|
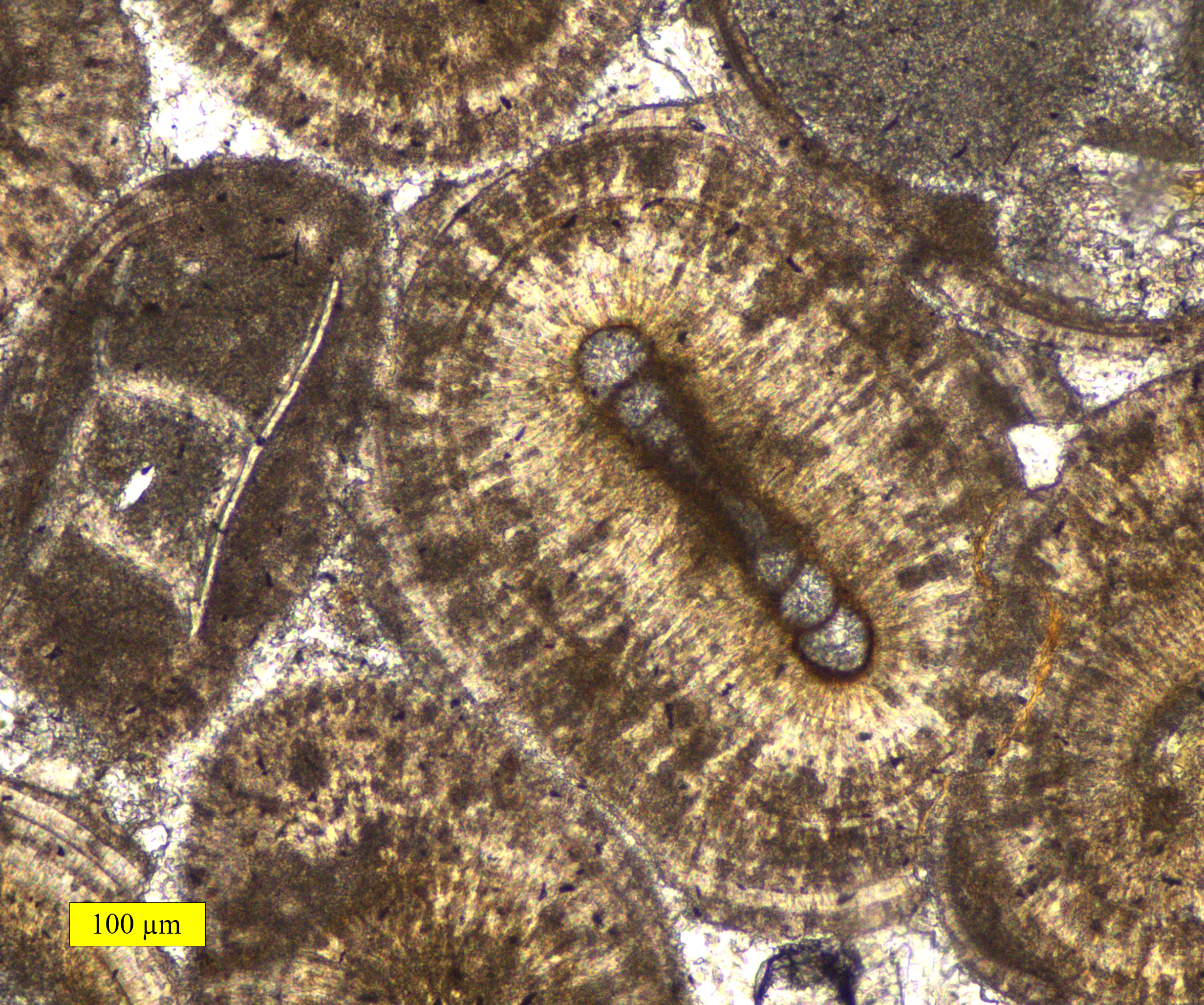
Ooid
Ooids are small (commonly ≤2 mm in diameter), spheroidal, "coated" (layered) sedimentary grains, usually composed of calcium carbonate, but sometimes made up of iron- or phosphate-based minerals. Ooids usually form on the sea floor, most commonly in shallow tropical seas (around the Bahamas, for example, or in the Persian Gulf). After being buried under additional sediment, these ooid grains can be cemented together to form a sedimentary rock called an ''oolite''. Oolites usually consist of calcium carbonate; these belong to the limestone rock family. Pisoids are similar to ooids, but are larger than 2 mm in diameter, often considerably larger, as with the pisoids in the hot springs at Carlsbad (Karlovy Vary) in the Czech Republic. Formation An ooid forms as a series of concentric layers around a nucleus. The layers contain crystals arranged radially, tangentially or randomly. The nucleus can be a shell fragment, quartz grain or any other small fragment. Most mode ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Limestone
Limestone ( calcium carbonate ) is a type of carbonate sedimentary rock which is the main source of the material lime. It is composed mostly of the minerals calcite and aragonite, which are different crystal forms of . Limestone forms when these minerals precipitate out of water containing dissolved calcium. This can take place through both biological and nonbiological processes, though biological processes, such as the accumulation of corals and shells in the sea, have likely been more important for the last 540 million years. Limestone often contains fossils which provide scientists with information on ancient environments and on the evolution of life. About 20% to 25% of sedimentary rock is carbonate rock, and most of this is limestone. The remaining carbonate rock is mostly dolomite, a closely related rock, which contains a high percentage of the mineral dolomite, . ''Magnesian limestone'' is an obsolete and poorly-defined term used variously for dolomite, for lime ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Oolite
Oolite or oölite (''egg stone'') is a sedimentary rock formed from ooids, spherical grains composed of concentric layers. The name derives from the Ancient Greek word for egg (ᾠόν). Strictly, oolites consist of ooids of diameter 0.25–2 millimetres; rocks composed of ooids larger than 2 mm are called pisolites. The term ''oolith'' can refer to oolite or individual ooids. Composition Ooids are most commonly composed of calcium carbonate (calcite or aragonite), but can be composed of phosphate, clays, chert, dolomite or iron minerals, including hematite. Dolomitic and chert ooids are most likely the result of the replacement of the original texture in limestone. Oolitic hematite occurs at Red Mountain near Birmingham, Alabama, along with oolitic limestone. They are usually formed in warm, supersaturated, shallow, highly agitated marine water intertidal environments, though some are formed in inland lakes. The mechanism of formation starts with a small fra ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Oolitic Aragonite Sand
Oolitic aragonite sand is composed of the calcium carbonate mineral, aragonite, with an egg-like shape (" oolitic" from the Ancient Greek word ᾠόν for "egg") and sand grain size. This sand type forms in tropical waters through precipitation, sedimentation, and microbial activity, and is indicative of high energy environments. The production of oolitic aragonite sand in the Bahamas surpasses anyplace else in the world. Changes in seawater chemistry and paleoenvironments can be interpreted by the sand's chemical composition and structure. Formation Oolitic aragonite sand forms in high-salinity waters that are turbulent, shallow, and warm. The sand starts to form around a nucleus of calcium carbonate, such as a peloid, shell fragment, or foraminifer. The nucleus is coated with a thin layer of crystalline carbonate to form the cortex of the ooid. Compared to other types of sand formation that involve the weathering and erosion of a larger rock by turbulent waters, oolitic ara ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Calcite Seas
A calcite sea is a sea in which low-magnesium calcite is the primary inorganic marine calcium carbonate precipitate. An aragonite sea is the alternate seawater chemistry in which aragonite and high-magnesium calcite are the primary inorganic carbonate precipitates. The Early Paleozoic and the Middle to Late Mesozoic oceans were predominantly calcite seas, whereas the Middle Paleozoic through the Early Mesozoic and the Cenozoic (including today) are characterized by aragonite seas. The most significant geological and biological effects of calcite sea conditions include rapid and widespread formation of carbonate hardgrounds, calcitic ooids, calcite cements, and the contemporaneous dissolution of aragonite shells in shallow warm seas. Hardgrounds were very common, for example, in the calcite seas of the Ordovician and Jurassic, but virtually absent from the aragonite seas of the Permian. Fossils of invertebrate organisms found in calcite sea deposits are usually dominated by ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Calcite
Calcite is a carbonate mineral and the most stable polymorph of calcium carbonate (CaCO3). It is a very common mineral, particularly as a component of limestone. Calcite defines hardness 3 on the Mohs scale of mineral hardness, based on scratch hardness comparison. Large calcite crystals are used in optical equipment, and limestone composed mostly of calcite has numerous uses. Other polymorphs of calcium carbonate are the minerals aragonite and vaterite. Aragonite will change to calcite over timescales of days or less at temperatures exceeding 300 °C, and vaterite is even less stable. Etymology Calcite is derived from the German ''Calcit'', a term from the 19th century that came from the Latin word for lime, ''calx'' (genitive calcis) with the suffix "-ite" used to name minerals. It is thus etymologically related to chalk. When applied by archaeologists and stone trade professionals, the term alabaster is used not just as in geology and mineralogy, where it is ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Jurassic
The Jurassic ( ) is a Geological period, geologic period and System (stratigraphy), stratigraphic system that spanned from the end of the Triassic Period million years ago (Mya) to the beginning of the Cretaceous Period, approximately Mya. The Jurassic constitutes the middle period of the Mesozoic, Mesozoic Era and is named after the Jura Mountains, where limestone strata from the period were first identified. The start of the Jurassic was marked by the major Triassic–Jurassic extinction event, associated with the eruption of the Central Atlantic magmatic province, Central Atlantic Magmatic Province. The beginning of the Toarcian Stage started around 183 million years ago and is marked by an extinction event associated with widespread Anoxic event, oceanic anoxia, ocean acidification, and elevated temperatures likely caused by the eruption of the Karoo-Ferrar, Karoo-Ferrar large igneous provinces. The end of the Jurassic, however, has no clear boundary with the Cretaceous and i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Calcite Sea
A calcite sea is a sea in which low-magnesium calcite is the primary inorganic marine calcium carbonate precipitate. An aragonite sea is the alternate seawater chemistry in which aragonite and high-magnesium calcite are the primary inorganic carbonate precipitates. The Early Paleozoic and the Middle to Late Mesozoic oceans were predominantly calcite seas, whereas the Middle Paleozoic through the Early Mesozoic and the Cenozoic (including today) are characterized by aragonite seas. The most significant geological and biological effects of calcite sea conditions include rapid and widespread formation of carbonate hardgrounds, calcitic ooids, calcite cements, and the contemporaneous dissolution of aragonite shells in shallow warm seas. Hardgrounds were very common, for example, in the calcite seas of the Ordovician and Jurassic, but virtually absent from the aragonite seas of the Permian. Fossils of invertebrate organisms found in calcite sea deposits are usually dominated by ei ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pisolite
A pisolite is a sedimentary rock made of pisoids, which are concretionary grains – typically of calcium carbonate which resemble ooids, but are more than 2 mm in diameter. These grains are approximately spherical and have concentric layers reaching 10 mm in diameter. The name derives from the Hellenic word for pea. Bauxites, limonites, and siderites often have a pisolitic structure. See also * Ooid * Oolite Oolite or oölite (''egg stone'') is a sedimentary rock formed from ooids, spherical grains composed of concentric layers. The name derives from the Ancient Greek word for egg (ᾠόν). Strictly, oolites consist of ooids of diameter 0.25� ... References Further reading * Sedimentary rocks {{petrology-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sedimentary Rock
Sedimentary rocks are types of rock that are formed by the accumulation or deposition of mineral or organic particles at Earth's surface, followed by cementation. Sedimentation is the collective name for processes that cause these particles to settle in place. The particles that form a sedimentary rock are called sediment, and may be composed of geological detritus (minerals) or biological detritus (organic matter). The geological detritus originated from weathering and erosion of existing rocks, or from the solidification of molten lava blobs erupted by volcanoes. The geological detritus is transported to the place of deposition by water, wind, ice or mass movement, which are called agents of denudation. Biological detritus was formed by bodies and parts (mainly shells) of dead aquatic organisms, as well as their fecal mass, suspended in water and slowly piling up on the floor of water bodies ( marine snow). Sedimentation may also occur as dissolved minerals precipita ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Marine Environment
Marine habitats are habitats that support marine life. Marine life depends in some way on the saltwater that is in the sea (the term ''marine'' comes from the Latin ''mare'', meaning sea or ocean). A habitat is an ecological or environmental area inhabited by one or more living species.Abercrombie, M., Hickman, C.J. and Johnson, M.L. 1966.''A Dictionary of Biology.'' Penguin Reference Books, London The marine environment supports many kinds of these habitats. Marine habitats can be divided into coastal and open ocean habitats. Coastal habitats are found in the area that extends from as far as the tide comes in on the shoreline out to the edge of the continental shelf. Most marine life is found in coastal habitats, even though the shelf area occupies only seven percent of the total ocean area. Open ocean habitats are found in the deep ocean beyond the edge of the continental shelf. Alternatively, marine habitats can be divided into pelagic and demersal zones. Pelagic ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bahamas
The Bahamas (), officially the Commonwealth of The Bahamas, is an island country within the Lucayan Archipelago of the West Indies in the North Atlantic. It takes up 97% of the Lucayan Archipelago's land area and is home to 88% of the archipelago's population. The archipelagic state consists of more than 3,000 islands, cays, and islets in the Atlantic Ocean, and is located north of Cuba and northwest of the island of Hispaniola (split between the Dominican Republic and Haiti) and the Turks and Caicos Islands, southeast of the U.S. state of Florida, and east of the Florida Keys. The capital is Nassau on the island of New Providence. The Royal Bahamas Defence Force describes The Bahamas' territory as encompassing of ocean space. The Bahama Islands were inhabited by the Lucayans, a branch of the Arawakan-speaking Taíno, for many centuries. Christopher Columbus was the first European to see the islands, making his first landfall in the "New World" in 1492 when he landed on the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ordovician
The Ordovician ( ) is a geologic period and system, the second of six periods of the Paleozoic Era. The Ordovician spans 41.6 million years from the end of the Cambrian Period million years ago (Mya) to the start of the Silurian Period Mya. The Ordovician, named after the Welsh tribe of the Ordovices, was defined by Charles Lapworth in 1879 to resolve a dispute between followers of Adam Sedgwick and Roderick Murchison, who were placing the same rock beds in North Wales in the Cambrian and Silurian systems, respectively. Lapworth recognized that the fossil fauna in the disputed strata were different from those of either the Cambrian or the Silurian systems, and placed them in a system of their own. The Ordovician received international approval in 1960 (forty years after Lapworth's death), when it was adopted as an official period of the Paleozoic Era by the International Geological Congress. Life continued to flourish during the Ordovician as it did in the earlier C ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |





.jpg)