|
Benzoquinone
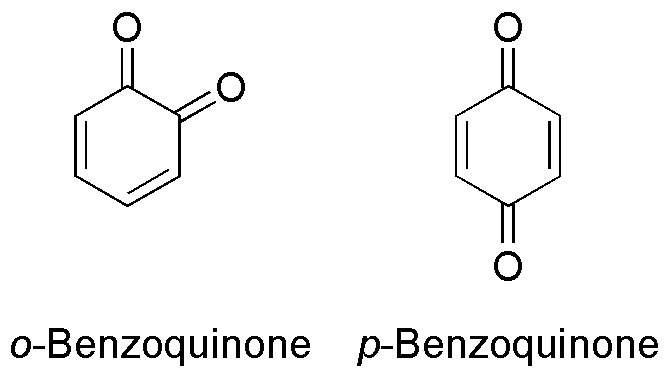
Benzoquinone (C6H4O2) is a quinone with a single benzene ring. There are 2 (out of 3 hypothetical) benzoquinones: * 1,4-Benzoquinone, most commonly, right image (also ''para''-benzoquinone, ''p''-benzoquinone, ''para''-quinone, or just quinone) * 1,2-Benzoquinone, less commonly, left image (also ''ortho''-benzoquinone, ''o''-benzoquinone, ''ortho''-quinone) *1,3-benzoquinone "does not exist, because its structure would be nonplanar and highly strained", though derivatives are known. An alkylated ''p''-benzoquinone has been found in the rhizomes of '' Iris kemaonensis''. See also * Arene substitution pattern Arene substitution patterns are part of organic chemistry IUPAC nomenclature and pinpoint the position of substituents other than hydrogen in relation to each other on an aromatic hydrocarbon. ''Ortho'', ''meta'', and ''para'' substitution * I ... References {{Chemistry index ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
1,4-Benzoquinone
1,4-Benzoquinone, commonly known as ''para''-quinone, is a chemical compound with the formula C6H4O2. In a pure state, it forms bright-yellow crystals with a characteristic irritating odor, resembling that of chlorine, bleach, and hot plastic or formaldehyde. This six-membered ring compound is the oxidized derivative of 1,4-hydroquinone. The molecule is multifunctional: it exhibits properties of a ketone, being able to form oximes; an oxidant, forming the dihydroxy derivative; and an alkene, undergoing addition reactions, especially those typical for α,β-unsaturated ketones. 1,4-Benzoquinone is sensitive toward both strong mineral acids and alkali, which cause condensation and decomposition of the compound. Preparation 1,4-Benzoquinone is prepared industrially by oxidation of hydroquinone, which can be obtained by several routes. One route involves oxidation of diisopropylbenzene and the Hock rearrangement. The net reaction can be represented as follows: :C6H4(CHMe2)2 + ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Quinone
The quinones are a class of organic compounds that are formally "derived from aromatic compounds benzene.html" ;"title="uch as benzene">uch as benzene or naphthalene] by conversion of an even number of –CH= groups into –C(=O)– groups with any necessary rearrangement of double bonds, resulting in "a fully Conjugated system, conjugated cyclic diketone, dione structure". The archetypical member of the class is 1,4-benzoquinone or cyclohexadienedione, often called simply "quinone" (thus the name of the class). Other important examples are 1,2-benzoquinone (''ortho''-quinone), 1,4-naphthoquinone and 9,10-anthraquinone. The name is derived from that of quinic acid (with the suffix "-one" indicating a ketone), since it is one of the compounds obtained upon oxidation of quinic acid. Quinic acid, like quinine is obtained from cinchona bark, called quinaquina in the indigenous languages of Peruvian tribes. Properties Quinones are oxidized derivatives of aromatic compounds and ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
1,2-Benzoquinone
1,2-Benzoquinone, also called ''ortho''-benzoquinone, is an organic compound with formula . It is one of the two isomers of quinone, the other being 1,4-Benzoquinone, 1,4-benzoquinone. It is a red volatile solid that is soluble in water and ethyl ether. It is rarely encountered because of its instability, but it is of fundamental interest as the parent compound of many derivative (chemistry), derivatives which are known. Structure The molecule has C symmetry. X-ray crystallography shows that the double bonds are localized, with alternatingly long and short C-C distances within the ring. The C=O distances of 1.21 Å are characteristic of ketones. Preparation and occurrence 1,2-Benzoquinone is produced on oxidation of catechol exposed to air in aqueous solution or by ortho oxidation of a phenol. It is a precursor to melanin. A strain of the bacterium ''Pseudomonas mendocina'' metabolism, metabolises benzoic acid, yielding 1,2-benzoquinone via catechol. References {{D ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Benzoquinones
Benzoquinone (C6H4O2) is a quinone with a single benzene ring. There are 2 (out of 3 hypothetical) benzoquinones: * 1,4-Benzoquinone, most commonly, right image (also ''para''-benzoquinone, ''p''-benzoquinone, ''para''-quinone, or just quinone) * 1,2-Benzoquinone, less commonly, left image (also ''ortho''-benzoquinone, ''o''-benzoquinone, ''ortho''-quinone) *1,3-benzoquinone "does not exist, because its structure would be nonplanar and highly strained", though derivatives are known. An alkylated ''p''-benzoquinone has been found in the rhizomes of '' Iris kemaonensis''. See also * Arene substitution pattern Arene substitution patterns are part of organic chemistry IUPAC nomenclature and pinpoint the position of substituents other than hydrogen in relation to each other on an aromatic hydrocarbon. ''Ortho'', ''meta'', and ''para'' substitution * I ... References {{Chemistry index ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Iris Kemaonensis
''Iris kemaonensis'', the Kumaon iris, is a plant species in the genus '' Iris'', it is also in the subgenus '' Iris'' and in the section ''Pseudoregelia''. It is a rhizomatous perennial, from Tibetan China, Bhutan, India, Kashmir and Nepal. It has light green or yellowish green leaves, that extend after flowering time. It has a short stem, 1–2 fragrant flowers that are purple, lilac, lilac-purple or pale purple. They also have darker coloured blotches or spots. It is cultivated as an ornamental plant in temperate regions. It is often known as ''Iris kumaonensis'', due to a publishing error. Description In China, there has been some confusion between ''Iris dolichosiphon'' (another ''Pseudoregelia iris'') and ''Iris kemaonensis'', they have similar flower forms, but ''Iris kemaonensis'' flowers are paler then ''Iris dolichosiphon'' but are strongly mottled, as well as a smaller perianth tube. It has short, thick rhizomes, which are gnarled and knobbly.British Iris Society ( ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Benzene Ring
Benzene is an organic chemical compound with the molecular formula C6H6. The benzene molecule is composed of six carbon atoms joined in a planar ring with one hydrogen atom attached to each. Because it contains only carbon and hydrogen atoms, benzene is classed as a hydrocarbon. Benzene is a natural constituent of petroleum and is one of the elementary petrochemicals. Due to the cyclic continuous pi bonds between the carbon atoms, benzene is classed as an aromatic hydrocarbon. Benzene is a colorless and highly flammable liquid with a sweet smell, and is partially responsible for the aroma of gasoline. It is used primarily as a precursor to the manufacture of chemicals with more complex structure, such as ethylbenzene and cumene, of which billions of kilograms are produced annually. Although benzene is a major industrial chemical, it finds limited use in consumer items because of its toxicity. History Discovery The word "''benzene''" derives from "''gum benzoin''" (benzoin re ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Rhizome
In botany and dendrology, a rhizome (; , ) is a modified subterranean plant stem that sends out roots and shoots from its nodes. Rhizomes are also called creeping rootstalks or just rootstalks. Rhizomes develop from axillary buds and grow horizontally. The rhizome also retains the ability to allow new shoots to grow upwards. A rhizome is the main stem of the plant that runs underground horizontally. A stolon is similar to a rhizome, but a stolon sprouts from an existing stem, has long internodes, and generates new shoots at the end, such as in the strawberry plant. In general, rhizomes have short internodes, send out roots from the bottom of the nodes, and generate new upward-growing shoots from the top of the nodes. A stem tuber is a thickened part of a rhizome or stolon that has been enlarged for use as a storage organ. In general, a tuber is high in starch, e.g. the potato, which is a modified stolon. The term "tuber" is often used imprecisely and is sometimes appli ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Arene Substitution Pattern
Arene substitution patterns are part of organic chemistry IUPAC nomenclature and pinpoint the position of substituents other than hydrogen in relation to each other on an aromatic hydrocarbon. ''Ortho'', ''meta'', and ''para'' substitution * In ''ortho''-substitution, two substituents occupy positions next to each other, which may be numbered 1 and 2. In the diagram, these positions are marked R and ''ortho''. * In ''meta''-substitution the substituents occupy positions 1 and 3 (corresponding to R and ''meta'' in the diagram). * In ''para''-substitution, the substituents occupy the opposite ends (positions 1 and 4, corresponding to R and ''para'' in the diagram). The toluidines serve as an example for these three types of substitution. Synthesis Electron donating groups, for example amino, hydroxyl, alkyl, and phenyl groups tend to be ''ortho''/''para''-directors, and electron withdrawing groups such as nitro, nitrile, and ketone groups, tend to be ''meta''-directors. Proper ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |


2.png)
