and oxo processes
In theZiegler process In organic chemistry, the Ziegler process (also called the Ziegler-Alfol synthesis) is a method for producing fatty alcohols from ethylene using an organoaluminium compound. The reaction produces linear primary alcohols with an even numbered carbon ...
, linear alcohols are produced from ethylene and triethylaluminium
Triethylaluminium is one of the simplest examples of an organoaluminium compound. Despite its name it has the formula Al2( C2H5)6 (abbreviated as Al2Et6 or TEA), as it exists as a dimer. This colorless liquid is pyrophoric. It is an industrially ...
followed by oxidation and hydrolysis. An idealized synthesis of 1-octanol
1-Octanol, also known as octan-1-ol, is the organic compound with the molecular formula CH3(CH2)7OH. It is a fatty alcohol. Many other isomers are also known generically as octanols. 1-Octanol is manufactured for the synthesis of esters for us ...
is shown:
:distillation
Distillation, or classical distillation, is the process of separating the components or substances from a liquid mixture by using selective boiling and condensation, usually inside an apparatus known as a still. Dry distillation is the heat ...
.
Many higher alcohols are produced by hydroformylation
Hydroformylation, also known as oxo synthesis or oxo process, is an industrial process for the production of aldehydes from alkenes. This chemical reaction entails the net addition of a formyl group (CHO) and a hydrogen atom to a carbon-carbon d ...
of alkenes followed by hydrogenation. When applied to a terminal alkene, as is common, one typically obtains a linear alcohol:
:Hydration reactions
Some low molecular weight alcohols of industrial importance are produced by the addition of water to alkenes. Ethanol, isopropanol, 2-butanol, and tert-butanol are produced by this general method. Two implementations are employed, the direct and indirect methods. The direct method avoids the formation of stable intermediates, typically using acid catalysts. In the indirect method, the alkene is converted to the sulfate ester, which is subsequently hydrolyzed. The direct hydration using ethylene ( ethylene hydration) or other alkenes from cracking of fractions of distilled crude oil. Hydration is also used industrially to produce the diolethylene glycol
Ethylene glycol (IUPAC name: ethane-1,2-diol) is an organic compound (a vicinal diol) with the formula . It is mainly used for two purposes, as a raw material in the manufacture of polyester fibers and for antifreeze formulations. It is an odo ...
from ethylene oxide
Ethylene oxide is an organic compound with the formula . It is a cyclic ether and the simplest epoxide: a three-membered ring consisting of one oxygen atom and two carbon atoms. Ethylene oxide is a colorless and flammable gas with a faintly sw ...
.
Biological routes
Ethanol is obtained by fermentation usingglucose
Glucose is a simple sugar with the molecular formula . Glucose is overall the most abundant monosaccharide, a subcategory of carbohydrates. Glucose is mainly made by plants and most algae during photosynthesis from water and carbon dioxide, u ...
produced from sugar from the hydrolysis
Hydrolysis (; ) is any chemical reaction in which a molecule of water breaks one or more chemical bonds. The term is used broadly for substitution, elimination, and solvation reactions in which water is the nucleophile.
Biological hydrolys ...
of starch, in the presence of yeast and temperature of less than 37 °C to produce ethanol. For instance, such a process might proceed by the conversion of sucrose by the enzyme invertase
Invertase is an enzyme that catalyzes the hydrolysis (breakdown) of sucrose (table sugar) into fructose and glucose. Alternative names for invertase include , saccharase, glucosucrase, beta-h-fructosidase, beta-fructosidase, invertin, sucrase, m ...
into glucose and fructose, then the conversion of glucose by the enzyme complex zymase
Zymase is an enzyme complex that catalyzes the fermentation of sugar into ethanol and carbon dioxide. It occurs naturally in yeasts. Zymase activity varies among yeast strains.
Zymase is also the brand name of the drug pancrelipase.
Cell-free ...
into ethanol and carbon dioxide.
Several species of the benign bacteria in the intestine use fermentation as a form of anaerobic metabolism
Anaerobic respiration is respiration using electron acceptors other than molecular oxygen (O2). Although oxygen is not the final electron acceptor, the process still uses a respiratory electron transport chain.
In aerobic organisms undergoing r ...
. This metabolic reaction produces ethanol as a waste product. Thus, human bodies contain some quantity of alcohol endogenously produced by these bacteria. In rare cases, this can be sufficient to cause " auto-brewery syndrome" in which intoxicating quantities of alcohol are produced.
Like ethanol, butanol
Butanol (also called butyl alcohol) is a four-carbon alcohol with a formula of C4 H9 O H, which occurs in five isomeric structures (four structural isomers), from a straight-chain primary alcohol to a branched-chain tertiary alcohol; all are a bu ...
can be produced by fermentation processes. ''Saccharomyces'' yeast are known to produce these higher alcohols at temperatures above . The bacterium ''Clostridium acetobutylicum
''Clostridium acetobutylicum'', ATCC 824, is a commercially valuable bacterium sometimes called the "Weizmann Organism", after Jewish Russian-born biochemist Chaim Weizmann. A senior lecturer at the University of Manchester, England, he used th ...
'' can feed on cellulose
Cellulose is an organic compound with the formula , a polysaccharide consisting of a linear chain of several hundred to many thousands of β(1→4) linked D-glucose units. Cellulose is an important structural component of the primary cell w ...
to produce butanol on an industrial scale.
Substitution
Primaryalkyl halide
The haloalkanes (also known as halogenoalkanes or alkyl halides) are alkanes containing one or more halogen substituents. They are a subset of the general class of halocarbons, although the distinction is not often made. Haloalkanes are widely us ...
s react with aqueous NaOH
Sodium hydroxide, also known as lye and caustic soda, is an inorganic compound with the formula NaOH. It is a white solid ionic compound consisting of sodium cations and hydroxide anions .
Sodium hydroxide is a highly caustic base and alkali ...
or KOH mainly to primary alcohols in nucleophilic aliphatic substitution
In chemistry, a nucleophilic substitution is a class of chemical reactions in which an electron-rich chemical species (known as a nucleophile) replaces a functional group within another electron-deficient molecule (known as the electrophile). Th ...
. (Secondary and especially tertiary alkyl halides will give the elimination (alkene) product instead). Grignard reagents react with carbonyl
In organic chemistry, a carbonyl group is a functional group composed of a carbon atom double-bonded to an oxygen atom: C=O. It is common to several classes of organic compounds, as part of many larger functional groups. A compound containi ...
groups to secondary and tertiary alcohols. Related reactions are the Barbier reaction
The Barbier reaction is an organometallic reaction between an alkyl halide (chloride, bromide, iodide), a carbonyl group and a metal. The reaction can be performed using magnesium, aluminium, zinc, indium, tin, samarium, barium or their salts. ...
and the Nozaki-Hiyama reaction.
Reduction
Aldehydes
In organic chemistry, an aldehyde () is an organic compound containing a functional group with the structure . The functional group itself (without the "R" side chain) can be referred to as an aldehyde but can also be classified as a formyl grou ...
or ketones are reduced with sodium borohydride
Sodium borohydride, also known as sodium tetrahydridoborate and sodium tetrahydroborate, is an inorganic compound with the formula Na BH4. This white solid, usually encountered as an aqueous basic solution, is a reducing agent that finds applica ...
or lithium aluminium hydride (after an acidic workup). Another reduction by aluminiumisopropylates is the Meerwein-Ponndorf-Verley reduction. Noyori asymmetric hydrogenation
In chemistry, the Noyori asymmetric hydrogenation refers to methodology for enantioselective reduction of ketones and related functional groups. This methodology was introduced by Ryoji Noyori, who shared the Nobel Prize in Chemistry in 2001 for c ...
is the asymmetric reduction of β-keto-esters.
Hydrolysis
Alkenes
In organic chemistry, an alkene is a hydrocarbon containing a carbon–carbon double bond.
Alkene is often used as synonym of olefin, that is, any hydrocarbon containing one or more double bonds.H. Stephen Stoker (2015): General, Organic, an ...
engage in an acid catalysed hydration reaction using concentrated sulfuric acid as a catalyst that gives usually secondary or tertiary alcohols. Formation of a secondary alcohol via alkene reduction and hydration is shown on the right:
:oxymercuration-reduction
The oxymercuration reaction is an electrophilic addition organic reaction that transforms an alkene into a neutral alcohol. In oxymercuration, the alkene reacts with mercuric acetate (AcO–Hg–OAc) in aqueous solution to yield the addition of an ...
of alkenes are more reliable in organic synthesis. Alkenes react with N-bromosuccinimide
''N''-Bromosuccinimide or NBS is a chemical reagent used in radical substitution, electrophilic addition, and electrophilic substitution reactions in organic chemistry. NBS can be a convenient source of Br•, the bromine radical.
Preparatio ...
and water in halohydrin formation reaction
In organic chemistry a halohydrin (also a haloalcohol or β-halo alcohol) is a functional group in which a halogen and a hydroxyl are bonded to adjacent carbon atoms, which otherwise bear only hydrogen or hydrocarbyl groups (e.g. 2-chloroethan ...
. Amine
In chemistry, amines (, ) are compounds and functional groups that contain a basic nitrogen atom with a lone pair. Amines are formally derivatives of ammonia (), wherein one or more hydrogen
Hydrogen is the chemical element wi ...
s can be converted to diazonium salt
Diazonium compounds or diazonium salts are a group of organic compounds sharing a common functional group where R can be any organic group, such as an alkyl or an aryl, and X is an inorganic or organic anion, such as a halide.
General properti ...
s, which are then hydrolyzed.
Reactions
Deprotonation
With aqueous p''K''a values of around 16–19, they are, in general, slightly weaker acids thanwater
Water (chemical formula ) is an Inorganic compound, inorganic, transparent, tasteless, odorless, and Color of water, nearly colorless chemical substance, which is the main constituent of Earth's hydrosphere and the fluids of all known living ...
. With strong bases such as sodium hydride
Sodium hydride is the chemical compound with the empirical formula Na H. This alkali metal hydride is primarily used as a strong yet combustible base in organic synthesis. NaH is a saline (salt-like) hydride, composed of Na+ and H− ions, in co ...
or sodium
Sodium is a chemical element with the symbol Na (from Latin ''natrium'') and atomic number 11. It is a soft, silvery-white, highly reactive metal. Sodium is an alkali metal, being in group 1 of the periodic table. Its only stable ...
they form salt
Salt is a mineral composed primarily of sodium chloride (NaCl), a chemical compound belonging to the larger class of salts; salt in the form of a natural crystalline mineral is known as rock salt or halite. Salt is present in vast quant ...
s called '' alkoxides'', with the general formula (where R is an alkyl
In organic chemistry, an alkyl group is an alkane missing one hydrogen.
The term ''alkyl'' is intentionally unspecific to include many possible substitutions.
An acyclic alkyl has the general formula of . A cycloalkyl is derived from a cycloa ...
and M is a metal
A metal (from Greek μέταλλον ''métallon'', "mine, quarry, metal") is a material that, when freshly prepared, polished, or fractured, shows a lustrous appearance, and conducts electricity and heat relatively well. Metals are typicall ...
).
: solvation
Solvation (or dissolution) describes the interaction of a solvent with dissolved molecules. Both ionized and uncharged molecules interact strongly with a solvent, and the strength and nature of this interaction influence many properties of the ...
. In the gas phase, alcohols are more acidic than in water. In DMSO, alcohols (and water) have a p''K''a of around 29–32. As a consequence, alkoxides (and hydroxide) are powerful bases and nucleophiles (e.g., for the Williamson ether synthesis
The Williamson ether synthesis is an organic reaction, forming an ether from an organohalide and a deprotonated alcohol (alkoxide). This reaction was developed by Alexander Williamson in 1850. Typically it involves the reaction of an alkoxide io ...
) in this solvent. In particular, or in DMSO can be used to generate significant equilibrium concentrations of acetylide ions through the deprotonation of alkynes (see Favorskii reaction
The Favorskii reaction is an organic chemistry reaction between an alkyne and a carbonyl group, under base (chemistry), basic conditions. The reaction was discovered in the early 1900s by the Russian chemist Alexei Yevgrafovich Favorskii.
When ...
).
Nucleophilic substitution
The OH group is not a goodleaving group In chemistry, a leaving group is defined by the IUPAC as an atom or group of atoms that detaches from the main or residual part of a substrate during a reaction or elementary step of a reaction. However, in common usage, the term is often limited ...
in nucleophilic substitution
In chemistry, a nucleophilic substitution is a class of chemical reactions in which an electron-rich chemical species (known as a nucleophile) replaces a functional group within another electron-deficient molecule (known as the electrophile). The ...
reactions, so neutral alcohols do not react in such reactions. However, if the oxygen is first protonated
In chemistry, protonation (or hydronation) is the adding of a proton (or hydron, or hydrogen cation), (H+) to an atom, molecule, or ion, forming a conjugate acid. (The complementary process, when a proton is removed from a Brønsted–Lowry acid, ...
to give , the leaving group (water
Water (chemical formula ) is an Inorganic compound, inorganic, transparent, tasteless, odorless, and Color of water, nearly colorless chemical substance, which is the main constituent of Earth's hydrosphere and the fluids of all known living ...
) is much more stable, and the nucleophilic substitution can take place. For instance, tertiary alcohols react with hydrochloric acid
Hydrochloric acid, also known as muriatic acid, is an aqueous solution of hydrogen chloride. It is a colorless solution with a distinctive pungent smell. It is classified as a strong acid
Acid strength is the tendency of an acid, symbol ...
to produce tertiary alkyl halide
The haloalkanes (also known as halogenoalkanes or alkyl halides) are alkanes containing one or more halogen substituents. They are a subset of the general class of halocarbons, although the distinction is not often made. Haloalkanes are widely us ...
s, where the hydroxyl group is replaced by a chlorine
Chlorine is a chemical element with the symbol Cl and atomic number 17. The second-lightest of the halogens, it appears between fluorine and bromine in the periodic table and its properties are mostly intermediate between them. Chlorine i ...
atom by unimolecular nucleophilic substitution. If primary or secondary alcohols are to be reacted with hydrochloric acid
Hydrochloric acid, also known as muriatic acid, is an aqueous solution of hydrogen chloride. It is a colorless solution with a distinctive pungent smell. It is classified as a strong acid
Acid strength is the tendency of an acid, symbol ...
, an activator such as zinc chloride
Zinc chloride is the name of inorganic chemical compounds with the formula ZnCl2 and its hydrates. Zinc chlorides, of which nine crystalline forms are known, are colorless or white, and are highly soluble in water. This salt is hygroscopic and e ...
is needed. In alternative fashion, the conversion may be performed directly using thionyl chloride
Thionyl chloride is an inorganic compound with the chemical formula . It is a moderately volatile, colourless liquid with an unpleasant acrid odour. Thionyl chloride is primarily used as a chlorinating reagent, with approximately per year bein ...
.
hydrobromic acid
Hydrobromic acid is a strong acid formed by dissolving the diatomic molecule hydrogen bromide (HBr) in water. "Constant boiling" hydrobromic acid is an aqueous solution that distills at and contains 47.6% HBr by mass, which is 8.77 mol/L. ...
or phosphorus tribromide
Phosphorus tribromide is a colourless liquid with the formula P Br3. The liquid fumes in moist air due to hydrolysis and has a penetrating odour. It is used in the laboratory for the conversion of alcohols to alkyl bromides.
Preparation
PBr3 ...
, for example:
: tributyltin hydride
Tributyltin hydride is an organotin compound with the formula (C4H9)3SnH. It is a colorless liquid that is soluble in organic solvents. The compound is used as a source of hydrogen atoms in organic synthesis.
Synthesis and characterization
The c ...
or a trimethylborane
Trimethylborane (TMB) is a toxic, pyrophoric gas with the formula B(CH3)3 (which can also be written as Me3B, with Me representing methyl).
Properties
As a liquid it is colourless. The strongest line in the infrared spectrum is at 1330 cm� ...
-water complex in a radical substitution
In organic chemistry, a radical-substitution reaction is a substitution reaction involving free radicals as a reactive intermediate.March Jerry; (1985). Advanced organic chemistry reactions, mechanisms and structure (3rd ed.). New York: John Wile ...
reaction.
Dehydration
Meanwhile, the oxygen atom has lone pairs of nonbonded electrons that render it weakly basic in the presence of strong acids such as sulfuric acid. For example, with methanol:elimination reaction
An elimination reaction is a type of organic reaction in which two substituents are removed from a molecule in either a one- or two-step mechanism. The one-step mechanism is known as the E2 reaction, and the two-step mechanism is known as the E1 r ...
to produce alkene
In organic chemistry, an alkene is a hydrocarbon containing a carbon–carbon double bond.
Alkene is often used as synonym of olefin, that is, any hydrocarbon containing one or more double bonds.H. Stephen Stoker (2015): General, Organic, an ...
s. The reaction, in general, obeys Zaitsev's Rule
In organic chemistry, Zaitsev's rule (or Saytzeff's rule, Saytzev's rule) is an empirical rule for predicting the favored alkene product(s) in elimination reactions. While at the University of Kazan, Russian chemist Alexander Zaitsev studied a v ...
, which states that the most stable (usually the most substituted) alkene is formed. Tertiary alcohols eliminate easily at just above room temperature, but primary alcohols require a higher temperature.
This is a diagram of acid catalysed dehydration of ethanol to produce ethylene:
 A more controlled elimination reaction requires the formation of the xanthate ester.
A more controlled elimination reaction requires the formation of the xanthate ester.
Protonolysis
Tertiary alcohols react with strong acids to generate carbocations. The reaction is related to their dehydration, e.g. isobutylene from tert-butyl alcohol. A special kind of dehydration reaction involvestriphenylmethanol
Triphenylmethanol (also known as triphenylcarbinol, TrOH) is an organic compound. It is a white crystalline solid that is insoluble in water and petroleum ether, but well soluble in ethanol, diethyl ether, and benzene. In strongly acidic solutions ...
and especially its amine-substituted derivatives. When treated with acid, these alcohols lose water to give stable carbocations, which are commercial dyes.
322px, Preparation of crystal violet by protonolysis of the tertiary alcohol.
Esterification
Alcohol and carboxylic acids react in the so-called Fischer esterification. The reaction usually requires acatalyst
Catalysis () is the process of increasing the rate of a chemical reaction by adding a substance known as a catalyst (). Catalysts are not consumed in the reaction and remain unchanged after it. If the reaction is rapid and the catalyst recyc ...
, such as concentrated sulfuric acid:
: tosyl
In organic chemistry, a toluenesulfonyl group (tosyl group, abbreviated Ts or Tos) is a univalent functional group with the chemical formula –. It consists of a Toluene, tolyl group, –, joined to a sulfonyl group, ––, with the open vale ...
(tosylate) esters are made by reaction of the alcohol with p- toluenesulfonyl chloride in pyridine.
Oxidation
Primary alcohols () can be oxidized either toaldehyde
In organic chemistry, an aldehyde () is an organic compound containing a functional group with the structure . The functional group itself (without the "R" side chain) can be referred to as an aldehyde but can also be classified as a formyl grou ...
s () or to carboxylic acids (). The oxidation of secondary alcohols () normally terminates at the ketone () stage. Tertiary alcohols () are resistant to oxidation.
The direct oxidation of primary alcohols to carboxylic acids
The oxidation of primary alcohols to carboxylic acids is an important oxidation reaction in organic chemistry.
When a primary alcohol is converted to a carboxylic acid, the terminal carbon atom increases its oxidation state by four. Oxidants able ...
normally proceeds via the corresponding aldehyde, which is transformed via an ''aldehyde hydrate'' () by reaction with water before it can be further oxidized to the carboxylic acid.
 Reagents useful for the transformation of primary alcohols to aldehydes are normally also suitable for the
Reagents useful for the transformation of primary alcohols to aldehydes are normally also suitable for the oxidation of secondary alcohols to ketones
The oxidation of secondary alcohols to ketones is an important oxidation reaction in organic chemistry.
When a secondary alcohol is oxidised, it is converted to a ketone. The hydrogen from the hydroxyl group is lost along with the hydrogen bond ...
. These include Collins reagent
Collins reagent is the complex of chromium(VI) oxide with pyridine in dichloromethane. This metal-pyridine complex, a red solid, is used to oxidize primary alcohols to the corresponding aldehydes and secondary alcohols to the corresponding keton ...
and Dess-Martin periodinane. The direct oxidation of primary alcohols to carboxylic acids can be carried out using potassium permanganate
Potassium permanganate is an inorganic compound with the chemical formula KMnO4. It is a purplish-black crystalline salt, that dissolves in water as K+ and , an intensely pink to purple solution.
Potassium permanganate is widely used in the c ...
or the Jones reagent
Jones may refer to:
People
*Jones (surname), a common Welsh and English surname
*List of people with surname Jones
* Jones (singer), a British singer-songwriter
Arts and entertainment
* Jones (''Animal Farm''), a human character in George Orwell ...
.
See also
*Enol
In organic chemistry, alkenols (shortened to enols) are a type of reactive structure or intermediate in organic chemistry that is represented as an alkene ( olefin) with a hydroxyl group attached to one end of the alkene double bond (). The t ...
* Ethanol fuel
Ethanol fuel is ethyl alcohol, the same type of alcohol found in alcoholic beverages, used as fuel. It is most often used as a motor fuel, mainly as a biofuel additive for gasoline. The first production car running entirely on ethanol was t ...
* Fatty alcohol
* Index of alcohol-related articles
* List of alcohols
* Lucas test
* Polyol
In organic chemistry, a polyol is an organic compound containing multiple hydroxyl groups (). The term "polyol" can have slightly different meanings depending on whether it is used in food science or polymer chemistry. Polyols containing two, thr ...
* Rubbing alcohol
Rubbing alcohol is either an isopropyl alcohol or an ethanol-based liquid, with isopropyl alcohol products being the most widely available. The comparable ''British Pharmacopoeia'' (''BP'') is surgical spirit. Rubbing alcohol is denatured and un ...
* Sugar alcohol
Sugar alcohols (also called polyhydric alcohols, polyalcohols, alditols or glycitols) are organic compounds, typically derived from sugars, containing one hydroxyl group (–OH) attached to each carbon atom. They are white, water-soluble solids ...
* Transesterification
In organic chemistry, transesterification is the process of exchanging the organic group R″ of an ester with the organic group R' of an alcohol. These reactions are often catalyzed by the addition of an acid or base catalyst. The reaction ca ...
Notes
Citations
General references
* {{Authority control Antiseptics Functional groups