Prolyl on:
[Wikipedia]
[Google]
[Amazon]
Proline (symbol Pro or P) is an organic acid classed as a

Proline MS Spectrum
Proline biosynthesis
{{Authority control AMPA receptor agonists Kainate receptor agonists NMDA receptor agonists Proteinogenic amino acids Glucogenic amino acids Glycine receptor agonists Cyclic amino acids Pyrrolidines Secondary amino acids Excitatory amino acids
proteinogenic amino acid
Proteinogenic amino acids are amino acids that are incorporated biosynthetically into proteins during translation. The word "proteinogenic" means "protein creating". Throughout known life, there are 22 genetically encoded (proteinogenic) amino aci ...
(used in the biosynthesis of proteins), although it does not contain the amino group
In chemistry, amines (, ) are compounds and functional groups that contain a basic nitrogen atom with a lone pair. Amines are formally derivatives of ammonia (), wherein one or more hydrogen atoms have been replaced by a substituent such a ...
but is rather a secondary amine
In chemistry, amines (, ) are compounds and functional groups that contain a basic nitrogen atom with a lone pair. Amines are formally derivatives of ammonia (), wherein one or more hydrogen atoms have been replaced by a substituent such a ...
. The secondary amine nitrogen is in the protonated form (NH2+) under biological conditions, while the carboxyl group
In organic chemistry, a carboxylic acid is an organic acid that contains a carboxyl group () attached to an R-group. The general formula of a carboxylic acid is or , with R referring to the alkyl, alkenyl, aryl, or other group. Carboxylic ...
is in the deprotonated
Deprotonation (or dehydronation) is the removal (transfer) of a proton (or hydron, or hydrogen cation), (H+) from a Brønsted–Lowry acid in an acid–base reaction.Henry Jakubowski, Biochemistry Online Chapter 2A3, https://employees.csbsju.edu ...
−COO− form. The "side chain" from the α carbon connects to the nitrogen forming a pyrrolidine
Pyrrolidine, also known as tetrahydropyrrole, is an organic compound with the molecular formula (CH2)4NH. It is a cyclic secondary amine, also classified as a saturated heterocycle. It is a colourless liquid that is miscible with water and most ...
loop, classifying it as a aliphatic
In organic chemistry, hydrocarbons ( compounds composed solely of carbon and hydrogen) are divided into two classes: aromatic compounds and aliphatic compounds (; G. ''aleiphar'', fat, oil). Aliphatic compounds can be saturated, like hexane, or ...
amino acid
Amino acids are organic compounds that contain both amino and carboxylic acid functional groups. Although hundreds of amino acids exist in nature, by far the most important are the alpha-amino acids, which comprise proteins. Only 22 alpha am ...
. It is non-essential in humans, meaning the body can synthesize it from the non-essential amino acid L-glutamate
Glutamic acid (symbol Glu or E; the ionic form is known as glutamate) is an α-amino acid that is used by almost all living beings in the biosynthesis of proteins. It is a non-essential nutrient for humans, meaning that the human body can syn ...
. It is encoded
In communications and information processing, code is a system of rules to convert information—such as a letter, word, sound, image, or gesture—into another form, sometimes shortened or secret, for communication through a communication ...
by all the codon
The genetic code is the set of rules used by living cells to translate information encoded within genetic material ( DNA or RNA sequences of nucleotide triplets, or codons) into proteins. Translation is accomplished by the ribosome, which links ...
s starting with CC (CCU, CCC, CCA, and CCG).
Proline is the only proteinogenic secondary amino acid Secondary amino acids are amino acids which do not contain the amino group but is rather a secondary amine. Secondary amino acids can be classified to cyclic acids such as proline and acyclic N-substituted amino acids.
In nature, proline, hydroxyp ...
which is a secondary amine, as the nitrogen atom is attached both to the α-carbon and to a chain of three carbons that together form a five-membered ring.
History and etymology
Proline was first isolated in 1900 byRichard Willstätter
Richard Martin Willstätter FRS(For) HFRSE (, 13 August 1872 – 3 August 1942) was a German organic chemist whose study of the structure of plant pigments, chlorophyll included, won him the 1915 Nobel Prize for Chemistry. Willstätter invented ...
who obtained the amino acid while studying ''N''-methylproline, and synthesized proline by the reaction of sodium salt of diethyl malonate
Diethyl malonate, also known as DEM, is the diethyl ester of malonic acid. It occurs naturally in grapes and strawberries as a colourless liquid with an apple-like odour, and is used in perfumes. It is also used to synthesize other compounds su ...
with 1,3-dibromopropane. The next year, Emil Fischer
Hermann Emil Louis Fischer (; 9 October 1852 – 15 July 1919) was a German chemist and 1902 recipient of the Nobel Prize in Chemistry. He discovered the Fischer esterification. He also developed the Fischer projection, a symbolic way of dra ...
isolated proline from casein
Casein ( , from Latin ''caseus'' "cheese") is a family of related phosphoproteins (CSN1S1, αS1, aS2, CSN2, β, K-casein, κ) that are commonly found in mammalian milk, comprising about 80% of the proteins in cow's milk and between 20% and 60% of ...
and the decomposition products of γ-phthalimido-propylmalonic ester, and published the synthesis of proline from phthalimide propylmalonic ester.
The name proline comes from pyrrolidine
Pyrrolidine, also known as tetrahydropyrrole, is an organic compound with the molecular formula (CH2)4NH. It is a cyclic secondary amine, also classified as a saturated heterocycle. It is a colourless liquid that is miscible with water and most ...
, one of its constituents.
Biosynthesis
Proline is biosynthetically derived from the amino acid L-glutamate
Glutamic acid (symbol Glu or E; the ionic form is known as glutamate) is an α-amino acid that is used by almost all living beings in the biosynthesis of proteins. It is a non-essential nutrient for humans, meaning that the human body can syn ...
. Glutamate-5-semialdehyde
Glutamate-5-semialdehyde is a non-proteinogenic amino acid involved in both the biosynthesis and degradation of proline and arginine (via ornithine), as well as in the biosynthesis of antibiotics, such as carbapenems. It is synthesized by the redu ...
is first formed by glutamate 5-kinase
In enzymology, a glutamate 5-kinase () is an enzyme that catalyzes the chemical reaction
:ATP + L-glutamate \rightleftharpoons ADP + L-glutamate 5-phosphate
Thus, the two substrates of this enzyme are ATP and L-glutamate, whereas its two pr ...
(ATP-dependent) and glutamate-5-semialdehyde dehydrogenase
In enzymology, a glutamate-5-semialdehyde dehydrogenase () is an enzyme that catalyzes the chemical reaction
:L-glutamate 5-semialdehyde + phosphate + NADP+ \rightleftharpoons L-glutamyl 5-phosphate + NADPH + H+
The 3 substrates of this enzyme ...
(which requires NADH or NADPH). This can then either spontaneously cyclize to form 1-pyrroline-5-carboxylic acid, which is reduced to proline by pyrroline-5-carboxylate reductase
In enzymology, a pyrroline-5-carboxylate reductase () is an enzyme that catalyzes the chemical reaction
:L-proline + NAD(P)+ \rightleftharpoons 1-pyrroline-5-carboxylate + NAD(P)H + H+
The 3 substrates of this enzyme are L-proline, NAD+, and ...
(using NADH or NADPH), or turned into ornithine
Ornithine is a non-proteinogenic amino acid that plays a role in the urea cycle. Ornithine is abnormally accumulated in the body in ornithine transcarbamylase deficiency. The radical is ornithyl.
Role in urea cycle
L-Ornithine is one of the produ ...
by ornithine aminotransferase
Ornithine aminotransferase (OAT) is an enzyme which is encoded in human by the OAT gene located on chromosome 10.
The OAT involved in the ultimate formation of the non-essential amino acid proline from the amino acid ornithine. Ornithine aminot ...
, followed by cyclisation by ornithine cyclodeaminase
The enzyme ornithine cyclodeaminase (EC 4.3.1.12) catalyzes the chemical reaction
L-ornithine \rightleftharpoons L-proline + NH4+
This enzyme belongs to the family of lyases, specifically ammonia lyases, which cleave carbon-nitrogen bonds. Th ...
to form proline.

Biological activity
L-Proline has been found to act as a weakagonist
An agonist is a chemical that activates a receptor to produce a biological response. Receptors are cellular proteins whose activation causes the cell to modify what it is currently doing. In contrast, an antagonist blocks the action of the ago ...
of the glycine receptor
The glycine receptor (abbreviated as GlyR or GLR) is the receptor of the amino acid neurotransmitter glycine. GlyR is an ionotropic receptor that produces its effects through chloride current. It is one of the most widely distributed inhibitory ...
and of both NMDA
''N''-methyl--aspartic acid or ''N''-methyl--aspartate (NMDA) is an amino acid derivative that acts as a specific agonist at the NMDA receptor mimicking the action of glutamate, the neurotransmitter which normally acts at that receptor. Unlike ...
and non-NMDA (AMPA
α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid, better known as AMPA, is a compound that is a specific agonist for the AMPA receptor, where it mimics the effects of the neurotransmitter glutamate.
There are several types of glutamatergic ...
/kainate
Kainic acid, or kainate, is an acid that naturally occurs in some seaweed. Kainic acid is a potent neuroexcitatory amino acid agonist that acts by activating receptors for glutamate, the principal excitatory neurotransmitter in the central nervo ...
) ionotropic glutamate receptor
Ionotropic glutamate receptors (iGluRs) are ligand-gated ion channels that are activated by the neurotransmitter glutamate. They mediate the majority of excitatory synaptic transmission throughout the central nervous system and are key players in ...
s. It has been proposed to be a potential endogenous
Endogenous substances and processes are those that originate from within a living system such as an organism, tissue, or cell.
In contrast, exogenous substances and processes are those that originate from outside of an organism.
For example, es ...
excitotoxin
In excitotoxicity, nerve cells suffer damage or death when the levels of otherwise necessary and safe neurotransmitters such as glutamate become pathologically high, resulting in excessive stimulation of receptors. For example, when glutamate re ...
. In plants
Plants are predominantly Photosynthesis, photosynthetic eukaryotes of the Kingdom (biology), kingdom Plantae. Historically, the plant kingdom encompassed all living things that were not animals, and included algae and fungi; however, all curr ...
, proline accumulation is a common physiological response to various stresses but is also part of the developmental program in generative tissues (e.g. pollen
Pollen is a powdery substance produced by seed plants. It consists of pollen grains (highly reduced microgametophytes), which produce male gametes (sperm cells). Pollen grains have a hard coat made of sporopollenin that protects the gametophyt ...
).
A diet rich in proline was linked to an increased risk of depression in humans in a study from 2022 that was tested on a limited pre-clinical trial on humans and primarily in other organisms. Results were significant in the other organisms.
Properties in protein structure
The distinctive cyclic structure of proline's side chain gives proline an exceptional conformational rigidity compared to other amino acids. It also affects the rate of peptide bond formation between proline and other amino acids. When proline is bound as an amide in a peptide bond, its nitrogen is not bound to any hydrogen, meaning it cannot act as ahydrogen bond
In chemistry, a hydrogen bond (or H-bond) is a primarily electrostatic force of attraction between a hydrogen (H) atom which is covalently bound to a more electronegative "donor" atom or group (Dn), and another electronegative atom bearing a ...
donor, but can be a hydrogen bond acceptor.
Peptide bond formation with incoming Pro-tRNAPro is considerably slower than with any other tRNAs, which is a general feature of ''N''-alkylamino acids. Peptide bond formation is also slow between an incoming tRNA and a chain ending in proline; with the creation of proline-proline bonds slowest of all.
The exceptional conformational rigidity of proline affects the secondary structure
Protein secondary structure is the three dimensional conformational isomerism, form of ''local segments'' of proteins. The two most common Protein structure#Secondary structure, secondary structural elements are alpha helix, alpha helices and beta ...
of proteins near a proline residue and may account for proline's higher prevalence in the proteins of thermophilic
A thermophile is an organism—a type of extremophile—that thrives at relatively high temperatures, between . Many thermophiles are archaea, though they can be bacteria or fungi. Thermophilic eubacteria are suggested to have been among the earl ...
organisms. Protein secondary structure
Protein secondary structure is the three dimensional form of ''local segments'' of proteins. The two most common secondary structural elements are alpha helices and beta sheets, though beta turns and omega loops occur as well. Secondary structure ...
can be described in terms of the dihedral angle
A dihedral angle is the angle between two intersecting planes or half-planes. In chemistry, it is the clockwise angle between half-planes through two sets of three atoms, having two atoms in common. In solid geometry, it is defined as the uni ...
s φ, ψ and ω of the protein backbone. The cyclic structure of proline's side chain locks the angle φ at approximately −65°.
Proline acts as a structural disruptor in the middle of regular secondary structure
Protein secondary structure is the three dimensional conformational isomerism, form of ''local segments'' of proteins. The two most common Protein structure#Secondary structure, secondary structural elements are alpha helix, alpha helices and beta ...
elements such as alpha helices
The alpha helix (α-helix) is a common motif in the secondary structure of proteins and is a right hand-helix conformation in which every backbone N−H group hydrogen bonds to the backbone C=O group of the amino acid located four residues ear ...
and beta sheet
The beta sheet, (β-sheet) (also β-pleated sheet) is a common motif of the regular protein secondary structure. Beta sheets consist of beta strands (β-strands) connected laterally by at least two or three backbone hydrogen bonds, forming a g ...
s; however, proline is commonly found as the first residue of an alpha helix
The alpha helix (α-helix) is a common motif in the secondary structure of proteins and is a right hand-helix conformation in which every backbone N−H group hydrogen bonds to the backbone C=O group of the amino acid located four residues e ...
and also in the edge strands of beta sheet
The beta sheet, (β-sheet) (also β-pleated sheet) is a common motif of the regular protein secondary structure. Beta sheets consist of beta strands (β-strands) connected laterally by at least two or three backbone hydrogen bonds, forming a g ...
s. Proline is also commonly found in turns (another kind of secondary structure), and aids in the formation of beta turns. This may account for the curious fact that proline is usually solvent-exposed, despite having a completely aliphatic
In organic chemistry, hydrocarbons ( compounds composed solely of carbon and hydrogen) are divided into two classes: aromatic compounds and aliphatic compounds (; G. ''aleiphar'', fat, oil). Aliphatic compounds can be saturated, like hexane, or ...
side chain.
Multiple prolines and/or hydroxyproline
(2''S'',4''R'')-4-Hydroxyproline, or L-hydroxyproline ( C5 H9 O3 N), is an amino acid, abbreviated as Hyp or O, ''e.g.'', in Protein Data Bank.
Structure and discovery
In 1902, Hermann Emil Fischer isolated hydroxyproline from hydrolyzed gelatin. ...
s in a row can create a polyproline helix
A polyproline helix is a type of protein secondary structure which occurs in proteins comprising repeating proline residues. A left-handed polyproline II helix (PPII, poly-Pro II) is formed when sequential residues all adopt (φ,ψ) backbone dihedr ...
, the predominant secondary structure
Protein secondary structure is the three dimensional conformational isomerism, form of ''local segments'' of proteins. The two most common Protein structure#Secondary structure, secondary structural elements are alpha helix, alpha helices and beta ...
in collagen
Collagen () is the main structural protein in the extracellular matrix found in the body's various connective tissues. As the main component of connective tissue, it is the most abundant protein in mammals, making up from 25% to 35% of the whole ...
. The hydroxylation
In chemistry, hydroxylation can refer to:
*(i) most commonly, hydroxylation describes a chemical process that introduces a hydroxyl group () into an organic compound.
*(ii) the ''degree of hydroxylation'' refers to the number of OH groups in a ...
of proline by prolyl hydroxylase
Procollagen-proline dioxygenase, commonly known as prolyl hydroxylase, is a member of the class of enzymes known as alpha-ketoglutarate-dependent hydroxylases. These enzymes catalyze the incorporation of oxygen into organic substrates through a mec ...
(or other additions of electron-withdrawing substituents such as fluorine
Fluorine is a chemical element with the symbol F and atomic number 9. It is the lightest halogen and exists at standard conditions as a highly toxic, pale yellow diatomic gas. As the most electronegative reactive element, it is extremely reacti ...
) increases the conformational stability of collagen
Collagen () is the main structural protein in the extracellular matrix found in the body's various connective tissues. As the main component of connective tissue, it is the most abundant protein in mammals, making up from 25% to 35% of the whole ...
significantly. Hence, the hydroxylation of proline is a critical biochemical process for maintaining the connective tissue
Connective tissue is one of the four primary types of animal tissue, along with epithelial tissue, muscle tissue, and nervous tissue. It develops from the mesenchyme derived from the mesoderm the middle embryonic germ layer. Connective tiss ...
of higher organisms. Severe diseases such as scurvy
Scurvy is a disease resulting from a lack of vitamin C (ascorbic acid). Early symptoms of deficiency include weakness, feeling tired and sore arms and legs. Without treatment, decreased red blood cells, gum disease, changes to hair, and bleeding ...
can result from defects in this hydroxylation, e.g., mutations in the enzyme prolyl hydroxylase or lack of the necessary ascorbate (vitamin C) cofactor.
''Cis''–''trans'' isomerization
Peptide bond
In organic chemistry, a peptide bond is an amide type of covalent chemical bond linking two consecutive alpha-amino acids from C1 (carbon number one) of one alpha-amino acid and N2 (nitrogen number two) of another, along a peptide or protein cha ...
s to proline, and to other ''N''-substituted amino acids (such as sarcosine
Sarcosine, also known as ''N''-methylglycine, or monomethylglycine, is a monopeptide with the formula CH3N(H)CH2CO2H. It exists at neutral pH as the zwitterion CH3N+(H)2CH2CO2−, which can be obtained as a white, water-soluble powder. Like some ...
), are able to populate both the ''cis
Cis or cis- may refer to:
Places
* Cis, Trentino, in Italy
* In Poland:
** Cis, Świętokrzyskie Voivodeship, south-central
** Cis, Warmian-Masurian Voivodeship, north
Math, science and biology
* cis (mathematics) (cis(''θ'')), a trigonome ...
'' and ''trans
Trans- is a Latin prefix meaning "across", "beyond", or "on the other side of".
Used alone, trans may refer to:
Arts, entertainment, and media
* Trans (festival), a former festival in Belfast, Northern Ireland, United Kingdom
* ''Trans'' (film ...
'' isomers. Most peptide bonds overwhelmingly adopt the ''trans'' isomer (typically 99.9% under unstrained conditions), chiefly because the amide hydrogen (''trans'' isomer) offers less steric repulsion to the preceding Cα atom than does the following Cα atom (''cis'' isomer). By contrast, the ''cis'' and ''trans'' isomers of the X-Pro peptide bond (where X represents any amino acid) both experience steric clashes with the neighboring substitution and have a much lower energy difference. Hence, the fraction of X-Pro peptide bonds in the ''cis'' isomer under unstrained conditions is significantly elevated, with ''cis'' fractions typically in the range of 3-10%. However, these values depend on the preceding amino acid, with Gly and aromatic residues yielding increased fractions of the ''cis'' isomer. ''Cis'' fractions up to 40% have been identified for aromatic–proline peptide bonds.
From a kinetic standpoint, ''cis''–''trans'' proline isomerization
In chemistry, isomerization or isomerisation is the process in which a molecule, polyatomic ion or molecular fragment is transformed into an isomer with a different chemical structure. Enolization is an example of isomerization, as is tautomeriz ...
is a very slow process that can impede the progress of protein folding
Protein folding is the physical process by which a protein chain is translated to its native three-dimensional structure, typically a "folded" conformation by which the protein becomes biologically functional. Via an expeditious and reproduci ...
by trapping one or more proline residues crucial for folding in the non-native isomer, especially when the native protein requires the ''cis'' isomer. This is because proline residues are exclusively synthesized in the ribosome
Ribosomes ( ) are macromolecular machines, found within all cells, that perform biological protein synthesis (mRNA translation). Ribosomes link amino acids together in the order specified by the codons of messenger RNA (mRNA) molecules to ...
as the ''trans'' isomer form. All organisms possess prolyl isomerase
Prolyl isomerase (also known as peptidylprolyl isomerase or PPIase) is an enzyme () found in both prokaryotes and eukaryotes that interconverts the ''cis'' and ''trans'' isomers of peptide bonds with the amino acid proline. Proline has an unusua ...
enzyme
Enzymes () are proteins that act as biological catalysts by accelerating chemical reactions. The molecules upon which enzymes may act are called substrates, and the enzyme converts the substrates into different molecules known as products. A ...
s to catalyze this isomerization, and some bacteria
Bacteria (; singular: bacterium) are ubiquitous, mostly free-living organisms often consisting of one biological cell. They constitute a large domain of prokaryotic microorganisms. Typically a few micrometres in length, bacteria were among ...
have specialized prolyl isomerases associated with the ribosome. However, not all prolines are essential for folding, and protein folding may proceed at a normal rate despite having non-native conformers of many X–Pro peptide bonds.
Uses
Proline and its derivatives are often used as asymmetric catalysts in proline organocatalysis reactions. The CBS reduction and proline catalysedaldol condensation
An aldol condensation is a condensation reaction in organic chemistry in which two carbonyl moieties (of aldehydes or ketones) react to form a β-hydroxyaldehyde or β-hydroxyketone (an aldol reaction), and this is then followed by dehydration to ...
are prominent examples.
In brewing, proteins rich in proline combine with polyphenols to produce haze (turbidity).
L-Proline is an osmoprotectant
Osmoprotectants or compatible solutes are small organic molecules with neutral charge and low toxicity at high concentrations that act as osmolytes and help organisms survive extreme osmotic stress. Osmoprotectants can be placed in three chemical c ...
and therefore is used in many pharmaceutical and biotechnological applications.
The growth medium
A growth medium or culture medium is a solid, liquid, or semi-solid designed to support the growth of a population of microorganisms or cells via the process of cell proliferation or small plants like the moss ''Physcomitrella patens''. Differen ...
used in plant tissue culture
Plant tissue culture is a collection of techniques used to maintain or grow plant cells, tissues or organs under sterile conditions on a nutrient culture medium of known composition. It is widely used to produce clones of a plant in a method known ...
may be supplemented with proline. This can increase growth, perhaps because it helps the plant tolerate the stresses of tissue culture. For proline's role in the stress response of plants, see .
Specialties
Proline is one of the two amino acids that do not follow along with the typicalRamachandran plot
In biochemistry, a Ramachandran plot (also known as a Rama plot, a Ramachandran diagram or a �,ψplot), originally developed in 1963 by G. N. Ramachandran, C. Ramakrishnan, and V. Sasisekharan, is a way to visualize energetically allowed region ...
, along with glycine
Glycine (symbol Gly or G; ) is an amino acid that has a single hydrogen atom as its side chain. It is the simplest stable amino acid (carbamic acid is unstable), with the chemical formula NH2‐ CH2‐ COOH. Glycine is one of the proteinogeni ...
. Due to the ring formation connected to the beta carbon, the ''ψ'' and ''φ'' angles about the peptide bond have fewer allowable degrees of rotation. As a result, it is often found in "turns" of proteins as its free entropy (Δ''S'') is not as comparatively large to other amino acids and thus in a folded form vs. unfolded form, the change in entropy is smaller. Furthermore, proline is rarely found in α and β structures as it would reduce the stability of such structures, because its side chain α-nitrogen can only form one nitrogen bond.
Additionally, proline is the only amino acid that does not form a red-purple colour when developed by spraying with ninhydrin
Ninhydrin (2,2-dihydroxyindane-1,3-dione) is an organic compound with the formula C6H4(CO)2C(OH)2. It is used to detect ammonia and amines. Upon reaction with these amines, ninhydrin gets converted into deep blue or purple derivatives, which are ...
for uses in chromatography
In chemical analysis, chromatography is a laboratory technique for the separation of a mixture into its components. The mixture is dissolved in a fluid solvent (gas or liquid) called the ''mobile phase'', which carries it through a system (a ...
. Proline, instead, produces an orange-yellow colour.
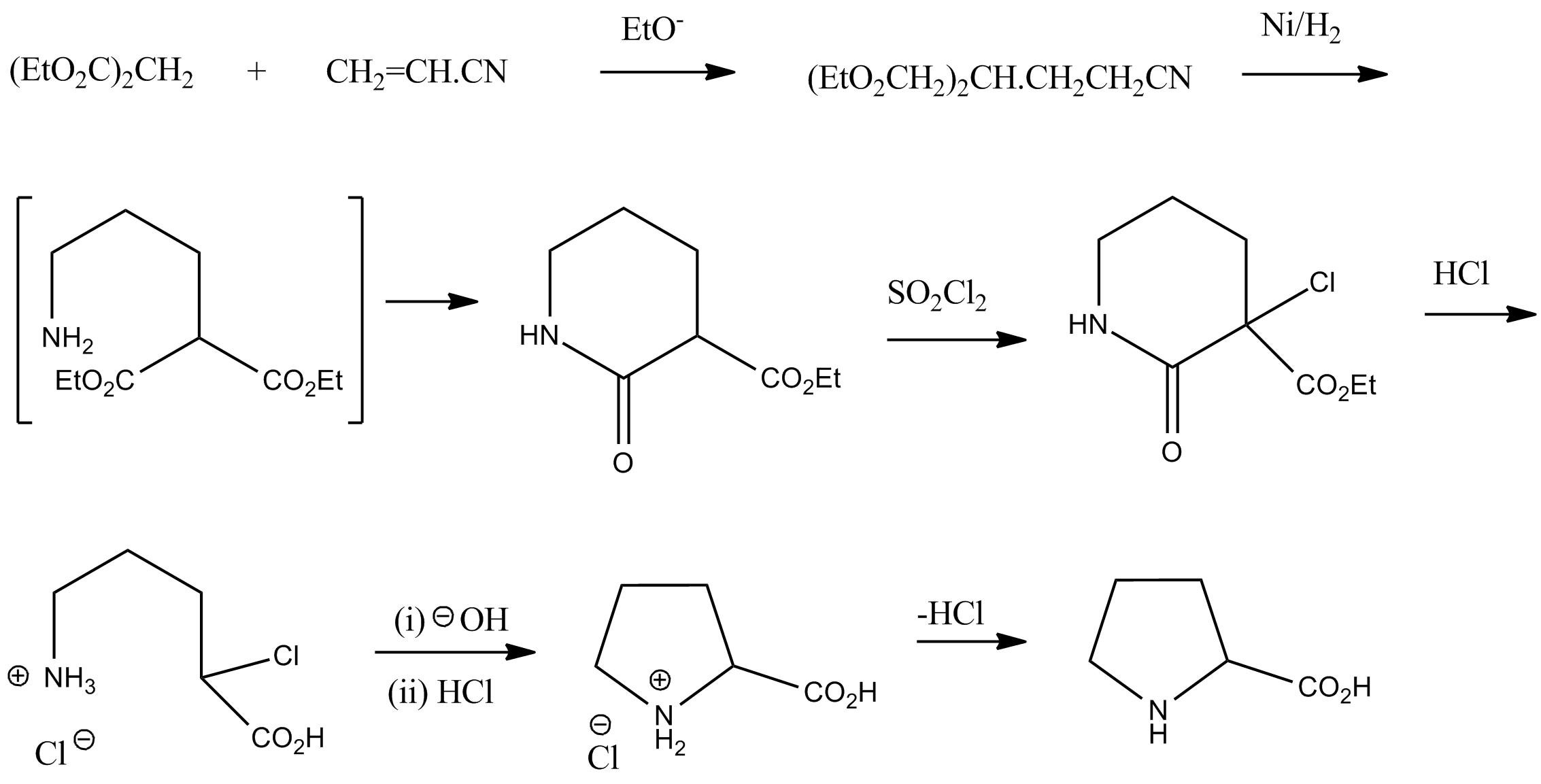
Synthesis
Racemic
In chemistry, a racemic mixture, or racemate (), is one that has equal amounts of left- and right-handed enantiomers of a chiral molecule or salt. Racemic mixtures are rare in nature, but many compounds are produced industrially as racemates. ...
proline can be synthesized from diethyl malonate
Diethyl malonate, also known as DEM, is the diethyl ester of malonic acid. It occurs naturally in grapes and strawberries as a colourless liquid with an apple-like odour, and is used in perfumes. It is also used to synthesize other compounds su ...
and acrylonitrile
Acrylonitrile is an organic compound with the formula and the structure . It is a colorless, volatile liquid although commercial samples can be yellow due to impurities. It has a pungent odor of garlic or onions. In terms of its molecular ...
:Vogel, ''Practical Organic Chemistry'' 5th edition
:
See also
*Hyperprolinemia
Hyperprolinemia is a condition which occurs when the amino acid proline is not broken down properly by the enzymes proline oxidase or 1-Pyrroline-5-carboxylate dehydrogenase, pyrroline-5-carboxylate dehydrogenase, causing a buildup of proline in t ...
* Inborn error of metabolism
Inborn errors of metabolism form a large class of genetic diseases involving congenital disorders of enzyme activities. The majority are due to defects of single genes that code for enzymes that facilitate conversion of various substances (substrat ...
* Prolidase deficiency
Prolidase deficiency (PD) is an extremely uncommon autosomal recessive disorder associated with collagen metabolism that affects connective tissues and thus a diverse array of organ systems more broadly, though it is extremely inconsistent in it ...
* Prolinol
Prolinol is a chiral amino-alcohol that is used as a chiral building block in organic synthesis. It exists as two enantiomers: the D and L forms.
Preparation
Prolinol is obtained by reduction of the amino acid proline using lithium aluminium hydri ...
References
Further reading
* .External links
Proline MS Spectrum
Proline biosynthesis
{{Authority control AMPA receptor agonists Kainate receptor agonists NMDA receptor agonists Proteinogenic amino acids Glucogenic amino acids Glycine receptor agonists Cyclic amino acids Pyrrolidines Secondary amino acids Excitatory amino acids