|
Tetrafluorohydrazine
Tetrafluorohydrazine or perfluorohydrazine, , is a colourless, reactive inorganic gas. It is a fluorinated analog of hydrazine. It is a highly hazardous chemical that explodes in the presence of organic materials. Tetrafluorohydrazine is manufactured from nitrogen trifluoride using an iron catalyst or iron(II) fluoride. It is used in some chemical syntheses, as a precursor or a catalyst. Tetrafluorohydrazine was considered for use as a high-energy liquid oxidizer in some never-flown rocket fuel formulas in 1959. Properties Tetrafluorohydrazine is in equilibrium with its radical monomer nitrogen difluoride. : N2F4 2 NF2• At room temperature N2F4 is mostly associated with only 0.7% in the form of NF2 at 5mm Hg pressure. When the temperature rises to 225 °C, it mostly dissociates with 99% in the form of NF2. molecule dimensions and angles The energy needed to break the N-N bond in N2F4 is 20.8 kcal/mol, with an entropy change of 38.6 eu. For comparison, the dis ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nitrogen Difluoride
Nitrogen difluoride, also known as difluoroamino, is a reactive radical molecule with formula . This small molecule is in equilibrium with its dimer dinitrogen tetrafluoride. : As the temperature increases the proportion of increases. The molecule is unusual in that it has an odd number of electrons, yet is stable enough to study experimentally. Properties The energy needed to break the N–N bond in is , with an entropy change of 38.6 eu. molecule dimensions and angles For comparison, the dissociation energy of the N–N bond is in , in , and in . The enthalpy of formation of (Δ''H''f) is . At room temperature is mostly associated with only 0.7% in the form of at pressure. When the temperature rises to 225 °C, it mostly dissociates with 99% in the form of . In , the N–F bond length is 1.3494 Å and the angle subtended at F–N–F is 103.33°. In the infrared spectrum the N–F bond in has a symmetrical stretching frequency of 1075 cm� ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrazine
Hydrazine is an inorganic compound with the chemical formula . It is a simple pnictogen hydride, and is a colourless flammable liquid with an ammonia-like odour. Hydrazine is highly toxic unless handled in solution as, for example, hydrazine hydrate (). Hydrazine is mainly used as a foaming agent in preparing polymer foams, but applications also include its uses as a precursor to polymerization catalysts, pharmaceuticals, and agrochemicals, as well as a long-term storable propellant for in-space spacecraft propulsion. Additionally, hydrazine is used in various rocket fuels and to prepare the gas precursors used in air bags. Hydrazine is used within both nuclear and conventional electrical power plant steam cycles as an oxygen scavenger to control concentrations of dissolved oxygen in an effort to reduce corrosion. the world hydrazine hydrate market amounted to $350 million. About two million tons of hydrazine hydrate were used in foam blowing agents in 2015. Hydrazines r ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
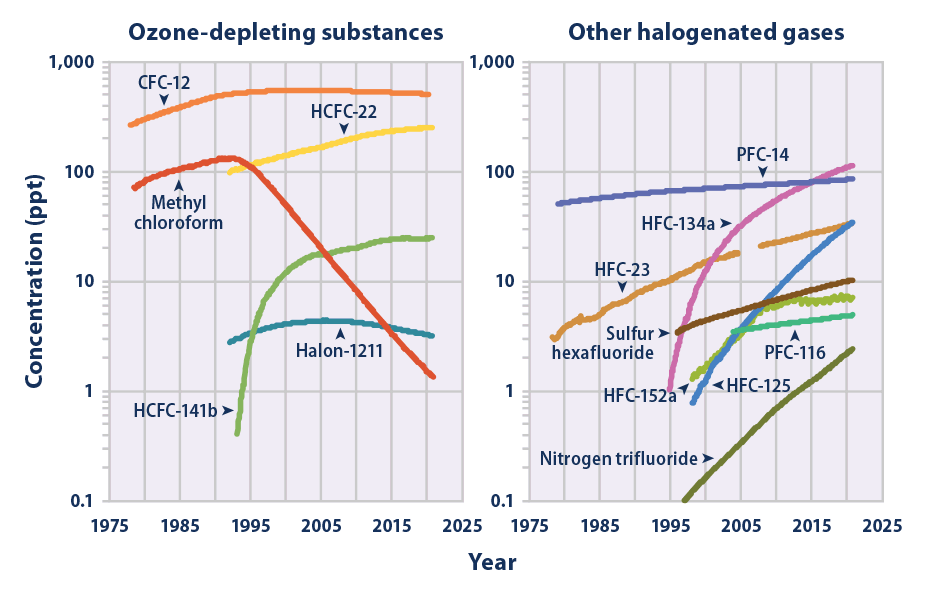
Nitrogen Trifluoride
Nitrogen trifluoride () is an inorganic, colorless, non-flammable, toxic gas with a slightly musty odor. It finds increasing use within the manufacturing of flat-panel displays, photovoltaics, LEDs and other microelectronics. Nitrogen trifluoride is also an extremely strong and long-lived greenhouse gas. Its atmospheric burden exceeded 2 parts per trillion during 2019 and has doubled every five years since the late 20th century. Synthesis and reactivity Nitrogen trifluoride did not exist in significant quantities on Earth prior to its synthesis by humans. It is a rare example of a binary fluoride that can be prepared directly from the elements only at very uncommon conditions, such as an electric discharge. After first attempting the synthesis in 1903, Otto Ruff prepared nitrogen trifluoride by the electrolysis of a molten mixture of ammonium fluoride and hydrogen fluoride. It proved to be far less reactive than the other nitrogen trihalides nitrogen trichloride, nitrogen trib ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nitrogen Fluorides
Nitrogen is the chemical element with the symbol N and atomic number 7. Nitrogen is a nonmetal and the lightest member of group 15 of the periodic table, often called the pnictogens. It is a common element in the universe, estimated at seventh in total abundance in the Milky Way and the Solar System. At standard temperature and pressure, two atoms of the element bond to form N2, a colorless and odorless diatomic gas. N2 forms about 78% of Earth's atmosphere, making it the most abundant uncombined element. Nitrogen occurs in all organisms, primarily in amino acids (and thus proteins), in the nucleic acids ( DNA and RNA) and in the energy transfer molecule adenosine triphosphate. The human body contains about 3% nitrogen by mass, the fourth most abundant element in the body after oxygen, carbon, and hydrogen. The nitrogen cycle describes the movement of the element from the air, into the biosphere and organic compounds, then back into the atmosphere. Many industrially importan ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrazine
Hydrazine is an inorganic compound with the chemical formula . It is a simple pnictogen hydride, and is a colourless flammable liquid with an ammonia-like odour. Hydrazine is highly toxic unless handled in solution as, for example, hydrazine hydrate (). Hydrazine is mainly used as a foaming agent in preparing polymer foams, but applications also include its uses as a precursor to polymerization catalysts, pharmaceuticals, and agrochemicals, as well as a long-term storable propellant for in-space spacecraft propulsion. Additionally, hydrazine is used in various rocket fuels and to prepare the gas precursors used in air bags. Hydrazine is used within both nuclear and conventional electrical power plant steam cycles as an oxygen scavenger to control concentrations of dissolved oxygen in an effort to reduce corrosion. the world hydrazine hydrate market amounted to $350 million. About two million tons of hydrazine hydrate were used in foam blowing agents in 2015. Hydrazines r ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Inorganic Compound
In chemistry, an inorganic compound is typically a chemical compound that lacks carbon–hydrogen bonds, that is, a compound that is not an organic compound. The study of inorganic compounds is a subfield of chemistry known as '' inorganic chemistry''. Inorganic compounds comprise most of the Earth's crust, although the compositions of the deep mantle remain active areas of investigation. Some simple carbon compounds are often considered inorganic. Examples include the allotropes of carbon (graphite, diamond, buckminsterfullerene, etc.), carbon monoxide, carbon dioxide, carbides, and the following salts of inorganic anions: carbonates, cyanides, cyanates, and thiocyanates. Many of these are normal parts of mostly organic systems, including organisms; describing a chemical as inorganic does not necessarily mean that it does not occur within living things. History Friedrich Wöhler's conversion of ammonium cyanate into urea in 1828 is often cited as the starting point of modern ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Rocket Oxidizers
A rocket (from it, rocchetto, , bobbin/spool) is a vehicle that uses jet propulsion to accelerate without using the surrounding air. A rocket engine produces thrust by reaction to exhaust expelled at high speed. Rocket engines work entirely from propellant carried within the vehicle; therefore a rocket can fly in the vacuum of space. Rockets work more efficiently in a vacuum and incur a loss of thrust due to the opposing pressure of the atmosphere. Multistage rockets are capable of attaining escape velocity from Earth and therefore can achieve unlimited maximum altitude. Compared with airbreathing engines, rockets are lightweight and powerful and capable of generating large accelerations. To control their flight, rockets rely on momentum, airfoils, auxiliary reaction engines, gimballed thrust, momentum wheels, deflection of the exhaust stream, propellant flow, spin, or gravity. Rockets for military and recreational uses date back to at least 13th-century China. Significant ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Standard Enthalpy Of Formation
In chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of enthalpy during the formation of 1 mole of the substance from its constituent elements in their reference state, with all substances in their standard states. The standard pressure value is recommended by IUPAC, although prior to 1982 the value 1.00 atm (101.325 kPa) was used. There is no standard temperature. Its symbol is . The superscript Plimsoll on this symbol indicates that the process has occurred under standard conditions at the specified temperature (usually 25 °C or 298.15 K). Standard states are as follows: # For a gas: the hypothetical state it would have assuming it obeyed the ideal gas equation at a pressure of 1 bar # For a gaseous or solid solute present in a diluted ideal solution: the hypothetical state of concentration of the solute of exactly one mole per liter (1 M) at a pressure of 1 bar extrapolated fro ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dinitrogen Dioxide
Dinitrogen dioxide is an inorganic compound having molecular formula . Many structural isomers are possible. The covalent bonding pattern O=N–N=O (a non-cyclic dimer of nitric oxide (NO)) is predicted to be the most stable isomer based on ab initio calculations and is the only one that has been experimentally produced. In the solid form, the molecules have ''C''2v symmetry: the entire structure is planar, with the two oxygen atoms ''cis Cis or cis- may refer to: Places * Cis, Trentino, in Italy * In Poland: ** Cis, Świętokrzyskie Voivodeship, south-central ** Cis, Warmian-Masurian Voivodeship, north Math, science and biology * cis (mathematics) (cis(''θ'')), a trigonome ...'' across the N–N bond. The O–N distance is 1.15 Å, the N–N distance is 2.33 Å, and the O=N–N angle is 95°. References * * {{Oxides Nitrogen oxides Azines (hydrazine derivatives) ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dinitrogen Tetroxide
Dinitrogen tetroxide, commonly referred to as nitrogen tetroxide (NTO), and occasionally (usually among ex-USSR/Russia rocket engineers) as amyl, is the chemical compound N2O4. It is a useful reagent in chemical synthesis. It forms an equilibrium mixture with nitrogen dioxide. Its molar mass is 92.011 g/mol. Dinitrogen tetroxide is a powerful oxidizer that is hypergolic (spontaneously reacts) upon contact with various forms of hydrazine, which has made the pair a common bipropellant for rockets. Structure and properties Dinitrogen tetroxide could be regarded as two nitro groups (-NO2) bonded together. It forms an equilibrium mixture with nitrogen dioxide. The molecule is planar with an N-N bond distance of 1.78Å and N-O distances of 1.19Å. The N-N distance corresponds to a weak bond, since it is significantly longer than the average N-N single bond length of 1.45Å. This exceptionally weak σ bond (amounting to overlapping of the ''sp''2 hybrid orbitals of the two NO2 units) ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Entropy Unit
The entropy unit is a non-S.I. unit of thermodynamic entropy, usually denoted "e.u." and equal to one calorie The calorie is a unit of energy. For historical reasons, two main definitions of "calorie" are in wide use. The large calorie, food calorie, or kilogram calorie was originally defined as the amount of heat needed to raise the temperature of on ... per kelvin per mole, or 4.184 joules per kelvin per mole. Entropy units are primarily used in chemistry to describe enthalpy changes. Sources {{Thermodynamics-stub Units of measurement ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Rocket Fuel
Rocket propellant is the reaction mass of a rocket. This reaction mass is ejected at the highest achievable velocity from a rocket engine to produce thrust. The energy required can either come from the propellants themselves, as with a chemical rocket, or from an external source, as with ion engines. Overview Rockets create thrust by expelling mass rear-ward, at high velocity. The thrust produced can be calculated by multiplying the mass flow rate of the propellants by their exhaust velocity relative to the rocket (specific impulse). A rocket can be thought of as being accelerated by the pressure of the combusting gases against the combustion chamber and nozzle, not by "pushing" against the air behind or below it. Rocket engines perform best in outer space because of the lack of air pressure on the outside of the engine. In space it is also possible to fit a longer nozzle without suffering from flow separation. Most chemical propellants release energy through redox chemistry, m ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |