|
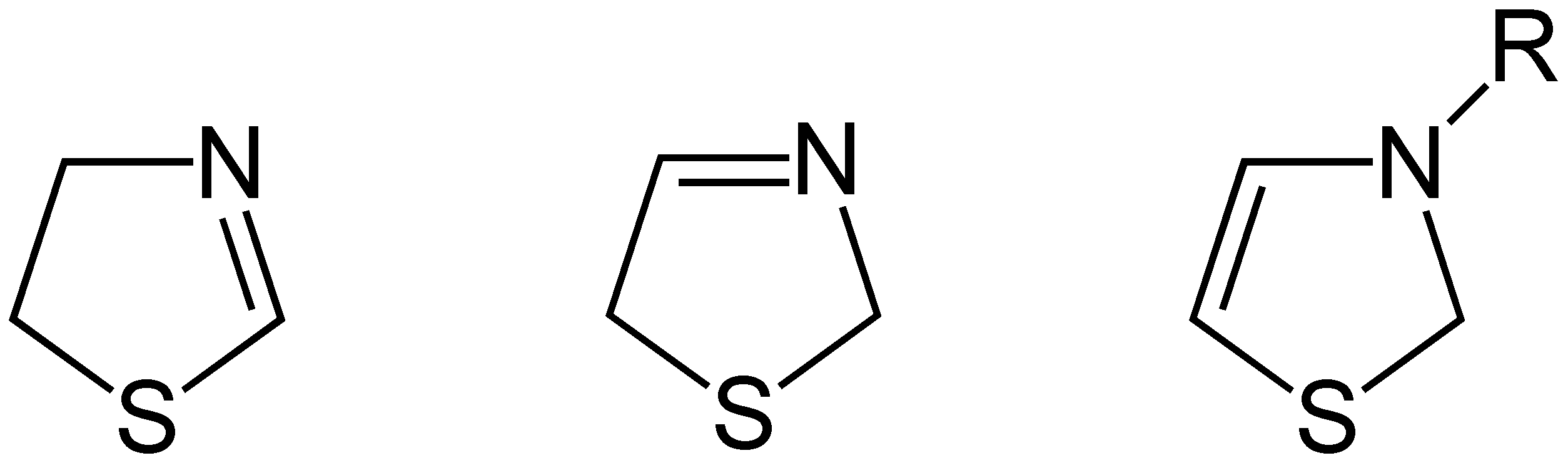
Thiazolidine
Thiazolidine is a heterocyclic organic compound with the formula (CH2)3(NH)S. It is a 5-membered saturated ring with a thioether group and an amine group in the 1 and 3 positions. It is a sulfur analog of oxazolidine. Thiazolidine is a colorless liquid. Derivatives, thiazolidines, are known. For example, the drug pioglitazone contains a thiazolidine ring. Another drug that contains a thiazolidine ring is the antibiotic penicillin. Preparation Thiazolidine is prepared as it was in its first reported synthesis, by the condensation of cysteamine and formaldehyde. Other thiazolidines may be synthesized by similar condensations. A notable derivative is 4-carboxythiazolidine, derived from formaldehyde and cysteine. Derivatives N-Methyl-2-thiazolidinethione is an accelerator for the vulcanization of chloroprene rubbers. Thiazolidines functionalized with carbonyls at the 2 and 4 positions, the thiazolidinediones, are drugs used in the treatment of diabetes mellitus type 2. Rhodanin ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Thiazolidinedione
The thiazolidinediones , abbreviated as TZD, also known as glitazones after the prototypical drug ciglitazone, are a class of heterocyclic compounds consisting of a five-membered C3NS ring. The term usually refers to a family of drugs used in the treatment of diabetes mellitus type 2 that were introduced in the late 1990s. Mechanism of action Thiazolidinediones or TZDs act by activating PPARs (peroxisome proliferator-activated receptors), a group of nuclear receptors In the field of molecular biology, nuclear receptors are a class of proteins responsible for sensing steroids, thyroid hormones, vitamins, and certain other molecules. These receptors work with other proteins to regulate the expression of specif ..., specific for ''PPARγ'' (PPAR-gamma, PPARG). They are thus the PPARG agonists subset of PPAR agonists. The endogenous ligands for these receptors are free fatty acids (FFAs) and eicosanoids. When activated, the receptor binds to DNA in complex with the retinoid X ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pioglitazone
Pioglitazone, sold under the brand name Actos among others, is an anti-diabetic medication used to treat type 2 diabetes. It may be used with metformin, a sulfonylurea, or insulin. Use is recommended together with exercise and diet. It is not recommended in type 1 diabetes. It is taken by mouth. Common side effects include headaches, muscle pains, inflammation of the throat, and swelling. Serious side effects may include bladder cancer, low blood sugar, heart failure, and osteoporosis. Use is not recommended in pregnancy or breastfeeding. It is in the thiazolidinedione (TZD) class and works by improving sensitivity of tissues to insulin. Pioglitazone was patented in 1985, and came into medical use in 1999. It is available as a generic medication. In 2020, it was the 168th most commonly prescribed medication in the United States, with more than 3million prescriptions. It was withdrawn in France and Germany in 2011. Medical uses Pioglitazone is used to lower blood glucose level ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Thiazoline
Thiazolines (or dihydrothiazoles) are a group of isomeric 5-membered heterocyclic compounds containing both sulfur and nitrogen in the ring. Although unsubstituted thiazolines are rarely encountered themselves, their derivatives are more common and some are bioactive. For example, in a common post-translational modification, cysteine residues are converted into thiazolines. The name thiazoline originates from the Hantzsch–Widman nomenclature. Isomers Three structural isomers of thiazoline exist depending on the position of the double bond. These forms do not readily interconvert and hence are not tautomers. Of these 2-thiazoline is the most common. A fourth structure exists in which the N and S atoms are adjacent; this known as isothiazoline. Synthesis Thiazolines were first prepared by dialkylation of thioamides by Richard Willstatter in 1909. 2-Thiazolines are commonly prepared from 2-aminoethanethiols (e.g. cysteamine). They may also be synthesized via the Asinger r ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Heterocyclic
A heterocyclic compound or ring structure is a cyclic compound that has atoms of at least two different elements as members of its ring(s). Heterocyclic chemistry is the branch of organic chemistry dealing with the synthesis, properties, and applications of these heterocycles. Examples of heterocyclic compounds include all of the nucleic acids, the majority of drugs, most biomass (cellulose and related materials), and many natural and synthetic dyes. More than half of known compounds are heterocycles. 59% of US FDA-approved drugs contain nitrogen heterocycles. Classification The study of heterocyclic chemistry focuses especially on unsaturated derivatives, and the preponderance of work and applications involves unstrained 5- and 6-membered rings. Included are pyridine, thiophene, pyrrole, and furan. Another large class of heterocycles refers to those fused to benzene rings. For example, the fused benzene derivatives of pyridine, thiophene, pyrrole, and furan are quinol ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Rhodanine
Rhodanine is a 5-membered heterocyclic organic compound possessing a thiazolidine core. It was discovered in 1877 by Marceli Nencki who named it ''"Rhodaninsaure"'' in reference to its synthesis from ammonium rhodanide (known as ammonium thiocyanate to modern chemists) and chloroacetic acid in water. Rhodanines can also be prepared by the reaction of carbon disulfide, ammonia, and chloroacetic acid, which proceeds via an intermediate dithiocarbamate In organic chemistry, a dithiocarbamate is a functional group with the general formula and structure . It is the analog of a carbamate in which both oxygen atoms are replaced by sulfur atoms (when only 1 oxygen is replaced the result is thioca .... : Derivatives Some rhodanine derivatives have pharmacological properties; for instance, epalrestat is used to treat diabetic neuropathy. However, most are promiscuous binders with poor selectivity; as a result, this class of compounds is viewed with suspicion by medicinal chemis ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
N-Methyl-2-thiazolidinethione
''N''-Methyl-2-thiazolidinethione is the organosulfur compound with the formula C2H4S(NCH3)CS. It is classified as a heterocycle called a thiazolidine. It is a colorless or off-white solid. It has gained attention as a proposed low toxicity replacement for ethylenethioureas, which are used as accelerators for the vulcanization of chloroprene rubbers. The compound is prepared by reaction of ''N''-methylethanolamine and carbon disulfide. See also *Mercaptobenzothiazole 2-Mercaptobenzothiazole is an organosulfur compound with the formula . It is used in the sulfur vulcanization of rubber. Structure The molecule is planar with a C=S double bond, so the name ''mercapto''benzothiazole is a misnomer. It is not a th ... - a structurally similar, but aromatic, vulcanization accelerator References {{DEFAULTSORT:Methyl-2-thiazolidinethione, N- Thiazolidines Dithiocarbamates ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cysteamine
Cysteamine is a chemical compound that can be biosynthesized in mammals, including humans, by the degradation of coenzyme A. The intermediate pantetheine is broken down into cysteamine and pantothenic acid. It is the biosynthetic precursor to the neurotransmitter hypotaurine. It is a stable aminothiol, i.e., an organic compound containing both an amine and a thiol functional groups. Cysteamine is a white, water-soluble solid. It is often used as salts of the ammonium derivative SCH2CH2NH3sup>+ including the hydrochloride, phosphocysteamine, and bitartrate. As a medication, cysteamine, sold under the brand name Cystagon among others, is indicated to treat cystinosis. Medical uses Cysteamine is used to treat cystinosis. It is available by mouth (capsule and extended release capsule) and in eye drops. When applied topically it can scavenge free radicals and lighten skin that's been darkened as a result of post-inflammatory hyperpigmentation, sun exposure and Melasma. Tent ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Thiazole
Thiazole, or 1,3-thiazole, is a heterocyclic compound that contains both sulfur and nitrogen. The term 'thiazole' also refers to a large family of derivatives. Thiazole itself is a pale yellow liquid with a pyridine-like odor and the molecular formula C3H3NS. The thiazole ring is notable as a component of the vitamin thiamine (B1). Molecular and electronic structure Thiazoles are members of the azoles, heterocycles that include imidazoles and oxazoles. Thiazole can also be considered a functional group. Oxazoles are related compounds, with sulfur replaced by oxygen. Thiazoles are structurally similar to imidazoles, with the thiazole sulfur replaced by nitrogen. Thiazole rings are planar and aromatic. Thiazoles are characterized by larger pi-electron delocalization than the corresponding oxazoles and have therefore greater aromaticity. This aromaticity is evidenced by the chemical shift of the ring protons in proton NMR spectroscopy (between 7.27 and 8.77 ppm), clearly indicating ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Diabetes Mellitus Type 2
Type 2 diabetes, formerly known as adult-onset diabetes, is a form of diabetes mellitus that is characterized by high blood sugar, insulin resistance, and relative lack of insulin. Common symptoms include increased thirst, frequent urination, and unexplained weight loss. Symptoms may also include increased hunger, feeling tired, and sores that do not heal. Often symptoms come on slowly. Long-term complications from high blood sugar include heart disease, strokes, diabetic retinopathy which can result in blindness, kidney failure, and poor blood flow in the limbs which may lead to amputations. The sudden onset of hyperosmolar hyperglycemic state may occur; however, ketoacidosis is uncommon. Type 2 diabetes primarily occurs as a result of obesity and lack of exercise. Some people are genetically more at risk than others. Type 2 diabetes makes up about 90% of cases of diabetes, with the other 10% due primarily to type 1 diabetes and gestational diabetes. In type 1 diabet ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chloroprene
Chloroprene is the common name for 2-chlorobuta-1,3-diene (IUPAC name) with the chemical formula CH2=CCl−CH=CH2. Chloroprene is a colorless volatile liquid, almost exclusively used as a monomer for the production of the polymer polychloroprene, better known as neoprene, a type of synthetic rubber. History Although it may have been discovered earlier, chloroprene was largely developed by DuPont during the early 1930s, specifically with the formation of neoprene in mind. The chemists Elmer K. Bolton, Wallace Carothers, Arnold Collins and Ira Williams are generally accredited with its development and commercialisation although the work was based upon that of Julius Arthur Nieuwland, with whom they collaborated. Production Chloroprene is produced in three steps from 1,3-butadiene: (i) chlorination, (ii) isomerization of part of the product stream, and (iii) dehydrochlorination of 3,4-dichlorobut-1-ene. Chlorine adds to 1,3-butadiene to afford a mixture of 3,4-dichlorobut-1-ene ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cysteine
Cysteine (symbol Cys or C; ) is a semiessential proteinogenic amino acid with the formula . The thiol side chain in cysteine often participates in enzymatic reactions as a nucleophile. When present as a deprotonated catalytic residue, sometimes the symbol Cyz is used. The deprotonated form can generally be described by the symbol Cym as well. The thiol is susceptible to oxidation to give the disulfide derivative cystine, which serves an important structural role in many proteins. In this case, the symbol Cyx is sometimes used. When used as a food additive, it has the E number E920. Cysteine is encoded by the codons UGU and UGC. The sulfur-containing amino acids cysteine and methionine are more easily oxidized than the other amino acids. Structure Like other amino acids (not as a residue of a protein), cysteine exists as a zwitterion. Cysteine has chirality in the older / notation based on homology to - and -glyceraldehyde. In the newer ''R''/''S'' system of designating chi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Formaldehyde
Formaldehyde ( , ) (systematic name methanal) is a naturally occurring organic compound with the formula and structure . The pure compound is a pungent, colourless gas that polymerises spontaneously into paraformaldehyde (refer to section Forms below), hence it is stored as an aqueous solution (formalin), which is also used to store animal specimens. It is the simplest of the aldehydes (). The common name of this substance comes from its similarity and relation to formic acid. Formaldehyde is an important precursor to many other materials and chemical compounds. In 1996, the installed capacity for the production of formaldehyde was estimated at 8.7 million tons per year. It is mainly used in the production of industrial resins, e.g., for particle board and coatings. Forms Formaldehyde is more complicated than many simple carbon compounds in that it adopts several diverse forms. These compounds can often be used interchangeably and can be interconverted. *Molecular formald ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |