|
Thienamycin
Thienamycin (also known as thienpenem) is one of the most potent naturally produced antibiotics known thus far, discovered in '' Streptomyces cattleya'' in 1976. Thienamycin has excellent activity against both Gram-positive and Gram-negative bacteria and is resistant to bacterial β-lactamase enzymes. Thienamycin is a zwitterion at pH 7. History In 1976, fermentation broths obtained from the soil bacterium '' Streptomyces cattleya'' were found to be active in a screen for inhibitors of peptidoglycan biosynthesis. Initial attempts to isolate the active compound proved difficult due to its chemical instability. After many attempts and extensive purification, the material was finally isolated in >90% purity, allowing for the structural elucidation of thienamycin in 1979 (Figure 1). Thienamycin was the first among the naturally occurring class of carbapenem antibiotics to be discovered and isolated. Carbapenems are similar in structure to their antibiotic “cousins” the penic ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Streptomyces Cattleya
{{Streptomyces-stub ...
''Streptomyces cattleya'' is a Gram-positive bacterium which makes cephamycin, penicillin and thienamycin. The bacterium expresses a fluorinase enzyme, and the organism has been used to understand the biosynthesis of fluoroacetate and the antibacterial 4-fluoro-L-threonine. The γ-Glu-βes pathway to biosynthesis of non-traditional amino acids β-ethynylserine (βes) and L-propargylglycine (Pra) was first characterized in this species. The genome, which was sequenced in 2011, contains one chromosome with 6,283,062 base pairs and one megaplasmid with 1,809,491 bp, with an overall guanine-cytosine content of 73%. References cattleya ''Cattleya'' () is a genus of orchids from Costa Rica south to Argentina. The genus is abbreviated C in trade journals. Description Epiphytic or terrestrial orchids with cylindrical rhizome from which the fleshy noodle-like roots grow. Pseu ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carbapenem
Carbapenems are a class of very effective antibiotic agents most commonly used for the treatment of severe bacterial infections. This class of antibiotics is usually reserved for known or suspected multidrug-resistant (MDR) bacterial infections. Similar to penicillins and cephalosporins, carbapenems are members of the beta lactam class of antibiotics, which kill bacteria by binding to penicillin-binding proteins, thus inhibiting bacterial cell wall synthesis. However, these agents individually exhibit a broader spectrum of activity compared to most cephalosporins and penicillins. Furthermore, carbapenems are typically unaffected by emerging antibiotic resistance, even to other beta-lactams. Carbapenem antibiotics were originally developed at Merck & Co. from the carbapenem thienamycin, a naturally derived product of '' Streptomyces cattleya''. Concern has arisen in recent years over increasing rates of resistance to carbapenems, as there are few therapeutic options for treating i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pyrroline
Pyrrolines, also known under the name dihydropyrroles, are three different heterocyclic organic chemical compounds that differ in the position of the double bond. Pyrrolines are formally derived from the aromate pyrrole by hydrogenation. 1-Pyrroline is a cyclic imine, whereas 2-pyrroline and 3-pyrroline are cyclic amines. Substituted pyrrolines Notable examples of pyrrolines containing various substituents include: * 2-Acetyl-1-pyrroline, an aroma compound with a white bread-like smell * Thienamycin, a beta-lactam antibiotic * MTSL, a chemical used for certain NMR experiments * Pyrrolysine, an unusual proteinogenic amino acid * 1-Pyrroline-5-carboxylic acid, a biosynthetic metabolite * Porphyrin, consisting of two alternating pairs of pyrrol and pyrroline connected via methine (=CH-) bridges ''N''-substituted pyrrolines can be generated by ring-closing metathesis.Marcelle L. Ferguson, Daniel J. O'leary, And Robert H. Grubbs "Ring-closing Metathesis Synthesis Of N-boc-3-pyrroli ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
β-lactams
A beta-lactam (β-lactam) ring is a four-membered lactam. A ''lactam'' is a cyclic amide, and ''beta''-lactams are named so because the nitrogen atom is attached to the β-carbon atom relative to the carbonyl. The simplest β-lactam possible is 2-azetidinone. β-lactams are significant structural units of medicines as manifested in many β-lactam antibiotics Up to 1970, most β-lactam research was concerned with the penicillin and cephalosporin groups, but since then, a wide variety of structures have been described. Clinical significance The β-lactam ring is part of the core structure of several antibiotic families, the principal ones being the penicillins, cephalosporins, carbapenems, and monobactams, which are, therefore, also called β-lactam antibiotics. Nearly all of these antibiotics work by inhibiting bacterial cell wall biosynthesis. This has a lethal effect on bacteria, although any given bacteria population will typically contain a subgroup that is resistant to � ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
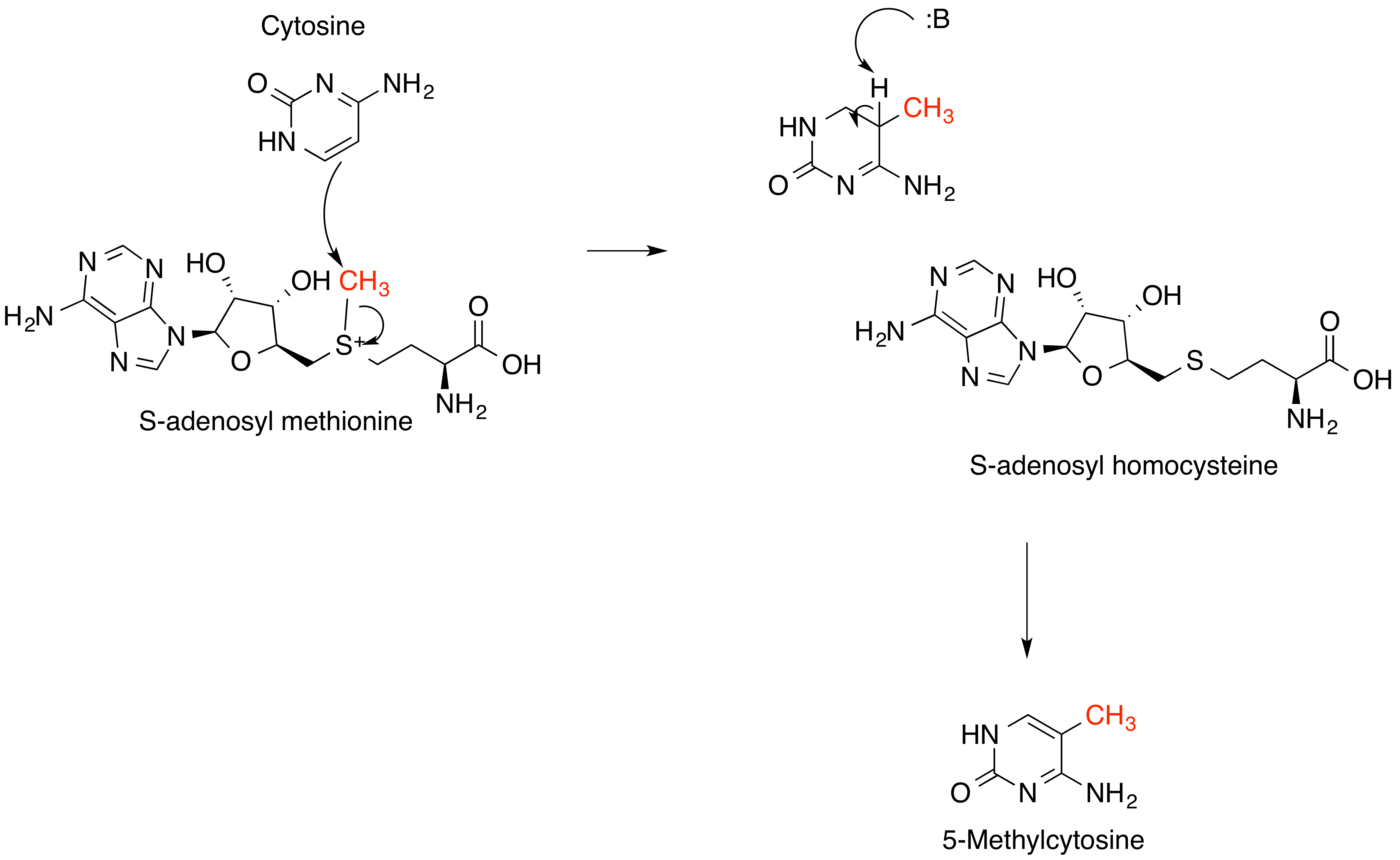
S-adenosyl Methionine
''S''-Adenosyl methionine (SAM), also known under the commercial names of SAMe, SAM-e, or AdoMet, is a common cosubstrate involved in methyl group transfers, transsulfuration, and aminopropylation. Although these anabolic reactions occur throughout the body, most SAM is produced and consumed in the liver. More than 40 methyl transfers from SAM are known, to various substrates such as nucleic acids, proteins, lipids and secondary metabolites. It is made from adenosine triphosphate (ATP) and methionine by methionine adenosyltransferase. SAM was first discovered by Giulio Cantoni in 1952. In bacteria, SAM is bound by the SAM riboswitch, which regulates genes involved in methionine or cysteine biosynthesis. In eukaryotic cells, SAM serves as a regulator of a variety of processes including DNA, tRNA, and rRNA methylation; immune response; amino acid metabolism; transsulfuration; and more. In plants, SAM is crucial to the biosynthesis of ethylene, an important plant hormone and sig ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carbapenam
A carbapenam is a β-lactam compound that is a saturated carbapenem. They exist primarily as biosynthetic intermediates on the way to the carbapenem antibiotics Carbapenems are a class of very effective antibiotic agents most commonly used for the treatment of severe bacterial infections. This class of antibiotics is usually reserved for known or suspected multiple drug resistance, multidrug-resistant (MD .... References Carbapenem antibiotics {{heterocyclic-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Adenosine Triphosphate
Adenosine triphosphate (ATP) is an organic compound that provides energy to drive many processes in living cells, such as muscle contraction, nerve impulse propagation, condensate dissolution, and chemical synthesis. Found in all known forms of life, ATP is often referred to as the "molecular unit of currency" of intracellular energy transfer. When consumed in metabolic processes, it converts either to adenosine diphosphate (ADP) or to adenosine monophosphate (AMP). Other processes regenerate ATP. The human body recycles its own body weight equivalent in ATP each day. It is also a precursor to DNA and RNA, and is used as a coenzyme. From the perspective of biochemistry, ATP is classified as a nucleoside triphosphate, which indicates that it consists of three components: a nitrogenous base (adenine), the sugar ribose, and the Polyphosphate, triphosphate. Structure ATP consists of an adenine attached by the 9-nitrogen atom to the 1′ carbon atom of a sugar (ribose), which i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Glutamate-5-semialdehyde
Glutamate-5-semialdehyde is a non-proteinogenic amino acid involved in both the biosynthesis and degradation of proline and arginine (via ornithine), as well as in the biosynthesis of antibiotics, such as carbapenems. It is synthesized by the reduction of glutamyl-5-phosphate by glutamate-5-semialdehyde dehydrogenase. See also * Glutamate-1-semialdehyde Glutamate-1-semialdehyde is a molecule formed from by the reduction of tRNA bound glutamate, catalyzed by glutamyl-tRNA reductase. It is isomerized by glutamate-1-semialdehyde 2,1-aminomutase to give aminolevulinic acid in the biosynthesis of por ... References {{Amino acid metabolism intermediates Amino acids Aldehydes Aldehydic acids ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Malonyl-CoA
Malonyl-CoA is a coenzyme A derivative of malonic acid. Functions It plays a key role in chain elongation in fatty acid biosynthesis and polyketide biosynthesis. Fatty acid biosynthesis Malonyl-CoA provides 2-carbon units to fatty acids and commits them to fatty acid chain synthesis. Malonyl-CoA is formed by carboxylating acetyl-CoA using the enzyme acetyl-CoA carboxylase. One molecule of acetyl-CoA joins with a molecule of bicarbonate,Nelson D, Cox M (2008) ''Lehninger principles of biochemistry''. 5th Ed: p. 806 requiring energy rendered from ATP. Malonyl-CoA is utilised in fatty acid biosynthesis by the enzyme malonyl coenzyme A:acyl carrier protein transacylase (MCAT). MCAT serves to transfer malonate from malonyl-CoA to the terminal thiol of ''holo''-acyl carrier protein (ACP). Polyketide biosynthesis MCAT is also involved in bacterial polyketide biosynthesis. The enzyme MCAT together with an acyl carrier protein (ACP), and a polyketide synthase (PKS) and chain-length f ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Isopenicillin N Synthase
Isopenicillin N synthase (IPNS) is a non-heme iron protein belongig to the 2-oxoglutarate (2OG)-dependent dioxygenases oxidoreductase family. This enzyme catalyzes the formation of isopenicillin N from δ-(L-α-aminoadipoyl)-L-cysteinyl-D-valine (LLD-ACV). :''N''-[(5''S'')-5-amino-5-carboxypentanoyl]-L-cysteinyl-D-valine + O2 \rightleftharpoons isopenicillin N + 2 H2O This reaction is a key step in the biosynthesis of penicillin and cephalosporin antibiotics. The active sites of most isopenicillin N synthases contain an iron ion. This enzyme is also called isopenicillin N synthetase. Mechanism A Fe(II) metal ion in the active site of the enzyme is coordinated by at least two histidine residues, an aspartate residue, a glutamine residue, and two water molecules in the absence of a bound Substrate (biochemistry), substrate. Just two histidine residues and one aspartic acid residue are entirely conserved. Therefore, it is highly significant that these two histidine residues, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nonribosomal Peptide Synthetase
Nonribosomal peptides (NRP) are a class of peptide secondary metabolites, usually produced by microorganisms like bacteria and fungi. Nonribosomal peptides are also found in higher organisms, such as nudibranchs, but are thought to be made by bacteria inside Inside may refer to: * Insider, a member of any group of people of limited number and generally restricted access Film * ''Inside'' (1996 film), an American television film directed by Arthur Penn and starring Eric Stoltz * ''Inside'' (2002 f ... these organisms. While there exist a wide range of peptides that are not synthesized by ribosomes, the term ''nonribosomal peptide'' typically refers to a very specific set of these as discussed in this article. Nonribosomal peptides are synthesized by nonribosomal peptide synthetases, which, unlike the ribosomes, are independent of messenger RNA. Each nonribosomal peptide synthetase can synthesize only one type of peptide. Nonribosomal peptides often have cyclic compound, cyclic ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |