
A fertilizer (
American English
American English, sometimes called United States English or U.S. English, is the set of varieties of the English language native to the United States. English is the most widely spoken language in the United States and in most circumstances i ...
) or fertiliser (
British English
British English (BrE, en-GB, or BE) is, according to Lexico, Oxford Dictionaries, "English language, English as used in Great Britain, as distinct from that used elsewhere". More narrowly, it can refer specifically to the English language in ...
;
see spelling differences) is any material of natural or synthetic origin that is applied to soil or to plant tissues to supply
plant nutrients. Fertilizers may be distinct from
liming materials or other non-nutrient
soil amendments
A soil conditioner is a product which is added to soil to improve the soil’s physical qualities, usually its fertility (ability to provide nutrition for plants) and sometimes its mechanics. In general usage, the term "soil conditioner" is often ...
. Many sources of fertilizer exist, both natural and industrially produced.
[ For most modern agricultural practices, fertilization focuses on three main macro nutrients: ]nitrogen
Nitrogen is the chemical element with the symbol N and atomic number 7. Nitrogen is a nonmetal and the lightest member of group 15 of the periodic table, often called the pnictogens. It is a common element in the universe, estimated at se ...
(N), phosphorus
Phosphorus is a chemical element with the symbol P and atomic number 15. Elemental phosphorus exists in two major forms, white phosphorus and red phosphorus, but because it is highly reactive, phosphorus is never found as a free element on Ear ...
(P), and potassium
Potassium is the chemical element with the symbol K (from Neo-Latin ''kalium'') and atomic number19. Potassium is a silvery-white metal that is soft enough to be cut with a knife with little force. Potassium metal reacts rapidly with atmosph ...
(K) with occasional addition of supplements like rock flour
Rock flour, or glacial flour, consists of fine-grained, silt-sized particles of rock, generated by mechanical grinding of bedrock by glacial erosion or by artificial grinding to a similar size. Because the material is very small, it becomes suspe ...
for micronutrients. Farmers apply these fertilizers in a variety of ways: through dry or pelletized or liquid application processes, using large agricultural equipment or hand-tool methods.
Historically fertilization came from natural or organic sources: compost, animal manure, human manure, harvested minerals, crop rotation
Crop rotation is the practice of growing a series of different types of crops in the same area across a sequence of growing seasons. It reduces reliance on one set of nutrients, pest and weed pressure, and the probability of developing resistant ...
s and byproducts of human-nature industries (i.e. fish processing waste, or bloodmeal from animal slaughter
Animal slaughter is the killing of animals, usually referring to killing domestic livestock. It is estimated that each year 80 billion land animals are slaughtered for food. Most animals are slaughtered for food; however, they may also be slau ...
). However, starting in the 19th century, after innovations in plant nutrition
Plant nutrition is the study of the chemical elements and compounds necessary for plant growth and reproduction, plant metabolism and their external supply. In its absence the plant is unable to complete a normal life cycle, or that the element i ...
, an agricultural industry
Agriculture or farming is the practice of cultivating plants and livestock. Agriculture was the key development in the rise of sedentary human civilization, whereby farming of domesticated species created food surpluses that enabled people to ...
developed around synthetically created fertilizers. This transition was important in transforming the global food system, allowing for larger-scale industrial agriculture
Industrial agriculture is a form of modern farming that refers to the industrialized production of crops and animals and animal products like eggs or milk. The methods of industrial agriculture include innovation in agricultural machinery and f ...
with large crop yields.
Nitrogen-fixing
Nitrogen fixation is a chemical process by which molecular nitrogen (), with a strong triple covalent bond, in the air is converted into ammonia () or related nitrogenous compounds, typically in soil or aquatic systems but also in industry. Atm ...
chemical processes such as the Haber process
The Haber process, also called the Haber–Bosch process, is an artificial nitrogen fixation process and is the main industrial procedure for the production of ammonia today. It is named after its inventors, the German chemists Fritz Haber and ...
at the beginning of the 20th century, amplified by production capacity created during World War II led to a boom in using nitrogen fertilizers. In the latter half of the 20th century, increased the use of nitrogen fertilizers (800% increase between 1961 and 2019) have been a crucial component of the increased productivity of conventional food systems
The term food system describes the interconnected systems and processes that influence nutrition, food, health, community development, and agriculture. A food system includes all processes and infrastructure involved in feeding a population: growi ...
(more than 30% per capita) as part of the so-called "Green Revolution
The Green Revolution, also known as the Third Agricultural Revolution, was a period of technology transfer initiatives that saw greatly increased crop yields and agricultural production. These changes in agriculture began in developed countrie ...
". The use of artificial and industrially-applied fertilizers has led to a number of environmental impacts, creating water pollution
Water pollution (or aquatic pollution) is the contamination of water bodies, usually as a result of human activities, so that it negatively affects its uses. Water bodies include lakes, rivers, oceans, aquifers, reservoirs and groundwater. ...
and Eutrophication
Eutrophication is the process by which an entire body of water, or parts of it, becomes progressively enriched with minerals and nutrients, particularly nitrogen and phosphorus. It has also been defined as "nutrient-induced increase in phytopla ...
caused by nutritional runoff, carbon
Carbon () is a chemical element with the symbol C and atomic number 6. It is nonmetallic and tetravalent—its atom making four electrons available to form covalent chemical bonds. It belongs to group 14 of the periodic table. Carbon mak ...
and other emissions from fertilizer production and mining, and contamination and pollution of soil. Various types of sustainable agriculture
Sustainable agriculture is farming in sustainable ways meeting society's present food and textile needs, without compromising the ability for current or future generations to meet their needs. It can be based on an understanding of ecosystem ser ...
practices can be implemented to reduce the environmental impact of fertilizer uses alongside other environmental impacts of agriculture.
History
 Management of soil fertility has preoccupied farmers for thousands of years. Egyptians, Romans, Babylonians, and early Germans are all recorded as using minerals or manure to enhance the productivity of their farms.
Management of soil fertility has preoccupied farmers for thousands of years. Egyptians, Romans, Babylonians, and early Germans are all recorded as using minerals or manure to enhance the productivity of their farms.[ The science of plant nutrition started well before the work of German chemist Justus von Liebig although his name is most mentioned. Nicolas Théodore de Saussure and scientific colleagues at the time were quick to disprove the simplifications of Justus von Liebig. There was a complex scientific understanding of plant nutrition, where the role of humus and organo-mineral interactions were central, and which was in line with more recent discoveries from 1990 onwards. Prominent scientists on whom Justus von Liebig drew were ]Carl Ludwig Sprenger
__NOTOC__
Carl Ludwig Sprenger was a German botanist, born on 30 November 1846 at Güstrow, Mecklenburg and died 13 December 1917 on the island of Corfu (Kérkyra).
Sprenger lived in Naples from 1877 to 1907, and was a partner in the horticultu ...
and Hermann Hellriegel. In this field, a 'knowledge erosion' took place, partly driven by an intermingling of economics and research. John Bennet Lawes
Sir John Bennet Lawes, 1st Baronet, FRS (28 December 1814 – 31 August 1900) was an English entrepreneur and agricultural scientist. He founded an experimental farm at his home at Rothamsted Manor that eventually became Rothamsted Research, ...
, an English entrepreneur
Entrepreneurship is the creation or extraction of economic value. With this definition, entrepreneurship is viewed as change, generally entailing risk beyond what is normally encountered in starting a business, which may include other values t ...
, began to experiment on the effects of various manures on plants growing in pots in 1837, and a year or two later the experiments were extended to crops in the field. One immediate consequence was that in 1842 he patented a manure formed by treating phosphates with sulfuric acid, and thus was the first to create the artificial manure industry. In the succeeding year he enlisted the services of Joseph Henry Gilbert
Sir Joseph Henry Gilbert, Fellow of the Royal Society (1 August 1817 – 23 December 1901) was an English chemist, noteworthy for his long career spent improving the methods of practical agriculture. He was a fellow of the Royal Society.
Life
He ...
; together they performed crop experiments at the Institute of Arable Crops Research.
The Birkeland–Eyde process
The Birkeland–Eyde process was one of the competing industrial processes in the beginning of nitrogen-based fertilizer production. It is a multi-step nitrogen fixation reaction that uses electrical arcs to react atmospheric nitrogen (N2) with ox ...
was one of the competing industrial processes in the beginning of nitrogen-based fertilizer production. This process was used to fix atmospheric nitrogen
Nitrogen is the chemical element with the symbol N and atomic number 7. Nitrogen is a nonmetal and the lightest member of group 15 of the periodic table, often called the pnictogens. It is a common element in the universe, estimated at se ...
(N2) into nitric acid
Nitric acid is the inorganic compound with the formula . It is a highly corrosive mineral acid. The compound is colorless, but older samples tend to be yellow cast due to decomposition into oxides of nitrogen. Most commercially available nitri ...
(HNO3), one of several chemical processes generally referred to as nitrogen fixation
Nitrogen fixation is a chemical process by which molecular nitrogen (), with a strong triple covalent bond, in the air is converted into ammonia () or related nitrogenous compounds, typically in soil or aquatic systems but also in industry. Atmo ...
. The resultant nitric acid was then used as a source of nitrate (NO3−). A factory based on the process was built in Rjukan
Rjukan () is a town and the administrative centre of Tinn municipality in Telemark, Norway. It is situated in Vestfjorddalen, between Møsvatn and Lake Tinn, and got its name after Rjukan Falls west of the town. The Tinn municipality council gra ...
and Notodden
Notodden () is a city and municipality in Vestfold og Telemark county, Norway. It is part of the traditional region of Øst-Telemark. The administrative centre of the municipality is the city of Notodden.
Notodden was separated from the munic ...
in Norway, combined with the building of large hydroelectric power
Hydroelectricity, or hydroelectric power, is electricity generated from hydropower (water power). Hydropower supplies one sixth of the world's electricity, almost 4500 TWh in 2020, which is more than all other renewable sources combined an ...
facilities.
The 1910s and 1920s witnessed the rise of the Haber process
The Haber process, also called the Haber–Bosch process, is an artificial nitrogen fixation process and is the main industrial procedure for the production of ammonia today. It is named after its inventors, the German chemists Fritz Haber and ...
and the Ostwald process The Ostwald process is a chemical process used for making nitric acid (HNO3). Wilhelm Ostwald developed the process, and he patented it in 1902. The Ostwald process is a mainstay of the modern chemical industry, and it provides the main raw materi ...
. The Haber process produces ammonia (NH3) from methane
Methane ( , ) is a chemical compound with the chemical formula (one carbon atom bonded to four hydrogen atoms). It is a group-14 hydride, the simplest alkane, and the main constituent of natural gas. The relative abundance of methane on Ea ...
(CH4) (natural gas
Natural gas (also called fossil gas or simply gas) is a naturally occurring mixture of gaseous hydrocarbons consisting primarily of methane in addition to various smaller amounts of other higher alkanes. Low levels of trace gases like carbo ...
) gas and molecular nitrogen (N2) from the air. The ammonia from the Haber process is then partially converted into nitric acid
Nitric acid is the inorganic compound with the formula . It is a highly corrosive mineral acid. The compound is colorless, but older samples tend to be yellow cast due to decomposition into oxides of nitrogen. Most commercially available nitri ...
(HNO3) in the Ostwald process The Ostwald process is a chemical process used for making nitric acid (HNO3). Wilhelm Ostwald developed the process, and he patented it in 1902. The Ostwald process is a mainstay of the modern chemical industry, and it provides the main raw materi ...
. After World War II, Nitrogen production plants that had ramped up for war-time bomb manufacturing were pivoted towards agriculture uses.tonnes
The tonne ( or ; symbol: t) is a unit of mass equal to 1000 kilograms. It is a non-SI unit accepted for use with SI. It is also referred to as a metric ton to distinguish it from the non-metric units of the short ton (United States c ...
of nitrogen per year.hectare
The hectare (; SI symbol: ha) is a non-SI metric unit of area equal to a square with 100- metre sides (1 hm2), or 10,000 m2, and is primarily used in the measurement of land. There are 100 hectares in one square kilometre. An acre is ...
() requires of phosphate
In chemistry, a phosphate is an anion, salt, functional group or ester derived from a phosphoric acid. It most commonly means orthophosphate, a derivative of orthophosphoric acid .
The phosphate or orthophosphate ion is derived from phosph ...
fertilizer to be applied; soybean crops require about half, as 20–25 kg per hectare. Yara International
Yara International ASA is a Norwegian chemical company. It produces, distributes, and sells nitrogen-based mineral fertilizers and related industrial products. Its product line also includes phosphate and potash-based mineral fertilizers, as well ...
is the world's largest producer of nitrogen-based fertilizers.
Mechanism

 Fertilizers enhance the growth of plants. This goal is met in two ways, the traditional one being additives that provide nutrients. The second mode by which some fertilizers act is to enhance the effectiveness of the soil by modifying its water retention and aeration. This article, like many on fertilizers, emphasises the nutritional aspect.
Fertilizers typically provide, in varying proportions:
Fertilizers enhance the growth of plants. This goal is met in two ways, the traditional one being additives that provide nutrients. The second mode by which some fertilizers act is to enhance the effectiveness of the soil by modifying its water retention and aeration. This article, like many on fertilizers, emphasises the nutritional aspect.
Fertilizers typically provide, in varying proportions:[
* three main macronutrients:
** ]Nitrogen
Nitrogen is the chemical element with the symbol N and atomic number 7. Nitrogen is a nonmetal and the lightest member of group 15 of the periodic table, often called the pnictogens. It is a common element in the universe, estimated at se ...
(N): leaf growth
** Phosphorus
Phosphorus is a chemical element with the symbol P and atomic number 15. Elemental phosphorus exists in two major forms, white phosphorus and red phosphorus, but because it is highly reactive, phosphorus is never found as a free element on Ear ...
(P): development of roots, flowers, seeds, fruit;
** Potassium
Potassium is the chemical element with the symbol K (from Neo-Latin ''kalium'') and atomic number19. Potassium is a silvery-white metal that is soft enough to be cut with a knife with little force. Potassium metal reacts rapidly with atmosph ...
(K): strong stem growth, movement of water in plants, promotion of flowering and fruiting;
* three secondary macronutrients: calcium
Calcium is a chemical element with the symbol Ca and atomic number 20. As an alkaline earth metal, calcium is a reactive metal that forms a dark oxide-nitride layer when exposed to air. Its physical and chemical properties are most similar t ...
(Ca), magnesium
Magnesium is a chemical element with the symbol Mg and atomic number 12. It is a shiny gray metal having a low density, low melting point and high chemical reactivity. Like the other alkaline earth metals (group 2 of the periodic ta ...
(Mg), and sulfur (S);
* micronutrients: copper
Copper is a chemical element with the symbol Cu (from la, cuprum) and atomic number 29. It is a soft, malleable, and ductile metal with very high thermal and electrical conductivity. A freshly exposed surface of pure copper has a pinkis ...
(Cu), iron
Iron () is a chemical element with Symbol (chemistry), symbol Fe (from la, Wikt:ferrum, ferrum) and atomic number 26. It is a metal that belongs to the first transition series and group 8 element, group 8 of the periodic table. It is, Abundanc ...
(Fe), manganese
Manganese is a chemical element with the symbol Mn and atomic number 25. It is a hard, brittle, silvery metal, often found in minerals in combination with iron. Manganese is a transition metal with a multifaceted array of industrial alloy use ...
(Mn), molybdenum (Mo), zinc
Zinc is a chemical element with the symbol Zn and atomic number 30. Zinc is a slightly brittle metal at room temperature and has a shiny-greyish appearance when oxidation is removed. It is the first element in group 12 (IIB) of the periodi ...
(Zn), boron (B). Of occasional significance are silicon
Silicon is a chemical element with the symbol Si and atomic number 14. It is a hard, brittle crystalline solid with a blue-grey metallic luster, and is a tetravalent metalloid and semiconductor. It is a member of group 14 in the periodic ta ...
(Si), cobalt
Cobalt is a chemical element with the symbol Co and atomic number 27. As with nickel, cobalt is found in the Earth's crust only in a chemically combined form, save for small deposits found in alloys of natural meteoric iron. The free element, p ...
(Co), and vanadium (V).
The nutrients required for healthy plant life are classified according to the elements, but the elements are not used as fertilizers. Instead compounds containing these elements are the basis of fertilizers. The macro-nutrients are consumed in larger quantities and are present in plant tissue in quantities from 0.15% to 6.0% on a dry matter
The dry matter or dry weight is a measurement of the mass of something when completely dried.
Analysis of food
The dry matter of plant and animal material consists of all its constituents excluding water. The dry matter of food includes carboh ...
(DM) (0% moisture) basis. Plants are made up of four main elements: hydrogen, oxygen, carbon, and nitrogen. Carbon, hydrogen and oxygen are widely available as water and carbon dioxide. Although nitrogen makes up most of the atmosphere, it is in a form that is unavailable to plants. Nitrogen is the most important fertilizer since nitrogen is present in protein
Proteins are large biomolecules and macromolecules that comprise one or more long chains of amino acid residues. Proteins perform a vast array of functions within organisms, including catalysing metabolic reactions, DNA replication, res ...
s, DNA and other components (e.g., chlorophyll). To be nutritious to plants, nitrogen must be made available in a "fixed" form. Only some bacteria and their host plants (notably legumes) can fix atmospheric nitrogen (N2) by converting it to ammonia
Ammonia is an inorganic compound of nitrogen and hydrogen with the formula . A stable binary hydride, and the simplest pnictogen hydride, ammonia is a colourless gas with a distinct pungent smell. Biologically, it is a common nitrogenous wa ...
. Phosphate is required for the production of DNA and ATP, the main energy carrier in cells, as well as certain lipids.
Microbiological considerations
Two sets of enzymatic reactions are highly relevant to the efficiency of nitrogen-based fertilizers.
;Urease
The first is the hydrolysis (reaction with water) of urea. Many soil
Soil, also commonly referred to as earth or dirt
Dirt is an unclean matter, especially when in contact with a person's clothes, skin, or possessions. In such cases, they are said to become dirty.
Common types of dirt include:
* Debri ...
bacteria possess the enzyme urease
Ureases (), functionally, belong to the superfamily of amidohydrolases and phosphotriesterases. Ureases are found in numerous bacteria, fungi, algae, plants, and some invertebrates, as well as in soils, as a soil enzyme. They are nickel-contai ...
, which catalyzes conversion of urea to ammonium ion (NH4+) and bicarbonate ion (HCO3−).
;Ammonia oxidation
Ammonia-oxidizing bacteria (AOB), such as species of ''Nitrosomonas
''Nitrosomonas'' is a genus of Gram-negative bacteria, belonging to the Betaproteobacteria. It is one of the five genera of ammonia-oxidizing bacteria and, as an obligate chemolithoautotroph, uses ammonia (NH3) as an energy source and carbon di ...
'', oxidize ammonia to nitrite, a process termed nitrification
''Nitrification'' is the biological oxidation of ammonia to nitrite followed by the oxidation of the nitrite to nitrate occurring through separate organisms or direct ammonia oxidation to nitrate in comammox bacteria. The transformation of am ...
. Nitrite-oxidizing bacteria, especially ''Nitrobacter
''Nitrobacter'' is a genus comprising rod-shaped, gram-negative, and chemoautotrophic bacteria. The name ''Nitrobacter'' derives from the Latin neuter gender noun ''nitrum, nitri'', alkalis; the Ancient Greek noun βακτηρία'','' βακτ� ...
'', oxidize nitrite to nitrate, which is extremely mobile and is a major cause of eutrophication
Eutrophication is the process by which an entire body of water, or parts of it, becomes progressively enriched with minerals and nutrients, particularly nitrogen and phosphorus. It has also been defined as "nutrient-induced increase in phytopla ...
.
Classification
Fertilizers are classified in several ways. They are classified according to whether they provide a single nutrient (e.g., K, P, or N), in which case they are classified as "straight fertilizers". "multinutrient fertilizers" (or "complex fertilizers") provide two or more nutrients, for example N and P. Fertilizers are also sometimes classified as inorganic (the topic of most of this article) versus organic. Inorganic fertilizers exclude carbon-containing materials except ureas 220 px, B vitamin, is a urea.">B_vitamins.html" ;"title="Biotin, a water-soluble B vitamins">B vitamin, is a urea.
In chemistry, ureas are a class of organic compounds with the formula (R2N)2CO where R = H, alkyl, aryl, etc. Thus, in addition to ...
. Organic fertilizers are usually (recycled) plant- or animal-derived matter. Inorganic are sometimes called synthetic fertilizers since various chemical treatments are required for their manufacture.
Single nutrient ("straight") fertilizers
The main nitrogen-based straight fertilizer is ammonia or its solutions. Ammonium nitrate
Ammonium nitrate is a chemical compound with the chemical formula . It is a white crystalline salt consisting of ions of ammonium and nitrate. It is highly soluble in water and hygroscopic as a solid, although it does not form hydrates. It is ...
(NH4NO3) is also widely used. Urea
Urea, also known as carbamide, is an organic compound with chemical formula . This amide has two amino groups (–) joined by a carbonyl functional group (–C(=O)–). It is thus the simplest amide of carbamic acid.
Urea serves an important ...
is another popular source of nitrogen, having the advantage that it is solid and non-explosive, unlike ammonia and ammonium nitrate, respectively. A few percent of the nitrogen fertilizer market (4% in 2007)superphosphate Triple superphosphate is a component of fertilizer that primarily consists of monocalcium phosphate, Ca(H2PO4)2. Triple superphosphate is obtained by treating phosphate rock with phosphoric acid. Traditional routes for extraction of phosphate roc ...
s. "Single superphosphate" (SSP) consists of 14–18% P2O5, again in the form of Ca(H2PO4)2, but also phosphogypsum
Phosphogypsum (PG) is the calcium sulfate hydrate formed as a by-product of the production of fertilizer from phosphate rock. It is mainly composed of gypsum (CaSO4·2H2O). Although gypsum is a widely used material in the construction industry, p ...
(). Triple superphosphate Triple superphosphate is a component of fertilizer that primarily consists of monocalcium phosphate, Ca(H2PO4)2. Triple superphosphate is obtained by treating phosphate rock with phosphoric acid. Traditional routes for extraction of phosphate roc ...
(TSP) typically consists of 44–48% of P2O5 and no gypsum. A mixture of single superphosphate and triple superphosphate is called double superphosphate. More than 90% of a typical superphosphate fertilizer is water-soluble.
The main potassium-based straight fertilizer is muriate of potash (MOP). Muriate of potash
Potash () includes various mined and manufactured salts that contain potassium in water-soluble form. consists of 95–99% KCl, and is typically available as 0-0-60 or 0-0-62 fertilizer.
Multinutrient fertilizers
These fertilizers are common. They consist of two or more nutrient components.
;Binary (NP, NK, PK) fertilizers
Major two-component fertilizers provide both nitrogen and phosphorus to the plants. These are called NP fertilizers. The main NP fertilizers are monoammonium phosphate
Ammonium dihydrogen phosphate (ADP), also known as monoammonium phosphate (MAP) is a chemical compound with the chemical formula (NH4)(H2PO4). ADP is a major ingredient of agricultural fertilizers and some fire extinguishers. It also has sign ...
(MAP) and diammonium phosphate
Diammonium phosphate (DAP; IUPAC name diammonium hydrogen phosphate; chemical formula (NH4)2(HPO4) is one of a series of water-soluble ammonium phosphate salts that can be produced when ammonia reacts with phosphoric acid.
Solid diammonium phosp ...
(DAP). The active ingredient in MAP is NH4H2PO4. The active ingredient in DAP is (NH4)2HPO4. About 85% of MAP and DAP fertilizers are soluble in water.
;NPK fertilizers
NPK fertilizers are three-component fertilizers providing nitrogen, phosphorus, and potassium. There exist two types of NPK fertilizers: compound and blends. Compound NPK fertilizers contain chemically bound ingredients, while blended NPK fertilizers are physical mixtures of single nutrient components.
NPK rating is a rating system describing the amount of nitrogen, phosphorus, and potassium in a fertilizer. NPK ratings consist of three numbers separated by dashes (e.g., 10-10-10 or 16-4-8) describing the chemical content of fertilizers. The first number represents the percentage of nitrogen in the product; the second number, P2O5; the third, K2O. Fertilizers do not actually contain P2O5 or K2O, but the system is a conventional shorthand for the amount of the phosphorus (P) or potassium (K) in a fertilizer. A bag of fertilizer labeled 16-4-8 contains of nitrogen (16% of the 50 pounds), an amount of phosphorus equivalent to that in 2 pounds of P2O5 (4% of 50 pounds), and 4 pounds of K2O (8% of 50 pounds). Most fertilizers are labeled according to this N-P-K convention, although Australian convention, following an N-P-K-S system, adds a fourth number for sulfur, and uses elemental values for all values including P and K.
Micronutrients
Micronutrients
Micronutrients are essential dietary elements required by organisms in varying quantities throughout life to orchestrate a range of physiological functions to maintain health. Micronutrient requirements differ between organisms; for example, huma ...
are consumed in smaller quantities and are present in plant tissue on the order of parts-per-million
In science and engineering, the parts-per notation is a set of pseudo-units to describe small values of miscellaneous dimensionless quantities, e.g. mole fraction or mass fraction. Since these fractions are quantity-per-quantity measures, they ...
(ppm), ranging from 0.15 to 400 ppm or less than 0.04% dry matter.zinc
Zinc is a chemical element with the symbol Zn and atomic number 30. Zinc is a slightly brittle metal at room temperature and has a shiny-greyish appearance when oxidation is removed. It is the first element in group 12 (IIB) of the periodi ...
, molybdenum, iron
Iron () is a chemical element with Symbol (chemistry), symbol Fe (from la, Wikt:ferrum, ferrum) and atomic number 26. It is a metal that belongs to the first transition series and group 8 element, group 8 of the periodic table. It is, Abundanc ...
, and manganese
Manganese is a chemical element with the symbol Mn and atomic number 25. It is a hard, brittle, silvery metal, often found in minerals in combination with iron. Manganese is a transition metal with a multifaceted array of industrial alloy use ...
.[ These elements are provided as water-soluble salts. Iron presents special problems because it converts to insoluble (bio-unavailable) compounds at moderate soil pH and phosphate concentrations. For this reason, iron is often administered as a ]chelate complex
Chelation is a type of bonding of ions and molecules to metal ions. It involves the formation or presence of two or more separate coordinate bonds between a polydentate (multiple bonded) ligand and a single central metal atom. These ligands are ...
, e.g., the EDTA
Ethylenediaminetetraacetic acid (EDTA) is an aminopolycarboxylic acid with the formula H2N(CH2CO2H)2sub>2. This white, water-soluble solid is widely used to bind to iron (Fe2+/Fe3+) and calcium ions (Ca2+), forming water-soluble complexes ev ...
or EDDHA
EDDHA or ethylenediamine-''N'',''N′''-bis(2-hydroxyphenylacetic acid) is a chelating agent. Like EDTA, it binds metal ions as a hexadentate ligand, using two amines, two phenolate centers, and two carboxylates as the six binding sites. The ...
derivatives. The micronutrient needs depend on the plant and the environment. For example, sugar beets appear to require boron, and legumes require cobalt
Cobalt is a chemical element with the symbol Co and atomic number 27. As with nickel, cobalt is found in the Earth's crust only in a chemically combined form, save for small deposits found in alloys of natural meteoric iron. The free element, p ...
,
Environment
Synthetic fertilizer used in agriculture has wide-reaching environmental consequences. According to the Intergovernmental Panel on Climate Change (IPCC) Special Report on Climate Change and Land, production of these fertilizers and associated land use
Land use involves the management and modification of natural environment or wilderness into built environment such as settlements and semi-natural habitats such as arable fields, pastures, and managed woods. Land use by humans has a long ...
practices are drivers of global warming
In common usage, climate change describes global warming—the ongoing increase in global average temperature—and its effects on Earth's climate system. Climate change in a broader sense also includes previous long-term changes to E ...
. The use of fertilizer has also led to a number of direct environmental consequences: agricultural runoff
Agricultural pollution refers to biotic and abiotic byproducts of farming practices that result in contamination or degradation of the environment and surrounding ecosystems, and/or cause injury to humans and their economic interests. The pol ...
which leads to downstream effects like ocean dead zones and waterway contamination, soil microbiome
Soil microbiology is the study of microorganisms in soil, their functions, and how they affect soil properties. It is believed that between two and four billion years ago, the first ancient bacteria and microorganisms came about on Earth's ocea ...
degradation, and accumulation of toxins in ecosystems. Indirect environmental impacts include: the environmental impacts of fracking for natural gas
Natural gas (also called fossil gas or simply gas) is a naturally occurring mixture of gaseous hydrocarbons consisting primarily of methane in addition to various smaller amounts of other higher alkanes. Low levels of trace gases like carbo ...
used in the Haber process
The Haber process, also called the Haber–Bosch process, is an artificial nitrogen fixation process and is the main industrial procedure for the production of ammonia today. It is named after its inventors, the German chemists Fritz Haber and ...
, the agricultural boom is partially responsible for the rapid growth in human population and large-scale industrial agricultural practices are associated with habitat destruction
Habitat destruction (also termed habitat loss and habitat reduction) is the process by which a natural habitat becomes incapable of supporting its native species. The organisms that previously inhabited the site are displaced or dead, thereby ...
, pressure on biodiversity and agricultural soil loss.
In order to mitigate environmental and food security
Food security speaks to the availability of food in a country (or geography) and the ability of individuals within that country (geography) to access, afford, and source adequate foodstuffs. According to the United Nations' Committee on World ...
concerns, the international community has included food systems in Sustainable Development Goal 2 which focuses on creating a climate-friendly and sustainable food production system.[United Nations (2017) Resolution adopted by the General Assembly on 6 July 2017, Work of the Statistical Commission pertaining to the 2030 Agenda for Sustainable Development]
A/RES/71/313
Most policy and regulatory approaches to address these issues focus on pivoting agricultural practices towards sustainable or regenerative agricultural practices: these use less synthetic fertilizers, better soil management
Soil management is the application of operations, practices, and treatments to protect soil and enhance its performance (such as soil fertility or soil mechanics). It includes soil conservation, soil amendment, and optimal soil health. In agricu ...
(for example no-till agriculture) and more organic fertilizers.
Production
The production of synthetic, or inorganic, fertilizers requires prepared chemicals, whereas organic fertilizers are derived from the organic processes of plants and animals in biological processes using biochemicals.
Nitrogen fertilizers
 Nitrogen fertilizers are made from
Nitrogen fertilizers are made from ammonia
Ammonia is an inorganic compound of nitrogen and hydrogen with the formula . A stable binary hydride, and the simplest pnictogen hydride, ammonia is a colourless gas with a distinct pungent smell. Biologically, it is a common nitrogenous wa ...
(NH3) produced by the Haber–Bosch process
The Haber process, also called the Haber–Bosch process, is an artificial nitrogen fixation process and is the main industrial procedure for the production of ammonia today. It is named after its inventors, the German chemists Fritz Haber and C ...
.natural gas
Natural gas (also called fossil gas or simply gas) is a naturally occurring mixture of gaseous hydrocarbons consisting primarily of methane in addition to various smaller amounts of other higher alkanes. Low levels of trace gases like carbo ...
(CH4) usually supplies the hydrogen, and the nitrogen (N2) is derived from the air. This ammonia is used as a feedstock for all other nitrogen fertilizers, such as anhydrous ammonium nitrate (NH4NO3) and urea
Urea, also known as carbamide, is an organic compound with chemical formula . This amide has two amino groups (–) joined by a carbonyl functional group (–C(=O)–). It is thus the simplest amide of carbamic acid.
Urea serves an important ...
(CO(NH2)2).
Deposits of sodium nitrate (NaNO3) ( Chilean saltpeter) are also found in the Atacama desert
The Atacama Desert ( es, Desierto de Atacama) is a desert plateau in South America covering a 1,600 km (990 mi) strip of land on the Pacific coast, west of the Andes Mountains. The Atacama Desert is the driest nonpolar desert in th ...
in Chile
Chile, officially the Republic of Chile, is a country in the western part of South America. It is the southernmost country in the world, and the closest to Antarctica, occupying a long and narrow strip of land between the Andes to the east a ...
and was one of the original (1830) nitrogen-rich fertilizers used. It is still mined for fertilizer. Nitrates are also produced from ammonia by the Ostwald process The Ostwald process is a chemical process used for making nitric acid (HNO3). Wilhelm Ostwald developed the process, and he patented it in 1902. The Ostwald process is a mainstay of the modern chemical industry, and it provides the main raw materi ...
.
Phosphate fertilizers
 Phosphate fertilizers are obtained by extraction from
Phosphate fertilizers are obtained by extraction from phosphate rock
Phosphorite, phosphate rock or rock phosphate is a non-detrital sedimentary rock that contains high amounts of phosphate minerals. The phosphate content of phosphorite (or grade of phosphate rock) varies greatly, from 4% to 20% phosphorus pentox ...
, which contains two principal phosphorus-containing minerals, fluorapatite Ca5(PO4)3F (CFA) and hydroxyapatite Ca5(PO4)3OH. These minerals are converted into water-soluble phosphate salts by treatment with sulfuric (H2SO4) or phosphoric acid
Phosphoric acid (orthophosphoric acid, monophosphoric acid or phosphoric(V) acid) is a colorless, odorless phosphorus-containing solid, and inorganic compound with the chemical formula . It is commonly encountered as an 85% aqueous solution, w ...
s (H3PO4). The large production of sulfuric acid is primarily motivated by this application. In the nitrophosphate process
The nitrophosphate process (also known as the Odda process) is a method for the industrial production of nitrogen fertilizers invented by Erling Johnson in the municipality of Odda, Norway around 1927.
The process involves acidifying phosphate ro ...
or Odda process (invented in 1927), phosphate rock with up to a 20% phosphorus (P) content is dissolved with nitric acid
Nitric acid is the inorganic compound with the formula . It is a highly corrosive mineral acid. The compound is colorless, but older samples tend to be yellow cast due to decomposition into oxides of nitrogen. Most commercially available nitri ...
(HNO3) to produce a mixture of phosphoric acid (H3PO4) and calcium nitrate
Calcium nitrate, also called ''Norgessalpeter'' (Norwegian salpeter), is an inorganic compound with the formula Ca(NO3)2(H2O)x. The anhydrous compound, which is rarely encountered, absorbs moisture from the air to give the tetrahydrate. Both anhyd ...
(Ca(NO3)2). This mixture can be combined with a potassium fertilizer to produce a ''compound fertilizer'' with the three macronutrients N, P and K in easily dissolved form.
Potassium fertilizers
Potash
Potash () includes various mined and manufactured salts that contain potassium in water-soluble form. is a mixture of potassium minerals used to make potassium (chemical symbol: K) fertilizers. Potash is soluble in water, so the main effort in producing this nutrient from the ore involves some purification steps; e.g., to remove sodium chloride (NaCl) (common salt
Salt is a mineral composed primarily of sodium chloride (NaCl), a chemical compound belonging to the larger class of salts; salt in the form of a natural crystalline mineral is known as rock salt or halite. Salt is present in vast quant ...
). Sometimes potash is referred to as K2O, as a matter of convenience to those describing the potassium content. In fact, potash fertilizers are usually potassium chloride, potassium sulfate
Potassium sulfate (US) or potassium sulphate (UK), also called sulphate of potash (SOP), arcanite, or archaically potash of sulfur, is the inorganic compound with formula K2SO4, a white water-soluble solid. It is commonly used in fertilizers, prov ...
, potassium carbonate
Potassium carbonate is the inorganic compound with the formula K2 CO3. It is a white salt, which is soluble in water. It is deliquescent, often appearing as a damp or wet solid. Potassium carbonate is mainly used in the production of soap and gl ...
, or potassium nitrate
Potassium nitrate is a chemical compound with the chemical formula . This alkali metal nitrate salt is also known as Indian saltpetre (large deposits of which were historically mined in India). It is an ionic salt of potassium ions K+ and nitr ...
.[
]
NPK fertilizers
There are four major routes for manufacturing NPK fertilizers (named for their main ingredients: nitrogen (N), phosphorus (P), and potassium (K)):
# steam granulation,
# chemical granulation,
# compaction,
# bulk blending.
The first three processes are used to produce compound NPKs. During steam granulation raw materials are mixed and further granulated using steam as binding agent. Chemical granulation process is based on chemical reactions between liquid raw materials (such as phosphoric acid
Phosphoric acid (orthophosphoric acid, monophosphoric acid or phosphoric(V) acid) is a colorless, odorless phosphorus-containing solid, and inorganic compound with the chemical formula . It is commonly encountered as an 85% aqueous solution, w ...
, sulfuric acid, ammonia
Ammonia is an inorganic compound of nitrogen and hydrogen with the formula . A stable binary hydride, and the simplest pnictogen hydride, ammonia is a colourless gas with a distinct pungent smell. Biologically, it is a common nitrogenous wa ...
) and solid raw materials (such as potassium chloride, recycle material). Compaction implements high pressure to agglomerate dry powder materials. Lastly, bulk blends are produced by mixing straight fertilizers.
Organic fertilizers

 "
"Organic fertilizer
Organic fertilizers are fertilizers that are naturally produced. Fertilizers are materials that can be added to soil or plants, in order to provide nutrients and sustain growth. Typical organic fertilizers include all animal waste including mea ...
s" can describe those fertilizers with an organic – biologic – origin—that is, fertilizers derived from living or formerly living materials. Organic fertilizers can also describe commercially available and frequently packaged products that strive to follow the expectations and restrictions adopted by “organic agriculture
Organic farming, also known as ecological farming or biological farming,Labelling, article 30 o''Regulation (EU) 2018/848 of the European Parliament and of the Council of 30 May 2018 on organic production and labelling of organic products and re ...
” and ” environmentally friendly" gardening – related systems of food and plant production that significantly limit or strictly avoid the use of synthetic fertilizers and pesticides. The "organic fertilizer" ''products'' typically contain both some organic materials as well as acceptable additives such as nutritive rock powders, ground sea shells (crab, oyster, etc.), other prepared products such as seed meal or kelp, and cultivated microorganisms and derivatives.
Fertilizers of an organic origin (the first definition) include animal wastes, plant wastes from agriculture, seaweed, compost, and treated sewage sludge
Sewage sludge is the residual, semi-solid material that is produced as a by-product during sewage treatment of industrial or municipal wastewater. The term " septage" also refers to sludge from simple wastewater treatment but is connected to s ...
(biosolid
Biosolids are solid organic matter recovered from a sewage treatment process and used as fertilizer. In the past, it was common for farmers to use animal manure to improve their soil fertility. In the 1920s, the farming community began also to use ...
s). Beyond manures, animal sources can include products from the slaughter of animals – bloodmeal, bone meal
Bone meal is a mixture of finely and coarsely ground animal bones and slaughter-house waste products. It is used as a dietary supplement to supply calcium (Ca) and phosphorus (P) to monogastric livestock in the form of hydroxiapathite. As a slo ...
, feather meal, hides, hoofs, and horns all are typical components.peat
Peat (), also known as turf (), is an accumulation of partially decayed vegetation or organic matter. It is unique to natural areas called peatlands, bogs, mires, moors, or muskegs. The peatland ecosystem covers and is the most efficient ...
is the most widely used packaged organic soil amendment. It is an immature form of coal and improves the soil by aeration and absorbing water but confers no nutritional value to the plants. It is therefore not a fertilizer as defined in the beginning of the article, but rather an amendment. Coir
Coir (), also called coconut fibre, is a natural fibre extracted from the outer husk of coconut and used in products such as floor mats, doormats, brushes, and mattresses. Coir is the fibrous material found between the hard, internal shell an ...
, (derived from coconut husks), bark, and sawdust when added to soil all act similarly (but not identically) to peat and are also considered organic soil amendments – or texturizers – because of their limited nutritive inputs. Some organic additives can have a reverse effect on nutrients – fresh sawdust can consume soil nutrients as it breaks down, and may lower soil pH – but these same organic texturizers (as well as compost, etc.) may increase the availability of nutrients through improved cation exchange, or through increased growth of microorganisms that in turn increase availability of certain plant nutrients. Organic fertilizers such as composts and manures may be distributed locally without going into industry production, making actual consumption more difficult to quantify.
Application
 Fertilizers are commonly used for growing all crops, with application rates depending on the soil fertility, usually as measured by a
Fertilizers are commonly used for growing all crops, with application rates depending on the soil fertility, usually as measured by a soil test
Soil test may refer to one or more of a wide variety of soil analysis conducted for one of several possible reasons. Possibly the most widely conducted soil tests are those done to estimate the plant-available concentrations of plant nutrients, i ...
and according to the particular crop. Legumes, for example, fix nitrogen from the atmosphere and generally do not require nitrogen fertilizer.
Liquid vs solid
Fertilizers are applied to crops both as solids and as liquid. About 90% of fertilizers are applied as solids. The most widely used solid inorganic fertilizers are urea
Urea, also known as carbamide, is an organic compound with chemical formula . This amide has two amino groups (–) joined by a carbonyl functional group (–C(=O)–). It is thus the simplest amide of carbamic acid.
Urea serves an important ...
, diammonium phosphate and potassium chloride.prill
A prill is a small aggregate or globule of a material, most often a dry sphere, formed from a melted liquid through spray crystallization. Prilled is a term used in mining and manufacturing to refer to a product that has been pelletized. ANFO e ...
s, a solid globule. Liquid fertilizers comprise anhydrous ammonia, aqueous solutions of ammonia, aqueous solutions of ammonium nitrate or urea. These concentrated products may be diluted with water to form a concentrated liquid fertilizer (e.g., UAN
UAN is a solution of urea and ammonium nitrate in water used as a fertilizer.
Uan or UAN may also refer to:
* Adapa, an alternate name for the first of the Mesopotamian seven sages
* Autonomous University of Nayarit ((in Spanish: ), a Mexican pu ...
). Advantages of liquid fertilizer are its more rapid effect and easier coverage.[ The addition of fertilizer to irrigation water is called " fertigation".][
]
Urea
Urea is highly soluble in water and is therefore also very suitable for use in fertilizer solutions (in combination with ammonium nitrate: UAN), e.g., in 'foliar feed' fertilizers. For fertilizer use, granules are preferred over prills because of their narrower particle size distribution, which is an advantage for mechanical application.
Urea is usually spread at rates of between 40 and 300 kg/ha (35 to 270 lbs/acre) but rates vary. Smaller applications incur lower losses due to leaching. During summer, urea is often spread just before or during rain to minimize losses from volatilization (a process wherein nitrogen is lost to the atmosphere as ammonia gas).
Because of the high nitrogen concentration in urea, it is very important to achieve an even spread. Drilling must not occur on contact with or close to seed, due to the risk of germination damage. Urea dissolves in water for application as a spray or through irrigation systems.
In grain and cotton crops, urea is often applied at the time of the last cultivation before planting. In high rainfall areas and on sandy soils (where nitrogen can be lost through leaching) and where good in-season rainfall is expected, urea can be side- or top-dressed during the growing season. Top-dressing is also popular on pasture and forage crops. In cultivating sugarcane, urea is side-dressed after planting, and applied to each ratoon crop.
Because it absorbs moisture from the atmosphere, urea is often stored in closed containers.
Overdose or placing urea near seed is harmful.
Slow- and controlled-release fertilizers
Foliar application
Foliar fertilizers are applied directly to leaves. This method is almost invariably used to apply water-soluble straight nitrogen fertilizers and used especially for high-value crops such as fruits. Urea is the most common foliar fertilizer.[
]
Chemicals that affect nitrogen uptake
 Various chemicals are used to enhance the efficiency of nitrogen-based fertilizers. In this way farmers can limit the polluting effects of nitrogen run-off.
Various chemicals are used to enhance the efficiency of nitrogen-based fertilizers. In this way farmers can limit the polluting effects of nitrogen run-off. Nitrification
''Nitrification'' is the biological oxidation of ammonia to nitrite followed by the oxidation of the nitrite to nitrate occurring through separate organisms or direct ammonia oxidation to nitrate in comammox bacteria. The transformation of am ...
inhibitors (also known as nitrogen stabilizers) suppress the conversion of ammonia into nitrate, an anion that is more prone to leaching. 1-Carbamoyl-3-methylpyrazole (CMP), dicyandiamide
2-Cyanoguanidine is a nitrile derived from guanidine. It is a dimer of cyanamide, from which it can be prepared. 2-Cyanoguanidine is a colourless solid that is soluble in water, acetone, and alcohol, but not nonpolar organic solvents.
Production ...
, nitrapyrin
Nitrapyrin is an organic compound with the formula ClC5H3NCCl3, and is described as a white crystalline solid with a sweet odor.It is used as a nitrification inhibitor and bactericide, which is applied to soils for the growing of agricultural crop ...
(2-chloro-6-trichloromethylpyridine) and 3,4-Dimethylpyrazole phosphate (DMPP
Dimethylphenylpiperazinium (DMPP) is a nicotinic acetylcholine receptor agonist which is selective for the ganglionic subtype. One of the earliest reports on the pharmacology of DMPP, describing it as a ganglion-stimulating, hypertensive agent, ...
) are popular.urease
Ureases (), functionally, belong to the superfamily of amidohydrolases and phosphotriesterases. Ureases are found in numerous bacteria, fungi, algae, plants, and some invertebrates, as well as in soils, as a soil enzyme. They are nickel-contai ...
s. A popular inhibitor of ureases is N-(n-butyl)thiophosphoric triamide (NBPT
''N''-(''n''-Butyl)thiophosphoric triamide (NBPT) is the organophosphorus compound with the formula SP(NH2)2(NHC4H9). A white solid, NBPT is an "enhanced efficiency fertilizer", intended to limit the release of nitrogen-containing gases followi ...
).
Overfertilization
Careful use of fertilization technologies is important because excess nutrients can be detrimental. Fertilizer burn
Fertilizer burns occur when the use of too much fertilizer, the wrong type of fertilizer, or too little water with a fertilizer causes damage to a plant. Although fertilizer is used to help a plant grow by providing nutrients, too much will result ...
can occur when too much fertilizer is applied, resulting in damage or even death of the plant. Fertilizers vary in their tendency to burn roughly in accordance with their salt index
Salt is a mineral composed primarily of sodium chloride (NaCl), a chemical compound belonging to the larger class of salts; salt in the form of a natural crystalline mineral is known as rock salt or halite. Salt is present in vast quantiti ...
.
Statistics
 Recently nitrogen fertilizers have plateaued in most developed countries. China although has become the largest producer and consumer of nitrogen fertilizers. Africa has little reliance on nitrogen fertilizers. Agricultural and chemical minerals are very important in industrial use of fertilizers, which is valued at approximately $200 billion.
Recently nitrogen fertilizers have plateaued in most developed countries. China although has become the largest producer and consumer of nitrogen fertilizers. Africa has little reliance on nitrogen fertilizers. Agricultural and chemical minerals are very important in industrial use of fertilizers, which is valued at approximately $200 billion.[Vasant Gowariker, V. N. Krishnamurthy, Sudha Gowariker, Manik Dhanorkar, Kalyani Paranjape "The Fertilizer Encyclopedia" 2009, John Wiley & Sons. . Online . ]Global market
In economics, a market is a composition of systems, institutions, procedures, social relations or infrastructures whereby parties engage in exchange. While parties may exchange goods and services by barter, most markets rely on sellers offering ...
value is likely to rise to more than US$185 billion until 2019. The European fertilizer market will grow to earn revenues of approx. €15.3 billion in 2018.
Data on the fertilizer consumption per hectare arable land
Arable land (from the la, arabilis, "able to be ploughed") is any land capable of being ploughed and used to grow crops.''Oxford English Dictionary'', "arable, ''adj''. and ''n.''" Oxford University Press (Oxford), 2013. Alternatively, for th ...
in 2012 are published by The World Bank
The World Bank Group (WBG) is a family of five international organizations that make leveraged loans to developing countries. It is the largest and best-known development bank in the world and an observer at the United Nations Development G ...
. The diagram below shows fertilizer consumption by the European Union (EU) countries as kilograms per hectare (pounds per acre). The total consumption of fertilizer in the EU is 15.9 million tons for 105 million hectare arable land area
Environmental effects
Use of fertilizers are beneficial in providing nutrients to plants although they have some negative environmental effects. The large growing consumption of fertilizers can affect soil, surface water, and groundwater due to dispersion of mineral use. For each ton of phosphoric acid produced by the processing of phosphate rock, five tons of waste are generated. This waste takes the form of impure, useless, radioactive solid called
For each ton of phosphoric acid produced by the processing of phosphate rock, five tons of waste are generated. This waste takes the form of impure, useless, radioactive solid called phosphogypsum
Phosphogypsum (PG) is the calcium sulfate hydrate formed as a by-product of the production of fertilizer from phosphate rock. It is mainly composed of gypsum (CaSO4·2H2O). Although gypsum is a widely used material in the construction industry, p ...
. Estimates range from 100,000,000 and 280,000,000 tons of phosphogypsum waste are produced annually worldwide.
Water
 Phosphorus and nitrogen fertilizers when commonly used have major environmental effects. This is due to high rainfalls causing the fertilizers to be washed into waterways.
Phosphorus and nitrogen fertilizers when commonly used have major environmental effects. This is due to high rainfalls causing the fertilizers to be washed into waterways.eutrophic
Eutrophication is the process by which an entire body of water, or parts of it, becomes progressively enriched with minerals and nutrients, particularly nitrogen and phosphorus. It has also been defined as "nutrient-induced increase in phytoplan ...
. The main contributor to eutrophication is phosphate, which is normally a limiting nutrient; high concentrations promote the growth of cyanobacteria and algae, the demise of which consumes oxygen.[ Cyanobacteria blooms (' algal blooms') can also produce harmful toxins that can accumulate in the food chain, and can be harmful to humans.]ocean
The ocean (also the sea or the world ocean) is the body of salt water that covers approximately 70.8% of the surface of Earth and contains 97% of Earth's water. An ocean can also refer to any of the large bodies of water into which the wo ...
s, especially in coastal zones, lake
A lake is an area filled with water, localized in a basin, surrounded by land, and distinct from any river or other outlet that serves to feed or drain the lake. Lakes lie on land and are not part of the ocean, although, like the much large ...
s and river
A river is a natural flowing watercourse, usually freshwater, flowing towards an ocean, sea, lake or another river. In some cases, a river flows into the ground and becomes dry at the end of its course without reaching another body of w ...
s. The resulting lack of dissolved oxygen greatly reduces the ability of these areas to sustain oceanic fauna
Fauna is all of the animal life present in a particular region or time. The corresponding term for plants is ''flora'', and for fungi, it is ''funga''. Flora, fauna, funga and other forms of life are collectively referred to as ''Biota (ecology ...
. The number of oceanic dead zones near inhabited coastlines is increasing.
As of 2006, the application of nitrogen fertilizer is being increasingly controlled in northwestern Europegroundwater
Groundwater is the water present beneath Earth's surface in rock and soil pore spaces and in the fractures of rock formations. About 30 percent of all readily available freshwater in the world is groundwater. A unit of rock or an unconsolidated ...
can be broken down by natural processes.
Nitrate pollution
Only a fraction of the nitrogen-based fertilizers is converted to plant matter. The remainder accumulates in the soil or is lost as run-off.water solubility
An aqueous solution is a solution in which the solvent is water. It is mostly shown in chemical equations by appending (aq) to the relevant chemical formula. For example, a solution of table salt, or sodium chloride (NaCl), in water would be rep ...
of nitrate leads to increased runoff
Runoff, run-off or RUNOFF may refer to:
* RUNOFF, the first computer text-formatting program
* Runoff or run-off, another name for bleed, printing that lies beyond the edges to which a printed sheet is trimmed
* Runoff or run-off, a stock marke ...
into surface water as well as leaching
Leaching is the loss or extraction of certain materials from a carrier into a liquid (usually, but not always a solvent). and may refer to:
* Leaching (agriculture), the loss of water-soluble plant nutrients from the soil; or applying a small amou ...
into groundwater, thereby causing groundwater pollution
Groundwater pollution (also called groundwater contamination) occurs when pollutants are released to the ground and make their way into groundwater. This type of water pollution can also occur naturally due to the presence of a minor and unwante ...
. The excessive use of nitrogen-containing fertilizers (be they synthetic or natural) is particularly damaging, as much of the nitrogen that is not taken up by plants is transformed into nitrate which is easily leached.
Nitrate levels above 10 mg/L (10 ppm) in groundwater can cause 'blue baby syndrome
Blue baby syndrome can refer to conditions that cause cyanosis, or blueness of the skin, in babies as a result of low oxygen levels in the blood. This term has traditionally been applied to cyanosis as a result of:
#Cyanotic heart disease, whi ...
' (acquired methemoglobinemia). The nutrients, especially nitrates, in fertilizers can cause problems for natural habitats and for human health if they are washed off soil into watercourses or leached through soil into groundwater.
Soil
Acidification
Nitrogen-containing fertilizers can cause soil acidification
Soil acidification is the buildup of hydrogen cations, which reduces the soil pH. Chemically, this happens when a proton donor gets added to the soil. The donor can be an acid, such as nitric acid, sulfuric acid, or carbonic acid. It can also be a ...
when added. This may lead to decrease in nutrient availability which may be offset by liming.
Accumulation of toxic elements
=Cadmium
=
The concentration of cadmium
Cadmium is a chemical element with the symbol Cd and atomic number 48. This soft, silvery-white metal is chemically similar to the two other stable metals in group 12, zinc and mercury. Like zinc, it demonstrates oxidation state +2 in most of ...
in phosphorus-containing fertilizers varies considerably and can be problematic. For example, mono-ammonium phosphate fertilizer may have a cadmium content of as low as 0.14 mg/kg or as high as 50.9 mg/kg.Christmas island
Christmas Island, officially the Territory of Christmas Island, is an Australian external territory comprising the island of the same name. It is located in the Indian Ocean, around south of Java and Sumatra and around north-west of the ...
s). Continuous use of high-cadmium fertilizer can contaminate soil (as shown in New Zealand)plants
Plants are predominantly photosynthetic eukaryotes of the kingdom Plantae. Historically, the plant kingdom encompassed all living things that were not animals, and included algae and fungi; however, all current definitions of Plantae exclude ...
.European Commission
The European Commission (EC) is the executive of the European Union (EU). It operates as a cabinet government, with 27 members of the Commission (informally known as "Commissioners") headed by a President. It includes an administrative body ...
.[Wilfried Werner "Fertilizers, 6. Environmental Aspects" Ullmann's Encyclopedia of Industrial Chemistry, 2002, Wiley-VCH, Weinheim.]
=Fluoride
=
Phosphate rocks contain high levels of fluoride. Consequently, the widespread use of phosphate fertilizers has increased soil fluoride concentrations.
=Radioactive elements
=
The radioactive content of the fertilizers varies considerably and depends both on their concentrations in the parent mineral and on the fertilizer production process.
=Other metals
=
Steel industry wastes, recycled into fertilizers for their high levels of zinc
Zinc is a chemical element with the symbol Zn and atomic number 30. Zinc is a slightly brittle metal at room temperature and has a shiny-greyish appearance when oxidation is removed. It is the first element in group 12 (IIB) of the periodi ...
(essential to plant growth), wastes can include the following toxic metals: leadarsenic
Arsenic is a chemical element with the symbol As and atomic number 33. Arsenic occurs in many minerals, usually in combination with sulfur and metals, but also as a pure elemental crystal. Arsenic is a metalloid. It has various allotropes, ...
, cadmium
Cadmium is a chemical element with the symbol Cd and atomic number 48. This soft, silvery-white metal is chemically similar to the two other stable metals in group 12, zinc and mercury. Like zinc, it demonstrates oxidation state +2 in most of ...
,
Trace mineral depletion
Attention has been addressed to the decreasing concentrations of elements such as iron, zinc, copper and magnesium in many foods over the last 50–60 years.Intensive farming
Intensive agriculture, also known as intensive farming (as opposed to extensive farming), conventional, or industrial agriculture, is a type of agriculture, both of crop plants and of animals, with higher levels of input and output per unit of ...
practices, including the use of synthetic fertilizers are frequently suggested as reasons for these declines and organic farming is often suggested as a solution.zinc
Zinc is a chemical element with the symbol Zn and atomic number 30. Zinc is a slightly brittle metal at room temperature and has a shiny-greyish appearance when oxidation is removed. It is the first element in group 12 (IIB) of the periodi ...
, copper, manganese
Manganese is a chemical element with the symbol Mn and atomic number 25. It is a hard, brittle, silvery metal, often found in minerals in combination with iron. Manganese is a transition metal with a multifaceted array of industrial alloy use ...
, iron and molybdenum were identified as limiting the growth of broad-acre crops and pastures in the 1940s and 1950s.
Changes in soil biology
High levels of fertilizer may cause the breakdown of the symbiotic relationships between plant roots and mycorrhizal fungi.
Energy consumption and sustainability
In the US in 2004, 317 billion cubic feet of natural gas were consumed in the industrial production of ammonia, less than 1.5% of total U.S. annual consumption of natural gas.[IFA – Statistics – Fertilizer Indicators – Details – Raw material reserves, (2002–10)](_blank)
Ammonia is produced from natural gas
Natural gas (also called fossil gas or simply gas) is a naturally occurring mixture of gaseous hydrocarbons consisting primarily of methane in addition to various smaller amounts of other higher alkanes. Low levels of trace gases like carbo ...
and air.
Contribution to climate change
The greenhouse gases carbon dioxide
Carbon dioxide ( chemical formula ) is a chemical compound made up of molecules that each have one carbon atom covalently double bonded to two oxygen atoms. It is found in the gas state at room temperature. In the air, carbon dioxide is trans ...
, methane
Methane ( , ) is a chemical compound with the chemical formula (one carbon atom bonded to four hydrogen atoms). It is a group-14 hydride, the simplest alkane, and the main constituent of natural gas. The relative abundance of methane on Ea ...
and nitrous oxide
Nitrous oxide (dinitrogen oxide or dinitrogen monoxide), commonly known as laughing gas, nitrous, or nos, is a chemical compound, an oxide of nitrogen with the formula . At room temperature, it is a colourless non-flammable gas, and has ...
are produced during the manufacture of nitrogen fertilizer. is estimated as over 1% of global emissions. Nitrogen fertilizer can be converted by soil bacteria to nitrous oxide
Nitrous oxide (dinitrogen oxide or dinitrogen monoxide), commonly known as laughing gas, nitrous, or nos, is a chemical compound, an oxide of nitrogen with the formula . At room temperature, it is a colourless non-flammable gas, and has ...
, a greenhouse gas. Nitrous oxide emissions by humans, most of which are from fertilizer, between 2007 and 2016 have been estimated at 7 million tonnes per year, which is incompatible with limiting global warming to below 2 °C.
Atmosphere
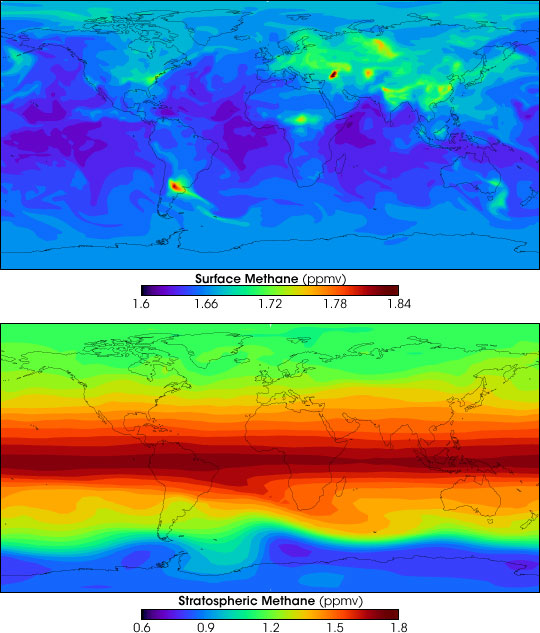 Through the increasing use of nitrogen fertilizer, which was used at a rate of about 110 million tons (of N) per year in 2012,
Through the increasing use of nitrogen fertilizer, which was used at a rate of about 110 million tons (of N) per year in 2012,nitrous oxide
Nitrous oxide (dinitrogen oxide or dinitrogen monoxide), commonly known as laughing gas, nitrous, or nos, is a chemical compound, an oxide of nitrogen with the formula . At room temperature, it is a colourless non-flammable gas, and has ...
(N2O) has become the third most important greenhouse gas after carbon dioxide and methane. It has a global warming potential
Global warming potential (GWP) is the heat absorbed by any greenhouse gas in the atmosphere, as a multiple of the heat that would be absorbed by the same mass of carbon dioxide (). GWP is 1 for . For other gases it depends on the gas and the time ...
296 times larger than an equal mass of carbon dioxide and it also contributes to stratospheric ozone depletion.
By changing processes and procedures, it is possible to mitigate some, but not all, of these effects on anthropogenic climate change
In common usage, climate change describes global warming—the ongoing increase in global average temperature—and its effects on Earth's climate system. Climate change in a broader sense also includes previous long-term changes to ...
.Methane emissions
Increasing methane emissions are a major contributor to the rising concentration of greenhouse gases in Earth's atmosphere, and are responsible for up to one-third of near-term global heating. During 2019, about 60% (360 million tons) of methane r ...
from crop fields (notably rice paddy fields) are increased by the application of ammonium-based fertilizers. These emissions contribute to global climate change as methane is a potent greenhouse gas.
Policy
Regulation
In Europe, problems with high nitrate concentrations in runoff are being addressed by the European Union's Nitrates Directive. Within Britain, farmers are encouraged to manage their land more sustainably in 'catchment-sensitive farming'. In the US, high concentrations of nitrate and phosphorus in runoff and drainage water are classified as nonpoint source pollutants due to their diffuse origin; this pollution is regulated at the state level. Oregon and Washington, both in the United States, have fertilizer registration programs with on-line databases listing chemical analyses of fertilizers.
In China, regulations have been implemented to control the use of N fertilizers in farming. In 2008, Chinese governments began to partially withdraw fertilizer subsidies, including subsidies to fertilizer transportation and to electricity and natural gas use in the industry. In consequence, the price of fertilizer has gone up and large-scale farms have begun to use less fertilizer. If large-scale farms keep reducing their use of fertilizer subsidies, they have no choice but to optimize the fertilizer they have which would therefore gain an increase in both grain yield and profit.
In March 2022, the United States Department of Agriculture announced a new $250M grant to promote American fertilizer production. Part of the Commodity Credit Corporation, the grant program will support fertilizer production that is independent of dominant fertilizer suppliers, made in America, and utilizing innovative production techniques to jumpstart future competition.
Two types of agricultural management practices include organic agriculture and conventional agriculture. The former encourages soil fertility using local resources to maximize efficiency. Organic agriculture avoids synthetic agrochemicals. Conventional agriculture uses all the components that organic agriculture does not use.
See also
* Agroecology
Agroecology (US: a-grō-ē-ˈkä-lə-jē) is an academic discipline that studies ecological processes applied to agricultural production systems. Bringing ecological principles to bear can suggest new management approaches in agroecosystems. Th ...
* Circulus (theory)
* Fertigation
* Food and Agriculture Organization
The Food and Agriculture Organization of the United Nations (FAO)french: link=no, Organisation des Nations unies pour l'alimentation et l'agriculture; it, Organizzazione delle Nazioni Unite per l'Alimentazione e l'Agricoltura is an intern ...
* History of organic farming
* Milorganite
* Leaf Color Chart
* Nutrient Recovery and Reuse
Resource recovery is using wastes as an input material to create valuable products as new outputs. The aim is to reduce the amount of waste generated, thereby reducing the need for landfill space, and optimising the values created from waste. Resou ...
* Phosphogypsum
Phosphogypsum (PG) is the calcium sulfate hydrate formed as a by-product of the production of fertilizer from phosphate rock. It is mainly composed of gypsum (CaSO4·2H2O). Although gypsum is a widely used material in the construction industry, p ...
* Soil defertilisation Soil defertilisation refers to the practice of reducing soil fertility in order to reduce the number of plants that can grow on that soil. It is often done on land not intended for agriculture, such as city parks.
Benefit
On land not intended fo ...
* Seaweed fertilizer
References
External links
Nitrogen for Feeding Our Food, Its Earthly Origin, Haber Process
International Fertilizer Industry Association (IFA)
Agriculture Guide, Complete Guide to Fertilizers and Fertilization
Nitrogen-Phosphorus-Potassium Values of Organic Fertilizers
{{Authority control
Horticulture
Climate change and agriculture
 A fertilizer (
A fertilizer (
 Phosphate fertilizers are obtained by extraction from
Phosphate fertilizers are obtained by extraction from  "
" Fertilizers are commonly used for growing all crops, with application rates depending on the soil fertility, usually as measured by a
Fertilizers are commonly used for growing all crops, with application rates depending on the soil fertility, usually as measured by a 

 Phosphorus and nitrogen fertilizers when commonly used have major environmental effects. This is due to high rainfalls causing the fertilizers to be washed into waterways. Agricultural run-off is a major contributor to the eutrophication of fresh water bodies. For example, in the US, about half of all the lakes are
Phosphorus and nitrogen fertilizers when commonly used have major environmental effects. This is due to high rainfalls causing the fertilizers to be washed into waterways. Agricultural run-off is a major contributor to the eutrophication of fresh water bodies. For example, in the US, about half of all the lakes are  Through the increasing use of nitrogen fertilizer, which was used at a rate of about 110 million tons (of N) per year in 2012, adding to the already existing amount of reactive nitrogen,
Through the increasing use of nitrogen fertilizer, which was used at a rate of about 110 million tons (of N) per year in 2012, adding to the already existing amount of reactive nitrogen,