|
Molybdopterin
Molybdopterins are a class of cofactors found in most molybdenum-containing and all tungsten-containing enzymes. Synonyms for molybdopterin are: MPT and pyranopterin-dithiolate. The nomenclature for this biomolecule can be confusing: Molybdopterin itself contains no molybdenum; rather, this is the name of the ligand (a '' pterin'') that will bind the active metal. After molybdopterin is eventually complexed with molybdenum, the complete ligand is usually called molybdenum cofactor. Molybdopterin consists of a pyranopterin, a complex heterocycle featuring a pyran fused to a pterin ring. In addition, the pyran ring features two thiolates, which serve as ligands in molybdo- and tungstoenzymes. In some cases, the alkyl phosphate group is replaced by an alkyl diphosphate nucleotide. Enzymes that contain the molybdopterin cofactor include xanthine oxidase, DMSO reductase, sulfite oxidase, and nitrate reductase. The only molybdenum-containing enzymes that do not feature molybd ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sulfite Oxidase
Sulfite oxidase () is an enzyme in the mitochondria of all eukaryotes, with exception of the yeasts. It oxidizes sulfite to sulfate and, via cytochrome c, transfers the electrons produced to the electron transport chain, allowing generation of ATP in oxidative phosphorylation. This is the last step in the metabolism of sulfur-containing compounds and the sulfate is excreted. Sulfite oxidase is a metallo-enzyme that utilizes a molybdopterin cofactor and a heme group (in a case of animals). It is one of the cytochromes ''b''5 and belongs to the enzyme super-family of molybdenum oxotransferases that also includes DMSO reductase, xanthine oxidase, and nitrite reductase. In mammals, the expression levels of sulfite oxidase is high in the liver, kidney, and heart, and very low in spleen, brain, skeletal muscle, and blood. Structure As a homodimer, sulfite oxidase contains two identical subunits with an N-terminal domain and a C-terminal domain. These two domains are connected b ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Molybdenum
Molybdenum is a chemical element with the symbol Mo and atomic number 42 which is located in period 5 and group 6. The name is from Neo-Latin ''molybdaenum'', which is based on Ancient Greek ', meaning lead, since its ores were confused with lead ores. Molybdenum minerals have been known throughout history, but the element was discovered (in the sense of differentiating it as a new entity from the mineral salts of other metals) in 1778 by Carl Wilhelm Scheele. The metal was first isolated in 1781 by Peter Jacob Hjelm. Molybdenum does not occur naturally as a free metal on Earth; it is found only in various oxidation states in minerals. The free element, a silvery metal with a grey cast, has the sixth-highest melting point of any element. It readily forms hard, stable carbides in alloys, and for this reason most of the world production of the element (about 80%) is used in steel alloys, including high-strength alloys and superalloys. Most molybdenum compounds have low so ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tungsten
Tungsten, or wolfram, is a chemical element with the symbol W and atomic number 74. Tungsten is a rare metal found naturally on Earth almost exclusively as compounds with other elements. It was identified as a new element in 1781 and first isolated as a metal in 1783. Its important ores include scheelite and wolframite, the latter lending the element its alternate name. The free element is remarkable for its robustness, especially the fact that it has the highest melting point of all known elements barring carbon (which sublimes at normal pressure), melting at . It also has the highest boiling point, at . Its density is , comparable with that of uranium and gold, and much higher (about 1.7 times) than that of lead. Polycrystalline tungsten is an intrinsically brittle and hard material (under standard conditions, when uncombined), making it difficult to work. However, pure single-crystalline tungsten is more ductile and can be cut with a hard-steel hacksaw. Tungsten o ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Radical SAM
Radical SAM is a designation for a superfamily of enzymes that use a + cluster">Fe-4Ssup>+ cluster to reductively cleave ''S''-adenosyl-L-methionine (SAM) to generate a radical, usually a 5′- deoxyadenosyl radical (5'-dAdo), as a critical intermediate. These enzymes utilize this radical intermediate to perform diverse transformations, often to functionalize unactivated C-H bonds. Radical SAM enzymes are involved in cofactor biosynthesis, enzyme activation, peptide modification, post-transcriptional and post-translational modifications, metalloprotein cluster formation, tRNA modification, lipid metabolism, biosynthesis of antibiotics and natural products etc. The vast majority of known radical SAM enzymes belong to the radical SAM superfamily, and have a cysteine-rich motif that matches or resembles CxxxCxxC. rSAMs comprise the largest superfamily of metal-containing enzymes. History and mechanism As of 2001, 645 unique radical SAM enzymes have been identified from 126 spec ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
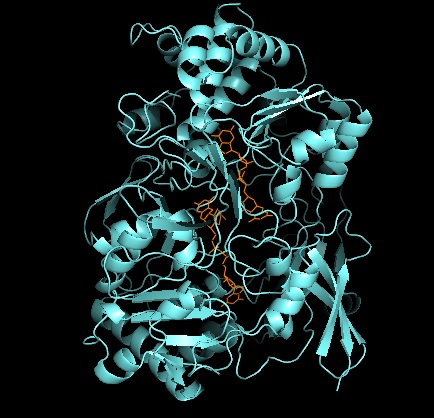
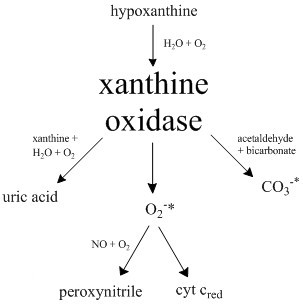
Xanthine Oxidase
Xanthine oxidase (XO, sometimes XAO) is a form of xanthine oxidoreductase, a type of enzyme that generates reactive oxygen species. These enzymes catalyze the oxidation of hypoxanthine to xanthine and can further catalyze the oxidation of xanthine to uric acid. These enzymes play an important role in the catabolism of purines in some species, including humans. Xanthine oxidase is defined as an ''enzyme activity'' (EC 1.17.3.2). The same protein, which in humans has the HGNC approved gene symbol ''XDH'', can also have xanthine dehydrogenase activity (EC 1.17.1.4). Most of the protein in the liver exists in a form with xanthine dehydrogenase activity, but it can be converted to xanthine oxidase by reversible sulfhydryl oxidation or by irreversible proteolytic modification. Reaction The following chemical reactions are catalyzed by xanthine oxidase: * hypoxanthine + H2O + O2 \rightleftharpoons xanthine + H2O2 * xanthine + H2O + O2 \rightleftharpoons uric acid + H2O2 * Xant ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dithiolene
Dithiolene metal complexes are complexes containing 1,2-dithiolene ligands. 1,2-Dithiolene ligands, a particular case of 1,2-dichalcogenolene species, are unsaturated bidentate ligand wherein the two donor atoms are sulfur. 1,2-Dithiolene metal complexes are often referred to as "metal dithiolenes", "metallodithiolenes" or "dithiolene complexes". Most molybdenum- and tungsten-containing proteins have dithiolene-like moieties at their active sites, which feature the so-called molybdopterin cofactor bound to the Mo or W. Dithiolene metal complexes have been studied since the 1960s when they were first popularized by Gerhard N. Schrauzer and Volker P. Mayweg, who prepared nickel bis(stilbene-1,2-dithiolate) (Ni(S2C2Ph2)2) by the reaction of nickel sulfide and diphenylacetylene. The structural, spectroscopic, and electrochemical properties of many related complexes have been described. Structure and bonding Dithiolene metal complexes can be found in coordination compounds where t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ethylbenzene Dehydrogenase
In enzymology, an ethylbenzene hydroxylase () is an enzyme that catalyzes the chemical reaction :ethylbenzene + H2O + acceptor \rightleftharpoons (S)-1-phenylethanol + reduced acceptor The 3 substrates of this enzyme are ethylbenzene, H2O, and acceptor, whereas its two products are (S)-1-phenylethanol and reduced acceptor. This enzyme belongs to the family of oxidoreductases, specifically those acting on CH or CH2 groups with other acceptors. The systematic name of this enzyme class is ethylbenzene:acceptor oxidoreductase. Other names in common use include ethylbenzene dehydrogenase, and ethylbenzene:(acceptor) oxidoreductase. This enzyme participates in ethylbenzene degradation by ''Aromatoleum aromaticum'', a denitrifying bacterium related to the genera ''Azoarcus'' and ''Thauera''. It is a molybdenum enzyme belonging to the DMSO reductase family. Molybdenum enzymes are distinguished by the presence of a unique active site containing molybdenum atom, one or two molybdopte ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Molybdenum Cofactor , a metal cluster that contains Fe, Mo, and S that is found in some nitrogenases
{{SIA ...
A molybdenum cofactor is a biochemical cofactor that contains molybdenum. Examples include: * Molybdopterin (or, strictly speaking, the molybdopterin-molybdenum-complex), the organophosphate-dithiolate ligand that binds molybdenum and tungsten in most molybdenum- (except nitrogenases) and all tungsten-containing proteins * FeMoco FeMoco ( cofactor) is the primary cofactor of nitrogenase. Nitrogenase is the enzyme that catalyzes the conversion of atmospheric nitrogen molecules N2 into ammonia (NH3) through the process known as nitrogen fixation. Containing iron and molybd ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cyclic Pyranopterin Monophosphate
Fosdenopterin (or cyclic pyranopterin monophosphate, cPMP), sold under the brand name Nulibry, is a medication used to reduce the risk of death due to a rare genetic disease known as molybdenum cofactor deficiency type A. The most common side effects include complications related to the intravenous line, fever, respiratory infections, vomiting, gastroenteritis, and diarrhea. Fosdenopterin was approved for medical use in the United States in February 2021, It is the first medication approved by the U.S. Food and Drug Administration (FDA) for the treatment of molybdenum cofactor deficiency type A. and in the European Union in September 2022. The US Food and Drug Administration considers it to be a first-in-class medication. Medical uses Fosdenopterin is indicated to reduce the risk of mortality in people with molybdenum cofactor deficiency (MoCD) type A. Mechanism of action People with molybdenum cofactor deficiency type A cannot produce cyclic pyranopterin monophosphate ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pterin
Pterin is a heterocyclic compound composed of a pteridine ring system, with a " keto group" (a lactam) and an amino group on positions 4 and 2 respectively. It is structurally related to the parent bicyclic heterocycle called pteridine. Pterins, as a group, are compounds related to pterin with additional substituents. Pterin itself is of no biological significance. Pterins were first discovered in the pigments of butterfly wings (hence the origin of their name, from the Greek ''pteron (πτερόν)'', wing) and perform many roles in coloration in the biological world. Chemistry Pterins exhibit a wide range of tautomerism in water, beyond what is assumed by just keto-enol tautomerism. For the unsubstituted pterin, at least five tautomers are commonly cited. For 6-methylpterin, seven tautomers are theoretically predicted to be important in solution. The pteridine ring system contains four nitrogen atoms, reducing its aromaticity to the point that it can be attacked by nu ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
DMSO Reductase
DMSO reductase is a molybdenum-containing enzyme that catalyzes reduction of dimethyl sulfoxide (DMSO) to dimethyl sulfide (DMS). This enzyme serves as the terminal reductase under anaerobic conditions in some bacteria, with DMSO being the terminal electron acceptor. During the course of the reaction, the oxygen atom in DMSO is transferred to molybdenum, and then reduced to water. DMSO reductase (DMSOR) and other members of the DMSO reductase family are unique to bacteria and archaea. Enzymes of this family in anaerobic oxidative phosphorylation and inorganic-donor-based lithotrophic respiration. These enzymes have been engineered to degrade oxoanions. DMSOR catalyzes the transfer of two electrons and one oxygen atom in the reaction: The active site of DMSOR contains molybdenum, which is otherwise rare in biology. Tertiary structure and active site As for other members of DMSO reductase family, the tertiary structure of DMSOR is composed of Mo-surrounding domains I-IV, wi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nitrate Reductase
Nitrate reductases are molybdoenzymes that reduce nitrate (NO) to nitrite (NO). This reaction is critical for the production of protein in most crop plants, as nitrate is the predominant source of nitrogen in fertilized soils. Types Eukaryotic Eukaryotic nitrate reductases are part of the sulfite oxidase family of molybdoenzymes. They transfer electrons from NADH or NADPH to nitrate. Prokaryotic Prokaryotic nitrate reductases belong to the DMSO reductase family of molybdoenzymes and have been classified into three groups, assimilatory nitrate reductases (Nas), respiratory nitrate reductase (Nar), and periplasmic nitrate reductases (Nap). The active site of these enzymes is a Mo ion that is bound to the four thiolate functions of two pterin molecules. The coordination sphere of the Mo is completed by one amino-acid side chain and oxygen and/or sulfur ligands. The exact environment of the Mo ion in certain of these enzymes (oxygen versus sulfur as a sixth molybdenum li ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |



2Ni(mnt)2.jpg)