|
Urushibara Nickel
Urushibara nickel is a nickel based hydrogenation catalyst, named after Yoshiyuki Urushibara. History It was discovered by Yoshiyuki Urushibara in 1951, while doing research on the reduction of estrone to estradiol. Preparation First nickel is precipitated in metallic form by reacting a solution of a nickel salt with an excess of zinc. This precipitated nickel contains relatively large amounts of zinc and zinc oxide. Then the catalyst is activated by digesting with either base or acid. There are different designations for differently prepared Urushibara nickel catalysts. The most common is U-Ni-A and U-Ni-B. U-Ni-A is prepared by digesting the precipitated nickel with an acid such as acetic acid. U-Ni-B is prepared by digesting with a base such as sodium hydroxide. After the digestion with acid most of the zinc and zinc oxide is dissolved from the catalyst, while after digestion with base it still contains considerable amounts of zinc and zinc oxide. It is also possible to ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nickel
Nickel is a chemical element with symbol Ni and atomic number 28. It is a silvery-white lustrous metal with a slight golden tinge. Nickel is a hard and ductile transition metal. Pure nickel is chemically reactive but large pieces are slow to react with air under standard conditions because a passivation layer of nickel oxide forms on the surface that prevents further corrosion. Even so, pure native nickel is found in Earth's crust only in tiny amounts, usually in ultramafic rocks, and in the interiors of larger nickel–iron meteorites that were not exposed to oxygen when outside Earth's atmosphere. Meteoric nickel is found in combination with iron, a reflection of the origin of those elements as major end products of supernova nucleosynthesis. An iron–nickel mixture is thought to compose Earth's outer and inner cores. Use of nickel (as natural meteoric nickel–iron alloy) has been traced as far back as 3500 BCE. Nickel was first isolated and classified as an e ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cobalt
Cobalt is a chemical element with the symbol Co and atomic number 27. As with nickel, cobalt is found in the Earth's crust only in a chemically combined form, save for small deposits found in alloys of natural meteoric iron. The free element, produced by reductive smelting, is a hard, lustrous, silver-gray metal. Cobalt-based blue pigments ( cobalt blue) have been used since ancient times for jewelry and paints, and to impart a distinctive blue tint to glass, but the color was for a long time thought to be due to the known metal bismuth. Miners had long used the name ''kobold ore'' (German for ''goblin ore'') for some of the blue-pigment-producing minerals; they were so named because they were poor in known metals, and gave poisonous arsenic-containing fumes when smelted. In 1735, such ores were found to be reducible to a new metal (the first discovered since ancient times), and this was ultimately named for the ''kobold''. Today, some cobalt is produced specifically from one of ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
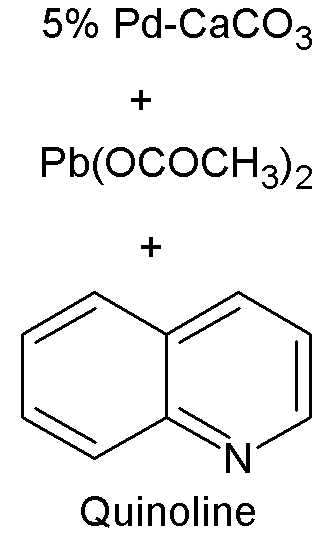
Lindlar Catalyst
A Lindlar catalyst is a heterogeneous catalyst that consists of palladium deposited on calcium carbonate or barium sulfate which is then poisoned with various forms of lead or sulfur. It is used for the hydrogenation of alkynes to alkenes (i.e. without further reduction into alkanes) and is named after its inventor Herbert Lindlar. Synthesis Lindlar catalyst is commercially available but may also be prepared by the reduction of palladium chloride in a slurry of calcium carbonate (CaCO3) followed by the addition of lead acetate. A variety of other "catalyst poisons" have been used, including lead oxide and quinoline. The palladium content of the supported catalyst is usually 5% by weight. Catalytic properties The catalyst is used for the hydrogenation of alkynes to alkenes (i.e. without further reduction into alkanes). The lead serves to deactivate the palladium sites, further deactivation of the catalyst with quinoline or 3,6-dithia-1,8-octanediol enhances its selectivity, pr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cobalt Boride
Cobalt borides are inorganic compounds with the general formula CoxBy. The two main cobalt borides are CoB and Co2B. These are refractory materials. Applications Materials science Cobalt borides are known to be exceptionally resistant to oxidation, a chemical property which makes them useful in the field of materials science. For instance, studies suggest cobalt boride can increase the lifespan of metal parts when used as a coating, imparting surfaces with higher corrosion and wear resistance. These properties have been exploited in the field of biomedical sciences for the design of specialized drug delivery systems. Renewable energy Cobalt boride has also been studied as a catalyst for hydrogen storage and fuel cell technologies. Organic synthesis Cobalt boride is also an effective hydrogenation catalyst used in organic synthesis. In one study, cobalt boride was found to be the most selective transition metal based catalyst available for the production of primary am ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nickel(II) Oxide
Nickel is a chemical element with symbol Ni and atomic number 28. It is a silvery-white lustrous metal with a slight golden tinge. Nickel is a hard and ductile transition metal. Pure nickel is chemically reactive but large pieces are slow to react with air under standard conditions because a passivation layer of nickel oxide forms on the surface that prevents further corrosion. Even so, pure native nickel is found in Earth's crust only in tiny amounts, usually in ultramafic rocks, and in the interiors of larger nickel–iron meteorites that were not exposed to oxygen when outside Earth's atmosphere. Meteoric nickel is found in combination with iron, a reflection of the origin of those elements as major end products of supernova nucleosynthesis. An iron–nickel mixture is thought to compose Earth's outer and inner cores. Use of nickel (as natural meteoric nickel–iron alloy) has been traced as far back as 3500 BCE. Nickel was first isolated and classified as an ele ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nickel Boride Catalyst
Nickel boride is the common name of materials composed chiefly of the elements nickel and boron that are widely used as catalysts in organic chemistry. Their approximate chemical composition is Ni2.5B, and they are often incorrectly denoted "" in organic chemistry publications. Nickel boride catalysts are typically prepared by reacting a salt of nickel with sodium borohydride. The composition and properties vary depending on the specific preparation method. The two most common forms, described and evaluated in detail by Herbert C. Brown and Charles Allan Brown in 1963, are known as P−1 nickel and P−2 nickel. These catalysts are usually obtained as black granules (P−1) or colloidal suspensions (P−2). They are air-stable, non-magnetic and non- pyrophoric, but slowly react with water to form nickel hydroxide . They are insoluble in all solvents, but react with concentrated mineral acids. They are claimed to be more effective hydrogenation catalysts than Raney nicke ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Alkene
In organic chemistry, an alkene is a hydrocarbon containing a carbon–carbon double bond. Alkene is often used as synonym of olefin, that is, any hydrocarbon containing one or more double bonds.H. Stephen Stoker (2015): General, Organic, and Biological Chemistry'. 1232 pages. Two general types of monoalkenes are distinguished: terminal and internal. Also called α-olefins, terminal alkenes are more useful. However, the International Union of Pure and Applied Chemistry (IUPAC) recommends using the name "alkene" only for acyclic hydrocarbons with just one double bond; alkadiene, alkatriene, etc., or polyene for acyclic hydrocarbons with two or more double bonds; cycloalkene, cycloalkadiene, etc. for cyclic ones; and "olefin" for the general class – cyclic or acyclic, with one or more double bonds. Acyclic alkenes, with only one double bond and no other functional groups (also known as mono-enes) form a homologous series of hydrocarbons with the general formula with '' ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Alkyne
\ce \ce Acetylene \ce \ce \ce Propyne \ce \ce \ce \ce 1-Butyne In organic chemistry, an alkyne is an unsaturated hydrocarbon containing at least one carbon—carbon triple bond. The simplest acyclic alkynes with only one triple bond and no other functional groups form a homologous series with the general chemical formula . Alkynes are traditionally known as acetylenes, although the name ''acetylene'' also refers specifically to , known formally as ethyne using IUPAC nomenclature. Like other hydrocarbons, alkynes are generally hydrophobic. Structure and bonding In acetylene, the H–C≡C bond angles are 180°. By virtue of this bond angle, alkynes are rod-like. Correspondingly, cyclic alkynes are rare. Benzyne cannot be isolated. The C≡C bond distance of 121 picometers is much shorter than the C=C distance in alkenes (134 pm) or the C–C bond in alkanes (153 pm). : The triple bond is very strong with a bond strength of 839 kJ/mol. The sigma bond contribute ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Functional Group
In organic chemistry, a functional group is a substituent or moiety in a molecule that causes the molecule's characteristic chemical reactions. The same functional group will undergo the same or similar chemical reactions regardless of the rest of the molecule's composition. This enables systematic prediction of chemical reactions and behavior of chemical compounds and the design of chemical synthesis. The reactivity of a functional group can be modified by other functional groups nearby. Functional group interconversion can be used in retrosynthetic analysis to plan organic synthesis. A functional group is a group of atoms in a molecule with distinctive chemical properties, regardless of the other atoms in the molecule. The atoms in a functional group are linked to each other and to the rest of the molecule by covalent bonds. For repeating units of polymers, functional groups attach to their nonpolar core of carbon atoms and thus add chemical character to carbon chains. Fun ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Amine
In chemistry, amines (, ) are compounds and functional groups that contain a basic nitrogen atom with a lone pair. Amines are formally derivatives of ammonia (), wherein one or more hydrogen atoms have been replaced by a substituent such as an alkyl or aryl group (these may respectively be called alkylamines and arylamines; amines in which both types of substituent are attached to one nitrogen atom may be called alkylarylamines). Important amines include amino acids, biogenic amines, trimethylamine, and aniline; Inorganic derivatives of ammonia are also called amines, such as monochloramine (). The substituent is called an amino group. Compounds with a nitrogen atom attached to a carbonyl group, thus having the structure , are called amides and have different chemical properties from amines. Classification of amines Amines can be classified according to the nature and number of substituents on nitrogen. Aliphatic amines contain only H and alkyl substituents. A ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Primary (chemistry)
Primary is a term used in organic chemistry to classify various types of compounds (e.g. alcohols, alkyl halides, amines) or reactive intermediates (e.g. alkyl radicals, carbocations). {{clear See also * Secondary (chemistry) * Tertiary (chemistry) * Quaternary (chemistry) Quaternary is a term used in organic chemistry to classify various types of compounds (e. g. amines and ammonium salts).Paula Yurkanis Bruice: ''Organic Chemistry'', Pearson Education Inc., 2004, 4. Ed., p. 78, 104, 893, and 912, . {{clear See ... References Chemical nomenclature ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
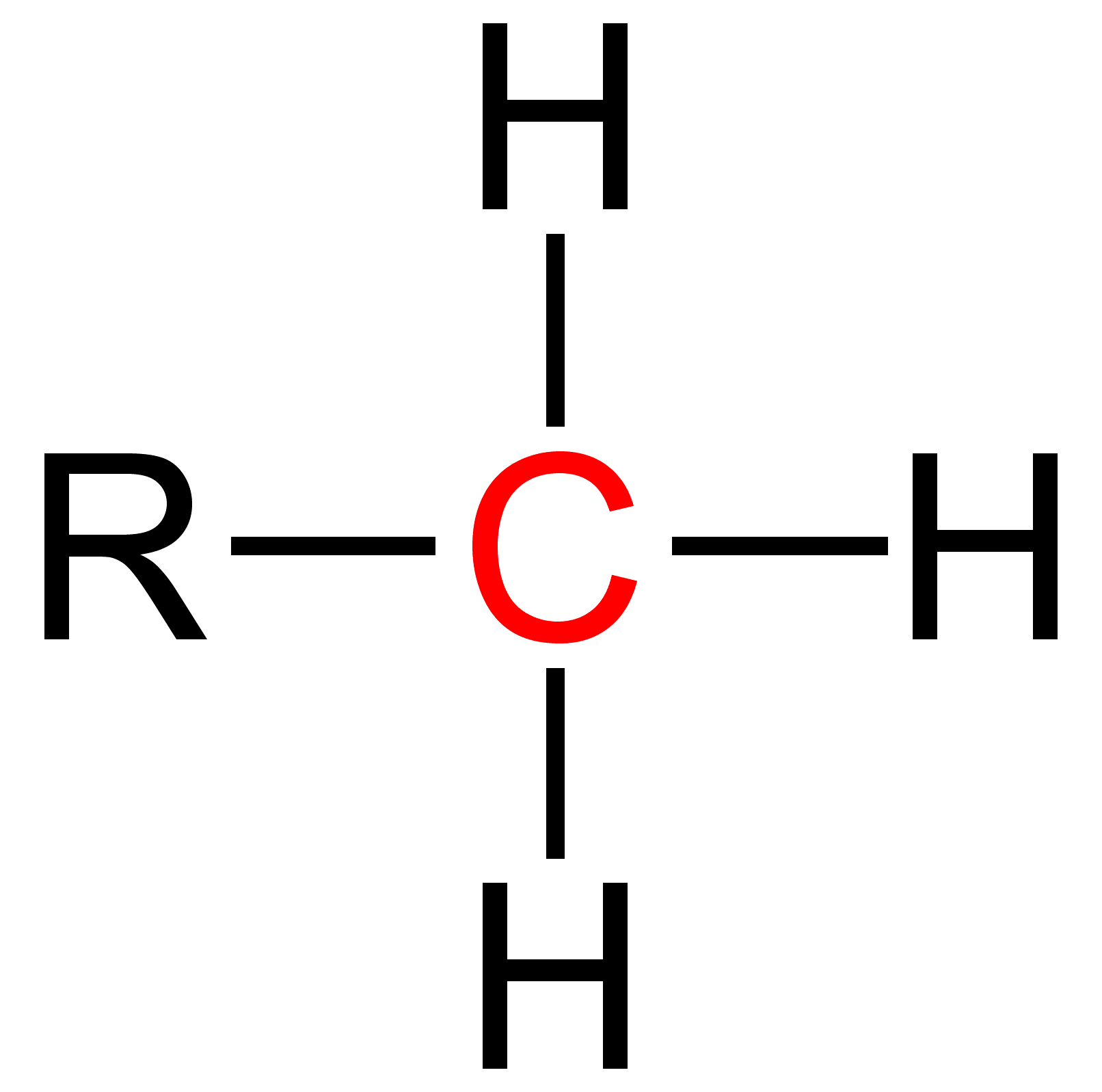
Nitrile Reduction
In nitrile reduction a nitrile is reduced to either an amine or an aldehyde with a suitable chemical reagent. Catalytic hydrogenation The catalytic hydrogenation of nitriles is often the most economical route available for the production of primary amines. Catalysts for the reaction often include group 10 metals such as Raney nickel, palladium black, or platinum dioxide. However, other catalysts, such as cobalt boride, also can be regioselective for primary amine production: : R-C≡N + 2 H2 → R-CH2NH2 A commercial application of this technology includes the production of hexamethylenediamine from adiponitrile, a precursor to Nylon 66. Depending on reaction conditions, reactive intermediate imines can also undergo attack by amine products to afford secondary and tertiary amines: : 2 R-C≡N + 4 H2 → (R-CH2)2NH + NH3 : 3 R-C≡N + 6 H2 → (R-CH2)3N + 2 NH3 Such reactions proceed via enamine intermediates. The most important reaction condition for selective p ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |