Silver Compounds on:
[Wikipedia]
[Google]
[Amazon]

 Silver and gold have rather low
Silver and gold have rather low
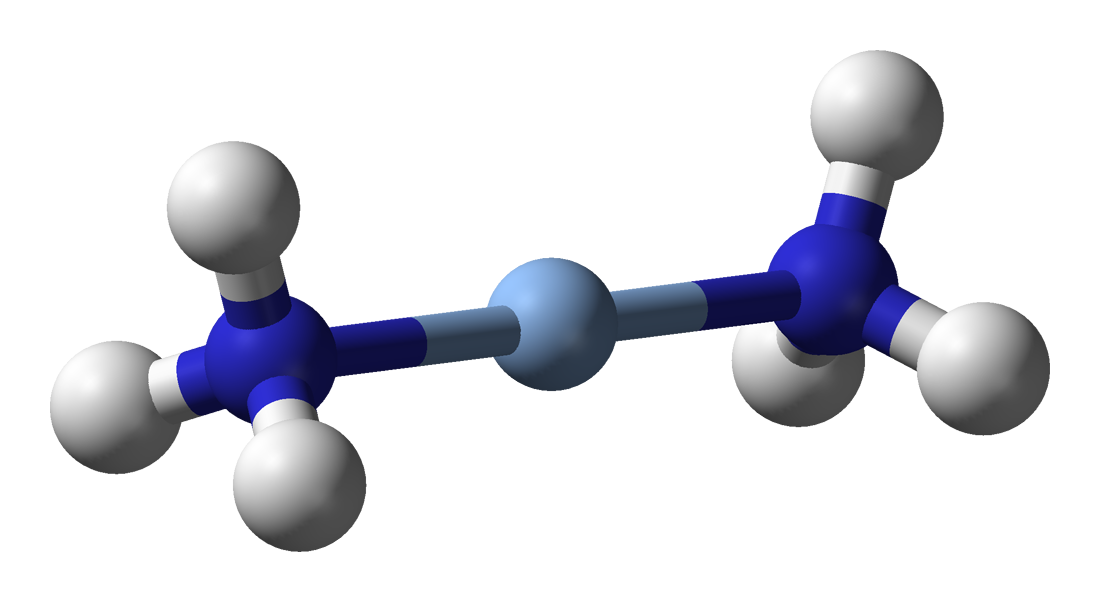 Silver complexes tend to be similar to those of its lighter homologue copper. Silver(III) complexes tend to be rare and very easily reduced to the more stable lower oxidation states, though they are slightly more stable than those of copper(III). For instance, the square planar periodate g(IO5OH)2sup>5− and tellurate g2sup>5− complexes may be prepared by oxidising silver(I) with alkaline
Silver complexes tend to be similar to those of its lighter homologue copper. Silver(III) complexes tend to be rare and very easily reduced to the more stable lower oxidation states, though they are slightly more stable than those of copper(III). For instance, the square planar periodate g(IO5OH)2sup>5− and tellurate g2sup>5− complexes may be prepared by oxidising silver(I) with alkaline

 Silver forms
Silver forms

Silver
Silver is a chemical element with the Symbol (chemistry), symbol Ag (from the Latin ', derived from the Proto-Indo-European wikt:Reconstruction:Proto-Indo-European/h₂erǵ-, ''h₂erǵ'': "shiny" or "white") and atomic number 47. A soft, whi ...
is a relatively unreactive metal, although it can form several compounds. The common oxidation state
In chemistry, the oxidation state, or oxidation number, is the hypothetical charge of an atom if all of its bonds to different atoms were fully ionic. It describes the degree of oxidation (loss of electrons) of an atom in a chemical compound. ...
s of silver are (in order of commonness): +1 (the most stable state; for example, silver nitrate
Silver nitrate is an inorganic compound with chemical formula . It is a versatile precursor to many other silver compounds, such as those used in photography. It is far less sensitive to light than the halides. It was once called ''lunar caustic ...
, AgNO3); +2 (highly oxidising; for example, silver(II) fluoride
Silver(II) fluoride is a chemical compound with the formula AgF2. It is a rare example of a silver(II) compound. Silver usually exists in its +1 oxidation state. It is used as a fluorinating agent.
Preparation
AgF2 can be synthesized by fluori ...
, AgF2); and even very rarely +3 (extreme oxidising; for example, potassium tetrafluoroargentate(III), KAgF4). The +3 state requires very strong oxidising agents to attain, such as fluorine or peroxodisulfate
The peroxydisulfate ion, , is an oxyanion, the anion of peroxydisulfuric acid. It is commonly referred to as persulfate, but this term also refers to the peroxomonosulfate ion, . It is also called ''peroxodisulfate''. Approximately 500,000 tons ...
, and some silver(III) compounds react with atmospheric moisture and attack glass.Greenwood and Earnshaw, p. 1188 Indeed, silver(III) fluoride is usually obtained by reacting silver or silver monofluoride with the strongest known oxidizing agent, krypton difluoride
Krypton difluoride, KrF2 is a chemical compound of krypton and fluorine. It was the first compound of krypton discovered. It is a volatile, colourless solid at room temperature. The structure of the KrF2 molecule is linear, with Kr−F distance ...
.Greenwood and Earnshaw, p. 903
Oxides and chalcogenides
Oxides
chemical affinities In chemical physics and physical chemistry, chemical affinity is the electronic property by which dissimilar chemical species are capable of forming chemical compounds. Chemical affinity can also refer to the tendency of an atom or compound to comb ...
for oxygen, lower than copper, and it is therefore expected that silver oxides are thermally quite unstable. Soluble silver(I) salts precipitate dark-brown silver(I) oxide
Silver oxide is the chemical compound with the formula Ag2O. It is a fine black or dark brown powder that is used to prepare other silver compounds.
Preparation
Silver oxide can be prepared by combining aqueous solutions of silver nitrate and ...
, Ag2O, upon the addition of alkali. (The hydroxide AgOH exists only in solution; otherwise it spontaneously decomposes to the oxide.) Silver(I) oxide is very easily reduced to metallic silver, and decomposes to silver and oxygen above 160 °C.Greenwood and Earnshaw, pp. 1181–82 This and other silver(I) compounds may be oxidized by the strong oxidizing agent peroxodisulfate
The peroxydisulfate ion, , is an oxyanion, the anion of peroxydisulfuric acid. It is commonly referred to as persulfate, but this term also refers to the peroxomonosulfate ion, . It is also called ''peroxodisulfate''. Approximately 500,000 tons ...
to black AgO, a mixed silver(I,III) oxide
Silver(I,III) oxide or tetrasilver tetroxide is the inorganic compound with the formula Ag4O4. It is a component of silver zinc batteries. It can be prepared by the slow addition of a silver(I) salt to a persulfate solution e.g. AgNO3 to a Na ...
of formula AgIAgIIIO2. Some other mixed oxides with silver in non-integral oxidation states, namely Ag2O3 and Ag3O4, are also known, as is Ag3O which behaves as a metallic conductor.
Other chalcogenides
Silver(I) sulfide
Silver sulfide is an inorganic compound with the formula . A dense black solid, it is the only sulfide of silver. It is useful as a photosensitizer in photography. It constitutes the tarnish that forms over time on silverware and other silver obje ...
, Ag2S, is very readily formed from its constituent elements and is the cause of the black tarnish on some old silver objects. It may also be formed from the reaction of hydrogen sulfide
Hydrogen sulfide is a chemical compound with the formula . It is a colorless chalcogen-hydride gas, and is poisonous, corrosive, and flammable, with trace amounts in ambient atmosphere having a characteristic foul odor of rotten eggs. The und ...
with silver metal or aqueous Ag+ ions. Many non-stoichiometric selenide A selenide is a chemical compound containing a selenium anion with oxidation number of −2 (Se2−), much as sulfur does in a sulfide. The chemistry of the selenides and sulfides is similar. Similar to sulfide, in aqueous solution, the selenide ion ...
s and tellurides
The telluride ion is the anion Te2− and its derivatives. It is analogous to the other chalcogenide anions, the lighter O2−, S2−, and Se2−, and the heavier Po2−.
In principle, Te2− is formed by the two-e− reduction of telluriu ...
are known; in particular, AgTe~3 is a low-temperature superconductor.
Halides
The only known dihalide of silver is the difluoride, AgF2, which can be obtained from the elements under heat. A strong yet thermally stable and therefore safe fluorinating agent, silver(II) fluoride is often used to synthesizehydrofluorocarbon
Hydrofluorocarbons (HFCs) are man-made organic compounds that contain fluorine and hydrogen atoms, and are the most common type of organofluorine compounds. Most are gases at room temperature and pressure. They are frequently used in air condi ...
s.Greenwood and Earnshaw, pp. 1183–85
In stark contrast to this, all four silver(I) halides are known. The fluoride
Fluoride (). According to this source, is a possible pronunciation in British English. is an inorganic, monatomic anion of fluorine, with the chemical formula (also written ), whose salts are typically white or colorless. Fluoride salts ...
, chloride
The chloride ion is the anion (negatively charged ion) Cl−. It is formed when the element chlorine (a halogen) gains an electron or when a compound such as hydrogen chloride is dissolved in water or other polar solvents. Chloride s ...
, and bromide
A bromide ion is the negatively charged form (Br−) of the element bromine, a member of the halogens group on the periodic table. Most bromides are colorless. Bromides have many practical roles, being found in anticonvulsants, flame-retardan ...
have the sodium chloride structure, but the iodide
An iodide ion is the ion I−. Compounds with iodine in formal oxidation state −1 are called iodides. In everyday life, iodide is most commonly encountered as a component of iodized salt, which many governments mandate. Worldwide, iodine def ...
has three known stable forms at different temperatures; that at room temperature is the cubic zinc blende
Sphalerite (sometimes spelled sphaelerite) is a sulfide mineral with the chemical formula . It is the most important ore of zinc. Sphalerite is found in a variety of deposit types, but it is primarily in sedimentary exhalative, Mississippi-Va ...
structure. They can all be obtained by the direct reaction of their respective elements. As the halogen group is descended, the silver halide gains more and more covalent character, solubility decreases, and the color changes from the white chloride to the yellow iodide as the energy required for ligand-metal charge transfer (X−Ag+ → XAg) decreases. The fluoride is anomalous, as the fluoride ion is so small that it has a considerable solvation
Solvation (or dissolution) describes the interaction of a solvent with dissolved molecules. Both ionized and uncharged molecules interact strongly with a solvent, and the strength and nature of this interaction influence many properties of the ...
energy and hence is highly water-soluble and forms di- and tetrahydrates. The other three silver halides are highly insoluble in aqueous solutions and are very commonly used in gravimetric analytical methods. All four are photosensitive Photosensitivity is the amount to which an object reacts upon receiving photons, especially visible light. In medicine, the term is principally used for abnormal reactions of the skin, and two types are distinguished, photoallergy and phototoxicity. ...
(though the monofluoride is so only to ultraviolet
Ultraviolet (UV) is a form of electromagnetic radiation with wavelength from 10 nm (with a corresponding frequency around 30 PHz) to 400 nm (750 THz), shorter than that of visible light, but longer than X-rays. UV radiati ...
light), especially the bromide and iodide which photodecompose to silver metal, and thus were used in traditional photography. The reaction involved is:Greenwood and Earnshaw, pp. 1185–87
:X− + ''hν'' → X + e− (excitation of the halide ion, which gives up its extra electron into the conduction band)
:Ag+ + e− → Ag (liberation of a silver ion, which gains an electron to become a silver atom)
The process is not reversible because the silver atom liberated is typically found at a crystal defect or an impurity site, so that the electron's energy is lowered enough that it is "trapped".
Other inorganic compounds
Whitesilver nitrate
Silver nitrate is an inorganic compound with chemical formula . It is a versatile precursor to many other silver compounds, such as those used in photography. It is far less sensitive to light than the halides. It was once called ''lunar caustic ...
, AgNO3, is a versatile precursor to many other silver compounds, especially the halides, and is much less sensitive to light. It was once called ''lunar caustic'' because silver was called ''luna'' by the ancient alchemists, who believed that silver was associated with the Moon. It is often used for gravimetric analysis, exploiting the insolubility of the heavier silver halides which it is a common precursor to. Silver nitrate is used in many ways in organic synthesis
Organic synthesis is a special branch of chemical synthesis and is concerned with the intentional construction of organic compounds. Organic molecules are often more complex than inorganic compounds, and their synthesis has developed into one o ...
, e.g. for deprotection
A protecting group or protective group is introduced into a molecule by chemical modification of a functional group to obtain chemoselectivity in a subsequent chemical reaction. It plays an important role in multistep organic synthesis.
In many ...
and oxidations. Ag+ binds alkene
In organic chemistry, an alkene is a hydrocarbon containing a carbon–carbon double bond.
Alkene is often used as synonym of olefin, that is, any hydrocarbon containing one or more double bonds.H. Stephen Stoker (2015): General, Organic, an ...
s reversibly, and silver nitrate has been used to separate mixtures of alkenes by selective absorption. The resulting adduct
An adduct (from the Latin ''adductus'', "drawn toward" alternatively, a contraction of "addition product") is a product of a direct addition of two or more distinct molecules, resulting in a single reaction product containing all atoms of all co ...
can be decomposed with ammonia
Ammonia is an inorganic compound of nitrogen and hydrogen with the formula . A stable binary hydride, and the simplest pnictogen hydride, ammonia is a colourless gas with a distinct pungent smell. Biologically, it is a common nitrogeno ...
to release the free alkene.
Yellow silver carbonate
Silver carbonate is the chemical compound with the formula Ag2CO3. This salt is yellow but typical samples are grayish due to the presence of elemental silver. It is poorly soluble in water, like most transition metal carbonates.
Preparation and ...
, Ag2CO3 can be easily prepared by reacting aqueous solutions of sodium carbonate with a deficiency of silver nitrate. Its principal use is for the production of silver powder for use in microelectronics. It is reduced with formaldehyde
Formaldehyde ( , ) ( systematic name methanal) is a naturally occurring organic compound with the formula and structure . The pure compound is a pungent, colourless gas that polymerises spontaneously into paraformaldehyde (refer to section ...
, producing silver free of alkali metals:Andreas Brumby et al. "Silver, Silver Compounds, and Silver Alloys" in Ullmann's Encyclopedia of Industrial Chemistry, Wiley-VCH, Weinheim, 2008.
:Ag2CO3 + CH2O → 2 Ag + 2 CO2 + H2
Silver carbonate is also used as a reagent in organic synthesis such as the Koenigs-Knorr reaction. In the Fétizon oxidation Fétizon oxidation is the oxidation of primary and secondary alcohols utilizing the compound silver(I) carbonate absorbed onto the surface of celite also known as Fétizon's reagent first employed by Marcel Fétizon in 1968. It is a mild reagent, ...
, silver carbonate on celite
Diatomaceous earth (), diatomite (), or kieselgur/kieselguhr is a naturally occurring, soft, siliceous sedimentary rock that can be crumbled into a fine white to off-white powder. It has a particle size ranging from more than 3 μm to les ...
acts as an oxidising agent
An oxidizing agent (also known as an oxidant, oxidizer, electron recipient, or electron acceptor) is a substance in a redox chemical reaction that gains or " accepts"/"receives" an electron from a (called the , , or ). In other words, an oxid ...
to form lactone
Lactones are cyclic carboxylic esters, containing a 1-oxacycloalkan-2-one structure (), or analogues having unsaturation or heteroatoms replacing one or more carbon atoms of the ring.
Lactones are formed by intramolecular esterification of the co ...
s from diols
A diol is a chemical compound containing two hydroxyl groups ( groups). An aliphatic diol is also called a glycol. This pairing of functional groups is pervasive, and many subcategories have been identified.
The most common industrial diol i ...
. It is also employed to convert alkyl
In organic chemistry, an alkyl group is an alkane missing one hydrogen.
The term ''alkyl'' is intentionally unspecific to include many possible substitutions.
An acyclic alkyl has the general formula of . A cycloalkyl is derived from a cycloal ...
bromides into alcohol
Alcohol most commonly refers to:
* Alcohol (chemistry), an organic compound in which a hydroxyl group is bound to a carbon atom
* Alcohol (drug), an intoxicant found in alcoholic drinks
Alcohol may also refer to:
Chemicals
* Ethanol, one of sev ...
s.
Silver fulminate
Silver fulminate (AgCNO) is the highly explosive silver salt of fulminic acid.
Silver fulminate is a primary explosive, but has limited use as such due to its extreme sensitivity to impact, heat, pressure, and electricity. The compound becomes ...
, AgCNO, a powerful, touch-sensitive explosive
An explosive (or explosive material) is a reactive substance that contains a great amount of potential energy that can produce an explosion if released suddenly, usually accompanied by the production of light, heat, sound, and pressure. An exp ...
used in percussion cap
The percussion cap or percussion primer, introduced in the early 1820s, is a type of single-use percussion ignition device for muzzle loader firearm locks enabling them to fire reliably in any weather condition. This crucial invention gave rise ...
s, is made by reaction of silver metal with nitric acid in the presence of ethanol
Ethanol (abbr. EtOH; also called ethyl alcohol, grain alcohol, drinking alcohol, or simply alcohol) is an organic compound. It is an alcohol with the chemical formula . Its formula can be also written as or (an ethyl group linked to a h ...
. Other dangerously explosive silver compounds are silver azide
Silver azide is the chemical compound with the formula . It is a silver(I) salt of hydrazoic acid. It forms a colorless crystals. It is a well-known explosive.
Structure and chemistry
Silver azide can be prepared by treating an aqueous solution ...
, AgN3, formed by reaction of silver nitrate
Silver nitrate is an inorganic compound with chemical formula . It is a versatile precursor to many other silver compounds, such as those used in photography. It is far less sensitive to light than the halides. It was once called ''lunar caustic ...
with sodium azide, and silver acetylide
Silver acetylide is an inorganic chemical compound with the formula Ag2C2, a metal acetylide. The compound can be regarded as a salt of the weak acid, acetylene. The salt's anion consists of two carbon atoms linked by a triple bond. The alternate ...
, Ag2C2, formed when silver reacts with acetylene
Acetylene ( systematic name: ethyne) is the chemical compound with the formula and structure . It is a hydrocarbon and the simplest alkyne. This colorless gas is widely used as a fuel and a chemical building block. It is unstable in its pur ...
gas in ammonia
Ammonia is an inorganic compound of nitrogen and hydrogen with the formula . A stable binary hydride, and the simplest pnictogen hydride, ammonia is a colourless gas with a distinct pungent smell. Biologically, it is a common nitrogeno ...
solution. In its most characteristic reaction, silver azide decomposes explosively, releasing nitrogen gas: given the photosensitivity of silver salts, this behaviour may be induced by shining a light on its crystals.Greenwood and Earnshaw, p. 1180
: 2 (s) → 3 (g) + 2 Ag (s)
Coordination compounds
 Silver complexes tend to be similar to those of its lighter homologue copper. Silver(III) complexes tend to be rare and very easily reduced to the more stable lower oxidation states, though they are slightly more stable than those of copper(III). For instance, the square planar periodate g(IO5OH)2sup>5− and tellurate g2sup>5− complexes may be prepared by oxidising silver(I) with alkaline
Silver complexes tend to be similar to those of its lighter homologue copper. Silver(III) complexes tend to be rare and very easily reduced to the more stable lower oxidation states, though they are slightly more stable than those of copper(III). For instance, the square planar periodate g(IO5OH)2sup>5− and tellurate g2sup>5− complexes may be prepared by oxidising silver(I) with alkaline peroxodisulfate
The peroxydisulfate ion, , is an oxyanion, the anion of peroxydisulfuric acid. It is commonly referred to as persulfate, but this term also refers to the peroxomonosulfate ion, . It is also called ''peroxodisulfate''. Approximately 500,000 tons ...
. The yellow diamagnetic gF4sup>− is much less stable, fuming in moist air and reacting with glass.
Silver(II) complexes are more common. Like the valence isoelectronic copper(II) complexes, they are usually square planar and paramagnetic, which is increased by the greater field splitting for 4d electrons than for 3d electrons. Aqueous Ag2+, produced by oxidation of Ag+ by ozone, is a very strong oxidising agent, even in acidic solutions: it is stabilized in phosphoric acid
Phosphoric acid (orthophosphoric acid, monophosphoric acid or phosphoric(V) acid) is a colorless, odorless phosphorus-containing solid, and inorganic compound with the chemical formula . It is commonly encountered as an 85% aqueous solutio ...
due to complex formation. Peroxodisulfate oxidation is generally necessary to give the more stable complexes with heterocyclic amine
In chemistry, amines (, ) are compounds and functional groups that contain a basic nitrogen atom with a lone pair. Amines are formally derivatives of ammonia (), wherein one or more hydrogen atoms have been replaced by a substituent su ...
s, such as g(py)4sup>2+ and g(bipy)2sup>2+: these are stable provided the counterion cannot reduce the silver back to the +1 oxidation state. gF4sup>2− is also known in its violet barium salt, as are some silver(II) complexes with ''N''- or ''O''-donor ligands such as pyridine carboxylates.Greenwood and Earnshaw, p. 1189
By far the most important oxidation state for silver in complexes is +1. The Ag+ cation is diamagnetic, like its homologues Cu+ and Au+, as all three have closed-shell electron configurations with no unpaired electrons: its complexes are colourless provided the ligands are not too easily polarized such as I−. Ag+ forms salts with most anions, but it is reluctant to coordinate to oxygen and thus most of these salts are insoluble in water: the exceptions are the nitrate, perchlorate, and fluoride. The tetracoordinate tetrahedral aqueous ion g(H2O)4sup>+ is known, but the characteristic geometry for the Ag+ cation is 2-coordinate linear. For example, silver chloride dissolves readily in excess aqueous ammonia to form g(NH3)2sup>+; silver salts are dissolved in photography due to the formation of the thiosulfate complex g(S2O3)2sup>3−; and cyanide
Cyanide is a naturally occurring, rapidly acting, toxic chemical that can exist in many different forms.
In chemistry, a cyanide () is a chemical compound that contains a functional group. This group, known as the cyano group, consists of ...
extraction for silver (and gold) works by the formation of the complex g(CN)2sup>−. Silver cyanide forms the linear polymer ; silver thiocyanate
Thiocyanate (also known as rhodanide) is the anion . It is the conjugate base of thiocyanic acid. Common derivatives include the colourless salts potassium thiocyanate and sodium thiocyanate. Mercury(II) thiocyanate was formerly used in pyro ...
has a similar structure, but forms a zigzag instead because of the sp3- hybridized sulfur atom. Chelating ligand
Chelation is a type of bonding of ions and molecules to metal ions. It involves the formation or presence of two or more separate coordinate bonds between a polydentate (multiple bonded) ligand and a single central metal atom. These ligands are ...
s are unable to form linear complexes and thus silver(I) complexes with them tend to form polymers; a few exceptions exist, such as the near-tetrahedral diphosphine
Diphosphane, or diphosphine, is an inorganic compound with the chemical formula P2H4. This colourless liquid is one of several binary phosphorus hydrides. It is the impurity that typically causes samples of phosphine to ignite in air.
Propert ...
and diarsine
Arsine (IUPAC name: arsane) is an inorganic compound with the formula As H3. This flammable, pyrophoric, and highly toxic pnictogen hydride gas is one of the simplest compounds of arsenic. Despite its lethality, it finds some applications in th ...
complexes g(L–L)2sup>+.Greenwood and Earnshaw, pp. 1195–96
Organometallic compounds
Under standard conditions, silver does not form simple carbonyls, due to the weakness of the Ag–C bond. A few are known at very low temperatures around 6–15 K, such as the green, planar paramagnetic Ag(CO)3, which dimerizes at 25–30 K, probably by forming Ag–Ag bonds. Additionally, the silver carbonylg(CO)
G, or g, is the seventh letter in the Latin alphabet, used in the modern English alphabet, the alphabets of other western European languages and others worldwide. Its name in English is ''gee'' (pronounced ), plural ''gees''.
History
Th ...
(OTeF5)4is known. Polymeric AgLX complexes with alkene
In organic chemistry, an alkene is a hydrocarbon containing a carbon–carbon double bond.
Alkene is often used as synonym of olefin, that is, any hydrocarbon containing one or more double bonds.H. Stephen Stoker (2015): General, Organic, an ...
s and alkyne
\ce
\ce
Acetylene
\ce
\ce
\ce
Propyne
\ce
\ce
\ce
\ce
1-Butyne
In organic chemistry, an alkyne is an unsaturated hydrocarbon containing at least one carbon—carbon triple bond. The simplest acyclic alkynes with only one triple bond and no ...
s are known, but their bonds are thermodynamically weaker than even those of the platinum
Platinum is a chemical element with the symbol Pt and atomic number 78. It is a dense, malleable, ductile, highly unreactive, precious, silverish-white transition metal. Its name originates from Spanish , a diminutive of "silver".
Pla ...
complexes (though they are formed more readily than those of the analogous gold complexes): they are also quite unsymmetrical, showing the weak ''π'' bonding in group 11. Ag–C ''σ'' bonds may also be formed by silver(I), like copper(I) and gold(I), but the simple alkyls and aryls of silver(I) are even less stable than those of copper(I) (which tend to explode under ambient conditions). For example, poor thermal stability is reflected in the relative decomposition temperatures of AgMe (−50 °C) and CuMe (−15 °C) as well as those of PhAg (74 °C) and PhCu (100 °C).Greenwood and Earnshaw, pp. 1199–200
The C–Ag bond is stabilized by perfluoroalkyl
A perfluorinated compound (PFC) or perfluoro compound is an organofluorine compound containing only carbon-fluorines and C−C bonds, as well as potentially heteroatoms. Perfluorinated compounds have properties that result from the presence of flu ...
ligands, for example in AgCF(CF3)2. Alkenylsilver compounds are also more stable than their alkylsilver counterparts. Silver- NHC complexes are easily prepared, and are commonly used to prepare other NHC complexes by displacing labile ligands. For example, the reaction of the bis(NHC)silver(I) complex with bis(acetonitrile)palladium dichloride
Bis(acetonitrile)palladium dichloride is the coordination complex with the formula PdCl2(NCCH3)2. It is the adduct of two acetonitrile ligands with palladium(II) chloride. It is a yellow-brown solid that is soluble in organic solvents. The comp ...
or chlorido(dimethyl sulfide)gold(I)
Chloro(dimethyl sulfide)gold(I) is a coordination complex of gold. It is a white solid. This compound is a common entry point into gold chemistry.
Structure
As for many other gold(I) complexes, the compound adopts a nearly linear (176.9°) geome ...
:
:
Intermetallic compounds
alloy
An alloy is a mixture of chemical elements of which at least one is a metal. Unlike chemical compounds with metallic bases, an alloy will retain all the properties of a metal in the resulting material, such as electrical conductivity, ductilit ...
s with most other elements on the periodic table. The elements from groups 1–3, except for hydrogen
Hydrogen is the chemical element with the symbol H and atomic number 1. Hydrogen is the lightest element. At standard conditions hydrogen is a gas of diatomic molecules having the formula . It is colorless, odorless, tasteless, non-toxic ...
, lithium
Lithium (from el, λίθος, lithos, lit=stone) is a chemical element with the symbol Li and atomic number 3. It is a soft, silvery-white alkali metal. Under standard conditions, it is the least dense metal and the least dense solid ...
, and beryllium
Beryllium is a chemical element with the symbol Be and atomic number 4. It is a steel-gray, strong, lightweight and brittle alkaline earth metal. It is a divalent element that occurs naturally only in combination with other elements to form m ...
, are very miscible with silver in the condensed phase and form intermetallic compounds; those from groups 4–9 are only poorly miscible; the elements in groups 10–14 (except boron and carbon
Carbon () is a chemical element with the symbol C and atomic number 6. It is nonmetallic and tetravalent—its atom making four electrons available to form covalent chemical bonds. It belongs to group 14 of the periodic table. Carbon makes ...
) have very complex Ag–M phase diagrams and form the most commercially important alloys; and the remaining elements on the periodic table have no consistency in their Ag–M phase diagrams. By far the most important such alloys are those with copper: most silver used for coinage and jewellery is in reality a silver–copper alloy, and the eutectic mixture
A eutectic system or eutectic mixture ( ) is a homogeneous mixture that has a melting point lower than those of the constituents. The lowest possible melting point over all of the mixing ratios of the constituents is called the ''eutectic temp ...
is used in vacuum brazing
Brazing is a metal-joining process in which two or more metal items are joined together by melting and flowing a filler metal into the joint, with the filler metal having a lower melting point than the adjoining metal.
Brazing differs from w ...
. The two metals are completely miscible as liquids but not as solids; their importance in industry comes from the fact that their properties tend to be suitable over a wide range of variation in silver and copper concentration, although most useful alloys tend to be richer in silver than the eutectic mixture (71.9% silver and 28.1% copper by weight, and 60.1% silver and 28.1% copper by atom).Ullmann, pp. 54–61
Most other binary alloys are of little use: for example, silver–gold alloys are too soft and silver–cadmium
Cadmium is a chemical element with the symbol Cd and atomic number 48. This soft, silvery-white metal is chemically similar to the two other stable metals in group 12, zinc and mercury. Like zinc, it demonstrates oxidation state +2 in most of ...
alloys too toxic. Ternary alloys have much greater importance: dental amalgams
Amalgam most commonly refers to:
* Amalgam (chemistry), mercury alloy
* Amalgam (dentistry), material of silver tooth fillings
** Bonded amalgam, used in dentistry
Amalgam may also refer to:
* Amalgam Comics, a publisher
* Amalgam Digital, an in ...
are usually silver–tin–mercury alloys, silver–copper–gold alloys are very important in jewellery (usually on the gold-rich side) and have a vast range of hardnesses and colours, silver–copper–zinc alloys are useful as low-melting brazing alloys, and silver–cadmium–indium
Indium is a chemical element with the symbol In and atomic number 49. Indium is the softest metal that is not an alkali metal. It is a silvery-white metal that resembles tin in appearance. It is a post-transition metal that makes up 0.21 par ...
(involving three adjacent elements on the periodic table) is useful in nuclear reactor
A nuclear reactor is a device used to initiate and control a fission nuclear chain reaction or nuclear fusion reactions. Nuclear reactors are used at nuclear power plants for electricity generation and in nuclear marine propulsion. Heat from nu ...
s because of its high thermal neutron capture cross-section
Cross section may refer to:
* Cross section (geometry)
** Cross-sectional views in architecture & engineering 3D
*Cross section (geology)
* Cross section (electronics)
* Radar cross section, measure of detectability
* Cross section (physics)
**Abs ...
, good conduction of heat, mechanical stability, and resistance to corrosion in hot water.
See also
*Copper compounds
Copper forms a rich variety of compounds, usually with oxidation states +1 and +2, which are often called ''cuprous'' and ''cupric'', respectively. Copper compounds, whether organic complexes or organometallics, promote or catalyse numerous che ...
* Gold compounds
Gold compounds are compounds by the element gold (Au). Although gold is the most noble of the noble metals, it still forms many diverse compounds. The oxidation state of gold in its compounds ranges from −1 to +5, but Au(I) and Au(III) dominate ...
References
{{Silver compounds Compounds Chemical compounds by element