Carbon (esports Team) on:
[Wikipedia]
[Google]
[Amazon]
Carbon () is a
 The
The
 The
The  At very high pressures, carbon forms the more compact allotrope,
At very high pressures, carbon forms the more compact allotrope,  Of the other discovered allotropes,
Of the other discovered allotropes,


 Carbon is the fourth most abundant chemical element in the
Carbon is the fourth most abundant chemical element in the
greatly upgraded database
for tracking
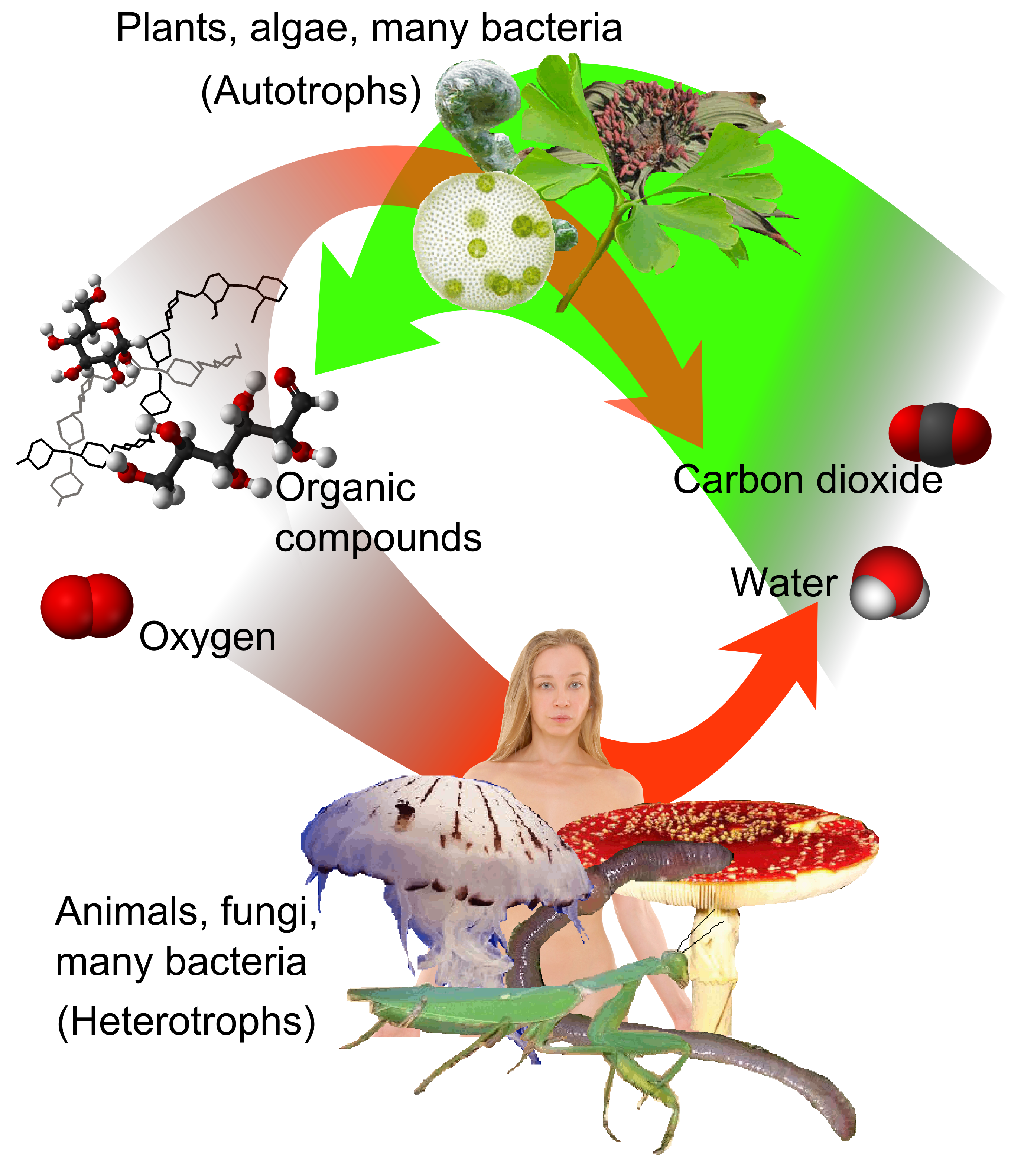
 Under terrestrial conditions, conversion of one element to another is very rare. Therefore, the amount of carbon on Earth is effectively constant. Thus, processes that use carbon must obtain it from somewhere and dispose of it somewhere else. The paths of carbon in the environment form the carbon cycle. For example, photosynthesis, photosynthetic plants draw
Under terrestrial conditions, conversion of one element to another is very rare. Therefore, the amount of carbon on Earth is effectively constant. Thus, processes that use carbon must obtain it from somewhere and dispose of it somewhere else. The paths of carbon in the environment form the carbon cycle. For example, photosynthesis, photosynthetic plants draw

 Carbon can form very long chains of interconnecting carbon–carbon bonds, a property that is called catenation. Carbon-carbon bonds are strong and stable. Through catenation, carbon forms a countless number of compounds. A tally of unique compounds shows that more contain carbon than do not.
A similar claim can be made for hydrogen because most organic compounds contain hydrogen chemically bonded to carbon or another common element like oxygen or nitrogen.
The simplest form of an organic molecule is the hydrocarbon—a large family of organic molecules that are composed of
Carbon can form very long chains of interconnecting carbon–carbon bonds, a property that is called catenation. Carbon-carbon bonds are strong and stable. Through catenation, carbon forms a countless number of compounds. A tally of unique compounds shows that more contain carbon than do not.
A similar claim can be made for hydrogen because most organic compounds contain hydrogen chemically bonded to carbon or another common element like oxygen or nitrogen.
The simplest form of an organic molecule is the hydrocarbon—a large family of organic molecules that are composed of
 It is important to note that in the cases above, each of the bonds to carbon contain less than two formal electron pairs. Thus, the formal electron count of these species does not exceed an octet. This makes them hypercoordinate but not hypervalent. Even in cases of alleged 10-C-5 species (that is, a carbon with five ligands and a formal electron count of ten), as reported by Akiba and co-workers, electronic structure calculations conclude that the electron population around carbon is still less than eight, as is true for other compounds featuring four-electron three-center bonding.
It is important to note that in the cases above, each of the bonds to carbon contain less than two formal electron pairs. Thus, the formal electron count of these species does not exceed an octet. This makes them hypercoordinate but not hypervalent. Even in cases of alleged 10-C-5 species (that is, a carbon with five ligands and a formal electron count of ten), as reported by Akiba and co-workers, electronic structure calculations conclude that the electron population around carbon is still less than eight, as is true for other compounds featuring four-electron three-center bonding.
 The English language, English name ''carbon'' comes from the Latin ''carbo'' for coal and charcoal, whence also comes the French language, French ''charbon'', meaning charcoal. In German language, German, Dutch language, Dutch and Danish language, Danish, the names for carbon are ''Kohlenstoff'', ''koolstof'' and ''kulstof'' respectively, all literally meaning
The English language, English name ''carbon'' comes from the Latin ''carbo'' for coal and charcoal, whence also comes the French language, French ''charbon'', meaning charcoal. In German language, German, Dutch language, Dutch and Danish language, Danish, the names for carbon are ''Kohlenstoff'', ''koolstof'' and ''kulstof'' respectively, all literally meaning  In 1722, René Antoine Ferchault de Réaumur demonstrated that iron was transformed into steel through the absorption of some substance, now known to be carbon. In 1772, Antoine Lavoisier showed that diamonds are a form of carbon; when he burned samples of charcoal and diamond and found that neither produced any water and that both released the same amount of
In 1722, René Antoine Ferchault de Réaumur demonstrated that iron was transformed into steel through the absorption of some substance, now known to be carbon. In 1772, Antoine Lavoisier showed that diamonds are a form of carbon; when he burned samples of charcoal and diamond and found that neither produced any water and that both released the same amount of
and Graphite: Mineral Commodity Summaries 2011 There are three types of natural graphite—amorphous, flake or crystalline flake, and vein or lump. Amorphous graphite is the lowest quality and most abundant. Contrary to science, in industry "amorphous" refers to very small crystal size rather than complete lack of crystal structure. Amorphous is used for lower value graphite products and is the lowest priced graphite. Large amorphous graphite deposits are found in China, Europe, Mexico and the United States. Flake graphite is less common and of higher quality than amorphous; it occurs as separate plates that crystallized in metamorphic rock. Flake graphite can be four times the price of amorphous. Good quality flakes can be processed into expandable graphite for many uses, such as flame retardants. The foremost deposits are found in Austria, Brazil, Canada, China, Germany and Madagascar. Vein or lump graphite is the rarest, most valuable, and highest quality type of natural graphite. It occurs in veins along intrusive contacts in solid lumps, and it is only commercially mined in Sri Lanka. According to the USGS, world production of natural graphite was 1.1 million tonnes in 2010, to which China contributed 800,000 t, India 130,000 t, Brazil 76,000 t, North Korea 30,000 t and Canada 25,000 t. No natural graphite was reported mined in the United States, but 118,000 t of synthetic graphite with an estimated value of $998 million was produced in 2009.
 The diamond supply chain is controlled by a limited number of powerful businesses, and is also highly concentrated in a small number of locations around the world (see figure).
Only a very small fraction of the diamond ore consists of actual diamonds. The ore is crushed, during which care has to be taken in order to prevent larger diamonds from being destroyed in this process and subsequently the particles are sorted by density. Today, diamonds are located in the diamond-rich density fraction with the help of X-ray fluorescence, after which the final sorting steps are done by hand. Before the use of X-rays became commonplace, the separation was done with grease belts; diamonds have a stronger tendency to stick to grease than the other minerals in the ore.
Historically diamonds were known to be found only in alluvial deposits in southern India. discussion on alluvial diamonds in India and elsewhere as well as earliest finds India led the world in diamond production from the time of their discovery in approximately the 9th century BC Ball was a Geologist in British service. Chapter I, Page 1 to the mid-18th century AD, but the commercial potential of these sources had been exhausted by the late 18th century and at that time India was eclipsed by Brazil where the first non-Indian diamonds were found in 1725.
Diamond production of primary deposits (kimberlites and lamproites) only started in the 1870s after the discovery of the diamond fields in South Africa. Production has increased over time and an accumulated total of over 4.5 billion carats have been mined since that date. Most commercially viable diamond deposits were in Russia, Botswana, Australia and the Democratic Republic of Congo. By 2005, Russia produced almost one-fifth of the global diamond output (mostly in Sakha Republic, Yakutia territory; for example, Mir Mine, Mir pipe and Udachnaya pipe) but the Argyle diamond mine, Argyle mine in Australia became the single largest source, producing 14 million carats in 2018. New finds, the Canada, Canadian mines at Diavik Diamond Mine, Diavik and Ekati Diamond Mine, Ekati, are expected to become even more valuable owing to their production of gem quality stones.
In the United States, diamonds have been found in Arkansas, Colorado and Montana. In 2004, a startling discovery of a microscopic diamond in the United States led to the January 2008 bulk-sampling of kimberlite pipes in a remote part of Montana.
The diamond supply chain is controlled by a limited number of powerful businesses, and is also highly concentrated in a small number of locations around the world (see figure).
Only a very small fraction of the diamond ore consists of actual diamonds. The ore is crushed, during which care has to be taken in order to prevent larger diamonds from being destroyed in this process and subsequently the particles are sorted by density. Today, diamonds are located in the diamond-rich density fraction with the help of X-ray fluorescence, after which the final sorting steps are done by hand. Before the use of X-rays became commonplace, the separation was done with grease belts; diamonds have a stronger tendency to stick to grease than the other minerals in the ore.
Historically diamonds were known to be found only in alluvial deposits in southern India. discussion on alluvial diamonds in India and elsewhere as well as earliest finds India led the world in diamond production from the time of their discovery in approximately the 9th century BC Ball was a Geologist in British service. Chapter I, Page 1 to the mid-18th century AD, but the commercial potential of these sources had been exhausted by the late 18th century and at that time India was eclipsed by Brazil where the first non-Indian diamonds were found in 1725.
Diamond production of primary deposits (kimberlites and lamproites) only started in the 1870s after the discovery of the diamond fields in South Africa. Production has increased over time and an accumulated total of over 4.5 billion carats have been mined since that date. Most commercially viable diamond deposits were in Russia, Botswana, Australia and the Democratic Republic of Congo. By 2005, Russia produced almost one-fifth of the global diamond output (mostly in Sakha Republic, Yakutia territory; for example, Mir Mine, Mir pipe and Udachnaya pipe) but the Argyle diamond mine, Argyle mine in Australia became the single largest source, producing 14 million carats in 2018. New finds, the Canada, Canadian mines at Diavik Diamond Mine, Diavik and Ekati Diamond Mine, Ekati, are expected to become even more valuable owing to their production of gem quality stones.
In the United States, diamonds have been found in Arkansas, Colorado and Montana. In 2004, a startling discovery of a microscopic diamond in the United States led to the January 2008 bulk-sampling of kimberlite pipes in a remote part of Montana.





 Carbon is essential to all known living systems, and without it life as we know it could not exist (see alternative biochemistry). The major economic use of carbon other than food and wood is in the form of hydrocarbons, most notably the fossil fuel methane gas and crude oil (petroleum). Petroleum, Crude oil is distillation, distilled in Oil refinery, refineries by the petrochemical industry to produce gasoline, kerosene, and other products. Cellulose is a natural, carbon-containing polymer produced by plants in the form of wood, cotton, linen, and hemp. Cellulose is used primarily for maintaining structure in plants. Commercially valuable carbon polymers of animal origin include wool, Cashmere wool, cashmere and silk. Plastics are made from synthetic carbon polymers, often with oxygen and nitrogen atoms included at regular intervals in the main polymer chain. The raw materials for many of these synthetic substances come from crude oil.
The uses of carbon and its compounds are extremely varied. It can form alloys with
Carbon is essential to all known living systems, and without it life as we know it could not exist (see alternative biochemistry). The major economic use of carbon other than food and wood is in the form of hydrocarbons, most notably the fossil fuel methane gas and crude oil (petroleum). Petroleum, Crude oil is distillation, distilled in Oil refinery, refineries by the petrochemical industry to produce gasoline, kerosene, and other products. Cellulose is a natural, carbon-containing polymer produced by plants in the form of wood, cotton, linen, and hemp. Cellulose is used primarily for maintaining structure in plants. Commercially valuable carbon polymers of animal origin include wool, Cashmere wool, cashmere and silk. Plastics are made from synthetic carbon polymers, often with oxygen and nitrogen atoms included at regular intervals in the main polymer chain. The raw materials for many of these synthetic substances come from crude oil.
The uses of carbon and its compounds are extremely varied. It can form alloys with
 Pure carbon has extremely low toxicity to humans and can be handled safely in the form of graphite or charcoal. It is resistant to dissolution or chemical attack, even in the acidic contents of the digestive tract. Consequently, once it enters into the body's tissues it is likely to remain there indefinitely. Carbon black was probably one of the first pigments to be used for tattooing, and Ötzi the Iceman was found to have carbon tattoos that survived during his life and for 5200 years after his death. Inhalation of coal dust or soot (carbon black) in large quantities can be dangerous, irritating lung tissues and causing the congestive human lung, lung disease, coalworker's pneumoconiosis. Diamond dust used as an abrasive can be harmful if ingested or inhaled. Microparticles of carbon are produced in diesel engine exhaust fumes, and may accumulate in the lungs. In these examples, the harm may result from contaminants (e.g., organic chemicals, heavy metals) rather than from the carbon itself.
Carbon generally has low toxicity to life, life on Earth; but carbon nanoparticles are deadly to ''Drosophila''.
Carbon may burn vigorously and brightly in the presence of air at high temperatures. Large accumulations of coal, which have remained inert for hundreds of millions of years in the absence of oxygen, may spontaneous combustion, spontaneously combust when exposed to air in coal mine waste tips, ship cargo holds and coal bunkers, and storage dumps.
In nuclear reactor, nuclear applications where graphite is used as a neutron moderator, accumulation of Wigner energy followed by a sudden, spontaneous release may occur. Annealing (metallurgy), Annealing to at least 250 °C can release the energy safely, although in the Windscale fire the procedure went wrong, causing other reactor materials to combust.
The great variety of carbon compounds include such lethal poisons as tetrodotoxin, the lectin ricin from seeds of the castor oil plant ''Ricinus communis'', cyanide (CN), and Carbon monoxide poisoning, carbon monoxide; and such essentials to life as glucose and protein.
Pure carbon has extremely low toxicity to humans and can be handled safely in the form of graphite or charcoal. It is resistant to dissolution or chemical attack, even in the acidic contents of the digestive tract. Consequently, once it enters into the body's tissues it is likely to remain there indefinitely. Carbon black was probably one of the first pigments to be used for tattooing, and Ötzi the Iceman was found to have carbon tattoos that survived during his life and for 5200 years after his death. Inhalation of coal dust or soot (carbon black) in large quantities can be dangerous, irritating lung tissues and causing the congestive human lung, lung disease, coalworker's pneumoconiosis. Diamond dust used as an abrasive can be harmful if ingested or inhaled. Microparticles of carbon are produced in diesel engine exhaust fumes, and may accumulate in the lungs. In these examples, the harm may result from contaminants (e.g., organic chemicals, heavy metals) rather than from the carbon itself.
Carbon generally has low toxicity to life, life on Earth; but carbon nanoparticles are deadly to ''Drosophila''.
Carbon may burn vigorously and brightly in the presence of air at high temperatures. Large accumulations of coal, which have remained inert for hundreds of millions of years in the absence of oxygen, may spontaneous combustion, spontaneously combust when exposed to air in coal mine waste tips, ship cargo holds and coal bunkers, and storage dumps.
In nuclear reactor, nuclear applications where graphite is used as a neutron moderator, accumulation of Wigner energy followed by a sudden, spontaneous release may occur. Annealing (metallurgy), Annealing to at least 250 °C can release the energy safely, although in the Windscale fire the procedure went wrong, causing other reactor materials to combust.
The great variety of carbon compounds include such lethal poisons as tetrodotoxin, the lectin ricin from seeds of the castor oil plant ''Ricinus communis'', cyanide (CN), and Carbon monoxide poisoning, carbon monoxide; and such essentials to life as glucose and protein.
Carbon
at ''The Periodic Table of Videos'' (University of Nottingham)
Carbon on Britannica
(archived 18 June 2010)
(archived 9 November 2001)
Carbon—Super Stuff. Animation with sound and interactive 3D-models.
(archived 9 November 2012) {{Carbon compounds Carbon, Chemical elements Allotropes of carbon Reactive nonmetals Polyatomic nonmetals Native element minerals Reducing agents Chemical elements with hexagonal planar structure
chemical element
A chemical element is a species of atoms that have a given number of protons in their nuclei, including the pure substance consisting only of that species. Unlike chemical compounds, chemical elements cannot be broken down into simpler sub ...
with the symbol
A symbol is a mark, sign, or word that indicates, signifies, or is understood as representing an idea, object, or relationship. Symbols allow people to go beyond what is known or seen by creating linkages between otherwise very different conc ...
C and atomic number
The atomic number or nuclear charge number (symbol ''Z'') of a chemical element is the charge number of an atomic nucleus. For ordinary nuclei, this is equal to the proton number (''n''p) or the number of protons found in the nucleus of every ...
6. It is nonmetal
In chemistry, a nonmetal is a chemical element that generally lacks a predominance of metallic properties; they range from colorless gases (like hydrogen) to shiny solids (like carbon, as graphite). The electrons in nonmetals behave differentl ...
lic and tetravalent
In chemistry, the valence (US spelling) or valency (British spelling) of an element is the measure of its combining capacity with other atoms when it forms chemical compounds or molecules.
Description
The combining capacity, or affinity of an ...
—its atom
Every atom is composed of a nucleus and one or more electrons bound to the nucleus. The nucleus is made of one or more protons and a number of neutrons. Only the most common variety of hydrogen has no neutrons.
Every solid, liquid, gas, and ...
making four electron
The electron ( or ) is a subatomic particle with a negative one elementary electric charge. Electrons belong to the first generation of the lepton particle family,
and are generally thought to be elementary particles because they have no kn ...
s available to form covalent
A covalent bond is a chemical bond that involves the sharing of electrons to form electron pairs between atoms. These electron pairs are known as shared pairs or bonding pairs. The stable balance of attractive and repulsive forces between atoms ...
chemical bond
A chemical bond is a lasting attraction between atoms or ions that enables the formation of molecules and crystals. The bond may result from the electrostatic force between oppositely charged ions as in ionic bonds, or through the sharing of ...
s. It belongs to group 14 of the periodic table
The periodic table, also known as the periodic table of the (chemical) elements, is a rows and columns arrangement of the chemical elements. It is widely used in chemistry, physics, and other sciences, and is generally seen as an icon of ch ...
. Carbon makes up about 0.025 percent of Earth's crust. Three isotopes
Isotopes are two or more types of atoms that have the same atomic number (number of protons in their nuclei) and position in the periodic table (and hence belong to the same chemical element), and that differ in nucleon numbers (mass numbers) ...
occur naturally, C and C being stable, while C is a radionuclide
A radionuclide (radioactive nuclide, radioisotope or radioactive isotope) is a nuclide that has excess nuclear energy, making it unstable. This excess energy can be used in one of three ways: emitted from the nucleus as gamma radiation; transfer ...
, decaying with a half-life
Half-life (symbol ) is the time required for a quantity (of substance) to reduce to half of its initial value. The term is commonly used in nuclear physics to describe how quickly unstable atoms undergo radioactive decay or how long stable ato ...
of about 5,730 years.
Carbon is one of the few elements known since antiquity.
Carbon is the 15th most abundant element in the Earth's crust, and the fourth most abundant element in the universe by mass after hydrogen
Hydrogen is the chemical element with the symbol H and atomic number 1. Hydrogen is the lightest element. At standard conditions hydrogen is a gas of diatomic molecules having the formula . It is colorless, odorless, tasteless, non-toxic, an ...
, helium
Helium (from el, ἥλιος, helios, lit=sun) is a chemical element with the symbol He and atomic number 2. It is a colorless, odorless, tasteless, non-toxic, inert, monatomic gas and the first in the noble gas group in the periodic table. ...
, and oxygen
Oxygen is the chemical element with the symbol O and atomic number 8. It is a member of the chalcogen group in the periodic table, a highly reactive nonmetal, and an oxidizing agent that readily forms oxides with most elements as wel ...
. Carbon's abundance, its unique diversity of organic compound
In chemistry, organic compounds are generally any chemical compounds that contain carbon-hydrogen or carbon-carbon bonds. Due to carbon's ability to catenate (form chains with other carbon atoms), millions of organic compounds are known. The ...
s, and its unusual ability to form polymer
A polymer (; Greek '' poly-'', "many" + ''-mer'', "part")
is a substance or material consisting of very large molecules called macromolecules, composed of many repeating subunits. Due to their broad spectrum of properties, both synthetic a ...
s at the temperatures commonly encountered on Earth
Earth is the third planet from the Sun and the only astronomical object known to harbor life. While large volumes of water can be found throughout the Solar System, only Earth sustains liquid surface water. About 71% of Earth's surfa ...
, enables this element to serve as a common element of all known life. It is the second most abundant element in the human body
The human body is the structure of a Human, human being. It is composed of many different types of Cell (biology), cells that together create Tissue (biology), tissues and subsequently organ systems. They ensure homeostasis and the life, viabi ...
by mass (about 18.5%) after oxygen.
The atoms of carbon can bond together in diverse ways, resulting in various allotropes of carbon
Carbon is capable of forming many allotropy, allotropes (structurally different forms of the same element) due to its Valence (chemistry), valency. Well-known forms of carbon include diamond and graphite. In recent decades, many more allotrope ...
. Well-known allotropes
Allotropy or allotropism () is the property of some chemical elements to exist in two or more different forms, in the same physical state, known as allotropes of the elements. Allotropes are different structural modifications of an element: the ...
include graphite
Graphite () is a crystalline form of the element carbon. It consists of stacked layers of graphene. Graphite occurs naturally and is the most stable form of carbon under standard conditions. Synthetic and natural graphite are consumed on large ...
, diamond
Diamond is a Allotropes of carbon, solid form of the element carbon with its atoms arranged in a crystal structure called diamond cubic. Another solid form of carbon known as graphite is the Chemical stability, chemically stable form of car ...
, amorphous carbon Amorphous carbon is free, reactive carbon that has no crystalline structure. Amorphous carbon materials may be stabilized by terminating dangling-π bonds with hydrogen. As with other amorphous solids, some short-range order can be observed. Amorp ...
and fullerenes
A fullerene is an allotrope of carbon whose molecule consists of carbon atoms connected by single and double bonds so as to form a closed or partially closed mesh, with fused rings of five to seven atoms. The molecule may be a hollow sphere, ...
. The physical properties
A physical property is any property that is measurable, whose value describes a state of a physical system. The changes in the physical properties of a system can be used to describe its changes between momentary states. Physical properties are o ...
of carbon vary widely with the allotropic form. For example, graphite is opaque
Opacity or opaque may refer to:
* Impediments to (especially, visible) light:
** Opacities, absorption coefficients
** Opacity (optics), property or degree of blocking the transmission of light
* Metaphors derived from literal optics:
** In lingui ...
and black while diamond is highly transparent
Transparency, transparence or transparent most often refer to:
* Transparency (optics), the physical property of allowing the transmission of light through a material
They may also refer to:
Literal uses
* Transparency (photography), a still, ...
. Graphite is soft enough to form a streak on paper (hence its name, from the Greek
Greek may refer to:
Greece
Anything of, from, or related to Greece, a country in Southern Europe:
*Greeks, an ethnic group.
*Greek language, a branch of the Indo-European language family.
**Proto-Greek language, the assumed last common ancestor ...
verb "γράφειν" which means "to write"), while diamond is the hardest naturally occurring material known. Graphite is a good electrical conductor
In physics and electrical engineering, a conductor is an object or type of material that allows the flow of charge (electric current) in one or more directions. Materials made of metal are common electrical conductors. Electric current is gener ...
while diamond has a low electrical conductivity
Electrical resistivity (also called specific electrical resistance or volume resistivity) is a fundamental property of a material that measures how strongly it resists electric current. A low resistivity indicates a material that readily allow ...
. Under normal conditions, diamond, carbon nanotube
A scanning tunneling microscopy image of a single-walled carbon nanotube
Rotating single-walled zigzag carbon nanotube
A carbon nanotube (CNT) is a tube made of carbon with diameters typically measured in nanometers.
''Single-wall carbon na ...
s, and graphene
Graphene () is an allotrope of carbon consisting of a single layer of atoms arranged in a hexagonal lattice nanostructure.
have the highest thermal conductivity, thermal conductivities of all known materials. All carbon allotropes are solids under normal conditions, with graphite being the most thermodynamically stable
In chemistry, chemical stability is the thermodynamic stability of a chemical system.
Thermodynamic stability occurs when a system is in its lowest energy state, or in chemical equilibrium with its environment. This may be a dynamic equilibriu ...
form at standard temperature and pressure. They are chemically resistant and require high temperature to react even with oxygen.
The most common oxidation state
In chemistry, the oxidation state, or oxidation number, is the hypothetical charge of an atom if all of its bonds to different atoms were fully ionic. It describes the degree of oxidation (loss of electrons) of an atom in a chemical compound. C ...
of carbon in inorganic compound
In chemistry, an inorganic compound is typically a chemical compound that lacks carbon–hydrogen bonds, that is, a compound that is not an organic compound. The study of inorganic compounds is a subfield of chemistry known as '' inorganic chemist ...
s is +4, while +2 is found in carbon monoxide
Carbon monoxide (chemical formula CO) is a colorless, poisonous, odorless, tasteless, flammable gas that is slightly less dense than air. Carbon monoxide consists of one carbon atom and one oxygen atom connected by a triple bond. It is the simple ...
and transition metal
In chemistry, a transition metal (or transition element) is a chemical element in the d-block of the periodic table (groups 3 to 12), though the elements of group 12 (and less often group 3) are sometimes excluded. They are the elements that can ...
carbonyl
In organic chemistry, a carbonyl group is a functional group composed of a carbon atom double-bonded to an oxygen atom: C=O. It is common to several classes of organic compounds, as part of many larger functional groups. A compound containing a ...
complexes. The largest sources of inorganic carbon are limestone
Limestone ( calcium carbonate ) is a type of carbonate sedimentary rock which is the main source of the material lime. It is composed mostly of the minerals calcite and aragonite, which are different crystal forms of . Limestone forms whe ...
s, dolomite Dolomite may refer to:
*Dolomite (mineral), a carbonate mineral
*Dolomite (rock), also known as dolostone, a sedimentary carbonate rock
*Dolomite, Alabama, United States, an unincorporated community
*Dolomite, California, United States, an unincor ...
s and carbon dioxide
Carbon dioxide (chemical formula ) is a chemical compound made up of molecules that each have one carbon atom covalently double bonded to two oxygen atoms. It is found in the gas state at room temperature. In the air, carbon dioxide is transpar ...
, but significant quantities occur in organic deposits of coal
Coal is a combustible black or brownish-black sedimentary rock, formed as rock strata called coal seams. Coal is mostly carbon with variable amounts of other elements, chiefly hydrogen, sulfur, oxygen, and nitrogen.
Coal is formed when dea ...
, peat
Peat (), also known as turf (), is an accumulation of partially decayed vegetation or organic matter. It is unique to natural areas called peatlands, bogs, mires, moors, or muskegs. The peatland ecosystem covers and is the most efficien ...
, oil
An oil is any nonpolar chemical substance that is composed primarily of hydrocarbons and is hydrophobic (does not mix with water) & lipophilic (mixes with other oils). Oils are usually flammable and surface active. Most oils are unsaturated ...
, and methane clathrate
Methane clathrate (CH4·5.75H2O) or (8CH4·46H2O), also called methane hydrate, hydromethane, methane ice, fire ice, natural gas hydrate, or gas hydrate, is a solid clathrate compound (more specifically, a clathrate hydrate) in which a large amou ...
s. Carbon forms a vast number of compounds, with about two hundred million having been described and indexed;
and yet that number is but a fraction of the number of theoretically possible compounds under standard conditions.
Characteristics
allotropes of carbon
Carbon is capable of forming many allotropy, allotropes (structurally different forms of the same element) due to its Valence (chemistry), valency. Well-known forms of carbon include diamond and graphite. In recent decades, many more allotrope ...
include graphite
Graphite () is a crystalline form of the element carbon. It consists of stacked layers of graphene. Graphite occurs naturally and is the most stable form of carbon under standard conditions. Synthetic and natural graphite are consumed on large ...
, one of the softest known substances, and diamond
Diamond is a Allotropes of carbon, solid form of the element carbon with its atoms arranged in a crystal structure called diamond cubic. Another solid form of carbon known as graphite is the Chemical stability, chemically stable form of car ...
, the hardest naturally occurring substance. It bonds readily with other small atom
Every atom is composed of a nucleus and one or more electrons bound to the nucleus. The nucleus is made of one or more protons and a number of neutrons. Only the most common variety of hydrogen has no neutrons.
Every solid, liquid, gas, and ...
s, including other carbon atoms, and is capable of forming multiple stable covalent
A covalent bond is a chemical bond that involves the sharing of electrons to form electron pairs between atoms. These electron pairs are known as shared pairs or bonding pairs. The stable balance of attractive and repulsive forces between atoms ...
bonds with suitable multivalent atoms. Carbon is a component element in the large majority of all chemical compounds
A chemical compound is a chemical substance composed of many identical molecules (or molecular entities) containing atoms from more than one chemical element held together by chemical bonds. A molecule consisting of atoms of only one element ...
, with about two hundred million examples having been described in the published chemical literature. Carbon also has the highest sublimation point of all elements. At atmospheric pressure
Atmospheric pressure, also known as barometric pressure (after the barometer), is the pressure within the atmosphere of Earth. The standard atmosphere (symbol: atm) is a unit of pressure defined as , which is equivalent to 1013.25 millibars, 7 ...
it has no melting point, as its triple point
In thermodynamics, the triple point of a substance is the temperature and pressure at which the three phases (gas, liquid, and solid) of that substance coexist in thermodynamic equilibrium.. It is that temperature and pressure at which the subli ...
is at and , so it sublimes at about .
Graphite is much more reactive than diamond at standard conditions, despite being more thermodynamically stable, as its delocalised pi system
In mathematics, a -system (or pi-system) on a set \Omega is a collection P of certain subsets of \Omega, such that
* P is non-empty.
* If A, B \in P then A \cap B \in P.
That is, P is a non-empty family of subsets of \Omega that is closed under ...
is much more vulnerable to attack. For example, graphite can be oxidised by hot concentrated nitric acid
Nitric acid is the inorganic compound with the formula . It is a highly corrosive mineral acid. The compound is colorless, but older samples tend to be yellow cast due to decomposition into oxides of nitrogen. Most commercially available nitri ...
at standard conditions to mellitic acid
Mellitic acid, also called graphitic acid or benzenehexacarboxylic acid, is an acid first discovered in 1799 by Martin Heinrich Klaproth in the mineral mellite (honeystone), which is the aluminium salt of the acid. It crystallizes in fine silky ne ...
, C6(CO2H)6, which preserves the hexagonal units of graphite while breaking up the larger structure.Greenwood and Earnshaw, pp. 289–292.
Carbon sublimes in a carbon arc, which has a temperature of about 5800 K (5,530 °C or 9,980 °F). Thus, irrespective of its allotropic form, carbon remains solid at higher temperatures than the highest-melting-point metals such as tungsten
Tungsten, or wolfram, is a chemical element with the symbol W and atomic number 74. Tungsten is a rare metal found naturally on Earth almost exclusively as compounds with other elements. It was identified as a new element in 1781 and first isolat ...
or rhenium
Rhenium is a chemical element with the symbol Re and atomic number 75. It is a silvery-gray, heavy, third-row transition metal in group 7 of the periodic table. With an estimated average concentration of 1 part per billion (ppb), rhenium is one ...
. Although thermodynamically prone to oxidation
Redox (reduction–oxidation, , ) is a type of chemical reaction in which the oxidation states of substrate change. Oxidation is the loss of electrons or an increase in the oxidation state, while reduction is the gain of electrons or a d ...
, carbon resists oxidation more effectively than elements such as iron
Iron () is a chemical element with symbol Fe (from la, ferrum) and atomic number 26. It is a metal that belongs to the first transition series and group 8 of the periodic table. It is, by mass, the most common element on Earth, right in f ...
and copper
Copper is a chemical element with the symbol Cu (from la, cuprum) and atomic number 29. It is a soft, malleable, and ductile metal with very high thermal and electrical conductivity. A freshly exposed surface of pure copper has a pinkis ...
, which are weaker reducing agents at room temperature.
Carbon is the sixth element, with a ground-state electron configuration
In atomic physics and quantum chemistry, the electron configuration is the distribution of electrons of an atom or molecule (or other physical structure) in atomic or molecular orbitals. For example, the electron configuration of the neon atom ...
of 1s22s22p2, of which the four outer electrons are valence electron
In chemistry and physics, a valence electron is an electron in the outer shell associated with an atom, and that can participate in the formation of a chemical bond if the outer shell is not closed. In a single covalent bond, a shared pair forms ...
s. Its first four ionisation energies, 1086.5, 2352.6, 4620.5 and 6222.7 kJ/mol, are much higher than those of the heavier group-14 elements. The electronegativity of carbon is 2.5, significantly higher than the heavier group-14 elements (1.8–1.9), but close to most of the nearby nonmetals, as well as some of the second- and third-row transition metal
In chemistry, a transition metal (or transition element) is a chemical element in the d-block of the periodic table (groups 3 to 12), though the elements of group 12 (and less often group 3) are sometimes excluded. They are the elements that can ...
s. Carbon's covalent radii are normally taken as 77.2 pm (C−C), 66.7 pm (C=C) and 60.3 pm (C≡C), although these may vary depending on coordination number and what the carbon is bonded to. In general, covalent radius decreases with lower coordination number and higher bond order.Greenwood and Earnshaw, pp. 276–8.
Carbon-based compounds form the basis of all known life on Earth
Earth is the third planet from the Sun and the only astronomical object known to harbor life. While large volumes of water can be found throughout the Solar System, only Earth sustains liquid surface water. About 71% of Earth's surfa ...
, and the carbon-nitrogen-oxygen cycle
The CNO cycle (for carbon–nitrogen–oxygen; sometimes called Bethe–Weizsäcker cycle after Hans Albrecht Bethe and Carl Friedrich von Weizsäcker) is one of the two known sets of fusion reactions by which stars convert hydrogen to helium, t ...
provides a small portion of the energy produced by the Sun
The Sun is the star at the center of the Solar System. It is a nearly perfect ball of hot plasma, heated to incandescence by nuclear fusion reactions in its core. The Sun radiates this energy mainly as light, ultraviolet, and infrared radi ...
, and most of the energy in larger star
A star is an astronomical object comprising a luminous spheroid of plasma (physics), plasma held together by its gravity. The List of nearest stars and brown dwarfs, nearest star to Earth is the Sun. Many other stars are visible to the naked ...
s (e.g. Sirius
Sirius is the list of brightest stars, brightest star in the night sky. Its name is derived from the Ancient Greek language, Greek word , or , meaning 'glowing' or 'scorching'. The star is designated α Canis Majoris, Latinisation ...
). Although it forms an extraordinary variety of compounds, most forms of carbon are comparatively unreactive under normal conditions. At standard temperature and pressure, it resists all but the strongest oxidizers. It does not react with sulfuric acid
Sulfuric acid (American spelling and the preferred IUPAC name) or sulphuric acid ( Commonwealth spelling), known in antiquity as oil of vitriol, is a mineral acid composed of the elements sulfur, oxygen and hydrogen, with the molecular formu ...
, hydrochloric acid
Hydrochloric acid, also known as muriatic acid, is an aqueous solution of hydrogen chloride. It is a colorless solution with a distinctive pungent smell. It is classified as a strong acid
Acid strength is the tendency of an acid, symbol ...
, chlorine
Chlorine is a chemical element with the Symbol (chemistry), symbol Cl and atomic number 17. The second-lightest of the halogens, it appears between fluorine and bromine in the periodic table and its properties are mostly intermediate betwee ...
or any alkalis
In chemistry, an alkali (; from ar, القلوي, al-qaly, lit=ashes of the saltwort) is a basic, ionic salt of an alkali metal or an alkaline earth metal. An alkali can also be defined as a base that dissolves in water. A solution of a so ...
. At elevated temperatures, carbon reacts with oxygen
Oxygen is the chemical element with the symbol O and atomic number 8. It is a member of the chalcogen group in the periodic table, a highly reactive nonmetal, and an oxidizing agent that readily forms oxides with most elements as wel ...
to form carbon oxides
In chemistry, an oxocarbon or oxide of carbon is a chemical compound consisting only of carbon and oxygen. The simplest and most common oxocarbons are carbon monoxide (CO) and carbon dioxide (). Many other stable (practically if not thermodynamica ...
and will rob oxygen from metal oxides to leave the elemental metal. This exothermic reaction
In thermochemistry, an exothermic reaction is a "reaction for which the overall standard enthalpy change Δ''H''⚬ is negative." Exothermic reactions usually release heat. The term is often confused with exergonic reaction, which IUPAC defines ...
is used in the iron and steel industry to smelt
Smelt may refer to:
* Smelting, chemical process
* The common name of various fish:
** Smelt (fish), a family of small fish, Osmeridae
** Australian smelt in the family Retropinnidae and species ''Retropinna semoni''
** Big-scale sand smelt ''At ...
iron and to control the carbon content of steel
Steel is an alloy made up of iron with added carbon to improve its strength and fracture resistance compared to other forms of iron. Many other elements may be present or added. Stainless steels that are corrosion- and oxidation-resistant ty ...
:
: + 4 C + 2 → 3 Fe + 4 .
Carbon reacts with sulfur
Sulfur (or sulphur in British English) is a chemical element with the symbol S and atomic number 16. It is abundant, multivalent and nonmetallic. Under normal conditions, sulfur atoms form cyclic octatomic molecules with a chemical formula ...
to form carbon disulfide
Carbon disulfide (also spelled as carbon disulphide) is a neurotoxic, colorless, volatile liquid with the formula and structure . The compound is used frequently as a building block in organic chemistry as well as an industrial and chemical non ...
, and it reacts with steam in the coal-gas reaction used in coal gasification Coal gasification is the process of producing syngas—a mixture consisting primarily of carbon monoxide (CO), hydrogen (H2), carbon dioxide (CO2), methane (CH4), and water vapour (H2O)—from coal and water, air and/or oxygen.
Historically, coal ...
:
:C + HO → CO + H.
Carbon combines with some metals at high temperatures to form metallic carbides, such as the iron carbide cementite
Cementite (or iron carbide) is a compound of iron and carbon, more precisely an intermediate transition metal carbide with the formula Fe3C. By weight, it is 6.67% carbon and 93.3% iron. It has an orthorhombic crystal structure. It is a hard, brit ...
in steel and tungsten carbide
Tungsten carbide (chemical formula: WC) is a chemical compound (specifically, a carbide) containing equal parts of tungsten and carbon atoms. In its most basic form, tungsten carbide is a fine gray powder, but it can be pressed and formed into ...
, widely used as an abrasive
An abrasive is a material, often a mineral, that is used to shape or finish a workpiece through rubbing which leads to part of the workpiece being worn away by friction. While finishing a material often means polishing it to gain a smooth, reflec ...
and for making hard tips for cutting tools.
The system of carbon allotropes spans a range of extremes:
Allotropes
Atomic carbon
Atomic carbon, systematically named carbon and λ0-methane, also called monocarbon, is a colourless gaseous inorganic chemical with the chemical formula C (also written . It is kinetically unstable at ambient temperature and pressure, being remo ...
is a very short-lived species and, therefore, carbon is stabilized in various multi-atomic structures with diverse molecular configurations called allotropes
Allotropy or allotropism () is the property of some chemical elements to exist in two or more different forms, in the same physical state, known as allotropes of the elements. Allotropes are different structural modifications of an element: the ...
. The three relatively well-known allotropes of carbon are amorphous carbon Amorphous carbon is free, reactive carbon that has no crystalline structure. Amorphous carbon materials may be stabilized by terminating dangling-π bonds with hydrogen. As with other amorphous solids, some short-range order can be observed. Amorp ...
, graphite
Graphite () is a crystalline form of the element carbon. It consists of stacked layers of graphene. Graphite occurs naturally and is the most stable form of carbon under standard conditions. Synthetic and natural graphite are consumed on large ...
, and diamond
Diamond is a Allotropes of carbon, solid form of the element carbon with its atoms arranged in a crystal structure called diamond cubic. Another solid form of carbon known as graphite is the Chemical stability, chemically stable form of car ...
. Once considered exotic, fullerene
A fullerene is an allotrope of carbon whose molecule consists of carbon atoms connected by single and double bonds so as to form a closed or partially closed mesh, with fused rings of five to seven atoms. The molecule may be a hollow sphere, ...
s are nowadays commonly synthesized and used in research; they include buckyball
Buckminsterfullerene is a type of fullerene with the formula C60. It has a cage-like fused-ring structure (truncated icosahedron) made of twenty hexagons and twelve pentagons, and resembles a soccer ball. Each of its 60 carbon atoms is bonded t ...
s,
carbon nanotube
A scanning tunneling microscopy image of a single-walled carbon nanotube
Rotating single-walled zigzag carbon nanotube
A carbon nanotube (CNT) is a tube made of carbon with diameters typically measured in nanometers.
''Single-wall carbon na ...
s,
carbon nanobud
In nanotechnology, a carbon nanobud is a material that combines carbon nanotubes and spheroidal fullerenes, both allotropes of carbon, forming "buds" attached to the tubes. Carbon nanobuds were discovered and synthesized in 2006.
In this mat ...
s and nanofibers
Nanofibers are fibers with diameters in the nanometer range (typically, between 1 nm and 1 μm). Nanofibers can be generated from different polymers and hence have different physical properties and application potentials. Examples of natural polyme ...
. Several other exotic allotropes have also been discovered, such as lonsdaleite
Lonsdaleite (named in honour of Kathleen Lonsdale), also called hexagonal diamond in reference to the crystal structure, is an allotrope of carbon with a hexagonal lattice, as opposed to the cubical lattice of conventional diamond. It is found i ...
,
glassy carbon
Glass-like carbon, often called glassy carbon or vitreous carbon, is a non-graphitizing, or nongraphitizable, carbon which combines glassy and ceramic properties with those of graphite. The most important properties are high temperature resis ...
, carbon nanofoam Carbon nanofoam is an allotrope of carbon discovered in 1997 by Andrei V. Rode and co-workers at the Australian National University in Canberra. It consists of a cluster-assembly of carbon atoms strung together in a loose three-dimensional web. The ...
and linear acetylenic carbon
Linear acetylenic carbon (LAC), also known as carbyne or Linear Carbon Chain (LCC), is an allotrope of carbon that has the chemical structure as a repeat unit, with alternating single and triple bonds. It would thus be the ultimate member of the ...
(carbyne).
Graphene
Graphene () is an allotrope of carbon consisting of a single layer of atoms arranged in a hexagonal lattice nanostructure.
is a two-dimensional sheet of carbon with the atoms arranged in a hexagonal lattice. As of 2009, graphene appears to be the strongest material ever tested.
*
The process of separating it from graphite
Graphite () is a crystalline form of the element carbon. It consists of stacked layers of graphene. Graphite occurs naturally and is the most stable form of carbon under standard conditions. Synthetic and natural graphite are consumed on large ...
will require some further technological development before it is economical for industrial processes.
If successful, graphene could be used in the construction of a space elevator
A space elevator, also referred to as a space bridge, star ladder, and orbital lift, is a proposed type of planet-to-space transportation system, often depicted in science fiction. The main component would be a cable (also called a tether) anc ...
. It could also be used to safely store hydrogen for use in a hydrogen based engine in cars.
 The
The amorphous
In condensed matter physics and materials science, an amorphous solid (or non-crystalline solid, glassy solid) is a solid that lacks the long-range order that is characteristic of a crystal.
Etymology
The term comes from the Greek ''a'' ("wi ...
form is an assortment of carbon atoms in a non-crystalline, irregular, glassy state, not held in a crystalline macrostructure. It is present as a powder, and is the main constituent of substances such as charcoal
Charcoal is a lightweight black carbon residue produced by strongly heating wood (or other animal and plant materials) in minimal oxygen to remove all water and volatile constituents. In the traditional version of this pyrolysis process, cal ...
, lampblack
Carbon black (subtypes are acetylene black, channel black, furnace black, lamp black and thermal black) is a material produced by the incomplete combustion of coal and coal tar, vegetable matter, or petroleum products, including fuel oil, fluid ...
(soot
Soot ( ) is a mass of impure carbon particles resulting from the incomplete combustion of hydrocarbons. It is more properly restricted to the product of the gas-phase combustion process but is commonly extended to include the residual pyrolysed ...
) and activated carbon
Activated carbon, also called activated charcoal, is a form of carbon commonly used to filter contaminants from water and air, among many other uses. It is processed (activated) to have small, low-volume pores that increase the surface area avail ...
. At normal pressures, carbon takes the form of graphite, in which each atom is bonded trigonally to three others in a plane composed of fused hexagon
In geometry, a hexagon (from Ancient Greek, Greek , , meaning "six", and , , meaning "corner, angle") is a six-sided polygon. The total of the internal angles of any simple polygon, simple (non-self-intersecting) hexagon is 720°.
Regular hexa ...
al rings, just like those in aromatic hydrocarbon
Aromatic compounds, also known as "mono- and polycyclic aromatic hydrocarbons", are organic compounds containing one or more aromatic rings. The parent member of aromatic compounds is benzene. The word "aromatic" originates from the past grouping ...
s. The resulting network is 2-dimensional, and the resulting flat sheets are stacked and loosely bonded through weak van der Waals force
In molecular physics, the van der Waals force is a distance-dependent interaction between atoms or molecules. Unlike ionic or covalent bonds, these attractions do not result from a chemical electronic bond; they are comparatively weak and th ...
s. This gives graphite its softness and its cleaving properties (the sheets slip easily past one another). Because of the delocalization of one of the outer electrons of each atom to form a π-cloud, graphite conducts electricity
Electricity is the set of physical phenomena associated with the presence and motion of matter that has a property of electric charge. Electricity is related to magnetism, both being part of the phenomenon of electromagnetism, as described ...
, but only in the plane of each covalently bonded
A covalent bond is a chemical bond that involves the sharing of electrons to form electron pairs between atoms. These electron pairs are known as shared pairs or bonding pairs. The stable balance of attractive and repulsive forces between atoms ...
sheet. This results in a lower bulk electrical conductivity
Electrical resistivity (also called specific electrical resistance or volume resistivity) is a fundamental property of a material that measures how strongly it resists electric current. A low resistivity indicates a material that readily allow ...
for carbon than for most metal
A metal (from Greek μέταλλον ''métallon'', "mine, quarry, metal") is a material that, when freshly prepared, polished, or fractured, shows a lustrous appearance, and conducts electricity and heat relatively well. Metals are typicall ...
s. The delocalization also accounts for the energetic stability of graphite over diamond at room temperature.
diamond
Diamond is a Allotropes of carbon, solid form of the element carbon with its atoms arranged in a crystal structure called diamond cubic. Another solid form of carbon known as graphite is the Chemical stability, chemically stable form of car ...
, having nearly twice the density of graphite. Here, each atom is bonded tetrahedrally to four others, forming a 3-dimensional network of puckered six-membered rings of atoms. Diamond has the same cubic structure
In crystallography, the cubic (or isometric) crystal system is a crystal system where the unit cell is in the shape of a cube. This is one of the most common and simplest shapes found in crystals and minerals.
There are three main varieties of ...
as silicon
Silicon is a chemical element with the symbol Si and atomic number 14. It is a hard, brittle crystalline solid with a blue-grey metallic luster, and is a tetravalent metalloid and semiconductor. It is a member of group 14 in the periodic tab ...
and germanium
Germanium is a chemical element with the symbol Ge and atomic number 32. It is lustrous, hard-brittle, grayish-white and similar in appearance to silicon. It is a metalloid in the carbon group that is chemically similar to its group neighbors s ...
, and because of the strength of the carbon-carbon bonds, it is the hardest naturally occurring substance measured by resistance to scratching. Contrary to the popular belief that ''"diamonds are forever"'', they are thermodynamically unstable (Δf''G''°(diamond, 298 K) = 2.9 kJ/mol) under normal conditions (298 K, 105 Pa) and should theoretically transform into graphite
Graphite () is a crystalline form of the element carbon. It consists of stacked layers of graphene. Graphite occurs naturally and is the most stable form of carbon under standard conditions. Synthetic and natural graphite are consumed on large ...
.
But due to a high activation energy
In chemistry and physics, activation energy is the minimum amount of energy that must be provided for compounds to result in a chemical reaction. The activation energy (''E''a) of a reaction is measured in joules per mole (J/mol), kilojoules pe ...
barrier, the transition into graphite is so slow at normal temperature that it is unnoticeable. However, at very high temperatures diamond will turn into graphite, and diamonds can burn up in a house fire. The bottom left corner of the phase diagram for carbon has not been scrutinized experimentally. Although a computational study employing density functional theory
Density-functional theory (DFT) is a computational quantum mechanical modelling method used in physics, chemistry and materials science to investigate the electronic structure (or nuclear structure) (principally the ground state) of many-body ...
methods reached the conclusion that as and , diamond becomes more stable than graphite by approximately 1.1 kJ/mol, more recent and definitive experimental and computational studies show that graphite is more stable than diamond for , without applied pressure, by 2.7 kJ/mol at ''T'' = 0 K and 3.2 kJ/mol at ''T'' = 298.15 K. Under some conditions, carbon crystallizes as lonsdaleite
Lonsdaleite (named in honour of Kathleen Lonsdale), also called hexagonal diamond in reference to the crystal structure, is an allotrope of carbon with a hexagonal lattice, as opposed to the cubical lattice of conventional diamond. It is found i ...
, a hexagonal
In geometry, a hexagon (from Greek , , meaning "six", and , , meaning "corner, angle") is a six-sided polygon. The total of the internal angles of any simple (non-self-intersecting) hexagon is 720°.
Regular hexagon
A '' regular hexagon'' has ...
crystal
A crystal or crystalline solid is a solid material whose constituents (such as atoms, molecules, or ions) are arranged in a highly ordered microscopic structure, forming a crystal lattice that extends in all directions. In addition, macros ...
lattice with all atoms covalently bonded and properties similar to those of diamond.
Fullerene
A fullerene is an allotrope of carbon whose molecule consists of carbon atoms connected by single and double bonds so as to form a closed or partially closed mesh, with fused rings of five to seven atoms. The molecule may be a hollow sphere, ...
s are a synthetic crystalline formation with a graphite-like structure, but in place of flat hexagonal cells only, some of the cells of which fullerenes are formed may be pentagons, nonplanar hexagons, or even heptagons of carbon atoms. The sheets are thus warped into spheres, ellipses, or cylinders. The properties of fullerenes (split into buckyballs, buckytubes, and nanobuds) have not yet been fully analyzed and represent an intense area of research in nanomaterials
*
Nanomaterials describe, in principle, materials of which a single unit is sized (in at least one dimension) between 1 and 100 nm (the usual definition of nanoscale).
Nanomaterials research takes a materials science-based approach to nan ...
. The names ''fullerene'' and ''buckyball'' are given after Richard Buckminster Fuller
Richard Buckminster Fuller (; July 12, 1895 – July 1, 1983) was an American architect, systems theorist, writer, designer, inventor, philosopher, and futurist. He styled his name as R. Buckminster Fuller in his writings, publishing more t ...
, popularizer of geodesic dome
A geodesic dome is a hemispherical thin-shell structure (lattice-shell) based on a geodesic polyhedron. The triangular elements of the dome are structurally rigid and distribute the structural stress throughout the structure, making geodesic dom ...
s, which resemble the structure of fullerenes. The buckyballs are fairly large molecules formed completely of carbon bonded trigonally, forming spheroid
A spheroid, also known as an ellipsoid of revolution or rotational ellipsoid, is a quadric surface obtained by rotating an ellipse about one of its principal axes; in other words, an ellipsoid with two equal semi-diameters. A spheroid has cir ...
s (the best-known and simplest is the soccerball-shaped C buckminsterfullerene
Buckminsterfullerene is a type of fullerene with the formula C60. It has a cage-like fused-ring structure (truncated icosahedron) made of twenty hexagons and twelve pentagons, and resembles a soccer ball. Each of its 60 carbon atoms is bonded ...
). Carbon nanotubes (buckytubes) are structurally similar to buckyballs, except that each atom is bonded trigonally in a curved sheet that forms a hollow cylinder
A cylinder (from ) has traditionally been a three-dimensional solid, one of the most basic of curvilinear geometric shapes. In elementary geometry, it is considered a prism with a circle as its base.
A cylinder may also be defined as an infin ...
. Nanobuds were first reported in 2007 and are hybrid buckytube/buckyball materials (buckyballs are covalently bonded to the outer wall of a nanotube) that combine the properties of both in a single structure.
 Of the other discovered allotropes,
Of the other discovered allotropes, carbon nanofoam Carbon nanofoam is an allotrope of carbon discovered in 1997 by Andrei V. Rode and co-workers at the Australian National University in Canberra. It consists of a cluster-assembly of carbon atoms strung together in a loose three-dimensional web. The ...
is a ferromagnetic
Ferromagnetism is a property of certain materials (such as iron) which results in a large observed magnetic permeability, and in many cases a large magnetic coercivity allowing the material to form a permanent magnet. Ferromagnetic materials ...
allotrope discovered in 1997. It consists of a low-density cluster-assembly of carbon atoms strung together in a loose three-dimensional web, in which the atoms are bonded trigonally in six- and seven-membered rings. It is among the lightest known solids, with a density of about 2 kg/m. Similarly, glassy carbon
Glass-like carbon, often called glassy carbon or vitreous carbon, is a non-graphitizing, or nongraphitizable, carbon which combines glassy and ceramic properties with those of graphite. The most important properties are high temperature resis ...
contains a high proportion of closed porosity
Porosity or void fraction is a measure of the void (i.e. "empty") spaces in a material, and is a fraction of the volume of voids over the total volume, between 0 and 1, or as a percentage between 0% and 100%. Strictly speaking, some tests measure ...
, but contrary to normal graphite, the graphitic layers are not stacked like pages in a book, but have a more random arrangement. Linear acetylenic carbon
Linear acetylenic carbon (LAC), also known as carbyne or Linear Carbon Chain (LCC), is an allotrope of carbon that has the chemical structure as a repeat unit, with alternating single and triple bonds. It would thus be the ultimate member of the ...
has the chemical structure −(C≡C)− . Carbon in this modification is linear with ''sp'' orbital hybridization
In chemistry, orbital hybridisation (or hybridization) is the concept of mixing atomic orbitals to form new ''hybrid orbitals'' (with different energies, shapes, etc., than the component atomic orbitals) suitable for the pairing of electrons to f ...
, and is a polymer
A polymer (; Greek '' poly-'', "many" + ''-mer'', "part")
is a substance or material consisting of very large molecules called macromolecules, composed of many repeating subunits. Due to their broad spectrum of properties, both synthetic a ...
with alternating single and triple bonds. This carbyne is of considerable interest to nanotechnology
Nanotechnology, also shortened to nanotech, is the use of matter on an atomic, molecular, and supramolecular scale for industrial purposes. The earliest, widespread description of nanotechnology referred to the particular technological goal o ...
as its Young's modulus
Young's modulus E, the Young modulus, or the modulus of elasticity in tension or compression (i.e., negative tension), is a mechanical property that measures the tensile or compressive stiffness of a solid material when the force is applied leng ...
is 40 times that of the hardest known material – diamond.
In 2015, a team at the North Carolina State University
North Carolina State University (NC State) is a public land-grant research university in Raleigh, North Carolina. Founded in 1887 and part of the University of North Carolina system, it is the largest university in the Carolinas. The universit ...
announced the development of another allotrope they have dubbed Q-carbon Amorphous carbon is free, reactive carbon that has no crystalline structure. Amorphous carbon materials may be stabilized by terminating dangling-π bonds with hydrogen. As with other amorphous solids, some short-range order can be observed. Amorph ...
, created by a high energy low duration laser pulse on amorphous carbon dust. Q-carbon is reported to exhibit ferromagnetism, fluorescence
Fluorescence is the emission of light by a substance that has absorbed light or other electromagnetic radiation. It is a form of luminescence. In most cases, the emitted light has a longer wavelength, and therefore a lower photon energy, tha ...
, and a hardness superior to diamonds.
In the vapor phase, some of the carbon is in the form of dicarbon
Diatomic carbon (systematically named dicarbon and 1λ2,2λ2-ethene), is a green, gaseous inorganic chemical with the chemical formula C=C (also written 2or C2). It is kinetically unstable at ambient temperature and pressure, being removed throug ...
(). When excited, this gas glows green.
Occurrence


 Carbon is the fourth most abundant chemical element in the
Carbon is the fourth most abundant chemical element in the observable universe
The observable universe is a ball-shaped region of the universe comprising all matter that can be observed from Earth or its space-based telescopes and exploratory probes at the present time, because the electromagnetic radiation from these obj ...
by mass after hydrogen, helium, and oxygen. Carbon is abundant in the Sun
The Sun is the star at the center of the Solar System. It is a nearly perfect ball of hot plasma, heated to incandescence by nuclear fusion reactions in its core. The Sun radiates this energy mainly as light, ultraviolet, and infrared radi ...
, star
A star is an astronomical object comprising a luminous spheroid of plasma (physics), plasma held together by its gravity. The List of nearest stars and brown dwarfs, nearest star to Earth is the Sun. Many other stars are visible to the naked ...
s, comet
A comet is an icy, small Solar System body that, when passing close to the Sun, warms and begins to release gases, a process that is called outgassing. This produces a visible atmosphere or coma, and sometimes also a tail. These phenomena ar ...
s, and in the atmospheres
The standard atmosphere (symbol: atm) is a unit of pressure defined as Pa. It is sometimes used as a ''reference pressure'' or ''standard pressure''. It is approximately equal to Earth's average atmospheric pressure at sea level.
History
The s ...
of most planet
A planet is a large, rounded astronomical body that is neither a star nor its remnant. The best available theory of planet formation is the nebular hypothesis, which posits that an interstellar cloud collapses out of a nebula to create a you ...
s. Some meteorite
A meteorite is a solid piece of debris from an object, such as a comet, asteroid, or meteoroid, that originates in outer space and survives its passage through the atmosphere to reach the surface of a planet or Natural satellite, moon. When the ...
s contain microscopic diamonds that were formed when the Solar System
The Solar SystemCapitalization of the name varies. The International Astronomical Union, the authoritative body regarding astronomical nomenclature, specifies capitalizing the names of all individual astronomical objects but uses mixed "Solar S ...
was still a protoplanetary disk
A protoplanetary disk is a rotating circumstellar disc of dense gas and dust surrounding a young newly formed star, a T Tauri star, or Herbig Ae/Be star. The protoplanetary disk may also be considered an accretion disk for the star itself, be ...
.
Microscopic diamonds may also be formed by the intense pressure and high temperature at the sites of meteorite impacts.
In 2014 NASA
The National Aeronautics and Space Administration (NASA ) is an independent agency of the US federal government responsible for the civil space program, aeronautics research, and space research.
NASA was established in 1958, succeeding t ...
announced greatly upgraded database
for tracking
polycyclic aromatic hydrocarbons
A polycyclic aromatic hydrocarbon (PAH) is a class of organic compounds that is composed of multiple aromatic rings. The simplest representative is naphthalene, having two aromatic rings and the three-ring compounds anthracene and phenanthrene. P ...
(PAHs) in the universe
The universe is all of space and time and their contents, including planets, stars, galaxies, and all other forms of matter and energy. The Big Bang theory is the prevailing cosmological description of the development of the universe. Acc ...
. More than 20% of the carbon in the universe may be associated with PAHs, complex compounds of carbon and hydrogen without oxygen. These compounds figure in the PAH world hypothesis
The PAH world hypothesis is a speculative hypothesis that proposes that polycyclic aromatic hydrocarbons (PAHs), known to be abundant in the universe, including in comets, and assumed to be abundant in the primordial soup of the early Earth, played ...
where they are hypothesized to have a role in abiogenesis
In biology, abiogenesis (from a- 'not' + Greek bios 'life' + genesis 'origin') or the origin of life is the natural process by which life has arisen from non-living matter, such as simple organic compounds. The prevailing scientific hypothes ...
and formation of life
Life is a quality that distinguishes matter that has biological processes, such as signaling and self-sustaining processes, from that which does not, and is defined by the capacity for growth, reaction to stimuli, metabolism, energ ...
. PAHs seem to have been formed "a couple of billion years" after the Big Bang
The Big Bang event is a physical theory that describes how the universe expanded from an initial state of high density and temperature. Various cosmological models of the Big Bang explain the evolution of the observable universe from the ...
, are widespread throughout the universe, and are associated with new stars and exoplanets
An exoplanet or extrasolar planet is a planet outside the Solar System. The first possible evidence of an exoplanet was noted in 1917 but was not recognized as such. The first confirmation of detection occurred in 1992. A different planet, init ...
.
It has been estimated that the solid earth as a whole contains 730 ppm of carbon, with 2000 ppm in the core and 120 ppm in the combined mantle and crust. Since the mass of the earth is , this would imply 4360 million gigatonne
The tonne ( or ; symbol: t) is a unit of mass equal to 1000 kilograms. It is a non-SI unit accepted for use with SI. It is also referred to as a metric ton to distinguish it from the non-metric units of the short ton (United States c ...
s of carbon. This is much more than the amount of carbon in the oceans or atmosphere (below).
In combination with oxygen
Oxygen is the chemical element with the symbol O and atomic number 8. It is a member of the chalcogen group in the periodic table, a highly reactive nonmetal, and an oxidizing agent that readily forms oxides with most elements as wel ...
in carbon dioxide
Carbon dioxide (chemical formula ) is a chemical compound made up of molecules that each have one carbon atom covalently double bonded to two oxygen atoms. It is found in the gas state at room temperature. In the air, carbon dioxide is transpar ...
, carbon is found in the Earth's atmosphere (approximately 900 gigatonnes of carbon — each ppm corresponds to 2.13 Gt) and dissolved in all water bodies (approximately 36,000 gigatonnes of carbon). Carbon in the biosphere
The biosphere (from Greek βίος ''bíos'' "life" and σφαῖρα ''sphaira'' "sphere"), also known as the ecosphere (from Greek οἶκος ''oîkos'' "environment" and σφαῖρα), is the worldwide sum of all ecosystems. It can also be ...
has been estimated at 550 gigatonnes but with a large uncertainty, due mostly to a huge uncertainty in the amount of terrestrial deep subsurface bacteria
The deep biosphere is the part of the biosphere that resides below the first few meters of the surface. It extends down at least 5 kilometers below the continental surface and 10.5 kilometers below the sea surface, at temperatures that ...
. Hydrocarbons
In organic chemistry, a hydrocarbon is an organic compound consisting entirely of hydrogen and carbon. Hydrocarbons are examples of group 14 hydrides. Hydrocarbons are generally colourless and hydrophobic, and their odors are usually weak or ex ...
(such as coal
Coal is a combustible black or brownish-black sedimentary rock, formed as rock strata called coal seams. Coal is mostly carbon with variable amounts of other elements, chiefly hydrogen, sulfur, oxygen, and nitrogen.
Coal is formed when dea ...
, petroleum
Petroleum, also known as crude oil, or simply oil, is a naturally occurring yellowish-black liquid mixture of mainly hydrocarbons, and is found in geological formations. The name ''petroleum'' covers both naturally occurring unprocessed crud ...
, and natural gas
Natural gas (also called fossil gas or simply gas) is a naturally occurring mixture of gaseous hydrocarbons consisting primarily of methane in addition to various smaller amounts of other higher alkanes. Low levels of trace gases like carbo ...
) contain carbon as well. Coal
Coal is a combustible black or brownish-black sedimentary rock, formed as rock strata called coal seams. Coal is mostly carbon with variable amounts of other elements, chiefly hydrogen, sulfur, oxygen, and nitrogen.
Coal is formed when dea ...
"reserves" (not "resources") amount to around 900 gigatonnes with perhaps 18,000 Gt of resources. Oil reserves
An oil is any polarity (chemistry), nonpolar chemical substance that is composed primarily of Hydrocarbon, hydrocarbons and is hydrophobe, hydrophobic (does not mix with water) & lipophilicity, lipophilic (mixes with other oils). Oils are usu ...
are around 150 gigatonnes. Proven sources of natural gas are about (containing about 105 gigatonnes of carbon), but studies estimate another of "unconventional" deposits such as shale gas
Shale gas is an unconventional natural gas that is found trapped within shale formations. Since the 1990s a combination of horizontal drilling and hydraulic fracturing has made large volumes of shale gas more economical to produce, and some a ...
, representing about 540 gigatonnes of carbon.
Carbon is also found in methane hydrates
Methane clathrate (CH4·5.75H2O) or (8CH4·46H2O), also called methane hydrate, hydromethane, methane ice, fire ice, natural gas hydrate, or gas hydrate, is a solid clathrate compound (more specifically, a clathrate hydrate) in which a large amou ...
in polar regions and under the seas. Various estimates put this carbon between 500, 2500 Gt, or 3,000 Gt.
In the past, quantities of hydrocarbons were greater. According to one source, in the period from 1751 to 2008 about 347 gigatonnes of carbon were released as carbon dioxide to the atmosphere from burning of fossil fuels. Another source puts the amount added to the atmosphere for the period since 1750 at 879 Gt, and the total going to the atmosphere, sea, and land (such as peat bogs
A bog or bogland is a wetland that accumulates peat as a deposit of dead plant materials often mosses, typically sphagnum moss. It is one of the four main types of wetlands. Other names for bogs include mire, mosses, quagmire, and muskeg; a ...
) at almost 2,000 Gt.
Carbon is a constituent (about 12% by mass) of the very large masses of carbonate rock (limestone
Limestone ( calcium carbonate ) is a type of carbonate sedimentary rock which is the main source of the material lime. It is composed mostly of the minerals calcite and aragonite, which are different crystal forms of . Limestone forms whe ...
, dolomite Dolomite may refer to:
*Dolomite (mineral), a carbonate mineral
*Dolomite (rock), also known as dolostone, a sedimentary carbonate rock
*Dolomite, Alabama, United States, an unincorporated community
*Dolomite, California, United States, an unincor ...
, marble and so on). Coal
Coal is a combustible black or brownish-black sedimentary rock, formed as rock strata called coal seams. Coal is mostly carbon with variable amounts of other elements, chiefly hydrogen, sulfur, oxygen, and nitrogen.
Coal is formed when dea ...
is very rich in carbon (anthracite contains 92–98%) and is the largest commercial source of mineral carbon, accounting for 4,000 gigatonnes or 80% of fossil fuel.
As for individual carbon allotropes, graphite is found in large quantities in the United States (mostly in New York (state), New York and Texas), Russia, Mexico, Greenland, and India. Natural diamonds occur in the rock kimberlite, found in ancient volcano, volcanic "necks", or "pipes". Most diamond deposits are in Africa, notably in South Africa, Namibia, Botswana, the Republic of the Congo, and Sierra Leone. Diamond deposits have also been found in Arkansas, Canada, the Russian Arctic, Brazil, and in Northern and Western Australia. Diamonds are now also being recovered from the ocean floor off the Cape of Good Hope. Diamonds are found naturally, but about 30% of all industrial diamonds used in the U.S. are now manufactured.
Carbon-14 is formed in upper layers of the troposphere and the stratosphere at altitudes of 9–15 km by a reaction that is precipitated by cosmic rays. Thermal neutrons are produced that collide with the nuclei of nitrogen-14, forming carbon-14 and a proton. As such, of atmospheric carbon dioxide contains carbon-14.
Carbon-rich asteroids are relatively preponderant in the outer parts of the asteroid belt in the Solar System
The Solar SystemCapitalization of the name varies. The International Astronomical Union, the authoritative body regarding astronomical nomenclature, specifies capitalizing the names of all individual astronomical objects but uses mixed "Solar S ...
. These asteroids have not yet been directly sampled by scientists. The asteroids can be used in hypothetical Asteroid mining, space-based carbon mining, which may be possible in the future, but is currently technologically impossible.
Isotopes
Isotopes of carbon are atomic nucleus, atomic nuclei that contain six protons plus a number of neutrons (varying from 2 to 16). Carbon has two stable, naturally occurring isotopes. The isotope carbon-12 (C) forms 98.93% of the carbon on Earth, while carbon-13 (C) forms the remaining 1.07%. The concentration of C is further increased in biological materials because biochemical reactions discriminate against C. In 1961, the International Union of Pure and Applied Chemistry (IUPAC) adopted the isotope carbon-12 as the basis for atomic weights. Identification of carbon in nuclear magnetic resonance (NMR) experiments is done with the isotope C. Carbon-14 (C) is a naturally occurring radioisotope, created in the upper atmosphere (lower stratosphere and upper troposphere) by interaction of nitrogen with cosmic rays. It is found in trace amounts on Earth of 1 part per 10^12, trillion (0.0000000001%) or more, mostly confined to the atmosphere and superficial deposits, particularly ofpeat
Peat (), also known as turf (), is an accumulation of partially decayed vegetation or organic matter. It is unique to natural areas called peatlands, bogs, mires, moors, or muskegs. The peatland ecosystem covers and is the most efficien ...
and other organic materials. This isotope decays by 0.158 MeV beta decay, β emission. Because of its relatively short half-life
Half-life (symbol ) is the time required for a quantity (of substance) to reduce to half of its initial value. The term is commonly used in nuclear physics to describe how quickly unstable atoms undergo radioactive decay or how long stable ato ...
of 5730 years, C is virtually absent in ancient rocks. The amount of C in the atmosphere and in living organisms is almost constant, but decreases predictably in their bodies after death. This principle is used in radiocarbon dating, invented in 1949, which has been used extensively to determine the age of carbonaceous materials with ages up to about 40,000 years.
There are 15 known isotopes of carbon and the shortest-lived of these is C which decays through proton emission and alpha decay and has a half-life of 1.98739 × 10 s. The exotic C exhibits a nuclear halo, which means its radius is appreciably larger than would be expected if the Atomic nucleus, nucleus were a sphere of constant density.
Formation in stars
Formation of the carbon atomic nucleus occurs within a giant star, giant or supergiant star through the triple-alpha process. This requires a nearly simultaneous collision of three alpha particles (helium
Helium (from el, ἥλιος, helios, lit=sun) is a chemical element with the symbol He and atomic number 2. It is a colorless, odorless, tasteless, non-toxic, inert, monatomic gas and the first in the noble gas group in the periodic table. ...
nuclei), as the products of further nuclear fusion reactions of helium with hydrogen or another helium nucleus produce isotopes of lithium, lithium-5 and isotopes of beryllium, beryllium-8 respectively, both of which are highly unstable and decay almost instantly back into smaller nuclei. The triple-alpha process happens in conditions of temperatures over 100 megakelvins and helium concentration that the rapid expansion and cooling of the early universe prohibited, and therefore no significant carbon was created during the Big Bang
The Big Bang event is a physical theory that describes how the universe expanded from an initial state of high density and temperature. Various cosmological models of the Big Bang explain the evolution of the observable universe from the ...
.
According to current physical cosmology theory, carbon is formed in the interiors of stars on the horizontal branch.
When massive stars die as supernova, the carbon is scattered into space as dust. This dust becomes component material for the formation of the Metallicity, next-generation star systems with accreted planets. The Solar System
The Solar SystemCapitalization of the name varies. The International Astronomical Union, the authoritative body regarding astronomical nomenclature, specifies capitalizing the names of all individual astronomical objects but uses mixed "Solar S ...
is one such star system with an abundance of carbon, enabling the existence of life as we know it. It is the opinion of most scholars that all the carbon in the Solar System and the Milky Way comes from dying stars.
The CNO cycle is an additional hydrogen fusion mechanism that powers stars, wherein carbon operates as a catalyst.
Rotational transitions of various isotopic forms of carbon monoxide (for example, CO, CO, and CO) are detectable in the submillimetre astronomy, submillimeter wavelength range, and are used in the study of Star formation, newly forming stars in molecular clouds.
Carbon cycle
carbon dioxide
Carbon dioxide (chemical formula ) is a chemical compound made up of molecules that each have one carbon atom covalently double bonded to two oxygen atoms. It is found in the gas state at room temperature. In the air, carbon dioxide is transpar ...
from the atmosphere (or seawater) and build it into biomass, as in the Calvin cycle, a process of carbon fixation. Some of this biomass is eaten by animals, while some carbon is exhaled by animals as carbon dioxide. The carbon cycle is considerably more complicated than this short loop; for example, some carbon dioxide is dissolved in the oceans; if bacteria do not consume it, dead plant or animal matter may become petroleum
Petroleum, also known as crude oil, or simply oil, is a naturally occurring yellowish-black liquid mixture of mainly hydrocarbons, and is found in geological formations. The name ''petroleum'' covers both naturally occurring unprocessed crud ...
or coal
Coal is a combustible black or brownish-black sedimentary rock, formed as rock strata called coal seams. Coal is mostly carbon with variable amounts of other elements, chiefly hydrogen, sulfur, oxygen, and nitrogen.
Coal is formed when dea ...
, which releases carbon when burned.
Compounds
Organic compounds
 Carbon can form very long chains of interconnecting carbon–carbon bonds, a property that is called catenation. Carbon-carbon bonds are strong and stable. Through catenation, carbon forms a countless number of compounds. A tally of unique compounds shows that more contain carbon than do not.
A similar claim can be made for hydrogen because most organic compounds contain hydrogen chemically bonded to carbon or another common element like oxygen or nitrogen.
The simplest form of an organic molecule is the hydrocarbon—a large family of organic molecules that are composed of
Carbon can form very long chains of interconnecting carbon–carbon bonds, a property that is called catenation. Carbon-carbon bonds are strong and stable. Through catenation, carbon forms a countless number of compounds. A tally of unique compounds shows that more contain carbon than do not.
A similar claim can be made for hydrogen because most organic compounds contain hydrogen chemically bonded to carbon or another common element like oxygen or nitrogen.
The simplest form of an organic molecule is the hydrocarbon—a large family of organic molecules that are composed of hydrogen
Hydrogen is the chemical element with the symbol H and atomic number 1. Hydrogen is the lightest element. At standard conditions hydrogen is a gas of diatomic molecules having the formula . It is colorless, odorless, tasteless, non-toxic, an ...
atoms bonded to a chain of carbon atoms. A hydrocarbon backbone can be substituted by other atoms, known as heteroatoms. Common heteroatoms that appear in organic compounds include oxygen, nitrogen, sulfur, phosphorus, and the nonradioactive halogens, as well as the metals lithium and magnesium. Organic compounds containing bonds to metal are known as organometallic compounds (''see below''). Certain groupings of atoms, often including heteroatoms, recur in large numbers of organic compounds. These collections, known as Functional group, ''functional groups'', confer common reactivity patterns and allow for the systematic study and categorization of organic compounds. Chain length, shape and functional groups all affect the properties of organic molecules.
In most stable compounds of carbon (and nearly all stable ''organic'' compounds), carbon obeys the octet rule and is ''tetravalent'', meaning that a carbon atom forms a total of four covalent bonds (which may include double and triple bonds). Exceptions include a small number of stabilized ''carbocations'' (three bonds, positive charge), ''radicals'' (three bonds, neutral), ''carbanions'' (three bonds, negative charge) and ''carbenes'' (two bonds, neutral), although these species are much more likely to be encountered as unstable, reactive intermediates.
Carbon occurs in all known organic material, organic life and is the basis of organic chemistry. When united with hydrogen
Hydrogen is the chemical element with the symbol H and atomic number 1. Hydrogen is the lightest element. At standard conditions hydrogen is a gas of diatomic molecules having the formula . It is colorless, odorless, tasteless, non-toxic, an ...
, it forms various hydrocarbons that are important to industry as refrigerants, lubricants, solvents, as chemical feedstock for the manufacture of plastics and petrochemicals, and as fossil fuels.
When combined with oxygen and hydrogen, carbon can form many groups of important biological compounds including sugars, lignans, chitins, Alcohol (chemistry), alcohols, fats, and aromatic esters, carotenoids and terpenes. With nitrogen it forms alkaloids, and with the addition of sulfur also it forms antibiotics, amino acids, and rubber products. With the addition of phosphorus to these other elements, it forms DNA and RNA, the chemical-code carriers of life, and adenosine triphosphate (ATP), the most important energy-transfer molecule in all living cells.
Inorganic compounds
Commonly carbon-containing compounds which are associated with minerals or which do not contain bonds to the other carbon atoms, halogens, or hydrogen, are treated separately from classical organic compounds; the definition is not rigid, and the classification of some compounds can vary from author to author (see reference articles above). Among these are the simple oxides of carbon. The most prominent oxide iscarbon dioxide
Carbon dioxide (chemical formula ) is a chemical compound made up of molecules that each have one carbon atom covalently double bonded to two oxygen atoms. It is found in the gas state at room temperature. In the air, carbon dioxide is transpar ...
(). This was once the principal constituent of the paleoatmosphere, but is a minor component of the Atmosphere of Earth, Earth's atmosphere today. Dissolved in water (molecule), water, it forms carbonic acid (), but as most compounds with multiple single-bonded oxygens on a single carbon it is unstable. Through this intermediate, though, resonance-stabilized carbonate ions are produced. Some important minerals are carbonates, notably calcite. Carbon disulfide () is similar. Nevertheless, due to its physical properties and its association with organic synthesis, carbon disulfide is sometimes classified as an ''organic'' solvent.
The other common oxide is carbon monoxide
Carbon monoxide (chemical formula CO) is a colorless, poisonous, odorless, tasteless, flammable gas that is slightly less dense than air. Carbon monoxide consists of one carbon atom and one oxygen atom connected by a triple bond. It is the simple ...
(CO). It is formed by incomplete combustion, and is a colorless, odorless gas. The molecules each contain a triple bond and are fairly polar molecule, polar, resulting in a tendency to bind permanently to hemoglobin molecules, displacing oxygen, which has a lower binding affinity. Cyanide (CN), has a similar structure, but behaves much like a halide ion (pseudohalogen). For example, it can form the nitride cyanogen molecule ((CN)), similar to diatomic halides. Likewise, the heavier analog of cyanide, cyaphide (CP), is also considered inorganic, though most simple derivatives are highly unstable. Other uncommon oxides are carbon suboxide (), the unstable dicarbon monoxide (CO), carbon trioxide (CO), cyclopentanepentone (CO),
cyclohexanehexone (CO), and mellitic anhydride (CO). However, mellitic anhydride is the triple acyl anhydride of mellitic acid; moreover, it contains a benzene ring. Thus, many chemists consider it to be organic.
With reactive metal
A metal (from Greek μέταλλον ''métallon'', "mine, quarry, metal") is a material that, when freshly prepared, polished, or fractured, shows a lustrous appearance, and conducts electricity and heat relatively well. Metals are typicall ...
s, such as tungsten
Tungsten, or wolfram, is a chemical element with the symbol W and atomic number 74. Tungsten is a rare metal found naturally on Earth almost exclusively as compounds with other elements. It was identified as a new element in 1781 and first isolat ...
, carbon forms either carbides (C) or acetylides () to form alloys with high melting points. These anions are also associated with methane and acetylene, both very weak acids. With an electronegativity of 2.5, carbon prefers to form covalent bonds. A few carbides are covalent lattices, like Silicon carbide, carborundum (SiC), which resembles diamond
Diamond is a Allotropes of carbon, solid form of the element carbon with its atoms arranged in a crystal structure called diamond cubic. Another solid form of carbon known as graphite is the Chemical stability, chemically stable form of car ...
. Nevertheless, even the most polar and salt-like of carbides are not completely ionic compounds.Greenwood and Earnshaw, pp. 297–301
Organometallic compounds
Organometallic compounds by definition contain at least one carbon-metal covalent bond. A wide range of such compounds exist; major classes include simple alkyl-metal compounds (for example, tetraethyllead), η-alkene compounds (for example, Zeise's salt), and η-allyl compounds (for example, allylpalladium chloride dimer); metallocenes containing cyclopentadienyl ligands (for example, ferrocene); and transition metal carbene complexes. Many metal carbonyls and Cyanometalate, metal cyanides exist (for example, tetracarbonylnickel and potassium ferricyanide); some workers consider metal carbonyl and cyanide complexes without other carbon ligands to be purely inorganic, and not organometallic. However, most organometallic chemists consider metal complexes with any carbon ligand, even 'inorganic carbon' (e.g., carbonyls, cyanides, and certain types of carbides and acetylides) to be organometallic in nature. Metal complexes containing organic ligands without a carbon-metal covalent bond (e.g., metal carboxylates) are termed ''metalorganic'' compounds. While carbon is understood to strongly prefer formation of four covalent bonds, other exotic bonding schemes are also known. Carboranes are highly stable dodecahedral derivatives of the [B12H12]2- unit, with one BH replaced with a CH+. Thus, the carbon is bonded to five boron atoms and one hydrogen atom. The cation [(PhPAu)C] contains an octahedral carbon bound to six phosphine-gold fragments. This phenomenon has been attributed to the aurophilicity of the gold ligands, which provide additional stabilization of an otherwise labile species. In nature, the iron-molybdenum cofactor (FeMoco) responsible for microbial nitrogen fixation likewise has an octahedral carbon center (formally a carbide, C(-IV)) bonded to six iron atoms. In 2016, it was confirmed that, in line with earlier theoretical predictions, the hexamethylbenzene, hexamethylbenzene dication contains a carbon atom with six bonds. More specifically, the dication could be described structurally by the formulation [MeC(η5-C5Me5)]2+, making it an "organic metallocene" in which a MeC3+ fragment is bonded to a η5-C5Me5− fragment through all five of the carbons of the ring. It is important to note that in the cases above, each of the bonds to carbon contain less than two formal electron pairs. Thus, the formal electron count of these species does not exceed an octet. This makes them hypercoordinate but not hypervalent. Even in cases of alleged 10-C-5 species (that is, a carbon with five ligands and a formal electron count of ten), as reported by Akiba and co-workers, electronic structure calculations conclude that the electron population around carbon is still less than eight, as is true for other compounds featuring four-electron three-center bonding.
It is important to note that in the cases above, each of the bonds to carbon contain less than two formal electron pairs. Thus, the formal electron count of these species does not exceed an octet. This makes them hypercoordinate but not hypervalent. Even in cases of alleged 10-C-5 species (that is, a carbon with five ligands and a formal electron count of ten), as reported by Akiba and co-workers, electronic structure calculations conclude that the electron population around carbon is still less than eight, as is true for other compounds featuring four-electron three-center bonding.
History and etymology
 The English language, English name ''carbon'' comes from the Latin ''carbo'' for coal and charcoal, whence also comes the French language, French ''charbon'', meaning charcoal. In German language, German, Dutch language, Dutch and Danish language, Danish, the names for carbon are ''Kohlenstoff'', ''koolstof'' and ''kulstof'' respectively, all literally meaning
The English language, English name ''carbon'' comes from the Latin ''carbo'' for coal and charcoal, whence also comes the French language, French ''charbon'', meaning charcoal. In German language, German, Dutch language, Dutch and Danish language, Danish, the names for carbon are ''Kohlenstoff'', ''koolstof'' and ''kulstof'' respectively, all literally meaning coal
Coal is a combustible black or brownish-black sedimentary rock, formed as rock strata called coal seams. Coal is mostly carbon with variable amounts of other elements, chiefly hydrogen, sulfur, oxygen, and nitrogen.
Coal is formed when dea ...
-substance.
Carbon was discovered in prehistory and was known in the forms of soot
Soot ( ) is a mass of impure carbon particles resulting from the incomplete combustion of hydrocarbons. It is more properly restricted to the product of the gas-phase combustion process but is commonly extended to include the residual pyrolysed ...
and charcoal
Charcoal is a lightweight black carbon residue produced by strongly heating wood (or other animal and plant materials) in minimal oxygen to remove all water and volatile constituents. In the traditional version of this pyrolysis process, cal ...
to the earliest human civilizations. Diamonds were known probably as early as 2500 BCE in China, while carbon in the form of charcoal
Charcoal is a lightweight black carbon residue produced by strongly heating wood (or other animal and plant materials) in minimal oxygen to remove all water and volatile constituents. In the traditional version of this pyrolysis process, cal ...
was made around Roman times by the same chemistry as it is today, by heating wood in a pyramid covered with clay to exclude air.
 In 1722, René Antoine Ferchault de Réaumur demonstrated that iron was transformed into steel through the absorption of some substance, now known to be carbon. In 1772, Antoine Lavoisier showed that diamonds are a form of carbon; when he burned samples of charcoal and diamond and found that neither produced any water and that both released the same amount of
In 1722, René Antoine Ferchault de Réaumur demonstrated that iron was transformed into steel through the absorption of some substance, now known to be carbon. In 1772, Antoine Lavoisier showed that diamonds are a form of carbon; when he burned samples of charcoal and diamond and found that neither produced any water and that both released the same amount of carbon dioxide
Carbon dioxide (chemical formula ) is a chemical compound made up of molecules that each have one carbon atom covalently double bonded to two oxygen atoms. It is found in the gas state at room temperature. In the air, carbon dioxide is transpar ...
per gram.
In 1779, Carl Wilhelm Scheele showed that graphite, which had been thought of as a form of lead, was instead identical with charcoal but with a small admixture of iron, and that it gave "aerial acid" (his name for carbon dioxide) when oxidized with nitric acid.
In 1786, the French scientists Claude Louis Berthollet, Gaspard Monge and C. A. Vandermonde confirmed that graphite was mostly carbon by oxidizing it in oxygen in much the same way Lavoisier had done with diamond. Some iron again was left, which the French scientists thought was necessary to the graphite structure. In their publication they proposed the name ''carbone'' (Latin ''carbonum'') for the element in graphite which was given off as a gas upon burning graphite. Antoine Lavoisier then listed carbon as an chemical element, element in his 1789 textbook.
A new allotrope of carbon, fullerene
A fullerene is an allotrope of carbon whose molecule consists of carbon atoms connected by single and double bonds so as to form a closed or partially closed mesh, with fused rings of five to seven atoms. The molecule may be a hollow sphere, ...
, that was discovered in 1985 includes nanostructured forms such as buckyball
Buckminsterfullerene is a type of fullerene with the formula C60. It has a cage-like fused-ring structure (truncated icosahedron) made of twenty hexagons and twelve pentagons, and resembles a soccer ball. Each of its 60 carbon atoms is bonded t ...
s and carbon nanotube, nanotubes.
Their discoverers – Robert Curl, Harold Kroto and Richard Smalley – received the Nobel Prize in Chemistry in 1996. The resulting renewed interest in new forms led to the discovery of further exotic allotropes, including glassy carbon
Glass-like carbon, often called glassy carbon or vitreous carbon, is a non-graphitizing, or nongraphitizable, carbon which combines glassy and ceramic properties with those of graphite. The most important properties are high temperature resis ...
, and the realization that "amorphous carbon Amorphous carbon is free, reactive carbon that has no crystalline structure. Amorphous carbon materials may be stabilized by terminating dangling-π bonds with hydrogen. As with other amorphous solids, some short-range order can be observed. Amorp ...
" is not strictly amorphous
In condensed matter physics and materials science, an amorphous solid (or non-crystalline solid, glassy solid) is a solid that lacks the long-range order that is characteristic of a crystal.
Etymology
The term comes from the Greek ''a'' ("wi ...
.
Production
Graphite
Commercially viable natural deposits of graphite occur in many parts of the world, but the most important sources economically are in China, India, Brazil and North Korea. Graphite deposits are of Metamorphic rock, metamorphic origin, found in association with quartz, mica and feldspars in schists, gneisses and metamorphosed sandstones andlimestone
Limestone ( calcium carbonate ) is a type of carbonate sedimentary rock which is the main source of the material lime. It is composed mostly of the minerals calcite and aragonite, which are different crystal forms of . Limestone forms whe ...
as lens (geology), lenses or vein (geology), veins, sometimes of a metre or more in thickness. Deposits of graphite in Borrowdale, Cumberland, England were at first of sufficient size and purity that, until the 19th century, pencils were made by sawing blocks of natural graphite into strips before encasing the strips in wood. Today, smaller deposits of graphite are obtained by crushing the parent rock and floating the lighter graphite out on water.USGS Minerals Yearbook: Graphite, 2009and Graphite: Mineral Commodity Summaries 2011 There are three types of natural graphite—amorphous, flake or crystalline flake, and vein or lump. Amorphous graphite is the lowest quality and most abundant. Contrary to science, in industry "amorphous" refers to very small crystal size rather than complete lack of crystal structure. Amorphous is used for lower value graphite products and is the lowest priced graphite. Large amorphous graphite deposits are found in China, Europe, Mexico and the United States. Flake graphite is less common and of higher quality than amorphous; it occurs as separate plates that crystallized in metamorphic rock. Flake graphite can be four times the price of amorphous. Good quality flakes can be processed into expandable graphite for many uses, such as flame retardants. The foremost deposits are found in Austria, Brazil, Canada, China, Germany and Madagascar. Vein or lump graphite is the rarest, most valuable, and highest quality type of natural graphite. It occurs in veins along intrusive contacts in solid lumps, and it is only commercially mined in Sri Lanka. According to the USGS, world production of natural graphite was 1.1 million tonnes in 2010, to which China contributed 800,000 t, India 130,000 t, Brazil 76,000 t, North Korea 30,000 t and Canada 25,000 t. No natural graphite was reported mined in the United States, but 118,000 t of synthetic graphite with an estimated value of $998 million was produced in 2009.
Diamond
 The diamond supply chain is controlled by a limited number of powerful businesses, and is also highly concentrated in a small number of locations around the world (see figure).
Only a very small fraction of the diamond ore consists of actual diamonds. The ore is crushed, during which care has to be taken in order to prevent larger diamonds from being destroyed in this process and subsequently the particles are sorted by density. Today, diamonds are located in the diamond-rich density fraction with the help of X-ray fluorescence, after which the final sorting steps are done by hand. Before the use of X-rays became commonplace, the separation was done with grease belts; diamonds have a stronger tendency to stick to grease than the other minerals in the ore.
Historically diamonds were known to be found only in alluvial deposits in southern India. discussion on alluvial diamonds in India and elsewhere as well as earliest finds India led the world in diamond production from the time of their discovery in approximately the 9th century BC Ball was a Geologist in British service. Chapter I, Page 1 to the mid-18th century AD, but the commercial potential of these sources had been exhausted by the late 18th century and at that time India was eclipsed by Brazil where the first non-Indian diamonds were found in 1725.
Diamond production of primary deposits (kimberlites and lamproites) only started in the 1870s after the discovery of the diamond fields in South Africa. Production has increased over time and an accumulated total of over 4.5 billion carats have been mined since that date. Most commercially viable diamond deposits were in Russia, Botswana, Australia and the Democratic Republic of Congo. By 2005, Russia produced almost one-fifth of the global diamond output (mostly in Sakha Republic, Yakutia territory; for example, Mir Mine, Mir pipe and Udachnaya pipe) but the Argyle diamond mine, Argyle mine in Australia became the single largest source, producing 14 million carats in 2018. New finds, the Canada, Canadian mines at Diavik Diamond Mine, Diavik and Ekati Diamond Mine, Ekati, are expected to become even more valuable owing to their production of gem quality stones.
In the United States, diamonds have been found in Arkansas, Colorado and Montana. In 2004, a startling discovery of a microscopic diamond in the United States led to the January 2008 bulk-sampling of kimberlite pipes in a remote part of Montana.
The diamond supply chain is controlled by a limited number of powerful businesses, and is also highly concentrated in a small number of locations around the world (see figure).
Only a very small fraction of the diamond ore consists of actual diamonds. The ore is crushed, during which care has to be taken in order to prevent larger diamonds from being destroyed in this process and subsequently the particles are sorted by density. Today, diamonds are located in the diamond-rich density fraction with the help of X-ray fluorescence, after which the final sorting steps are done by hand. Before the use of X-rays became commonplace, the separation was done with grease belts; diamonds have a stronger tendency to stick to grease than the other minerals in the ore.
Historically diamonds were known to be found only in alluvial deposits in southern India. discussion on alluvial diamonds in India and elsewhere as well as earliest finds India led the world in diamond production from the time of their discovery in approximately the 9th century BC Ball was a Geologist in British service. Chapter I, Page 1 to the mid-18th century AD, but the commercial potential of these sources had been exhausted by the late 18th century and at that time India was eclipsed by Brazil where the first non-Indian diamonds were found in 1725.
Diamond production of primary deposits (kimberlites and lamproites) only started in the 1870s after the discovery of the diamond fields in South Africa. Production has increased over time and an accumulated total of over 4.5 billion carats have been mined since that date. Most commercially viable diamond deposits were in Russia, Botswana, Australia and the Democratic Republic of Congo. By 2005, Russia produced almost one-fifth of the global diamond output (mostly in Sakha Republic, Yakutia territory; for example, Mir Mine, Mir pipe and Udachnaya pipe) but the Argyle diamond mine, Argyle mine in Australia became the single largest source, producing 14 million carats in 2018. New finds, the Canada, Canadian mines at Diavik Diamond Mine, Diavik and Ekati Diamond Mine, Ekati, are expected to become even more valuable owing to their production of gem quality stones.
In the United States, diamonds have been found in Arkansas, Colorado and Montana. In 2004, a startling discovery of a microscopic diamond in the United States led to the January 2008 bulk-sampling of kimberlite pipes in a remote part of Montana.
Applications




 Carbon is essential to all known living systems, and without it life as we know it could not exist (see alternative biochemistry). The major economic use of carbon other than food and wood is in the form of hydrocarbons, most notably the fossil fuel methane gas and crude oil (petroleum). Petroleum, Crude oil is distillation, distilled in Oil refinery, refineries by the petrochemical industry to produce gasoline, kerosene, and other products. Cellulose is a natural, carbon-containing polymer produced by plants in the form of wood, cotton, linen, and hemp. Cellulose is used primarily for maintaining structure in plants. Commercially valuable carbon polymers of animal origin include wool, Cashmere wool, cashmere and silk. Plastics are made from synthetic carbon polymers, often with oxygen and nitrogen atoms included at regular intervals in the main polymer chain. The raw materials for many of these synthetic substances come from crude oil.
The uses of carbon and its compounds are extremely varied. It can form alloys with
Carbon is essential to all known living systems, and without it life as we know it could not exist (see alternative biochemistry). The major economic use of carbon other than food and wood is in the form of hydrocarbons, most notably the fossil fuel methane gas and crude oil (petroleum). Petroleum, Crude oil is distillation, distilled in Oil refinery, refineries by the petrochemical industry to produce gasoline, kerosene, and other products. Cellulose is a natural, carbon-containing polymer produced by plants in the form of wood, cotton, linen, and hemp. Cellulose is used primarily for maintaining structure in plants. Commercially valuable carbon polymers of animal origin include wool, Cashmere wool, cashmere and silk. Plastics are made from synthetic carbon polymers, often with oxygen and nitrogen atoms included at regular intervals in the main polymer chain. The raw materials for many of these synthetic substances come from crude oil.
The uses of carbon and its compounds are extremely varied. It can form alloys with iron
Iron () is a chemical element with symbol Fe (from la, ferrum) and atomic number 26. It is a metal that belongs to the first transition series and group 8 of the periodic table. It is, by mass, the most common element on Earth, right in f ...
, of which the most common is carbon steel. Graphite is combined with clays to form the 'lead' used in pencils used for writing and drawing. It is also used as a lubricant and a pigment, as a molding material in glass manufacture, in electrodes for dry Battery (electricity), batteries and in electroplating and electroforming, in brush (electric), brushes for electric motors and as a neutron moderator in nuclear reactors.
Charcoal is used as a drawing material in artwork, barbecue grilling, iron smelting, and in many other applications. Wood, coal and oil are used as fuel for production of energy and heating. Gem quality diamond
Diamond is a Allotropes of carbon, solid form of the element carbon with its atoms arranged in a crystal structure called diamond cubic. Another solid form of carbon known as graphite is the Chemical stability, chemically stable form of car ...
is used in jewelry, and industrial diamonds are used in drilling, cutting and polishing tools for machining metals and stone. Plastics are made from fossil hydrocarbons, and carbon fiber, made by pyrolysis of synthetic polyester fibers is used to reinforce plastics to form advanced, lightweight composite materials.
Carbon fiber is made by pyrolysis of extruded and stretched filaments of polyacrylonitrile (PAN) and other organic substances. The crystallographic structure and mechanical properties of the fiber depend on the type of starting material, and on the subsequent processing. Carbon fibers made from PAN have structure resembling narrow filaments of graphite, but thermal processing may re-order the structure into a continuous rolled sheet. The result is fibers with higher specific strength, specific tensile strength than steel.
Carbon black is used as the black pigment in printing ink, artist's oil paint and water colours, carbon paper, automotive finishes, India ink and laser printer toner. Carbon black is also used as a Filler (materials), filler in rubber products such as tyres and in plastic compounds. Activated charcoal is used as an absorption (chemistry), absorbent and adsorbent in Filter (chemistry), filter material in applications as diverse as gas masks, water purification, and kitchen extractor hoods, and in medicine to Absorption (chemistry), absorb toxins, poisons, or gases from the Human digestive system, digestive system. Carbon is used in Redox, chemical reduction at high temperatures. Coke (fuel), Coke is used to reduce iron ore into iron (smelting). Case hardening of steel is achieved by heating finished steel components in carbon powder. Carbides of silicon carbide, silicon, tungsten carbide, tungsten, boron carbide, boron and titanium carbide, titanium, are among the hardest known materials, and are used as abrasives in cutting and grinding tools. Carbon compounds make up most of the materials used in clothing, such as natural and synthetic textiles and leather, and almost all of the interior surfaces in the built environment other than glass, stone and metal.
Diamonds
Thediamond
Diamond is a Allotropes of carbon, solid form of the element carbon with its atoms arranged in a crystal structure called diamond cubic. Another solid form of carbon known as graphite is the Chemical stability, chemically stable form of car ...
industry falls into two categories: one dealing with gem-grade diamonds and the other, with industrial-grade diamonds. While a large trade in both types of diamonds exists, the two markets function dramatically differently.
Unlike precious metals such as gold or platinum, gem diamonds do not trade as a commodity: there is a substantial mark-up in the sale of diamonds, and there is not a very active market for resale of diamonds.
Industrial diamonds are valued mostly for their hardness and heat conductivity, with the gemological qualities of clarity and color being mostly irrelevant. About 80% of mined diamonds (equal to about 100 million carats or 20 tonnes annually) are unsuitable for use as gemstones are relegated for industrial use (known as ''bort)''. synthetic diamonds, invented in the 1950s, found almost immediate industrial applications; 3 billion carats (600 tonnes) of synthetic diamond is produced annually.
The dominant industrial use of diamond is in cutting, drilling, grinding, and polishing. Most of these applications do not require large diamonds; in fact, most diamonds of gem-quality except for their small size can be used industrially. Diamonds are embedded in drill tips or saw blades, or ground into a powder for use in grinding and polishing applications. Specialized applications include use in laboratories as containment for Pressure experiment, high pressure experiments (see diamond anvil cell), high-performance bearing (mechanical), bearings, and limited use in specialized windows. With the continuing advances in the production of synthetic diamonds, new applications are becoming feasible. Garnering much excitement is the possible use of diamond as a semiconductor suitable for integrated circuit, microchips, and because of its exceptional heat conductance property, as a heat sink in electronics.
Precautions
 Pure carbon has extremely low toxicity to humans and can be handled safely in the form of graphite or charcoal. It is resistant to dissolution or chemical attack, even in the acidic contents of the digestive tract. Consequently, once it enters into the body's tissues it is likely to remain there indefinitely. Carbon black was probably one of the first pigments to be used for tattooing, and Ötzi the Iceman was found to have carbon tattoos that survived during his life and for 5200 years after his death. Inhalation of coal dust or soot (carbon black) in large quantities can be dangerous, irritating lung tissues and causing the congestive human lung, lung disease, coalworker's pneumoconiosis. Diamond dust used as an abrasive can be harmful if ingested or inhaled. Microparticles of carbon are produced in diesel engine exhaust fumes, and may accumulate in the lungs. In these examples, the harm may result from contaminants (e.g., organic chemicals, heavy metals) rather than from the carbon itself.
Carbon generally has low toxicity to life, life on Earth; but carbon nanoparticles are deadly to ''Drosophila''.
Carbon may burn vigorously and brightly in the presence of air at high temperatures. Large accumulations of coal, which have remained inert for hundreds of millions of years in the absence of oxygen, may spontaneous combustion, spontaneously combust when exposed to air in coal mine waste tips, ship cargo holds and coal bunkers, and storage dumps.
In nuclear reactor, nuclear applications where graphite is used as a neutron moderator, accumulation of Wigner energy followed by a sudden, spontaneous release may occur. Annealing (metallurgy), Annealing to at least 250 °C can release the energy safely, although in the Windscale fire the procedure went wrong, causing other reactor materials to combust.
The great variety of carbon compounds include such lethal poisons as tetrodotoxin, the lectin ricin from seeds of the castor oil plant ''Ricinus communis'', cyanide (CN), and Carbon monoxide poisoning, carbon monoxide; and such essentials to life as glucose and protein.
Pure carbon has extremely low toxicity to humans and can be handled safely in the form of graphite or charcoal. It is resistant to dissolution or chemical attack, even in the acidic contents of the digestive tract. Consequently, once it enters into the body's tissues it is likely to remain there indefinitely. Carbon black was probably one of the first pigments to be used for tattooing, and Ötzi the Iceman was found to have carbon tattoos that survived during his life and for 5200 years after his death. Inhalation of coal dust or soot (carbon black) in large quantities can be dangerous, irritating lung tissues and causing the congestive human lung, lung disease, coalworker's pneumoconiosis. Diamond dust used as an abrasive can be harmful if ingested or inhaled. Microparticles of carbon are produced in diesel engine exhaust fumes, and may accumulate in the lungs. In these examples, the harm may result from contaminants (e.g., organic chemicals, heavy metals) rather than from the carbon itself.
Carbon generally has low toxicity to life, life on Earth; but carbon nanoparticles are deadly to ''Drosophila''.
Carbon may burn vigorously and brightly in the presence of air at high temperatures. Large accumulations of coal, which have remained inert for hundreds of millions of years in the absence of oxygen, may spontaneous combustion, spontaneously combust when exposed to air in coal mine waste tips, ship cargo holds and coal bunkers, and storage dumps.
In nuclear reactor, nuclear applications where graphite is used as a neutron moderator, accumulation of Wigner energy followed by a sudden, spontaneous release may occur. Annealing (metallurgy), Annealing to at least 250 °C can release the energy safely, although in the Windscale fire the procedure went wrong, causing other reactor materials to combust.
The great variety of carbon compounds include such lethal poisons as tetrodotoxin, the lectin ricin from seeds of the castor oil plant ''Ricinus communis'', cyanide (CN), and Carbon monoxide poisoning, carbon monoxide; and such essentials to life as glucose and protein.
See also
* Carbon chauvinism * Carbon detonation * Carbon footprint * Carbon star * Carbon planet * Gas carbon * Low-carbon economy * Timeline of carbon nanotubesReferences
Bibliography
* *External links
*Carbon
at ''The Periodic Table of Videos'' (University of Nottingham)
Carbon on Britannica
(archived 18 June 2010)
(archived 9 November 2001)
Carbon—Super Stuff. Animation with sound and interactive 3D-models.
(archived 9 November 2012) {{Carbon compounds Carbon, Chemical elements Allotropes of carbon Reactive nonmetals Polyatomic nonmetals Native element minerals Reducing agents Chemical elements with hexagonal planar structure