|
Polarization (corrosion)
In electrochemistry, polarization is a collective term for certain mechanical side-effects (of an electrochemical process) by which isolating barriers develop at the interface between electrode and electrolyte. These side-effects influence the reaction mechanisms, as well as the chemical kinetics of corrosion and metal deposition.. In a reaction we can displace the bonding electrons by attacking reagents. The electronic displacement in turn may be due to certain effects, some of which are permanent (inductive and mesomeric effects), and the others are temporary (electromeric effect). Those effects which are permanently operating in the molecule are known as polarization effects, and those effects which are brought into play by attacking reagent (and as the attacking reagent is removed, the electronic displacement disappears) are known as polarisability effects. The term 'polarization' derives from the early 19th-century discovery that electrolysis causes the elements in an electr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electrochemistry
Electrochemistry is the branch of physical chemistry concerned with the relationship between electrical potential difference, as a measurable and quantitative phenomenon, and identifiable chemical change, with the potential difference as an outcome of a particular chemical change, or vice versa. These reactions involve electrons moving via an electronically-conducting phase (typically an external electrical circuit, but not necessarily, as in electroless plating) between electrodes separated by an ionically conducting and electronically insulating electrolyte (or ionic species in a solution). When a chemical reaction is driven by an electrical potential difference, as in electrolysis, or if a potential difference results from a chemical reaction as in an electric battery or fuel cell, it is called an ''electrochemical'' reaction. Unlike in other chemical reactions, in electrochemical reactions electrons are not transferred directly between atoms, ions, or molecules, but via the af ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Reduction Potential
Redox potential (also known as oxidation / reduction potential, ''ORP'', ''pe'', ''E_'', or E_) is a measure of the tendency of a chemical species to acquire electrons from or lose electrons to an electrode and thereby be reduced or oxidised respectively. Redox potential is expressed in volts (V). Each species has its own intrinsic redox potential; for example, the more positive the reduction potential (reduction potential is more often used due to general formalism in electrochemistry), the greater the species' affinity for electrons and tendency to be reduced. Measurement and interpretation In aqueous solutions, redox potential is a measure of the tendency of the solution to either gain or lose electrons when it is subjected to change by introduction of a new species. A solution with a higher (more positive) reduction potential than the new species will have a tendency to gain electrons from the new species (i.e. to be reduced by oxidizing the new species) and a solution with ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Depolarizer
A depolarizer or depolariser, in electrochemistry, according to an IUPAC definition, is a synonym of electroactive substance, i.e., a substance which changes its oxidation state, or partakes in a formation or breaking of chemical bonds, in a charge-transfer step of an electrochemical reaction. In the battery industry, the term "depolarizer" has been used to denote a substance used in a primary cell to prevent buildup of hydrogen gas bubbles."McGraw-Hill Dictionary of Scientific & Technical Terms", McGraw-Hill, Inc., 2003. A battery depolarizer takes up electrons during discharge of the cell; therefore, it is always an oxidizing agent. The term "depolarizer" can be considered as outdated or misleading, since it is based on the concept of " polarization" which is hardly realistic in many cases. Polarization Under certain conditions for some electrochemical cells, especially if they use an aqueous electrolyte, hydrogen ions can be converted into hydrogen atoms and H2 molecules. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Depolarizer
A depolarizer or depolariser, in electrochemistry, according to an IUPAC definition, is a synonym of electroactive substance, i.e., a substance which changes its oxidation state, or partakes in a formation or breaking of chemical bonds, in a charge-transfer step of an electrochemical reaction. In the battery industry, the term "depolarizer" has been used to denote a substance used in a primary cell to prevent buildup of hydrogen gas bubbles."McGraw-Hill Dictionary of Scientific & Technical Terms", McGraw-Hill, Inc., 2003. A battery depolarizer takes up electrons during discharge of the cell; therefore, it is always an oxidizing agent. The term "depolarizer" can be considered as outdated or misleading, since it is based on the concept of " polarization" which is hardly realistic in many cases. Polarization Under certain conditions for some electrochemical cells, especially if they use an aqueous electrolyte, hydrogen ions can be converted into hydrogen atoms and H2 molecules. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electroplating
Electroplating, also known as electrochemical deposition or electrodeposition, is a process for producing a metal coating on a solid substrate through the reduction of cations of that metal by means of a direct electric current. The part to be coated acts as the cathode (negative electrode) of an electrolytic cell; the electrolyte is a solution of a salt of the metal to be coated; and the anode (positive electrode) is usually either a block of that metal, or of some inert conductive material. The current is provided by an external power supply. Electroplating is widely used in industry and decorative arts to improve the surface qualities of objects—such as resistance to abrasion and corrosion, lubricity, reflectivity, electrical conductivity, or appearance. It is used to build up thickness on undersized or worn-out parts, or to manufacture metal plates with complex shape, a process called electroforming. It is used to deposit copper and other conductors in forming printe ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electropolishing
Electropolishing, also known as electrochemical polishing, anodic polishing, or electrolytic polishing (especially in the metallography field), is an electrochemical process that removes material from a metallic workpiece, reducing the surface roughness by levelling micro-peaks and valleys, improving the surface finish. Electropolishing is often compared to, but distinctly different from, electrochemical machining. It is used to polish, passivate, and deburr metal parts. It is often described as the reverse of electroplating. It may be used in lieu of abrasive fine polishing in microstructural preparation. Mechanism Typically, the work-piece is immersed in a temperature-controlled bath of electrolyte and serves as the anode; it is connected to the positive terminal of a DC power supply, the negative terminal being attached to the cathode. A current passes from the anode, where metal on the surface is oxidised and dissolved in the electrolyte, to the cathode. At the cathode, a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
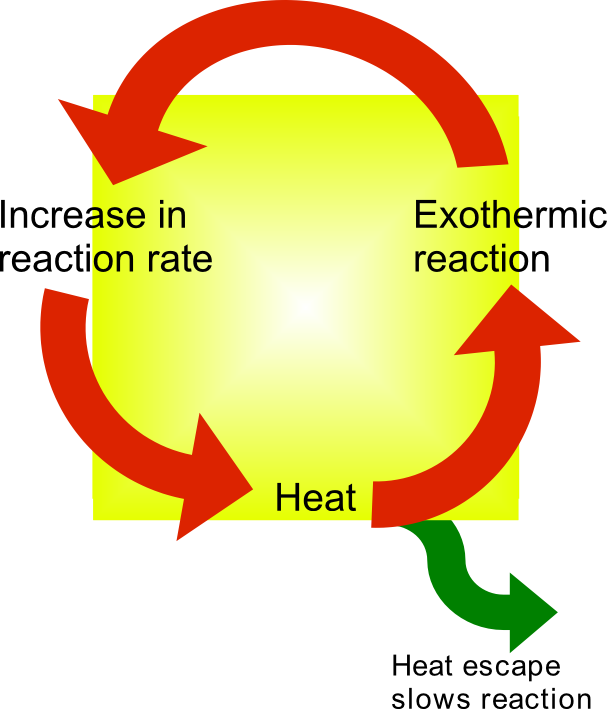
Thermal Runaway
Thermal runaway describes a process that is accelerated by increased temperature, in turn releasing energy that further increases temperature. Thermal runaway occurs in situations where an increase in temperature changes the conditions in a way that causes a further increase in temperature, often leading to a destructive result. It is a kind of uncontrolled positive feedback. In chemistry (and chemical engineering), thermal runaway is associated with strongly exothermic reactions that are accelerated by temperature rise. In electrical engineering, thermal runaway is typically associated with increased current flow and power dissipation. Thermal runaway can occur in civil engineering, notably when the heat released by large amounts of curing concrete is not controlled. In astrophysics, runaway nuclear fusion reactions in stars can lead to nova and several types of supernova explosions, and also occur as a less dramatic event in the normal evolution of solar-mass stars, the " he ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dendrite (metal)
A dendrite in metallurgy is a characteristic tree-like structure of crystals growing as molten metal solidifies, the shape produced by faster growth along energetically favourable crystallographic directions. This dendritic growth has large consequences in regard to material properties. Dendrites form in unary (one-component) systems as well as multi-component systems. The requirement is that the liquid (the molten material) be undercooled, aka supercooled, below the freezing point of the solid. Initially, a spherical solid nucleus grows in the undercooled melt. As the sphere grows, the spherical morphology becomes unstable and its shape becomes perturbed. The solid shape begins to express the preferred growth directions of the crystal. This growth direction may be due to anisotropy in the surface energy of the solid–liquid interface, or to the ease of attachment of atoms to the interface on different crystallographic planes, or both (for an example of the latter, see hopp ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electrochemical Cell
An electrochemical cell is a device capable of either generating electrical energy from chemical reactions or using electrical energy to cause chemical reactions. The electrochemical cells which generate an electric current are called voltaic or galvanic cells and those that generate chemical reactions, via electrolysis for example, are called electrolytic cells. A common example of a galvanic cell is a standard 1.5 volt cell meant for consumer use. A ''battery'' consists of one or more cells, connected in parallel, series or series-and-parallel pattern. Electrolytic cell An electrolytic cell is an electrochemical cell that drives a non-spontaneous redox reaction through the application of electrical energy. They are often used to decompose chemical compounds, in a process called electrolysis—the Greek word lysis means ''to break up''. Important examples of electrolysis are the decomposition of water into hydrogen and oxygen, and bauxite into aluminium and other chemic ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electromotive Force
In electromagnetism and electronics, electromotive force (also electromotance, abbreviated emf, denoted \mathcal or ) is an energy transfer to an electric circuit per unit of electric charge, measured in volts. Devices called electrical ''transducers'' provide an emf by converting other forms of energy into electrical energy. Other electrical equipment also produce an emf, such as batteries, which convert chemical energy, and generators, which convert mechanical energy. This energy conversion is achieved by physical forces applying physical work on electric charges. However, electromotive force itself is not a physical force, and for the current ISO/IEC standards consider the term deprecated, favoring the names source voltage or source tension instead (denoted U_s). An electronic–hydraulic analogy may view emf as the mechanical work done to water by a pump, which results in a pressure difference (analogous to voltage). In electromagnetic induction, emf can be defined ar ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ohm's Law
Ohm's law states that the current through a conductor between two points is directly proportional to the voltage across the two points. Introducing the constant of proportionality, the resistance, one arrives at the usual mathematical equation that describes this relationship: :I = \frac, where is the current through the conductor, ''V'' is the voltage measured ''across'' the conductor and ''R'' is the resistance of the conductor. More specifically, Ohm's law states that the ''R'' in this relation is constant, independent of the current. If the resistance is not constant, the previous equation cannot be called ''Ohm's law'', but it can still be used as a definition of static/DC resistance. Ohm's law is an empirical relation which accurately describes the conductivity of the vast majority of electrically conductive materials over many orders of magnitude of current. However some materials do not obey Ohm's law; these are called non-ohmic. The law was named after t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Concentration Polarization
Concentration polarization is a term used in the scientific fields of electrochemistry and membrane science. In electrochemistry In electrochemistry, concentration polarization denotes the part of the polarization of an electrolytic cell resulting from changes in the electrolyte concentration due to the passage of current through the electrode/solution interface. Here ''polarization'' is understood as the shift of the electrochemical potential difference across the cell from its equilibrium value. When the term is used in this sense, it is equivalent to “ concentration overpotential”. the changes in concentration (emergence of concentration gradients in the solution adjacent to the electrode surface) is the difference in the rate of electrochemical reaction at the electrode and the rate of ion migration in the solution from/to the surface. When a chemical species participating in an electrochemical electrode reaction is in short supply, the concentration of this species at the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |