|
Tiflorex Synthesis
Tiflorex (TFX), formerly known as flutiorex, is a stimulant amphetamine that was under development as an appetite suppressant in the 1970s, but appears to have been abandoned. It is structurally related to fenfluramine and 4-MTA. Tiflorex went to phase II clinical trials. The extended release formulation "TFX-SR" produced significant suppression of appetite. It also caused slightly more sleep disturbances and headaches than placebo, as well as mydriasis and a self-reported decrease in arousal. It had little effect on heart rate. Tifluorex is claimed to be a more potent anorectic than fenfluramine, with twice its potency in humans and 4 times its potency in rats. Pharmacology Pharmacodynamics The mechanism of action of tiflorex has apparently never been studied. Similar compounds such as fenfluramine, norfenfluramine and 4-MTA act as selective serotonin releasing agents and 5-HT2 receptor agonists. Fenfluramine in particular causes very similar side effects and app ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Racemic Mixture
In chemistry, a racemic mixture or racemate () is a mixture that has equal amounts (50:50) of left- and right-handed enantiomers of a chiral molecule or salt. Racemic mixtures are rare in nature, but many compounds are produced industrially as racemates. History The first known racemic mixture was racemic acid, which Louis Pasteur found to be a mixture of the two enantiomeric isomers of tartaric acid. He manually separated the crystals of a mixture, starting from an aqueous solution of the sodium ammonium salt of racemate tartaric acid. Pasteur benefited from the fact that ammonium tartrate salt gives enantiomeric crystals with distinct crystal forms (at 77 °F). Reasoning from the macroscopic scale down to the molecular, he reckoned that the molecules had to have non-superimposable mirror images. A sample with only a single enantiomer is an ''enantiomerically pure'' or ''enantiopure'' compound. Etymology The word ''racemic'' derives from Latin , meaning pertaining to a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
5-HT2 Receptor
The 5-HT2 receptors are a subfamily of 5-HT receptors that bind the endogenous neurotransmitter serotonin (5-hydroxytryptamine, 5-HT). The 5-HT2 subfamily consists of three G protein-coupled receptor G protein-coupled receptors (GPCRs), also known as seven-(pass)-transmembrane domain receptors, 7TM receptors, heptahelical receptors, serpentine receptors, and G protein-linked receptors (GPLR), form a large group of evolutionarily related ...s (GPCRs) which are coupled to Gq/G11 and mediate excitatory neurotransmission, including 5-HT2A, 5-HT2B, and 5-HT2C. For more information, please see the respective main articles of the individual subtypes: * 5-HT2A receptor * 5-HT2B receptor * 5-HT2C receptor See also * 5-HT1 receptor * 5-HT3 receptor * 5-HT4 receptor * 5-HT5 receptor * 5-HT6 receptor * 5-HT7 receptor * 5-HT2 antagonists References {{Serotonergics Serotonin receptors ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Trifluoromethyl Compounds
The trifluoromethyl group is a functional group that has the formula In science, a formula is a concise way of expressing information symbolically, as in a mathematical formula or a ''chemical formula''. The informal use of the term ''formula'' in science refers to the general construct of a relationship betwe ... . The naming of is group is derived from the methyl group (which has the formula ), by replacing each hydrogen atom by a fluorine atom. Some common examples are trifluoromethane , 1,1,1-trifluoroethane , and hexafluoroacetone . Compounds with this group are a subclass of the organofluorines. Properties The trifluoromethyl group has a significant electronegativity that is often described as being intermediate between the electronegativities of fluorine and chlorine. For this reason, trifluoromethyl-substituted compounds are often strong acids, such as trifluoromethanesulfonic acid and trifluoroacetic acid. Conversely, the trifluoromethyl group lowers the basicity of ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Abandoned Drugs
Abandon, abandoned, or abandonment may refer to: Common uses * Abandonment (emotional), a subjective emotional state in which people feel undesired, left behind, insecure, or discarded * Abandonment (legal), a legal term regarding property ** Child abandonment, the extralegal abandonment of children ** Lost, mislaid, and abandoned property, legal status of property after abandonment and rediscovery * Abandonment (mysticism) Art, entertainment, and media Film * ''Abandon'' (film), a 2002 film starring Katie Holmes * ''Abandoned'' (1949 film), starring Dennis O'Keefe * ''Abandoned'' (1955 film), the English language title of the Italian war film ''Gli Sbandati'' * ''Abandoned'' (2001 film), a Hungarian film * ''Abandoned'' (2010 film), starring Brittany Murphy * ''Abandoned'' (2015 film), a television movie about the shipwreck of the ''Rose-Noëlle'' in 1989 * ''Abandoned'' (2022 film), starring Emma Roberts * ''The Abandoned'' (1945 film), a 1945 Mexican film * ''The Aba ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Substituted Amphetamines
Substituted amphetamines, or simply amphetamines, are a class of compounds based upon the amphetamine structure; it includes all derivative compounds which are formed by replacing, or substituting, one or more hydrogen atoms in the amphetamine core structure with substituents. The compounds in this class span a variety of pharmacological subclasses, including stimulants, empathogens, and hallucinogens, among others. Examples of substituted amphetamines are amphetamine (itself), methamphetamine, ephedrine, cathinone, phentermine, mephentermine, tranylcypromine, bupropion, methoxyphenamine, selegiline, amfepramone (diethylpropion), pyrovalerone, MDMA (ecstasy), and DOM (STP). Some of amphetamine's substituted derivatives occur in nature, for example in the leaves of '' Ephedra'' and khat plants. Amphetamine was first produced at the end of the 19th century. By the 1930s, amphetamine and some of its derivative compounds found use as decongestants in the symptomat ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Anorectics
An anorectic is a drug that reduces appetite, resulting in lower food consumption, leading to weight loss. These substances work by affecting the central nervous system or certain neurotransmitters to create a feeling of fullness or reduce the desire to eat. The understanding of anorexiant effects is crucial in the development of interventions for weight management, eating disorders, and related health concerns. The anorexiant effect can be induced through diverse mechanisms, ranging from hormonal regulation to neural signaling. Ghrelin, leptin, and peptide YY are among the hormones involved in appetite control. Additionally, neurotransmitters such as serotonin and dopamine in the central nervous system contribute significantly to the regulation of food intake. By contrast, an appetite stimulant is referred to as orexigenic. The term is (from the Greek and ), and such drugs are also known as anorexigenic, anorexiant, or appetite suppressant. History Used on a short-term ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Formic Acid
Formic acid (), systematically named methanoic acid, is the simplest carboxylic acid. It has the chemical formula HCOOH and structure . This acid is an important intermediate in chemical synthesis and occurs naturally, most notably in some ants. Esters, salts, and the anion derived from formic acid are called formates. Industrially, formic acid is produced from methanol. Natural occurrence Formic acid, which has a pungent, penetrating odor, is found naturally in insects, weeds, fruits and vegetables, and forest emissions. It appears in most ants and in stingless bees of the genus '' Oxytrigona''. Wood ants from the genus ''Formica'' can spray formic acid on their prey or to defend the nest. The puss moth caterpillar (''Cerura vinula'') will spray it as well when threatened by predators. It is also found in the trichomes of stinging nettle (''Urtica dioica''). Apart from that, this acid is incorporated in many fruits such as pineapple (0.21 mg per 100 g), apple (2 mg per ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ethylamine
Ethylamine, also known as ethanamine, is an organic compound with the chemical formula, formula CH3CH2NH2. This colourless gas has a strong ammonia-like odor. It condenses just below room temperature to a liquid miscibility, miscible with virtually all solvents. It is a nucleophilic base (chemistry), base, as is typical for amines. Ethylamine is widely used in chemical industry and organic synthesis. It is a DEA list I chemical by 21 CFR § 1310.02. Synthesis Ethylamine is produced on a large scale by two processes. Most commonly ethanol and ammonia are combined in the presence of an oxide catalyst: :CH3CH2OH + NH3 → CH3CH2NH2 + H2O In this reaction, ethylamine is coproduced together with diethylamine and triethylamine. In aggregate, approximately 80M kilograms/year of these three amines are produced industrially.Karsten Eller, Erhard Henkes, Roland Rossbacher, Hartmut Höke, "Amines, Aliphatic" Ullmann's Encyclopedia of Industrial Chemistry, Wiley-VCH, Weinheim, 2005. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
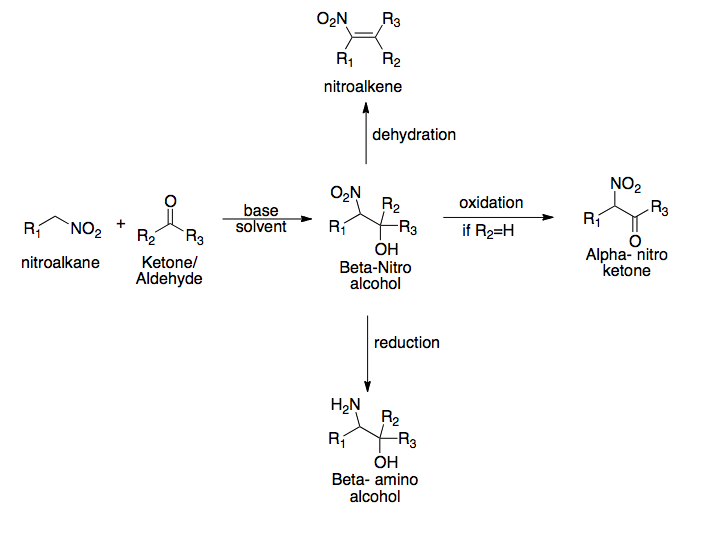
Henry Reaction
The Henry reaction is a classic carbon–carbon bond formation reaction in organic chemistry. Discovered in 1895 by the Belgian chemist Louis Henry (1834–1913), it is the combination of a nitroalkane and an aldehyde or ketone in the presence of a base to form β-nitro alcohols. This type of reaction is also referred to as a nitroaldol reaction (nitroalkane, aldehyde, and alcohol). It is nearly analogous to the aldol reaction that had been discovered 23 years prior that couples two carbonyl compounds to form β-hydroxy carbonyl compounds known as "aldols" (aldehyde and alcohol). The Henry reaction is a useful technique in the area of organic chemistry due to the synthetic utility of its corresponding products, as they can be easily converted to other useful synthetic intermediates. These conversions include subsequent dehydration to yield nitroalkenes, oxidation of the secondary alcohol to yield α-nitro ketones, or reduction of the nitro group to yield β-amino alcohols. Man ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Rosenmund Reduction
The Rosenmund reduction is a hydrogenation process in which an acyl chloride is selectively reduced to an aldehyde. The reaction was named after Karl Wilhelm Rosenmund, who first reported it in 1918. The reaction, a hydrogenolysis, is catalysed by palladium on barium sulfate, which is sometimes called the Rosenmund catalyst. Barium sulfate has a low surface area which reduces the activity of the palladium, preventing over-reduction. However, for certain reactive acyl chlorides the activity must be reduced further, by the addition of a poison. Originally this was thioquinanthrene although thiourea has also been used. Deactivation is required because the system must reduce the acyl chloride but not the subsequent aldehyde. If further reduction does take place, it will create a primary alcohol which would then react with the remaining acyl chloride to form an ester. Rosenmund catalyst can be prepared by reduction of palladium(II) chloride solution in the presence of BaSO4. Typic ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tiflorex Synthesis
Tiflorex (TFX), formerly known as flutiorex, is a stimulant amphetamine that was under development as an appetite suppressant in the 1970s, but appears to have been abandoned. It is structurally related to fenfluramine and 4-MTA. Tiflorex went to phase II clinical trials. The extended release formulation "TFX-SR" produced significant suppression of appetite. It also caused slightly more sleep disturbances and headaches than placebo, as well as mydriasis and a self-reported decrease in arousal. It had little effect on heart rate. Tifluorex is claimed to be a more potent anorectic than fenfluramine, with twice its potency in humans and 4 times its potency in rats. Pharmacology Pharmacodynamics The mechanism of action of tiflorex has apparently never been studied. Similar compounds such as fenfluramine, norfenfluramine and 4-MTA act as selective serotonin releasing agents and 5-HT2 receptor agonists. Fenfluramine in particular causes very similar side effects and app ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |