Benzene Hexachloride (other) on:
[Wikipedia]
[Google]
[Amazon]
Benzene is an
Historic Benzene Formulae Kekulé (original).png, Kekulé's 1872 modification of his 1865 theory, illustrating rapid alternation of double bondsCritics pointed out a problem with Kekulé's original (1865) structure for benzene: Whenever benzene underwent substitution at the ortho position, two distinguishable isomers should have resulted, depending on whether a double bond or a single bond existed between the carbon atoms to which the substituents were attached; however, no such isomers were observed. In 1872, Kekulé suggested that benzene had two complementary structures and that these forms rapidly interconverted, so that if there were a double bond between any pair of carbon atoms at one instant, that double bond would become a single bond at the next instant (and vice versa). To provide a mechanism for the conversion process, Kekulé proposed that the valency of an atom is determined by the frequency with which it collided with its neighbors in a molecule. As the carbon atoms in the benzene ring collided with each other, each carbon atom would collide twice with one neighbor during a given interval and then twice with its other neighbor during the next interval. Thus, a double bond would exist with one neighbor during the first interval and with the other neighbor during the next interval. Therefore, between the carbon atoms of benzene there were no fixed (i.e., constant) and distinct single or double bonds; instead, the bonds between the carbon atoms were identical. Se
pages 86–89
of Auguste Kekulé (1872) "Ueber einige Condensationsprodukte des Aldehyds" (On some condensation products of aldehydes), ''Liebig's Annalen der Chemie und Pharmacie'', 162(1): 77–124, 309–320. From p. 89: ''"Das einfachste Mittel aller Stöße eines Kohlenstoffatoms ergiebt sich aus der Summe der Stöße der beiden ersten Zeiteinheiten, die sich dann periodisch wiederholen. … man sieht daher, daß jedes Kohlenstoffatom mit den beiden anderen, … daß diese Verschiedenheit nur eine scheinbare, aber keine wirkliche ist."'' (The simplest average of all the collisions of a carbon atom
In 1845,

C6H5CH3 + H2 -> C6H6 + CH4
This irreversible reaction is accompanied by an equilibrium side reaction that produces
File:Benzene_uses.png, center, Major commodity chemicals and polymers derived from benzene. Clicking on the image loads the appropriate article, 600px, thumb
rect 39 660 435 807
 The most widely practiced example of this reaction is the
The most widely practiced example of this reaction is the  Approximately 24,700,000 tons were produced in 1999. Highly instructive but of far less industrial significance is the Friedel-Crafts alkylation of benzene (and many other aromatic rings) using an alkyl halide in the presence of a strong Lewis acid catalyst. Similarly, the Friedel-Crafts acylation is a related example of electrophilic aromatic substitution. The reaction involves the acylation of benzene (or many other aromatic rings) with an
Approximately 24,700,000 tons were produced in 1999. Highly instructive but of far less industrial significance is the Friedel-Crafts alkylation of benzene (and many other aromatic rings) using an alkyl halide in the presence of a strong Lewis acid catalyst. Similarly, the Friedel-Crafts acylation is a related example of electrophilic aromatic substitution. The reaction involves the acylation of benzene (or many other aromatic rings) with an 
 Benzene is classified as a
Benzene is classified as a
Benzene
at '' The Periodic Table of Videos'' (University of Nottingham)
International Chemical Safety Card 0015
*
Dept. of Health and Human Services: TR-289: Toxicology and Carcinogenesis Studies of Benzene
Video Podcast
of Sir John Cadogan giving a lecture on Benzene since Faraday, in 1991
Substance profile
*
NLM Hazardous Substances Databank – Benzene
{{Authority control Annulenes Aromatic hydrocarbons Aromatic solvents Carcinogens Commodity chemicals GABAA receptor positive allosteric modulators Hazardous air pollutants Hydrocarbon solvents IARC Group 1 carcinogens Immunotoxins Mutagens Occupational safety and health Petrochemicals Simple aromatic rings Six-membered rings Soil contamination Sweet-smelling chemicals Teratogens
organic
Organic may refer to:
* Organic, of or relating to an organism, a living entity
* Organic, of or relating to an anatomical organ
Chemistry
* Organic matter, matter that has come from a once-living organism, is capable of decay or is the product ...
chemical compound
A chemical compound is a chemical substance composed of many identical molecules (or molecular entities) containing atoms from more than one chemical element held together by chemical bonds. A molecule consisting of atoms of only one element ...
with the molecular formula C6H6. The benzene molecule
A molecule is a group of two or more atoms held together by attractive forces known as chemical bonds; depending on context, the term may or may not include ions which satisfy this criterion. In quantum physics, organic chemistry, and bioch ...
is composed of six carbon
Carbon () is a chemical element with the symbol C and atomic number 6. It is nonmetallic and tetravalent
In chemistry, the valence (US spelling) or valency (British spelling) of an element is the measure of its combining capacity with o ...
atoms joined in a planar ring
Ring may refer to:
* Ring (jewellery), a round band, usually made of metal, worn as ornamental jewelry
* To make a sound with a bell, and the sound made by a bell
:(hence) to initiate a telephone connection
Arts, entertainment and media Film and ...
with one hydrogen
Hydrogen is the chemical element with the symbol H and atomic number 1. Hydrogen is the lightest element. At standard conditions hydrogen is a gas of diatomic molecules having the formula . It is colorless, odorless, tasteless, non-toxic, an ...
atom attached to each. Because it contains only carbon and hydrogen atoms, benzene is classed as a hydrocarbon
In organic chemistry, a hydrocarbon is an organic compound consisting entirely of hydrogen and carbon. Hydrocarbons are examples of group 14 hydrides. Hydrocarbons are generally colourless and hydrophobic, and their odors are usually weak or ex ...
.
Benzene is a natural constituent of petroleum
Petroleum, also known as crude oil, or simply oil, is a naturally occurring yellowish-black liquid mixture of mainly hydrocarbons, and is found in geological formations. The name ''petroleum'' covers both naturally occurring unprocessed crud ...
and is one of the elementary petrochemical
Petrochemicals (sometimes abbreviated as petchems) are the chemical products obtained from petroleum by refining. Some chemical compounds made from petroleum are also obtained from other fossil fuels, such as coal or natural gas, or renewable sou ...
s. Due to the cyclic continuous pi bonds between the carbon atoms, benzene is classed as an aromatic hydrocarbon. Benzene is a colorless and highly flammable
A combustible material is something that can burn (i.e., ''combust'') in air. A combustible material is flammable if it ignites easily at ambient temperatures. In other words, a combustible material ignites with some effort and a flammable mat ...
liquid with a sweet smell, and is partially responsible for the aroma of gasoline
Gasoline (; ) or petrol (; ) (see ) is a transparent, petroleum-derived flammable liquid that is used primarily as a fuel in most spark-ignited internal combustion engines (also known as petrol engines). It consists mostly of organic co ...
. It is used primarily as a precursor
Precursor or Precursors may refer to:
*Precursor (religion), a forerunner, predecessor
** The Precursor, John the Baptist
Science and technology
* Precursor (bird), a hypothesized genus of fossil birds that was composed of fossilized parts of unr ...
to the manufacture of chemicals with more complex structure, such as ethylbenzene
Ethylbenzene is an organic compound with the formula . It is a highly flammable, colorless liquid with an odor similar to that of gasoline. This monocyclic aromatic hydrocarbon is important in the petrochemical industry as an reaction intermediat ...
and cumene, of which billions of kilograms are produced annually. Although benzene is a major industrial chemical
The chemical industry comprises the companies that produce industrial chemicals. Central to the modern world economy, it converts raw materials (oil, natural gas, air, water, metals, and minerals) into more than 70,000 different products. The ...
, it finds limited use in consumer items because of its toxicity.
History
Discovery
The word "''benzene''" derives from "''gum benzoin''" (benzoin resin
Benzoin or benjamin (corrupted pronunciation) is a balsamic resin obtained from the bark of several species of trees in the genus ''Styrax''. It is used in perfumes and some kinds of incense and as a flavoring and medicine (see tincture of benz ...
), an aromatic resin known to European pharmacists and perfumers since the 16th century as a product of southeast Asia. An acidic material was derived from benzoin by sublimation
Sublimation or sublimate may refer to:
* ''Sublimation'' (album), by Canvas Solaris, 2004
* Sublimation (phase transition), directly from the solid to the gas phase
* Sublimation (psychology), a mature type of defense mechanism
* Sublimate of mer ...
, and named "flowers of benzoin", or benzoic acid. The hydrocarbon derived from benzoic acid thus acquired the name benzin, benzol, or benzene. Michael Faraday
Michael Faraday (; 22 September 1791 – 25 August 1867) was an English scientist who contributed to the study of electromagnetism and electrochemistry. His main discoveries include the principles underlying electromagnetic inducti ...
first isolated and identified benzene in 1825 from the oily residue derived from the production of illuminating gas, giving it the name ''bicarburet of hydrogen''. In 1833, Eilhard Mitscherlich
Eilhard Mitscherlich (; 7 January 179428 August 1863) was a German chemist, who is perhaps best remembered today for his discovery of the phenomenon of crystallographic isomorphism in 1819.
Early life and work
Mitscherlich was born at Neuende ...
produced it by distilling benzoic acid
Benzoic acid is a white (or colorless) solid organic compound with the formula , whose structure consists of a benzene ring () with a carboxyl () substituent. It is the simplest aromatic carboxylic acid. The name is derived from gum benzoin, wh ...
(from gum benzoin) and lime
Lime commonly refers to:
* Lime (fruit), a green citrus fruit
* Lime (material), inorganic materials containing calcium, usually calcium oxide or calcium hydroxide
* Lime (color), a color between yellow and green
Lime may also refer to:
Botany ...
. He gave the compound the name ''benzin''. In 1836, the French chemist Auguste Laurent
Auguste Laurent (14 November 1807 – 15 April 1853) was a French chemist who helped in the founding of organic chemistry with his discoveries of anthracene, phthalic acid, and carbolic acid.
He devised a systematic nomenclature for organic chem ...
named the substance "phène"; this word has become the root of the English word "phenol
Phenol (also called carbolic acid) is an aromatic organic compound with the molecular formula . It is a white crystalline solid that is volatile. The molecule consists of a phenyl group () bonded to a hydroxy group (). Mildly acidic, it req ...
", which is hydroxylated
In chemistry, hydroxylation can refer to:
*(i) most commonly, hydroxylation describes a chemistry, chemical process that introduces a hydroxyl group () into an organic compound.
*(ii) the ''degree of hydroxylation'' refers to the number of OH gr ...
benzene, and " phenyl", the radical formed by abstraction of a hydrogen atom ( free radical H•) from benzene.
pages 86–89
of Auguste Kekulé (1872) "Ueber einige Condensationsprodukte des Aldehyds" (On some condensation products of aldehydes), ''Liebig's Annalen der Chemie und Pharmacie'', 162(1): 77–124, 309–320. From p. 89: ''"Das einfachste Mittel aller Stöße eines Kohlenstoffatoms ergiebt sich aus der Summe der Stöße der beiden ersten Zeiteinheiten, die sich dann periodisch wiederholen. … man sieht daher, daß jedes Kohlenstoffatom mit den beiden anderen, … daß diese Verschiedenheit nur eine scheinbare, aber keine wirkliche ist."'' (The simplest average of all the collisions of a carbon atom
n benzene
N, or n, is the fourteenth letter in the Latin alphabet, used in the modern English alphabet, the alphabets of other western European languages and others worldwide. Its name in English is ''en'' (pronounced ), plural ''ens''.
History
...
comes from the sum of the collisions during the first two units of time, which then periodically repeat. … thus one sees that each carbon atom collides equally often with the two others against which it bumps, ndthus stands in exactly the same relation with its two neighbors. The usual structural formula for benzene expresses, of course, only the collisions that occur during ''one'' unit of time, thus during one phase, and so one is led to the view hatdoubly substituted derivatives f benzene
F, or f, is the sixth letter in the Latin alphabet, used in the modern English alphabet, the alphabets of other western European languages and others worldwide. Its name in English is ''ef'' (pronounced ), and the plural is ''efs''.
Hist ...
must be different at positions 1,2 and 1,6 f the benzene ring
F, or f, is the sixth letter in the Latin alphabet, used in the modern English alphabet, the alphabets of other western European languages and others worldwide. Its name in English is ''ef'' (pronounced ), and the plural is ''efs''.
Hist ...
If the idea hat was
A hat is a head covering which is worn for various reasons, including protection against weather conditions, ceremonial reasons such as university graduation, religious reasons, safety, or as a fashion accessory. Hats which incorporate mecha ...
just presented—or a similar one—can be regarded as correct, then tfollows therefrom that this difference etween the bonds at positions 1,2 and 1,6is only an apparent ne not a real ne)
Charles Blachford Mansfield
Charles Blachford Mansfield (8 May 1819 – 26 February 1855) was a British chemist and author.
Early life
He was born on 8 May 1819 at Rowner, Hampshire, where his father, John Mansfield, was rector; his mother was Winifred, eldest daughter of ...
, working under August Wilhelm von Hofmann
August Wilhelm von Hofmann (8 April 18185 May 1892) was a German chemist who made considerable contributions to organic chemistry. His research on aniline helped lay the basis of the aniline-dye industry, and his research on coal tar laid the g ...
, isolated benzene from coal tar
Coal tar is a thick dark liquid which is a by-product of the production of coke and coal gas from coal. It is a type of creosote. It has both medical and industrial uses. Medicinally it is a topical medication applied to skin to treat psoriasi ...
. Four years later, Mansfield began the first industrial-scale production of benzene, based on the coal-tar method. Gradually, the sense developed among chemists that a number of substances were chemically related to benzene, comprising a diverse chemical family. In 1855, Hofmann used the word "aromatic
In chemistry, aromaticity is a chemical property of cyclic ( ring-shaped), ''typically'' planar (flat) molecular structures with pi bonds in resonance (those containing delocalized electrons) that gives increased stability compared to satur ...
" to designate this family relationship, after a characteristic property of many of its members. In 1997, benzene was detected in deep space.
Ring formula
The empirical formula for benzene was long known, but its highlypolyunsaturated
In nutrition, biology, and chemistry, fat usually means any ester of fatty acids, or a mixture of such compounds, most commonly those that occur in living beings or in food.
The term often refers specifically to triglycerides (triple e ...
structure, with just one hydrogen
Hydrogen is the chemical element with the symbol H and atomic number 1. Hydrogen is the lightest element. At standard conditions hydrogen is a gas of diatomic molecules having the formula . It is colorless, odorless, tasteless, non-toxic, an ...
atom for each carbon
Carbon () is a chemical element with the symbol C and atomic number 6. It is nonmetallic and tetravalent
In chemistry, the valence (US spelling) or valency (British spelling) of an element is the measure of its combining capacity with o ...
atom, was challenging to determine. Archibald Scott Couper
Archibald Scott Couper (; 31 March 1831 – 11 March 1892) was a Scottish chemist who proposed an early theory of chemical structure and bonding. He developed the concepts of tetravalent carbon atoms linking together to form large molecules ...
in 1858 and Johann Josef Loschmidt in 1861 suggested possible structures that contained multiple double bonds or multiple rings, but too little evidence was then available to help chemists decide on any particular structure.
In 1865, the German chemist Friedrich August Kekulé Friedrich may refer to:
Names
* Friedrich (surname), people with the surname ''Friedrich''
* Friedrich (given name), people with the given name ''Friedrich''
Other
* Friedrich (board game), a board game about Frederick the Great and the Seven Year ...
published a paper in French (for he was then teaching in Francophone Belgium) suggesting that the structure contained a ring of six carbon atoms with alternating single and double bonds. The next year he published a much longer paper in German on the same subject. On p. 100, Kekulé suggests that the carbon atoms of benzene could form a "chaîne fermée" (a closed chain, a loop). Kekulé used evidence that had accumulated in the intervening years—namely, that there always appeared to be only one isomer
In chemistry, isomers are molecules or polyatomic ions with identical molecular formulae – that is, same number of atoms of each element – but distinct arrangements of atoms in space. Isomerism is existence or possibility of isomers.
Iso ...
of any monoderivative of benzene, and that there always appeared to be exactly three isomers of every disubstituted derivative—now understood to correspond to the ortho, meta, and para patterns of arene substitution—to argue in support of his proposed structure. Kekulé's symmetrical ring could explain these curious facts, as well as benzene's 1:1 carbon-hydrogen ratio.
The new understanding of benzene, and hence of all aromatic compounds, proved to be so important for both pure and applied chemistry that in 1890 the German Chemical Society organized an elaborate appreciation in Kekulé's honor, celebrating the twenty-fifth anniversary of his first benzene paper. Here Kekulé spoke of the creation of the theory. He said that he had discovered the ring shape of the benzene molecule after having a reverie or day-dream of a snake biting its own tail (this is a common symbol in many ancient cultures known as the Ouroboros or endless knot). This vision, he said, came to him after years of studying the nature of carbon-carbon bonds. This was seven years after he had solved the problem of how carbon atoms could bond to up to four other atoms at the same time. Curiously, a similar, humorous depiction of benzene had appeared in 1886 in a pamphlet entitled ''Berichte der Durstigen Chemischen Gesellschaft'' (Journal of the Thirsty Chemical Society), a parody of the ''Berichte der Deutschen Chemischen Gesellschaft'', only the parody had monkeys seizing each other in a circle, rather than snakes as in Kekulé's anecdote. Some historians have suggested that the parody was a lampoon of the snake anecdote, possibly already well known through oral transmission even if it had not yet appeared in print. Kekulé's 1890 speech in which this anecdote appeared has been translated into English. If the anecdote is the memory of a real event, circumstances mentioned in the story suggest that it must have happened early in 1862.
In 1929, the cyclic nature of benzene was finally confirmed by the crystallographer Kathleen Lonsdale
Dame Kathleen Lonsdale ( Yardley; 28 January 1903 – 1 April 1971) was an Irish-born British pacifist, prison reformer and crystallographer. She proved, in 1929, that the benzene ring is flat by using X-ray diffraction methods to elucidate t ...
using X-ray diffraction
X-ray crystallography is the experimental science determining the atomic and molecular structure of a crystal, in which the crystalline structure causes a beam of incident X-rays to diffract into many specific directions. By measuring the angles ...
methods. Using large crystals of hexamethylbenzene
Hexamethylbenzene, also known as mellitene, is a hydrocarbon with the molecular formula C12H18 and the condensed structural formula C6(CH3)6. It is an aromatic compound and a derivative of benzene, where benzene's six hydrogen atoms have each ...
, a benzene derivative with the same core of six carbon atoms, Lonsdale obtained diffraction patterns. Through calculating more than thirty parameters, Lonsdale demonstrated that the benzene ring could not be anything but a flat hexagon, and provided accurate distances for all carbon-carbon bonds in the molecule.
Nomenclature
The German chemistWilhelm Körner
Wilhelm Körner, later a.k.a. Guglielmo Körner (April 20, 1839 in Cassel – March 29, 1925 in Milan), was a German chemist.
Life
Körner studied chemistry at Giessen, where he graduated in 1860. In 1866, he became assistant to Kekulé at ...
suggested the prefixes ortho-, meta-, para- to distinguish di-substituted benzene derivatives in 1867; however, he did not use the prefixes to distinguish the relative positions of the substituents on a benzene ring. It was the German chemist Carl Gräbe
Carl Gräbe (; 24 February 1841 – 19 January 1927) was a German industrial and academic chemist from Frankfurt am Main who held professorships in his field at Leipzig, Königsberg, and Geneva. He is known for the first synthesis of the ec ...
who, in 1869, first used the prefixes ortho-, meta-, para- to denote specific relative locations of the substituents on a di-substituted aromatic ring (viz, naphthalene). In 1870, the German chemist Viktor Meyer
Viktor Meyer (8 September 18488 August 1897) was a German chemist and significant contributor to both organic and inorganic chemistry. He is best known for inventing an apparatus for determining vapour densities, the Viktor Meyer apparatus, and ...
first applied Gräbe's nomenclature to benzene.
Early applications
In the 19th and early 20th centuries, benzene was used as an after-shave lotion because of its pleasant smell. Prior to the 1920s, benzene was frequently used as an industrial solvent, especially fordegreasing
Degreasing, often called defatting or fat trimming, is the removal of fatty acids from an object. In culinary science, degreasing is done with the intention of reducing the fat content of a meal.
Degreasing food
Degreasing is often used by diete ...
metal. As its toxicity became obvious, benzene was supplanted by other solvents, especially toluene
Toluene (), also known as toluol (), is a substituted aromatic hydrocarbon. It is a colorless, water-insoluble liquid with the smell associated with paint thinners. It is a mono-substituted benzene derivative, consisting of a methyl group (CH3) at ...
(methylbenzene), which has similar physical properties but is not as carcinogenic.
In 1903, Ludwig Roselius popularized the use of benzene to decaffeinate
Decaffeination is the removal of caffeine from coffee beans, cocoa, tea leaves, and other caffeine-containing materials. Decaffeinated drinks contain typically 1–2% of the original caffeine content, and sometimes as much as 20%. Decaffeinated ...
coffee
Coffee is a drink prepared from roasted coffee beans. Darkly colored, bitter, and slightly acidic, coffee has a stimulant, stimulating effect on humans, primarily due to its caffeine content. It is the most popular hot drink in the world.
S ...
. This discovery led to the production of Sanka
Sanka is a brand of instant decaffeinated coffee, sold around the world, and was one of the earliest decaffeinated varieties. Sanka is distributed in the United States by Kraft Heinz.
History
Decaffeinated coffee was developed in 1903 by a team ...
. This process was later discontinued. Benzene was historically used as a significant component in many consumer products such as liquid wrench
A liquid is a nearly incompressible fluid that conforms to the shape of its container but retains a (nearly) constant volume independent of pressure. As such, it is one of the four fundamental states of matter (the others being solid, gas, a ...
, several paint strippers, rubber cement
Rubber cement (cow gum in British English) is an adhesive made from elastic polymers (typically latex) mixed in a solvent such as acetone, hexane, heptane or toluene to keep it fluid enough to be used. This makes it part of the class of drying ad ...
s, spot removers, and other products. Manufacture of some of these benzene-containing formulations ceased in about 1950, although Liquid Wrench continued to contain significant amounts of benzene until the late 1970s.
Occurrence
Trace amounts of benzene are found in petroleum and coal. It is a byproduct of the incomplete combustion of many materials. For commercial use, untilWorld War II
World War II or the Second World War, often abbreviated as WWII or WW2, was a world war that lasted from 1939 to 1945. It involved the vast majority of the world's countries—including all of the great powers—forming two opposin ...
, much of benzene was obtained as a by-product of coke production (or "coke-oven light oil") for the steel
Steel is an alloy made up of iron with added carbon to improve its strength and fracture resistance compared to other forms of iron. Many other elements may be present or added. Stainless steels that are corrosion- and oxidation-resistant ty ...
industry. However, in the 1950s, increased demand for benzene, especially from the growing polymer
A polymer (; Greek '' poly-'', "many" + ''-mer'', "part")
is a substance or material consisting of very large molecules called macromolecules, composed of many repeating subunits. Due to their broad spectrum of properties, both synthetic a ...
s industry, necessitated the production of benzene from petroleum. Today, most benzene comes from the petrochemical industry
The petrochemical industry is concerned with the production and trade of petrochemicals. A major part is constituted by the plastics (polymer) industry. It directly interfaces with the petroleum industry, especially the downstream sector.
Compan ...
, with only a small fraction being produced from coal. Benzene molecules have been detected on Mars
Mars is the fourth planet from the Sun and the second-smallest planet in the Solar System, only being larger than Mercury (planet), Mercury. In the English language, Mars is named for the Mars (mythology), Roman god of war. Mars is a terr ...
.
Structure
X-ray diffraction
X-ray crystallography is the experimental science determining the atomic and molecular structure of a crystal, in which the crystalline structure causes a beam of incident X-rays to diffract into many specific directions. By measuring the angles ...
shows that all six carbon-carbon bonds in benzene are of the same length, at 140 picometre
The picometre (international spelling as used by the International Bureau of Weights and Measures; SI symbol: pm) or picometer (American spelling) is a unit of length in the International System of Units (SI), equal to , or one trillionth of ...
s (pm). The C–C bond lengths are greater than a double bond (135 pm) but shorter than a single bond (147 pm). This intermediate distance is caused by electron delocalization: the electrons for C=C bonding are distributed equally between each of the six carbon atoms. Benzene has 6 hydrogen atoms, fewer than the corresponding parent alkane
In organic chemistry, an alkane, or paraffin (a historical trivial name that also has other meanings), is an acyclic saturated hydrocarbon. In other words, an alkane consists of hydrogen and carbon atoms arranged in a tree structure in which ...
, hexane
Hexane () is an organic compound, a straight-chain alkane with six carbon atoms and has the molecular formula C6H14.
It is a colorless liquid, odorless when pure, and with boiling points approximately . It is widely used as a cheap, relatively ...
, which has 14. Benzene and cyclohexane have a similar structure, only the ring of delocalized electrons and the loss of one hydrogen per carbon distinguishes it from cyclohexane. The molecule is planar. The molecular orbital description involves the formation of three delocalized π orbitals spanning all six carbon atoms, while the valence bond description involves a superposition of resonance structures. It is likely that this stability contributes to the peculiar molecular and chemical properties known as aromaticity
In chemistry, aromaticity is a chemical property of cyclic ( ring-shaped), ''typically'' planar (flat) molecular structures with pi bonds in resonance (those containing delocalized electrons) that gives increased stability compared to saturate ...
. To accurately reflect the nature of the bonding, benzene is often depicted with a circle inside a hexagonal arrangement of carbon atoms.
Derivatives of benzene occur sufficiently often as a component of organic molecules, so much so that the Unicode
Unicode, formally The Unicode Standard,The formal version reference is is an information technology Technical standard, standard for the consistent character encoding, encoding, representation, and handling of Character (computing), text expre ...
Consortium has allocated a symbol in the Miscellaneous Technical block with the code U+232C (⌬) to represent it with three double bonds, and U+23E3 (⏣) for a delocalized version.
Benzene derivatives
Many important chemical compounds are derived from benzene by replacing one or more of its hydrogen atoms with anotherfunctional group
In organic chemistry, a functional group is a substituent or moiety in a molecule that causes the molecule's characteristic chemical reactions. The same functional group will undergo the same or similar chemical reactions regardless of the rest ...
. Examples of simple benzene derivatives are phenol
Phenol (also called carbolic acid) is an aromatic organic compound with the molecular formula . It is a white crystalline solid that is volatile. The molecule consists of a phenyl group () bonded to a hydroxy group (). Mildly acidic, it req ...
, toluene
Toluene (), also known as toluol (), is a substituted aromatic hydrocarbon. It is a colorless, water-insoluble liquid with the smell associated with paint thinners. It is a mono-substituted benzene derivative, consisting of a methyl group (CH3) at ...
, and aniline
Aniline is an organic compound with the formula C6 H5 NH2. Consisting of a phenyl group attached to an amino group, aniline is the simplest aromatic amine
In organic chemistry, an aromatic amine is an organic compound consisting of an aroma ...
, abbreviated PhOH, PhMe, and PhNH2, respectively. Linking benzene rings gives biphenyl
Biphenyl (also known as diphenyl, phenylbenzene, 1,1′-biphenyl, lemonene or BP) is an organic compound that forms colorless crystals. Particularly in older literature, compounds containing the functional group consisting of biphenyl less one ...
, C6H5–C6H5. Further loss of hydrogen gives "fused" aromatic hydrocarbons, such as naphthalene
Naphthalene is an organic compound with formula . It is the simplest polycyclic aromatic hydrocarbon, and is a white crystalline solid with a characteristic odor that is detectable at concentrations as low as 0.08 ppm by mass. As an aromati ...
, anthracene
Anthracene is a solid polycyclic aromatic hydrocarbon (PAH) of formula C14H10, consisting of three fused benzene rings. It is a component of coal tar. Anthracene is used in the Economic production, production of the red dye alizarin and other dyes ...
, phenanthrene
Phenanthrene is a polycyclic aromatic hydrocarbon (PAH) with formula C14H10, consisting of three fused benzene rings. It is a colorless, crystal-like solid, but can also appear yellow. Phenanthrene is used to make dyes, plastics and pesticides, e ...
, and pyrene
Pyrene is a polycyclic aromatic hydrocarbon (PAH) consisting of four fused benzene rings, resulting in a flat aromatic system. The chemical formula is . This yellow solid is the smallest peri-fused PAH (one where the rings are fused through mor ...
. The limit of the fusion process is the hydrogen-free allotrope of carbon, graphite
Graphite () is a crystalline form of the element carbon. It consists of stacked layers of graphene. Graphite occurs naturally and is the most stable form of carbon under standard conditions. Synthetic and natural graphite are consumed on large ...
.
In heterocycles, carbon atoms in the benzene ring are replaced with other elements. The most important variations contain nitrogen
Nitrogen is the chemical element with the symbol N and atomic number 7. Nitrogen is a nonmetal and the lightest member of group 15 of the periodic table, often called the pnictogens. It is a common element in the universe, estimated at se ...
. Replacing one CH with N gives the compound pyridine
Pyridine is a basic heterocyclic organic compound with the chemical formula . It is structurally related to benzene, with one methine group replaced by a nitrogen atom. It is a highly flammable, weakly alkaline, water-miscible liquid with a d ...
, C5H5N. Although benzene and pyridine are ''structurally'' related, benzene cannot be converted into pyridine. Replacement of a second CH bond with N gives, depending on the location of the second N, pyridazine, pyrimidine
Pyrimidine (; ) is an aromatic, heterocyclic, organic compound similar to pyridine (). One of the three diazines (six-membered heterocyclics with two nitrogen atoms in the ring), it has nitrogen atoms at positions 1 and 3 in the ring. The other ...
, or pyrazine.
Production
Four chemical processes contribute to industrial benzene production:catalytic reforming
Catalytic reforming is a chemical process used to convert petroleum refinery naphthas distilled from crude oil (typically having low octane ratings) into high-octane liquid products called reformates, which are premium blending stocks for high-o ...
, toluene
Toluene (), also known as toluol (), is a substituted aromatic hydrocarbon. It is a colorless, water-insoluble liquid with the smell associated with paint thinners. It is a mono-substituted benzene derivative, consisting of a methyl group (CH3) at ...
hydrodealkylation, toluene disproportionation, and steam cracking
Steam cracking is a petrochemical process in which saturated hydrocarbons are broken down into smaller, often unsaturated, hydrocarbons. It is the principal industrial method for producing the lighter alkenes (or commonly olefins), including ethe ...
etc. According to the ATSDR Toxicological Profile for benzene, between 1978 and 1981, catalytic reformates accounted for approximately 44–50% of the total U.S benzene production.
Catalytic reforming
In catalytic reforming, a mixture ofhydrocarbon
In organic chemistry, a hydrocarbon is an organic compound consisting entirely of hydrogen and carbon. Hydrocarbons are examples of group 14 hydrides. Hydrocarbons are generally colourless and hydrophobic, and their odors are usually weak or ex ...
s with boiling points between 60 and 200 °C is blended with hydrogen
Hydrogen is the chemical element with the symbol H and atomic number 1. Hydrogen is the lightest element. At standard conditions hydrogen is a gas of diatomic molecules having the formula . It is colorless, odorless, tasteless, non-toxic, an ...
gas and then exposed to a bifunctional
In organic chemistry, when a single organic molecule has two different functional groups, it is called a bifunctional molecule . A bifunctional molecule has the properties of two different types of functional groups, such as an alcohol (), amide ( ...
platinum chloride or rhenium chloride catalyst
Catalysis () is the process of increasing the rate of a chemical reaction by adding a substance known as a catalyst (). Catalysts are not consumed in the reaction and remain unchanged after it. If the reaction is rapid and the catalyst recyc ...
at 500–525 °C and pressures ranging from 8–50 atm. Under these conditions, aliphatic
In organic chemistry, hydrocarbons ( compounds composed solely of carbon and hydrogen) are divided into two classes: aromatic compounds and aliphatic compounds (; G. ''aleiphar'', fat, oil). Aliphatic compounds can be saturated, like hexane, or ...
hydrocarbons form rings and lose hydrogen to become aromatic hydrocarbons. The aromatic products of the reaction are then separated from the reaction mixture (or reformate) by extraction Extraction may refer to:
Science and technology
Biology and medicine
* Comedo extraction, a method of acne treatment
* Dental extraction, the surgical removal of a tooth from the mouth
Computing and information science
* Data extraction, the pro ...
with any one of a number of solvent
A solvent (s) (from the Latin '' solvō'', "loosen, untie, solve") is a substance that dissolves a solute, resulting in a solution. A solvent is usually a liquid but can also be a solid, a gas, or a supercritical fluid. Water is a solvent for ...
s, including diethylene glycol or sulfolane
Sulfolane (also ''tetramethylene sulfone'', systematic name: 1λ6-thiolane-1,1-dione) is an organosulfur compound, formally a cyclic sulfone, with the formula (CH2)4SO2. It is a colorless liquid commonly used in the chemical industry as a solvent ...
, and benzene is then separated from the other aromatics by distillation. The extraction step of aromatics from the reformate is designed to produce aromatics with lowest non-aromatic components. Recovery of the aromatics, commonly referred to as BTX (benzene, toluene and xylene isomers), involves such extraction and distillation steps.
In similar fashion to this catalytic reforming, UOP and BP commercialized a method from LPG (mainly propane and butane) to aromatics.
Toluene hydrodealkylation
Toluenehydrodealkylation
Hydrodealkylation is a chemical reaction that often involves reacting an aromatic hydrocarbon, such as toluene, in the presence of hydrogen gas to form a simpler aromatic hydrocarbon devoid of functional groups. An example is the conversion of 1,2, ...
converts toluene
Toluene (), also known as toluol (), is a substituted aromatic hydrocarbon. It is a colorless, water-insoluble liquid with the smell associated with paint thinners. It is a mono-substituted benzene derivative, consisting of a methyl group (CH3) at ...
to benzene. In this hydrogen-intensive process, toluene is mixed with hydrogen, then passed over a chromium
Chromium is a chemical element with the symbol Cr and atomic number 24. It is the first element in group 6. It is a steely-grey, lustrous, hard, and brittle transition metal.
Chromium metal is valued for its high corrosion resistance and hardne ...
, molybdenum
Molybdenum is a chemical element with the symbol Mo and atomic number 42 which is located in period 5 and group 6. The name is from Neo-Latin ''molybdaenum'', which is based on Ancient Greek ', meaning lead, since its ores were confused with lea ...
, or platinum
Platinum is a chemical element with the symbol Pt and atomic number 78. It is a dense, malleable, ductile, highly unreactive, precious, silverish-white transition metal. Its name originates from Spanish , a diminutive of "silver".
Platinu ...
oxide
An oxide () is a chemical compound that contains at least one oxygen atom and one other element in its chemical formula. "Oxide" itself is the dianion of oxygen, an O2– (molecular) ion. with oxygen in the oxidation state of −2. Most of the E ...
catalyst at 500–650 °C and 20–60 atm pressure. Sometimes, higher temperatures are used instead of a catalyst (at the similar reaction condition). Under these conditions, toluene undergoes dealkylation to benzene and methane
Methane ( , ) is a chemical compound with the chemical formula (one carbon atom bonded to four hydrogen atoms). It is a group-14 hydride, the simplest alkane, and the main constituent of natural gas. The relative abundance of methane on Eart ...
:
:biphenyl
Biphenyl (also known as diphenyl, phenylbenzene, 1,1′-biphenyl, lemonene or BP) is an organic compound that forms colorless crystals. Particularly in older literature, compounds containing the functional group consisting of biphenyl less one ...
(aka diphenyl) at higher temperature:
:2 +
If the raw material stream contains much non-aromatic components (paraffins or naphthenes), those are likely decomposed to lower hydrocarbons such as methane, which increases the consumption of hydrogen.
A typical reaction yield exceeds 95%. Sometimes, xylene
In organic chemistry, xylene or xylol (; IUPAC name: dimethylbenzene) are any of three organic compounds with the formula . They are derived from the substitution of two hydrogen atoms with methyl groups in a benzene ring; which hydrogens are sub ...
s and heavier aromatics are used in place of toluene, with similar efficiency.
This is often called "on-purpose" methodology to produce benzene, compared to conventional BTX (benzene-toluene-xylene) extraction processes.
Toluene disproportionation
Toluenedisproportionation
In chemistry, disproportionation, sometimes called dismutation, is a redox reaction in which one compound of intermediate oxidation state converts to two compounds, one of higher and one of lower oxidation states. More generally, the term can b ...
(TDP) is the conversion of toluene to benzene and xylene
In organic chemistry, xylene or xylol (; IUPAC name: dimethylbenzene) are any of three organic compounds with the formula . They are derived from the substitution of two hydrogen atoms with methyl groups in a benzene ring; which hydrogens are sub ...
.
Given that demand for ''para''-xylene ( ''p''-xylene) substantially exceeds demand for other xylene isomers, a refinement of the TDP process called Selective TDP (STDP) may be used. In this process, the xylene stream exiting the TDP unit is approximately 90% ''p''-xylene. In some systems, even the benzene-to-xylenes ratio is modified to favor xylenes.
Steam cracking
Steam cracking
Steam cracking is a petrochemical process in which saturated hydrocarbons are broken down into smaller, often unsaturated, hydrocarbons. It is the principal industrial method for producing the lighter alkenes (or commonly olefins), including ethe ...
is the process for producing ethylene
Ethylene (IUPAC name: ethene) is a hydrocarbon which has the formula or . It is a colourless, flammable gas with a faint "sweet and musky" odour when pure. It is the simplest alkene (a hydrocarbon with carbon-carbon double bonds).
Ethylene i ...
and other alkene
In organic chemistry, an alkene is a hydrocarbon containing a carbon–carbon double bond.
Alkene is often used as synonym of olefin, that is, any hydrocarbon containing one or more double bonds.H. Stephen Stoker (2015): General, Organic, an ...
s from aliphatic hydrocarbons
In organic chemistry, hydrocarbons ( compounds composed solely of carbon and hydrogen) are divided into two classes: aromatic compounds and aliphatic compounds (; G. ''aleiphar'', fat, oil). Aliphatic compounds can be saturated, like hexane, o ...
. Depending on the feedstock used to produce the olefins, steam cracking can produce a benzene-rich liquid by-product called ''pyrolysis gasoline
Pyrolysis gasoline or Pygas is a naphtha-range product with high aromatics content. It is a by-product of high temperature naphtha cracking during ethylene and propylene production. Also, it is a high octane number mixture that contains aromati ...
''. Pyrolysis gasoline can be blended with other hydrocarbons as a gasoline additive, or routed through an extraction process to recover BTX aromatics (benzene, toluene and xylenes).
Other methods
Although of no commercial significance, many other routes to benzene exist.Phenol
Phenol (also called carbolic acid) is an aromatic organic compound with the molecular formula . It is a white crystalline solid that is volatile. The molecule consists of a phenyl group () bonded to a hydroxy group (). Mildly acidic, it req ...
and halobenzene In organic chemistry, an aryl halide (also known as haloarene) is an aromatic compound in which one or more hydrogen atoms, directly bonded to an aromatic ring are replaced by a halide. The haloarene are different from haloalkanes because they exhi ...
s can be reduced with metals. Benzoic acid
Benzoic acid is a white (or colorless) solid organic compound with the formula , whose structure consists of a benzene ring () with a carboxyl () substituent. It is the simplest aromatic carboxylic acid. The name is derived from gum benzoin, wh ...
and its salts undergo decarboxylation
Decarboxylation is a chemical reaction that removes a carboxyl group and releases carbon dioxide (CO2). Usually, decarboxylation refers to a reaction of carboxylic acids, removing a carbon atom from a carbon chain. The reverse process, which is t ...
to benzene. The reaction of the diazonium compound
Diazonium compounds or diazonium salts are a group of organic compounds sharing a common functional group where R can be any organic group, such as an alkyl or an aryl, and X is an inorganic or organic anion, such as a halide.
General propert ...
derived from aniline
Aniline is an organic compound with the formula C6 H5 NH2. Consisting of a phenyl group attached to an amino group, aniline is the simplest aromatic amine
In organic chemistry, an aromatic amine is an organic compound consisting of an aroma ...
with hypophosphorus acid gives benzene. Alkyne trimerisation
In organic chemistry, an alkyne trimerisation is a +2+2nbsp; cycloaddition reaction in which three alkyne units () react to form a benzene ring. The reaction requires a metal catalyst. The process is of historic interest as well as being applic ...
of acetylene
Acetylene (systematic name: ethyne) is the chemical compound with the formula and structure . It is a hydrocarbon and the simplest alkyne. This colorless gas is widely used as a fuel and a chemical building block. It is unstable in its pure ...
gives benzene. Complete decarboxylation
Decarboxylation is a chemical reaction that removes a carboxyl group and releases carbon dioxide (CO2). Usually, decarboxylation refers to a reaction of carboxylic acids, removing a carbon atom from a carbon chain. The reverse process, which is t ...
of mellitic acid gives benzene.
Uses
Benzene is used mainly as an intermediate to make other chemicals, above allethylbenzene
Ethylbenzene is an organic compound with the formula . It is a highly flammable, colorless liquid with an odor similar to that of gasoline. This monocyclic aromatic hydrocarbon is important in the petrochemical industry as an reaction intermediat ...
(and other alkylbenzenes
The alkylbenzenes are derivatives of benzene, in which one or more hydrogen atoms are replaced by alkyl groups of different sizes. They are a subset of the aromatic hydrocarbons. The simplest member is toluene, in which a hydrogen atom of the benze ...
), cumene, cyclohexane
Cyclohexane is a cycloalkane with the molecular formula . Cyclohexane is non-polar. Cyclohexane is a colorless, flammable liquid with a distinctive detergent-like odor, reminiscent of cleaning products (in which it is sometimes used). Cyclohexan ...
, and nitrobenzene
Nitrobenzene is an organic compound with the chemical formula C6H5 NO2. It is a water-insoluble pale yellow oil with an almond-like odor. It freezes to give greenish-yellow crystals. It is produced on a large scale from benzene as a precursor t ...
. In 1988 it was reported that two-thirds of all chemicals on the American Chemical Society
The American Chemical Society (ACS) is a scientific society based in the United States that supports scientific inquiry in the field of chemistry. Founded in 1876 at New York University, the ACS currently has more than 155,000 members at all d ...
's lists contained at least one benzene ring. More than half of the entire benzene production is processed into ethylbenzene, a precursor to styrene, which is used to make polymers and plastics like polystyrene
Polystyrene (PS) is a synthetic polymer made from monomers of the aromatic hydrocarbon styrene. Polystyrene can be solid or foamed. General-purpose polystyrene is clear, hard, and brittle. It is an inexpensive resin per unit weight. It is a ...
. Some 20% of the benzene production is used to manufacture cumene, which is needed to produce phenol
Phenol (also called carbolic acid) is an aromatic organic compound with the molecular formula . It is a white crystalline solid that is volatile. The molecule consists of a phenyl group () bonded to a hydroxy group (). Mildly acidic, it req ...
and acetone for resins and adhesives. Cyclohexane
Cyclohexane is a cycloalkane with the molecular formula . Cyclohexane is non-polar. Cyclohexane is a colorless, flammable liquid with a distinctive detergent-like odor, reminiscent of cleaning products (in which it is sometimes used). Cyclohexan ...
consumes around 10% of the world's benzene production; it is primarily used in the manufacture of nylon fibers, which are processed into textiles and engineering plastics. Smaller amounts of benzene are used to make some types of rubber
Rubber, also called India rubber, latex, Amazonian rubber, ''caucho'', or ''caoutchouc'', as initially produced, consists of polymers of the organic compound isoprene, with minor impurities of other organic compounds. Thailand, Malaysia, and ...
s, lubricant
A lubricant (sometimes shortened to lube) is a substance that helps to reduce friction between surfaces in mutual contact, which ultimately reduces the heat generated when the surfaces move. It may also have the function of transmitting forces, t ...
s, dye
A dye is a colored substance that chemically bonds to the substrate to which it is being applied. This distinguishes dyes from pigments which do not chemically bind to the material they color. Dye is generally applied in an aqueous solution an ...
s, detergent
A detergent is a surfactant or a mixture of surfactants with cleansing properties when in dilute solutions. There are a large variety of detergents, a common family being the alkylbenzene sulfonates, which are soap-like compounds that are more ...
s, drug
A drug is any chemical substance that causes a change in an organism's physiology or psychology when consumed. Drugs are typically distinguished from food and substances that provide nutritional support. Consumption of drugs can be via insuffla ...
s, explosive
An explosive (or explosive material) is a reactive substance that contains a great amount of potential energy that can produce an explosion if released suddenly, usually accompanied by the production of light, heat, sound, and pressure. An expl ...
s, and pesticide
Pesticides are substances that are meant to control pests. This includes herbicide, insecticide, nematicide, molluscicide, piscicide, avicide, rodenticide, bactericide, insect repellent, animal repellent, microbicide, fungicide, and lampri ...
s. In 2013, the biggest consumer country of benzene was China, followed by the USA. Benzene production is currently expanding in the Middle East and in Africa, whereas production capacities in Western Europe and North America are stagnating.
Toluene
Toluene (), also known as toluol (), is a substituted aromatic hydrocarbon. It is a colorless, water-insoluble liquid with the smell associated with paint thinners. It is a mono-substituted benzene derivative, consisting of a methyl group (CH3) at ...
is now often used as a substitute for benzene, for instance as a fuel additive. The solvent-properties of the two are similar, but toluene is less toxic and has a wider liquid range. Toluene is also processed into benzene.
Benzene
Benzene is an organic chemical compound with the molecular formula C6H6. The benzene molecule is composed of six carbon atoms joined in a planar ring with one hydrogen atom attached to each. Because it contains only carbon and hydrogen atoms, ...
rect 665 60 1062 207 Ethylbenzene
Ethylbenzene is an organic compound with the formula . It is a highly flammable, colorless liquid with an odor similar to that of gasoline. This monocyclic aromatic hydrocarbon is important in the petrochemical industry as an reaction intermediat ...
rect 665 426 1062 579 Cumene
rect 665 795 1062 942 Cyclohexane
Cyclohexane is a cycloalkane with the molecular formula . Cyclohexane is non-polar. Cyclohexane is a colorless, flammable liquid with a distinctive detergent-like odor, reminiscent of cleaning products (in which it is sometimes used). Cyclohexan ...
rect 665 1161 1062 1317 Aniline
Aniline is an organic compound with the formula C6 H5 NH2. Consisting of a phenyl group attached to an amino group, aniline is the simplest aromatic amine
In organic chemistry, an aromatic amine is an organic compound consisting of an aroma ...
rect 665 1533 1062 1686 Chlorobenzene
rect 1215 345 1614 495 Acetone
Acetone (2-propanone or dimethyl ketone), is an organic compound with the formula . It is the simplest and smallest ketone (). It is a colorless, highly volatile and flammable liquid with a characteristic pungent odour.
Acetone is miscib ...
rect 1215 636 1614 783 Phenol
Phenol (also called carbolic acid) is an aromatic organic compound with the molecular formula . It is a white crystalline solid that is volatile. The molecule consists of a phenyl group () bonded to a hydroxy group (). Mildly acidic, it req ...
rect 1764 57 2163 210 Styrene
rect 1764 432 2163 585 Bisphenol A
Bisphenol A (BPA) is a chemical compound primarily used in the manufacturing of various plastics. It is a colourless solid which is soluble in most common organic solvents, but has very poor solubility in water. BPA is produced on an industrial s ...
rect 1764 1083 2163 1233 Adipic acid
Adipic acid or hexanedioic acid is the organic compound with the formula (CH2)4(COOH)2. From an industrial perspective, it is the most important dicarboxylic acid: about 2.5 billion kilograms of this white crystalline powder are produced annually, ...
rect 1764 1332 2163 1482 Caprolactam
Caprolactam (CPL) is an organic compound with the formula (CH2)5C(O)NH. This colourless solid is a lactam (a cyclic amide) of caproic acid. Global demand for this compound is approximately five million tons per year, and the vast majority is used ...
rect 2313 57 2712 207 Polystyrene
Polystyrene (PS) is a synthetic polymer made from monomers of the aromatic hydrocarbon styrene. Polystyrene can be solid or foamed. General-purpose polystyrene is clear, hard, and brittle. It is an inexpensive resin per unit weight. It is a ...
rect 2313 315 2712 462 Polycarbonate
Polycarbonates (PC) are a group of thermoplastic polymers containing carbonate groups in their chemical structures. Polycarbonates used in engineering are strong, tough materials, and some grades are optically transparent. They are easily work ...
rect 2313 570 2712 717 Epoxy resin
Epoxy is the family of basic components or cured end products of epoxy resins. Epoxy resins, also known as polyepoxides, are a class of reactive prepolymers and polymers which contain epoxide groups. The epoxide functional group is also coll ...
rect 2313 822 2712 975 Phenolic resin
rect 2313 1083 2712 1233 Nylon 6-6
Nylon 66 (loosely written nylon 6-6, nylon 6/6, nylon 6,6, or nylon 6:6) is a type of polyamide or nylon. It, and nylon 6, are the two most common for textile and plastic industries. Nylon 66 is made of two monomers each containing 6 carbon atoms, ...
rect 2313 1335 2712 1485 Nylon 6
Nylon 6 or polycaprolactam is a polymer, in particular semicrystalline polyamide. Unlike most other nylons, nylon 6 is not a condensation polymer, but instead is formed by ring-opening polymerization; this makes it a special case in the comparis ...
desc bottom-left
Component of gasoline
As agasoline
Gasoline (; ) or petrol (; ) (see ) is a transparent, petroleum-derived flammable liquid that is used primarily as a fuel in most spark-ignited internal combustion engines (also known as petrol engines). It consists mostly of organic co ...
(petrol) additive, benzene increases the octane rating
An octane rating, or octane number, is a standard measure of a fuel's ability to withstand compression in an internal combustion engine without detonating. The higher the octane number, the more compression the fuel can withstand before detonating ...
and reduces knocking. As a consequence, gasoline often contained several percent benzene before the 1950s, when tetraethyl lead replaced it as the most widely used antiknock additive. With the global phaseout of leaded gasoline, benzene has made a comeback as a gasoline additive in some nations. In the United States
The United States of America (U.S.A. or USA), commonly known as the United States (U.S. or US) or America, is a country primarily located in North America. It consists of 50 states, a federal district, five major unincorporated territorie ...
, concern over its negative health effects and the possibility of benzene entering the groundwater
Groundwater is the water present beneath Earth's surface in rock and soil pore spaces and in the fractures of rock formations. About 30 percent of all readily available freshwater in the world is groundwater. A unit of rock or an unconsolidate ...
has led to stringent regulation of gasoline's benzene content, with limits typically around 1%. European petrol specifications now contain the same 1% limit on benzene content. The United States Environmental Protection Agency
The Environmental Protection Agency (EPA) is an independent executive agency of the United States federal government tasked with environmental protection matters. President Richard Nixon proposed the establishment of EPA on July 9, 1970; it be ...
introduced new regulations in 2011 that lowered the benzene content in gasoline to 0.62%.
In many European languages, the word for petroleum or gasoline is an exact cognate of "benzene".
Reactions
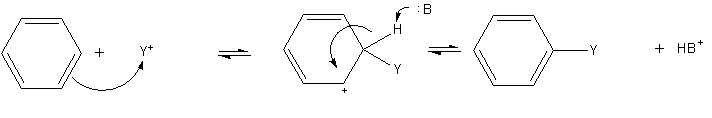
The most common reactions of benzene involve substitution of a proton by other groups. Electrophilic aromatic substitution is a general method of derivatizing benzene. Benzene is sufficiently nucleophilic that it undergoes substitution byacylium
In chemistry, an acyl group is a moiety derived by the removal of one or more hydroxyl groups from an oxoacid, including inorganic acids. It contains a double-bonded oxygen atom and an alkyl group (). In organic chemistry, the acyl group (IUPAC n ...
ions and alkyl carbocation
A carbocation is an ion with a positively charged carbon atom. Among the simplest examples are the methenium , methanium and vinyl cations. Occasionally, carbocations that bear more than one positively charged carbon atom are also encountere ...
s to give substituted derivatives.
: The most widely practiced example of this reaction is the
The most widely practiced example of this reaction is the ethylation
In organic chemistry, an ethyl group (abbr. Et) is an alkyl substituent with the formula , derived from ethane (). ''Ethyl'' is used in the International Union of Pure and Applied Chemistry's nomenclature of organic chemistry for a saturated t ...
of benzene.
:: Approximately 24,700,000 tons were produced in 1999. Highly instructive but of far less industrial significance is the Friedel-Crafts alkylation of benzene (and many other aromatic rings) using an alkyl halide in the presence of a strong Lewis acid catalyst. Similarly, the Friedel-Crafts acylation is a related example of electrophilic aromatic substitution. The reaction involves the acylation of benzene (or many other aromatic rings) with an
Approximately 24,700,000 tons were produced in 1999. Highly instructive but of far less industrial significance is the Friedel-Crafts alkylation of benzene (and many other aromatic rings) using an alkyl halide in the presence of a strong Lewis acid catalyst. Similarly, the Friedel-Crafts acylation is a related example of electrophilic aromatic substitution. The reaction involves the acylation of benzene (or many other aromatic rings) with an acyl chloride
In organic chemistry, an acyl chloride (or acid chloride) is an organic compound with the functional group . Their formula is usually written , where R is a side chain. They are reactive derivatives of carboxylic acids (). A specific example o ...
using a strong Lewis acid
A Lewis acid (named for the American physical chemist Gilbert N. Lewis) is a chemical species that contains an empty orbital which is capable of accepting an electron pair from a Lewis base to form a Lewis adduct. A Lewis base, then, is any sp ...
catalyst
Catalysis () is the process of increasing the rate of a chemical reaction by adding a substance known as a catalyst (). Catalysts are not consumed in the reaction and remain unchanged after it. If the reaction is rapid and the catalyst recyc ...
such as aluminium chloride or Iron(III) chloride
Iron(III) chloride is the inorganic compound with the formula . Also called ferric chloride, it is a common compound of iron in the +3 oxidation state. The anhydrous compound is a crystalline solid with a melting point of 307.6 °C. The col ...
.
Sulfonation, chlorination, nitration
Using electrophilic aromatic substitution, many functional groups are introduced onto the benzene framework. Sulfonation of benzene involves the use ofoleum
Oleum (Latin ''oleum'', meaning oil), or fuming sulfuric acid, is a term referring to solutions of various compositions of sulfur trioxide in sulfuric acid, or sometimes more specifically to disulfuric acid (also known as pyrosulfuric acid). Ole ...
, a mixture of sulfuric acid with sulfur trioxide
Sulfur trioxide (alternative spelling sulphur trioxide, also known as ''nisso sulfan'') is the chemical compound with the formula SO3. It has been described as "unquestionably the most important economically" sulfur oxide. It is prepared on an ind ...
. Sulfonated benzene derivatives are useful detergent
A detergent is a surfactant or a mixture of surfactants with cleansing properties when in dilute solutions. There are a large variety of detergents, a common family being the alkylbenzene sulfonates, which are soap-like compounds that are more ...
s. In nitration, benzene reacts with nitronium ions (NO2+), which is a strong electrophile produced by combining sulfuric and nitric acids. Nitrobenzene
Nitrobenzene is an organic compound with the chemical formula C6H5 NO2. It is a water-insoluble pale yellow oil with an almond-like odor. It freezes to give greenish-yellow crystals. It is produced on a large scale from benzene as a precursor t ...
is the precursor to aniline
Aniline is an organic compound with the formula C6 H5 NH2. Consisting of a phenyl group attached to an amino group, aniline is the simplest aromatic amine
In organic chemistry, an aromatic amine is an organic compound consisting of an aroma ...
. Chlorination is achieved with chlorine to give chlorobenzene in the presence of a Lewis acid catalyst such as aluminium tri-chloride.
Hydrogenation
Viahydrogenation
Hydrogenation is a chemical reaction between molecular hydrogen (H2) and another compound or element, usually in the presence of a Catalysis, catalyst such as nickel, palladium or platinum. The process is commonly employed to redox, reduce or S ...
, benzene and its derivatives convert to cyclohexane
Cyclohexane is a cycloalkane with the molecular formula . Cyclohexane is non-polar. Cyclohexane is a colorless, flammable liquid with a distinctive detergent-like odor, reminiscent of cleaning products (in which it is sometimes used). Cyclohexan ...
and derivatives. This reaction is achieved by the use of high pressures of hydrogen
Hydrogen is the chemical element with the symbol H and atomic number 1. Hydrogen is the lightest element. At standard conditions hydrogen is a gas of diatomic molecules having the formula . It is colorless, odorless, tasteless, non-toxic, an ...
in the presence of heterogeneous catalyst
In chemistry, heterogeneous catalysis is catalysis where the phase of catalysts differs from that of the reactants or products. The process contrasts with homogeneous catalysis where the reactants, products and catalyst exist in the same phase. Ph ...
s, such as finely divided nickel
Nickel is a chemical element with symbol Ni and atomic number 28. It is a silvery-white lustrous metal with a slight golden tinge. Nickel is a hard and ductile transition metal. Pure nickel is chemically reactive but large pieces are slow to ...
. Whereas alkenes can be hydrogenated near room temperatures, benzene and related compounds are more reluctant substrates, requiring temperatures >100 °C. This reaction is practiced on a large scale industrially. In the absence of the catalyst, benzene is impervious to hydrogen. Hydrogenation cannot be stopped to give cyclohexene or cyclohexadienes as these are superior substrates. Birch reduction
The Birch reduction is an organic reaction that is used to convert arenes to cyclohexadienes. The reaction is named after the Australian chemist Arthur Birch and involves the organic reduction of aromatic rings in an amine solvent (traditionally ...
, a non catalytic process, however selectively hydrogenates benzene to the diene.
Metal complexes
Benzene is an excellentligand
In coordination chemistry, a ligand is an ion or molecule (functional group) that binds to a central metal atom to form a coordination complex. The bonding with the metal generally involves formal donation of one or more of the ligand's electr ...
in the organometallic
Organometallic chemistry is the study of organometallic compounds, chemical compounds containing at least one chemical bond between a carbon atom of an organic molecule and a metal, including alkali, alkaline earth, and transition metals, and so ...
chemistry of low-valent metals. Important examples include the sandwich and half-sandwich complexes, respectively, Cr(C6H6)2 and uCl2(C6H6)sub>2.
Health effects
carcinogen
A carcinogen is any substance, radionuclide, or radiation that promotes carcinogenesis (the formation of cancer). This may be due to the ability to damage the genome or to the disruption of cellular metabolic processes. Several radioactive substan ...
, which increases the risk of cancer and other illnesses, and is also a notorious cause of bone marrow failure Bone marrow failure occurs in individuals who produce an insufficient amount of red blood cells, white blood cells or platelets. Red blood cells transport oxygen to be distributed throughout the body's tissue. White blood cells fight off infectio ...
. Substantial quantities of epidemiologic, clinical, and laboratory data link benzene to aplastic anemia, acute leukemia, bone marrow abnormalities and cardiovascular disease. The specific hematologic malignancies that benzene is associated with include: acute myeloid leukemia (AML), aplastic anemia, myelodysplastic syndrome (MDS), acute lymphoblastic leukemia (ALL), and chronic myeloid leukemia (CML).
The American Petroleum Institute
The American Petroleum Institute (API) is the largest U.S. trade association for the oil and natural gas industry. It claims to represent nearly 600 corporations involved in production, refinement, distribution, and many other aspects of the pet ...
(API) stated in 1948 that "it is generally considered that the only absolutely safe concentration for benzene is zero". There is no safe exposure level; even tiny amounts can cause harm. The US Department of Health and Human Services (DHHS) classifies benzene as a human carcinogen
A carcinogen is any substance, radionuclide, or radiation that promotes carcinogenesis (the formation of cancer). This may be due to the ability to damage the genome or to the disruption of cellular metabolic processes. Several radioactive substan ...
. Long-term exposure to excessive levels of benzene in the air causes leukemia
Leukemia ( also spelled leukaemia and pronounced ) is a group of blood cancers that usually begin in the bone marrow and result in high numbers of abnormal blood cells. These blood cells are not fully developed and are called ''blasts'' or ' ...
, a potentially fatal cancer
Cancer is a group of diseases involving abnormal cell growth with the potential to invade or spread to other parts of the body. These contrast with benign tumors, which do not spread. Possible signs and symptoms include a lump, abnormal b ...
of the blood-forming organs. In particular, acute myeloid leukemia or acute nonlymphocytic leukemia (AML & ANLL) is caused by benzene. IARC rated benzene as "known to be carcinogenic to humans" (Group 1).
As benzene is ubiquitous in gasoline and hydrocarbon fuels that are in use everywhere, human exposure to benzene is a global health problem. Benzene targets the liver, kidney, lung, heart and brain and can cause DNA strand breaks and chromosomal
A chromosome is a long DNA molecule with part or all of the genetic material of an organism. In most chromosomes the very long thin DNA fibers are coated with packaging proteins; in eukaryotic cells the most important of these proteins are ...
damage. Benzene causes cancer
Cancer is a group of diseases involving abnormal cell growth with the potential to invade or spread to other parts of the body. These contrast with benign tumors, which do not spread. Possible signs and symptoms include a lump, abnormal b ...
in animals including humans. Benzene has been shown to cause cancer in both sexes of multiple species of laboratory animals exposed via various routes.
Exposure to benzene
According to theAgency for Toxic Substances and Disease Registry
The Agency for Toxic Substances and Disease Registry (ATSDR) is a federal public health agency within the United States Department of Health and Human Services. The agency focuses on minimizing human health risks associated with exposure to haza ...
(ATSDR) (2007), benzene is both a synthetically-made and naturally occurring chemical from processes that include: volcanic eruptions, wild fires, synthesis of chemicals such as phenol
Phenol (also called carbolic acid) is an aromatic organic compound with the molecular formula . It is a white crystalline solid that is volatile. The molecule consists of a phenyl group () bonded to a hydroxy group (). Mildly acidic, it req ...
, production of synthetic fiber
Synthetic fibers or synthetic fibres (in British English; see spelling differences) are fibers made by humans through chemical synthesis, as opposed to natural fibers that are directly derived from living organisms, such as plants (like cotton) ...
s, and fabrication of rubber
Rubber, also called India rubber, latex, Amazonian rubber, ''caucho'', or ''caoutchouc'', as initially produced, consists of polymers of the organic compound isoprene, with minor impurities of other organic compounds. Thailand, Malaysia, and ...
s, lubricant
A lubricant (sometimes shortened to lube) is a substance that helps to reduce friction between surfaces in mutual contact, which ultimately reduces the heat generated when the surfaces move. It may also have the function of transmitting forces, t ...
s, pesticide
Pesticides are substances that are meant to control pests. This includes herbicide, insecticide, nematicide, molluscicide, piscicide, avicide, rodenticide, bactericide, insect repellent, animal repellent, microbicide, fungicide, and lampri ...
s, medications, and dye
A dye is a colored substance that chemically bonds to the substrate to which it is being applied. This distinguishes dyes from pigments which do not chemically bind to the material they color. Dye is generally applied in an aqueous solution an ...
s. The major sources of benzene exposure are tobacco
Tobacco is the common name of several plants in the genus '' Nicotiana'' of the family Solanaceae, and the general term for any product prepared from the cured leaves of these plants. More than 70 species of tobacco are known, but the ...
smoke, automobile service stations, exhaust from motor vehicles, and industrial emissions; however, ingestion and dermal absorption of benzene can also occur through contact with contaminated water. Benzene is hepatically metabolized and excreted in the urine
Urine is a liquid by-product of metabolism in humans and in many other animals. Urine flows from the kidneys through the ureters to the urinary bladder. Urination results in urine being excretion, excreted from the body through the urethra.
Cel ...
. Measurement of air and water levels of benzene is accomplished through collection via activated charcoal
"Activated" is a song by English singer Cher Lloyd. It was released on 22 July 2016 through Vixen Records. The song was made available to stream exclusively on ''Rolling Stone'' a day before to release (on 21 July 2016).
Background
In an interv ...
tubes, which are then analyzed with a gas chromatograph
Gas chromatography (GC) is a common type of chromatography used in analytical chemistry for separating and analyzing compounds that can be vaporized without decomposition. Typical uses of GC include testing the purity of a particular substance, ...
. The measurement of benzene in humans can be accomplished via urine
Urine is a liquid by-product of metabolism in humans and in many other animals. Urine flows from the kidneys through the ureters to the urinary bladder. Urination results in urine being excretion, excreted from the body through the urethra.
Cel ...
, blood
Blood is a body fluid in the circulatory system of humans and other vertebrates that delivers necessary substances such as nutrients and oxygen to the cells, and transports metabolic waste products away from those same cells. Blood in the c ...
, and breath test
A breath test is a type of test performed on air generated from the act of exhalation.
Types include:
*Breathalyzer – by far the most common usage of this term relates to the legal breath test to determine if a person is driving under the inf ...
s; however, all of these have their limitations because benzene is rapidly metabolized in the human body.
Exposure to benzene may lead progressively to aplastic anemia
Anemia or anaemia (British English) is a blood disorder in which the blood has a reduced ability to carry oxygen due to a lower than normal number of red blood cells, or a reduction in the amount of hemoglobin. When anemia comes on slowly, th ...
, leukaemia
Leukemia ( also spelled leukaemia and pronounced ) is a group of blood cancers that usually begin in the bone marrow and result in high numbers of abnormal blood cells. These blood cells are not fully developed and are called ''blasts'' or ' ...
, and multiple myeloma
Multiple myeloma (MM), also known as plasma cell myeloma and simply myeloma, is a cancer of plasma cells, a type of white blood cell that normally produces antibodies. Often, no symptoms are noticed initially. As it progresses, bone pain, an ...
.
OSHA
OSHA or Osha may refer to:
Work
* Occupational Safety and Health Administration, a federal agency of the United States that regulates workplace safety and health
* Occupational Safety and Health Act (United States) of 1970, a federal law in the Un ...
regulates levels of benzene in the workplace. The maximum allowable amount of benzene in workroom air during an 8-hour workday, 40-hour workweek is 1 ppm. As benzene can cause cancer
Cancer is a group of diseases involving abnormal cell growth with the potential to invade or spread to other parts of the body. These contrast with benign tumors, which do not spread. Possible signs and symptoms include a lump, abnormal b ...
, NIOSH
The National Institute for Occupational Safety and Health (NIOSH, ) is the United States federal agency responsible for conducting research and making recommendations for the prevention of work-related injury and illness. NIOSH is part of the C ...
recommends that all workers wear special breathing equipment when they are likely to be exposed to benzene at levels exceeding the recommended (8-hour) exposure limit of 0.1 ppm.
Benzene exposure limits
TheUnited States Environmental Protection Agency
The Environmental Protection Agency (EPA) is an independent executive agency of the United States federal government tasked with environmental protection matters. President Richard Nixon proposed the establishment of EPA on July 9, 1970; it be ...
has set a maximum contaminant level for benzene in drinking water
Drinking water is water that is used in drink or food preparation; potable water is water that is safe to be used as drinking water. The amount of drinking water required to maintain good health varies, and depends on physical activity level, a ...
at 0.0005 mg/L (5 ppb), as promulgated via the U.S. National Primary Drinking Water Regulations. This regulation is based on preventing benzene leukemogenesis
Leukemia ( also spelled leukaemia and pronounced ) is a group of blood cancers that usually begin in the bone marrow and result in high numbers of abnormal blood cells. These blood cells are not fully developed and are called ''blasts'' or '' ...
. The maximum contaminant level goal ( MCLG), a nonenforceable health goal that would allow an adequate margin of safety for the prevention of adverse effects, is zero benzene concentration in drinking water. The EPA requires that spills or accidental releases into the environment of 10 pounds (4.5 kg) or more of benzene be reported.
The U.S. Occupational Safety and Health Administration
The Occupational Safety and Health Administration'' (OSHA ) is a large regulatory agency of the United States Department of Labor that originally had federal visitorial powers to inspect and examine workplaces. Congress established the agenc ...
(OSHA) has set a permissible exposure limit of 1 part of benzene per million parts of air (1 ppm) in the workplace during an 8-hour workday, 40-hour workweek. The short term exposure limit for airborne benzene is 5 ppm for 15 minutes. These legal limits were based on studies demonstrating compelling evidence of health risk to workers exposed to benzene. The risk from exposure to 1 ppm for a working lifetime has been estimated as 5 excess leukemia deaths per 1,000 employees exposed. (This estimate assumes no threshold for benzene's carcinogenic effects.) OSHA has also established an action level of 0.5 ppm to encourage even lower exposures in the workplace.
The U.S. National Institute for Occupational Safety and Health (NIOSH) revised the Immediately Dangerous to Life and Health
The term immediately dangerous to life or health (IDLH) is defined by the US National Institute for Occupational Safety and Health (NIOSH) as exposure to airborne contaminants that is "likely to cause death or immediate or delayed permanent advers ...
(IDLH) concentration for benzene to 500 ppm. The current NIOSH definition for an IDLH condition, as given in the NIOSH Respirator Selection Logic, is one that poses a threat of exposure to airborne contaminants when that exposure is likely to cause death or immediate or delayed permanent adverse health effects or prevent escape from such an environment. The purpose of establishing an IDLH value is (1) to ensure that the worker can escape from a given contaminated environment in the event of failure of the respiratory protection equipment and (2) is considered a maximum level above which only a highly reliable breathing apparatus providing maximum worker protection is permitted. publication No. 2005-100. In September 1995, NIOSH issued a new policy for developing recommended exposure limits (RELs) for substances, including carcinogens. As benzene can cause cancer, NIOSH recommends that all workers wear special breathing equipment when they are likely to be exposed to benzene at levels exceeding the REL (10-hour) of 0.1 ppm. The NIOSH short-term exposure limit (STEL – 15 min) is 1 ppm.
American Conference of Governmental Industrial Hygienists (ACGIH) adopted Threshold Limit Values (TLVs) for benzene at 0.5 ppm TWA and 2.5 ppm STEL.
Toxicology
Biomarkers of exposure
Several tests can determine exposure to benzene. Benzene itself can be measured in breath, blood or urine, but such testing is usually limited to the first 24 hours post-exposure due to the relatively rapid removal of the chemical by exhalation or biotransformation. Most people in developed countries have measureable baseline levels of benzene and other aromatic petroleum hydrocarbons in their blood. In the body, benzene is enzymatically converted to a series of oxidation products including muconic acid, phenylmercapturic acid,phenol
Phenol (also called carbolic acid) is an aromatic organic compound with the molecular formula . It is a white crystalline solid that is volatile. The molecule consists of a phenyl group () bonded to a hydroxy group (). Mildly acidic, it req ...
, catechol, hydroquinone
Hydroquinone, also known as benzene-1,4-diol or quinol, is an aromatic organic compound that is a type of phenol, a derivative of benzene, having the chemical formula C6H4(OH)2. It has two hydroxyl groups bonded to a benzene ring in a ''para'' ...
and 1,2,4-trihydroxybenzene. Most of these metabolites have some value as biomarkers of human exposure, since they accumulate in the urine in proportion to the extent and duration of exposure, and they may still be present for some days after exposure has ceased. The current ACGIH biological exposure limits for occupational exposure are 500 μg/g creatinine for muconic acid and 25 μg/g creatinine for phenylmercapturic acid in an end-of-shift urine specimen.
Biotransformations
Even if it is not a common substrate for metabolism, benzene can be oxidized by bothbacteria
Bacteria (; singular: bacterium) are ubiquitous, mostly free-living organisms often consisting of one biological cell. They constitute a large domain of prokaryotic microorganisms. Typically a few micrometres in length, bacteria were among ...
and eukaryote
Eukaryotes () are organisms whose cells have a nucleus. All animals, plants, fungi, and many unicellular organisms, are Eukaryotes. They belong to the group of organisms Eukaryota or Eukarya, which is one of the three domains of life. Bacte ...
s. In bacteria, dioxygenase
Dioxygenases are oxidoreductase enzymes. Aerobic life, from simple single-celled bacteria species to complex eukaryotic organisms, has evolved to depend on the oxidizing power of dioxygen in various metabolic pathways. From energetic adenosine tri ...
enzyme can add an oxygen
Oxygen is the chemical element with the symbol O and atomic number 8. It is a member of the chalcogen group in the periodic table, a highly reactive nonmetal, and an oxidizing agent that readily forms oxides with most elements as wel ...
to the ring, and the unstable product is immediately reduced (by NADH) to a cyclic diol
A diol is a chemical compound containing two hydroxyl groups ( groups). An aliphatic diol is also called a glycol. This pairing of functional groups is pervasive, and many subcategories have been identified.
The most common industrial diol is e ...
with two double bonds, breaking the aromaticity. Next, the diol is newly reduced by NADH to catechol. The catechol is then metabolized to acetyl CoA and succinyl CoA
Succinyl-coenzyme A, abbreviated as succinyl-CoA () or SucCoA, is a thioester of succinic acid and coenzyme A.
Sources
It is an important intermediate in the citric acid cycle, where it is synthesized from α-ketoglutarate by α-ketoglutarate d ...
, used by organisms mainly in the citric acid cycle
The citric acid cycle (CAC)—also known as the Krebs cycle or the TCA cycle (tricarboxylic acid cycle)—is a series of chemical reactions to release stored energy through the oxidation of acetyl-CoA derived from carbohydrates, fats, and proteins ...
for energy production.
The pathway for the metabolism of benzene is complex and begins in the liver. Several enzymes are involved. These include cytochrome P450
Cytochromes P450 (CYPs) are a Protein superfamily, superfamily of enzymes containing heme as a cofactor (biochemistry), cofactor that functions as monooxygenases. In mammals, these proteins oxidize steroids, fatty acids, and xenobiotics, and are ...
2E1 (CYP2E1), quinine oxidoreductase (NQ01 or DT-diaphorase or NAD(P)H dehydrogenase (quinone 1)
NAD(P)H dehydrogenase uinone1 is an enzyme that in humans is encoded by the ''NQO1'' gene. This protein-coding gene is a member of the NAD(P)H dehydrogenase (quinone) family and encodes a 2-electron reductase (enzyme). This FAD-binding protein fo ...
), GSH, and myeloperoxidase (MPO). CYP2E1 is involved at multiple steps: converting benzene to oxepin (benzene oxide), phenol
Phenol (also called carbolic acid) is an aromatic organic compound with the molecular formula . It is a white crystalline solid that is volatile. The molecule consists of a phenyl group () bonded to a hydroxy group (). Mildly acidic, it req ...
to hydroquinone
Hydroquinone, also known as benzene-1,4-diol or quinol, is an aromatic organic compound that is a type of phenol, a derivative of benzene, having the chemical formula C6H4(OH)2. It has two hydroxyl groups bonded to a benzene ring in a ''para'' ...
, and hydroquinone to both benzenetriol and catechol. Hydroquinone, benzenetriol and catechol are converted to polyphenols. In the bone marrow, MPO converts these polyphenols to benzoquinones. These intermediates and metabolites induce genotoxicity by multiple mechanisms including inhibition of topoisomerase II (which maintains chromosome structure), disruption of microtubules
Microtubules are polymers of tubulin that form part of the cytoskeleton and provide structure and shape to eukaryotic cells. Microtubules can be as long as 50 micrometres, as wide as 23 to 27 nm and have an inner diameter between 11 an ...
(which maintains cellular structure and organization), generation of oxygen free radicals (unstable species) that may lead to point mutations, increasing oxidative stress, inducing DNA strand breaks, and altering DNA methylation
In the chemical sciences, methylation denotes the addition of a methyl group on a substrate, or the substitution of an atom (or group) by a methyl group. Methylation is a form of alkylation, with a methyl group replacing a hydrogen atom. These t ...
(which can affect gene expression). NQ01 and GSH shift metabolism away from toxicity. NQ01 metabolizes benzoquinone toward polyphenols (counteracting the effect of MPO). GSH is involved with the formation of } phenylmercapturic acid.
Genetic polymorphisms in these enzymes may induce loss of function or gain of function. For example, mutations in CYP2E1 increase activity and result in increased generation of toxic metabolites. NQ01 mutations result in loss of function and may result in decreased detoxification. Myeloperoxidase mutations result in loss of function and may result in decreased generation of toxic metabolites. GSH mutations or deletions result in loss of function and result in decreased detoxification. These genes may be targets for genetic screening for susceptibility to benzene toxicity.
Molecular toxicology
The paradigm of toxicological assessment of benzene is shifting towards the domain of molecular toxicology as it allows understanding of fundamental biological mechanisms in a better way. Glutathione seems to play an important role by protecting against benzene-induced DNA breaks and it is being identified as a new biomarker for exposure and effect. Benzene causes chromosomal aberrations in the peripheral blood leukocytes and bone marrow explaining the higher incidence of leukemia and multiple myeloma caused by chronic exposure. These aberrations can be monitored usingfluorescent in situ hybridization
Fluorescence ''in situ'' hybridization (FISH) is a molecular cytogenetic technique that uses fluorescent probes that bind to only particular parts of a nucleic acid sequence with a high degree of sequence complementarity. It was developed ...
(FISH) with DNA probes to assess the effects of benzene along with the hematological tests as markers of hematotoxicity. Benzene metabolism involves enzymes coded for by polymorphic genes. Studies have shown that genotype at these loci may influence susceptibility to the toxic effects of benzene exposure. Individuals carrying variant of NAD(P)H:quinone oxidoreductase 1 (NQO1), microsomal epoxide hydrolase (EPHX) and deletion of the glutathione S-transferase T1 (GSTT1) showed a greater frequency of DNA single-stranded breaks.
Biological oxidation and carcinogenic activity
One way of understanding the carcinogenic effects of benzene is by examining the products of biological oxidation. Pure benzene, for example, oxidizes in the body to produce an epoxide,benzene oxide
Oxepin is an oxygen-containing heterocycle consisting of a seven-membered ring with three double bonds. The parent C6H6O exists as an equilibrium mixture with benzene oxide. The oxepin–benzene oxide equilibrium is affected by the ring substit ...
, which is not excreted readily and can interact with DNA to produce harmful mutations.
Routes of exposure
Inhalation
Outdoor air may contain low levels of benzene from automobile service stations, wood smoke, tobacco smoke, the transfer of gasoline, exhaust from motor vehicles, and industrial emissions. About 50% of the entire nationwide (United States) exposure to benzene results from smoking tobacco or from exposure to tobacco smoke. After smoking 32 cigarettes per day, the smoker would take in about 1.8 milligrams (mg) of benzene. This amount is about 10 times the average daily intake of benzene by nonsmokers. Inhaled benzene is primarily expelled unchanged through exhalation. In a human study 16.4 to 41.6% of retained benzene was eliminated through the lungs within five to seven hours after a two- to three-hour exposure to 47 to 110 ppm and only 0.07 to 0.2% of the remaining benzene was excreted unchanged in the urine. After exposure to 63 to 405 mg/m3 of benzene for 1 to 5 hours, 51 to 87% was excreted in the urine as phenol over a period of 23 to 50 hours. In another human study, 30% of absorbed dermally applied benzene, which is primarily metabolized in the liver, was excreted as phenol in the urine.Exposure from soft drinks
Under specific conditions and in the presence of other chemicalsbenzoic acid
Benzoic acid is a white (or colorless) solid organic compound with the formula , whose structure consists of a benzene ring () with a carboxyl () substituent. It is the simplest aromatic carboxylic acid. The name is derived from gum benzoin, wh ...
(a preservative) and ascorbic acid
Vitamin C (also known as ascorbic acid and ascorbate) is a water-soluble vitamin found in citrus and other fruits and vegetables, also sold as a dietary supplement and as a topical 'serum' ingredient to treat melasma (dark pigment spots) an ...
(Vitamin C) may interact to produce benzene. In March 2006, the official Food Standards Agency
, type = Non-ministerial government department
, nativename =
, nativename_a =
, nativename_r =
, logo = Food Standards Agency.svg
, logo_width =
, logo_caption =
, seal =
, seal_width =
, seal_caption =
, picture =
, picture_width =
, ...
in United Kingdom
The United Kingdom of Great Britain and Northern Ireland, commonly known as the United Kingdom (UK) or Britain, is a country in Europe, off the north-western coast of the continental mainland. It comprises England, Scotland, Wales and North ...
conducted a survey of 150 brands of soft drinks. It found that four contained benzene levels above World Health Organization
The World Health Organization (WHO) is a specialized agency of the United Nations responsible for international public health. The WHO Constitution states its main objective as "the attainment by all peoples of the highest possible level of h ...
limits. The affected batches were removed from sale. Similar problems were reported by the FDA in the United States.
Contamination of water supply
In 2005, the water supply to the city ofHarbin
Harbin (; mnc, , v=Halbin; ) is a sub-provincial city and the provincial capital and the largest city of Heilongjiang province, People's Republic of China, as well as the second largest city by urban population after Shenyang and largest ...
in China with a population of almost nine million people, was cut off because of a major benzene exposure. Benzene leaked into the Songhua River
The Songhua Postal Romanization, or Sunghwa River (also Haixi or Xingal, russian: Сунгари ''Sungari'') is one of the primary List of rivers of China, rivers of China, and the longest tributary of the Amur. It flows about from the Chang ...
, which supplies drinking water to the city, after an explosion at a China National Petroleum Corporation (CNPC) factory in the city of Jilin on 13 November 2005.
When plastic water pipes are subject to high heat, the water may be contaminated with benzene.
Genocide
TheNazis
Nazism ( ; german: Nazismus), the common name in English for National Socialism (german: Nationalsozialismus, ), is the far-right totalitarian political ideology and practices associated with Adolf Hitler and the Nazi Party (NSDAP) in Na ...
used benzene administered via injection
Injection or injected may refer to:
Science and technology
* Injective function, a mathematical function mapping distinct arguments to distinct values
* Injection (medicine), insertion of liquid into the body with a syringe
* Injection, in broadca ...
as one of their many methods for killing
Killing, Killings, or The Killing may refer to:
Arts, entertainment, and media Films
* ''Killing'' (film), a 2018 Japanese film
* ''The Killing'' (film), a 1956 film noir directed by Stanley Kubrick Television
* ''The Killing'' (Danish TV serie ...
.
See also
*BTEX
In the petroleum refining and petrochemical industries, the initialism BTX refers to mixtures of benzene, toluene, and the three xylene isomers, all of which are aromatic hydrocarbons. The xylene isomers are distinguished by the designations ''or ...
* ''Industrial Union Department v. American Petroleum Institute
''Industrial Union Department v. American Petroleum Institute'' (also known as the Benzene Case), 448 U.S. 607 (1980), was a case decided by the Supreme Court of the United States. This case represented a challenge to the OSHA practice of regulat ...
''
* Six-membered aromatic rings with one carbon replaced by another element: borabenzene, silabenzene, germabenzene
Germabenzene (C5H6Ge) is the parent representative of a group of chemical compounds containing in their molecular structure a benzene ring with a carbon atom replaced by a germanium atom. Germabenzene itself has been studied theoretically, and syn ...
, stannabenzene
Stannabenzene (C5H6Sn) is the parent representative of a group of organotin compounds that are related to benzene with a carbon atom replaced by a tin atom. Stannabenzene itself has been studied by computational chemistry, but has not been isolat ...
, pyridine
Pyridine is a basic heterocyclic organic compound with the chemical formula . It is structurally related to benzene, with one methine group replaced by a nitrogen atom. It is a highly flammable, weakly alkaline, water-miscible liquid with a d ...
, phosphorine
Phosphorine (IUPAC name: phosphinine) is a heavier element analog of pyridine, containing a phosphorus atom instead of an aza- moiety. It is also called phosphabenzene and belongs to the phosphaalkene class. It is a colorless liquid that is m ...
, arsabenzene, bismabenzene
Bismabenzene () is the parent representative of a group of organobismuth compounds that are related to benzene with a carbon atom replaced by a bismuth atom. Bismabenzene itself has been synthesised but not isolated because it is too reactive, te ...
, pyrylium
Pyrylium is a cation (positive ion) with formula , consisting of a six-membered ring of five carbon atoms, each with one hydrogen atom, and one positively charged oxygen atom. The bonds in the ring are conjugated as in benzene, giving it an aroma ...
, thiopyrylium
Thiopyrylium is a cation with the chemical formula C5H5S+. It is analogous to the pyrylium cation with the oxygen atom replaced by a sulfur atom.
Thiopyrylium salts are less reactive than the analogous pyrylium salts due to the higher polarizabi ...
, selenopyrylium
Selenopyrylium is an aromatic heterocyclic compound consisting of a six-membered ring with five carbon atoms and a positively charged selenium atom.
Naming and numbering
Formerly it was named selenapyrylium. However, this is misleading as "selena ...
, telluropyrylium
Telluropyrylium is an aromatic heterocyclic compound consisting of a six member ring with five carbon atoms, and a positively charged tellurium atom. Derivatives of telluropyrylium are important in research of infrared dyes.
Naming and numbering ...
Explanatory notes
References
External links
Benzene
at '' The Periodic Table of Videos'' (University of Nottingham)
International Chemical Safety Card 0015
*
Dept. of Health and Human Services: TR-289: Toxicology and Carcinogenesis Studies of Benzene
Video Podcast
of Sir John Cadogan giving a lecture on Benzene since Faraday, in 1991
Substance profile
*
NLM Hazardous Substances Databank – Benzene
{{Authority control Annulenes Aromatic hydrocarbons Aromatic solvents Carcinogens Commodity chemicals GABAA receptor positive allosteric modulators Hazardous air pollutants Hydrocarbon solvents IARC Group 1 carcinogens Immunotoxins Mutagens Occupational safety and health Petrochemicals Simple aromatic rings Six-membered rings Soil contamination Sweet-smelling chemicals Teratogens