|
CyaA
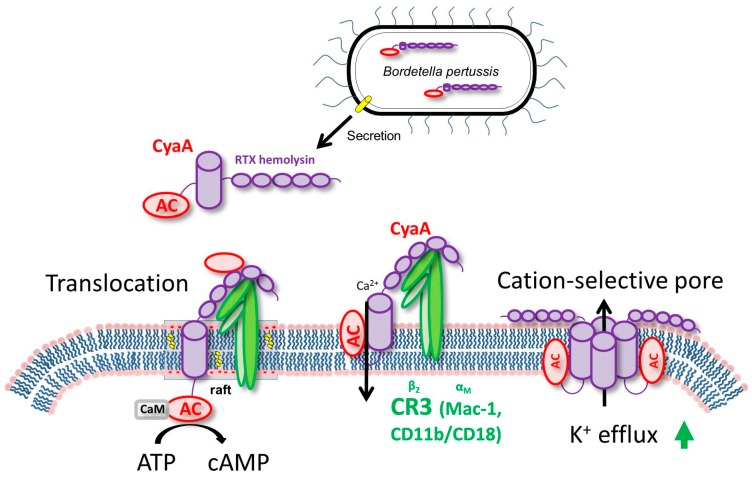
Adenylate cyclase toxin (CyaA) is released from bacterium Bordetella pertussis by the T1SS (Type 1 secretion system) and released in the host’s respiratory tract in order to suppress its early innate and subsequent adaptive immune defense. CyaA plays a particular role in the early phases of airway colonization. It is able to instantly ablate the bactericidal oxidative burst, along with the opsonophagocytic killing capacities of neutrophils and macrophages. As a result, it enables establishment of Bordetella infection of airway mucosa and promotes immune evasion of B. pertussis, by affecting the host’s immune cells. Structure The toxin is a 1706 residue-long polypeptide that consists of an N-terminal ~400 residue-long adenylate cyclase (AC) enzyme that is linked to a characteristic RTX hemolysin (Hly) moiety of ~1300 residues. This Hly moiety itself consists of four functional subdomains, comprising: (i) a hydrophobic pore-forming domain ; (ii) an activation domain, with the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Adenylate Cyclase
Adenylate cyclase (EC 4.6.1.1, also commonly known as adenyl cyclase and adenylyl cyclase, abbreviated AC) is an enzyme with systematic name ATP diphosphate-lyase (cyclizing; 3′,5′-cyclic-AMP-forming). It catalyzes the following reaction: :ATP = 3′,5′-cyclic AMP + diphosphate It has key regulatory roles in essentially all cells. It is the most polyphyletic known enzyme: six distinct classes have been described, all catalyzing the same reaction but representing unrelated gene families with no known sequence or structural homology. The best known class of adenylyl cyclases is class III or AC-III (Roman numerals are used for classes). AC-III occurs widely in eukaryotes and has important roles in many human tissues. All classes of adenylyl cyclase catalyse the conversion of adenosine triphosphate (ATP) to 3',5'-cyclic AMP (cAMP) and pyrophosphate.Magnesium ions are generally required and appear to be closely involved in the enzymatic mechanism. The cAMP produced by AC t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pertussis Toxin
Pertussis toxin (PT) is a protein-based AB5-type exotoxin produced by the bacterium ''Bordetella pertussis'', which causes whooping cough. PT is involved in the colonization of the respiratory tract and the establishment of infection. Research suggests PT may have a therapeutic role in treating a number of common human ailments, including hypertension, viral infection, and autoimmunity. History PT clearly plays a central role in the pathogenesis of pertussis although this was discovered only in the early 1980s. The appearance of pertussis is quite recent, compared with other epidemic infectious diseases. The earliest mention of pertussis, or whooping cough, is of an outbreak in Paris in 1414. This was published in Moulton's The Mirror of Health, in 1640. Another epidemic of pertussis took place in Paris in 1578 and was described by a contemporary observer, Guillaume de Baillou. Pertussis was well known throughout Europe by the middle of the 18th century. Jules Bordet and O ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bordetella Pertussis
''Bordetella pertussis'' is a Gram-negative, aerobic, pathogenic, encapsulated coccobacillus of the genus ''Bordetella'', and the causative agent of pertussis or whooping cough. Like '' B. bronchiseptica'', ''B. pertussis'' is motile and expresses a flagellum-like structure. Its virulence factors include pertussis toxin, adenylate cyclase toxin, filamentous hæmagglutinin, pertactin, fimbria, and tracheal cytotoxin. The bacterium is spread by airborne droplets; its incubation period is 7–10 days on average (range 6–20 days). Humans are the only known reservoir for ''B. pertussis''. The complete ''B. pertussis'' genome of 4,086,186 base pairs was published in 2003. Compared to its closest relative ''B. bronchiseptica'', the genome size is greatly reduced. This is mainly due to the adaptation to one host species (human) and the loss of capability of survival outside of a host body. History The disease pertussis was first described by French physician Guillaume de Baillo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
T1SS
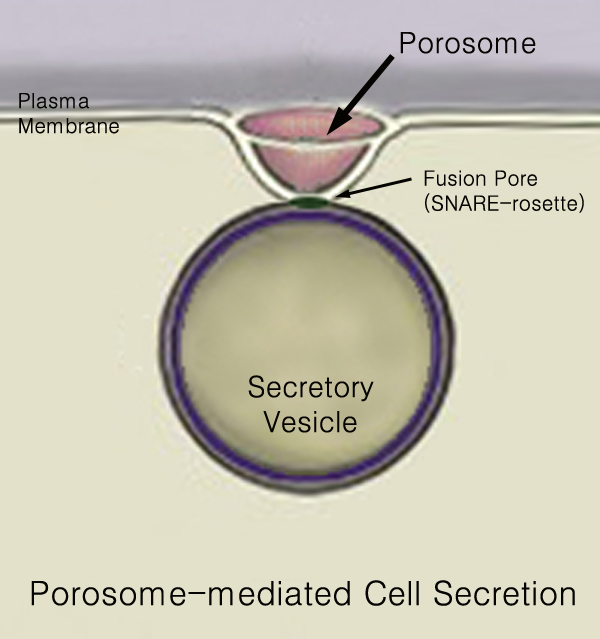
440px Secretion is the movement of material from one point to another, such as a secreted chemical substance from a cell or gland. In contrast, excretion is the removal of certain substances or waste products from a cell or organism. The classical mechanism of cell secretion is via secretory portals at the plasma membrane called porosomes. Porosomes are permanent cup-shaped lipoprotein structures embedded in the cell membrane, where secretory vesicles transiently dock and fuse to release intra-vesicular contents from the cell. Secretion in bacterial species means the transport or translocation of effector molecules for example: proteins, enzymes or toxins (such as cholera toxin in pathogenic bacteria e.g. ''Vibrio cholerae'') from across the interior (cytoplasm or cytosol) of a bacterial cell to its exterior. Secretion is a very important mechanism in bacterial functioning and operation in their natural surrounding environment for adaptation and survival. In eukaryotic cells M ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Neutrophil
Neutrophils (also known as neutrocytes or heterophils) are the most abundant type of granulocytes and make up 40% to 70% of all white blood cells in humans. They form an essential part of the innate immune system, with their functions varying in different animals. They are formed from stem cells in the bone marrow and Cellular differentiation, differentiated into #Subpopulations, subpopulations of neutrophil-killers and neutrophil-cagers. They are short-lived and highly mobile, as they can enter parts of tissue where other cells/molecules cannot. Neutrophils may be subdivided into segmented neutrophils and banded neutrophils (or Band cell, bands). They form part of the polymorphonuclear cells family (PMNs) together with basophils and eosinophils. The name ''neutrophil'' derives from staining characteristics on hematoxylin and eosin (H&E stain, H&E) histology, histological or cell biology, cytological preparations. Whereas basophilic white blood cells stain dark blue and eosinoph ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Macrophage
Macrophages (abbreviated as M φ, MΦ or MP) ( el, large eaters, from Greek ''μακρός'' (') = large, ''φαγεῖν'' (') = to eat) are a type of white blood cell of the immune system that engulfs and digests pathogens, such as cancer cells, microbes, cellular debris, and foreign substances, which do not have proteins that are specific to healthy body cells on their surface. The process is called phagocytosis, which acts to defend the host against infection and injury. These large phagocytes are found in essentially all tissues, where they patrol for potential pathogens by amoeboid movement. They take various forms (with various names) throughout the body (e.g., histiocytes, Kupffer cells, alveolar macrophages, microglia, and others), but all are part of the mononuclear phagocyte system. Besides phagocytosis, they play a critical role in nonspecific defense (innate immunity) and also help initiate specific defense mechanisms (adaptive immunity) by recruiting other immune ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hemolysin
Hemolysins or haemolysins are lipids and proteins that cause lysis of red blood cells by disrupting the cell membrane. Although the lytic activity of some microbe-derived hemolysins on red blood cells may be of great importance for nutrient acquisition, many hemolysins produced by pathogens do not cause significant destruction of red blood cells during infection. However, hemolysins are often capable of lysing red blood cells ''in vitro''. While most hemolysins are protein compounds, some are lipid biosurfactants. Properties Many bacteria produce hemolysins that can be detected in the laboratory. It is now believed that many clinically relevant fungi also produce hemolysins. Hemolysins can be identified by their ability to lyse red blood cells ''in vitro''. Not only are the erythrocytes affected by hemolysins, but there are also some effects among other blood cells, such as leucocytes (white blood cells). ''Escherichia coli'' hemolysin is potentially cytotoxic to monocytes ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Macrophage-1 Antigen
Macrophage-1 antigen (or integrin αMβ2 or macrophage integrin or Mac-1) is a complement receptor ("CR3") consisting of CD11b (integrin αM) and CD18 (integrin β2). The integrin α chain is noncovalently bound to the integrin β chain. It binds to iC3b and can be involved in cellular adhesion, binding to the intercellular adhesion molecule-1 (ICAM-1). CR3 causes phagocytosis and destruction of cells opsonized with iC3b. CR3 and CR4 are thought to exhibit overlapping functions; however, the distinct binding sites to iC3b suggests differences in their functions. Additionally, CR3 has been shown to have therapeutic promise. Function Macrophage-1 antigen (hereafter complement receptor 3 or CR3) (CD11b/CD18) is a human cell surface receptor found on B and T lymphocytes, polymorphonuclear leukocytes (mostly neutrophils), NK cells, and mononuclear phagocytes like macrophages. CR3 is a pattern recognition receptor, capable of recognizing and binding to many molecules found on the surf ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Talin (protein)
Talin is a high-molecular-weight cytoskeletal protein concentrated at regions of cell–substratum contact and, in lymphocytes, at cell–cell contacts. Discovered in 1983 by Keith Burridge and colleagues, talin is a ubiquitous cytosolic protein that is found in high concentrations in focal adhesions. It is capable of linking integrins to the actin cytoskeleton either directly or indirectly by interacting with vinculin and α-actinin. Also, talin-1 drives extravasation mechanism through engineered human microvasculature in microfluidic systems. Talin-1 is involved in each part of extravasation affecting adhesion, trans-endothelial migration and the invasion stages. Integrin receptors are involved in the attachment of adherent cells to the extracellular matrix and of lymphocytes to other cells. In these situations, talin codistributes with concentrations of integrins in the plasma membrane. Furthermore, in vitro binding studies suggest that integrins bind to talin, although ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Protein Kinase A
In cell biology, protein kinase A (PKA) is a family of enzymes whose activity is dependent on cellular levels of cyclic AMP (cAMP). PKA is also known as cAMP-dependent protein kinase (). PKA has several functions in the cell, including regulation of glycogen, sugar, and lipid metabolism. It should not be confused with 5'-AMP-activated protein kinase (AMP-activated protein kinase). History Protein kinase A, more precisely known as adenosine 3',5'-monophosphate (cyclic AMP)-dependent protein kinase, abbreviated to PKA, was discovered by chemists Edmond H. Fischer and Edwin G. Krebs in 1968. They won the Nobel Prize in Physiology or Medicine in 1992 for their work on phosphorylation and dephosphorylation and how it relates to PKA activity. PKA is one of the most widely researched protein kinases, in part because of its uniqueness; out of 540 different protein kinase genes that make up the human kinome, only one other protein kinase, casein kinase 2, is known to exist in a physio ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Calmodulin
Calmodulin (CaM) (an abbreviation for calcium-modulated protein) is a multifunctional intermediate calcium-binding messenger protein expressed in all eukaryotic cells. It is an intracellular target of the secondary messenger Ca2+, and the binding of Ca2+ is required for the activation of calmodulin. Once bound to Ca2+, calmodulin acts as part of a calcium signal transduction pathway by modifying its interactions with various target proteins such as kinases or phosphatases. Structure Calmodulin is a small, highly conserved protein that is 148 amino acids long (16.7 kDa). The protein has two approximately symmetrical globular domains (the N- and C- domains) each containing a pair of EF hand motifs separated by a flexible linker region for a total of four Ca2+ binding sites, two in each globular domain. In the Ca2+-free state, the helices that form the four EF-hands are collapsed in a compact orientation, and the central linker is disordered; in the Ca2+-saturated state, th ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |