hydroboration–oxidation reaction on:
[Wikipedia]
[Google]
[Amazon]
Hydroboration–oxidation reaction is a two-step hydration reaction that converts an 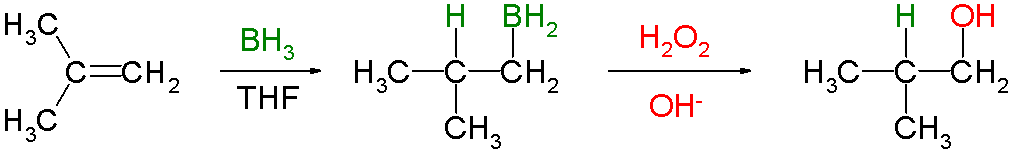
 Knowing that the group containing the boron will be replaced by a hydroxyl group, it can be seen that the initial hydroboration step determines the regioselectivity. Hydroboration proceeds in an anti-Markovnikov manner. The reaction sequence is also stereospecific, giving syn addition (on the same face of the alkene): the hydroboration is syn-selective and the oxidation replaces the boron with hydroxyl having the same geometric position. Thus 1-methylcyclopentene reacts with diborane predominantly to give ''trans''-1-hydroxy-2-methylcyclopentane—the newly added H and OH are ''cis'' to each other.
Until all hydrogens attached to boron have been transferred away, the boron group BH2 will continue adding to more alkenes. This means that one mole of hydroborane will undergo the reaction with three moles of alkene. Furthermore, it is not necessary for the hydroborane to have more than one hydrogen. For example, reagents of the type R2BH are commonly used, where R can represents the remainder of the molecule. Such modified hydroboration reagents include
Knowing that the group containing the boron will be replaced by a hydroxyl group, it can be seen that the initial hydroboration step determines the regioselectivity. Hydroboration proceeds in an anti-Markovnikov manner. The reaction sequence is also stereospecific, giving syn addition (on the same face of the alkene): the hydroboration is syn-selective and the oxidation replaces the boron with hydroxyl having the same geometric position. Thus 1-methylcyclopentene reacts with diborane predominantly to give ''trans''-1-hydroxy-2-methylcyclopentane—the newly added H and OH are ''cis'' to each other.
Until all hydrogens attached to boron have been transferred away, the boron group BH2 will continue adding to more alkenes. This means that one mole of hydroborane will undergo the reaction with three moles of alkene. Furthermore, it is not necessary for the hydroborane to have more than one hydrogen. For example, reagents of the type R2BH are commonly used, where R can represents the remainder of the molecule. Such modified hydroboration reagents include
 The 'H' atom in the reaction comes from B2H6, the 'O' atom comes from hydrogen peroxide (H2O2) whereas the O attached 'H' atom comes from the solvent (refer mechanism).
The 'H' atom in the reaction comes from B2H6, the 'O' atom comes from hydrogen peroxide (H2O2) whereas the O attached 'H' atom comes from the solvent (refer mechanism).
https://www.organic-chemistry.org/namedreactions/brown-hydroboration.shtm
{{DEFAULTSORT:Hydroboration-oxidation reaction Addition reactions
alkene
In organic chemistry, an alkene is a hydrocarbon containing a carbon–carbon double bond.
Alkene is often used as synonym of olefin, that is, any hydrocarbon containing one or more double bonds.H. Stephen Stoker (2015): General, Organic, an ...
into an alcohol. The process results in the syn addition of a hydrogen and a hydroxyl
In chemistry, a hydroxy or hydroxyl group is a functional group with the chemical formula and composed of one oxygen atom covalently bonded to one hydrogen atom. In organic chemistry, alcohols and carboxylic acids contain one or more hydroxy ...
group where the double bond
In chemistry, a double bond is a covalent bond between two atoms involving four bonding electrons as opposed to two in a single bond. Double bonds occur most commonly between two carbon atoms, for example in alkenes. Many double bonds exist betwee ...
had been. Hydroboration–oxidation is an anti-Markovnikov reaction, with the hydroxyl group attaching to the less-substituted carbon. The reaction thus provides a more stereospecific and complementary regiochemical alternative to other hydration reactions such as acid-catalyzed addition and the oxymercuration–reduction process. The reaction was first reported by Herbert C. Brown
Herbert Charles Brown (May 22, 1912 – December 19, 2004) was an American chemist and recipient of the 1979 Nobel Prize in Chemistry for his work with organoboranes.
Life and career
Brown was born Herbert Brovarnik in London, to Ukrainian Jewis ...
in the late 1950s and it was recognized in his receiving the Nobel Prize in Chemistry
)
, image = Nobel Prize.png
, alt = A golden medallion with an embossed image of a bearded man facing left in profile. To the left of the man is the text "ALFR•" then "NOBEL", and on the right, the text (smaller) "NAT•" then "M ...
in 1979.
The general form of the reaction is as follows:

Tetrahydrofuran
Tetrahydrofuran (THF), or oxolane, is an organic compound with the formula (CH2)4O. The compound is classified as heterocyclic compound, specifically a cyclic ether. It is a colorless, water- miscible organic liquid with low viscosity. It is ...
(THF) is the archetypal solvent
A solvent (s) (from the Latin '' solvō'', "loosen, untie, solve") is a substance that dissolves a solute, resulting in a solution. A solvent is usually a liquid but can also be a solid, a gas, or a supercritical fluid. Water is a solvent for ...
used for hydroboration.
Mechanism and scope
Hydroboration step
In the first step,borane
Trihydridoboron, also known as borane or borine, is an unstable and highly reactive molecule with the chemical formula . The preparation of borane carbonyl, BH3(CO), played an important role in exploring the chemistry of boranes, as it indicated ...
(BH3) adds to the double bond
In chemistry, a double bond is a covalent bond between two atoms involving four bonding electrons as opposed to two in a single bond. Double bonds occur most commonly between two carbon atoms, for example in alkenes. Many double bonds exist betwee ...
, transferring one of the hydrogen atoms to the carbon adjacent to the one that becomes bonded to the boron
Boron is a chemical element with the symbol B and atomic number 5. In its crystalline form it is a brittle, dark, lustrous metalloid; in its amorphous form it is a brown powder. As the lightest element of the ''boron group'' it has th ...
. This hydroboration
In organic chemistry, hydroboration refers to the addition of a hydrogen-boron bond to certain double and triple bonds involving carbon (, , , and ). This chemical reaction is useful in the organic synthesis of organic compounds.
Hydroboration p ...
is repeated two additional times, successively reacting each B–H bond so that three alkenes add to each BH3. The resulting trialkylborane is treated with hydrogen peroxide in the second step. This process replaces the B-C bonds with HO-C bonds. The boron reagent is converted to boric acid
Boric acid, more specifically orthoboric acid, is a compound of boron, oxygen, and hydrogen with formula . It may also be called hydrogen borate or boracic acid. It is usually encountered as colorless crystals or a white powder, that dissolve ...
. The reaction was originally described by H.C. Brown in 1957 for the conversion of 1-hexene into 1-hexanol.
9-BBN
9-Borabicyclo .3.1onane or 9-BBN is an organoborane compound. This colourless solid is used in organic chemistry as a hydroboration reagent. The compound exists as a hydride-bridged dimer, which easily cleaves in the presence of reducible substr ...
, catecholborane, and disiamylborane
Disiamylborane (bis(1,2-dimethylpropyl)borane, Sia2BH) is an organoborane used in organic synthesis. It is used for hydroboration–oxidation reactions of terminal alkynes, giving aldehydes via anti-Markovnikov hydration followed by tautomerizati ...
.
Oxidation step
In the second step of the reaction sequence, the nucleophilichydroperoxide
Hydroperoxides or peroxols are compounds containing the hydroperoxide functional group (ROOH). If the R is organic, the compounds are called organic hydroperoxides. Such compounds are a subset of organic peroxides, which have the formula ROOR. ...
anion
An ion () is an atom or molecule with a net electrical charge.
The charge of an electron is considered to be negative by convention and this charge is equal and opposite to the charge of a proton, which is considered to be positive by conve ...
attacks the boron atom. Alkyl migration to oxygen gives the alkyl borane with retention of stereochemistry (in reality, the reaction occurs via the trialkyl borate
A borate is any of several boron oxyanions, negative ions consisting of boron and oxygen, such as orthoborate , metaborate , or tetraborate ; or any salt with such anions, such as sodium metaborate, and disodium tetraborate . The name also re ...
B(OR)3, rather than the monoalkyl borinic ester BH2OR).
Alkyne hydroboration
A hydroboration reaction also takes place on alkynes. Again the mode of action is ''syn'' and secondary reaction products arealdehyde
In organic chemistry, an aldehyde () is an organic compound containing a functional group with the structure . The functional group itself (without the "R" side chain) can be referred to as an aldehyde but can also be classified as a formyl grou ...
s from terminal alkynes and ketones from internal alkynes. In order to prevent hydroboration across both the pi-bonds, a bulky borane like disiamyl (di-sec-iso-amyl) borane is used.
Alternative oxidations
Use of other oxidants instead of hydrogen peroxide can lead to carbonyl products rather than alcohols from alkenes. ''N''-Methylmorpholine ''N''-oxide with catalytictetrapropylammonium perruthenate
Tetrapropylammonium perruthenate (TPAP or TPAPR) is the chemical compound described by the formula N(C3H7)4RuO4. Sometimes known as the Ley–Griffith reagent, this ruthenium compound is used as a reagent in organic synthesis. This salt consists ...
converts the alkylborane into a carbonyl, thus a ketone or aldehyde
In organic chemistry, an aldehyde () is an organic compound containing a functional group with the structure . The functional group itself (without the "R" side chain) can be referred to as an aldehyde but can also be classified as a formyl grou ...
product depending on what other groups were attached to that carbon in the original alkene. Various dichromates or related chromium(VI) reagents give ketones as well, but give carboxylic acids instead of aldehydes for terminal alkenes.
References
External links
* Organic Chemistry Portal. Hydroboration (including recent literature)https://www.organic-chemistry.org/namedreactions/brown-hydroboration.shtm
{{DEFAULTSORT:Hydroboration-oxidation reaction Addition reactions