|
Tetra-n-butylammonium Bromide
Tetrabutylammonium bromide (TBAB) is a quaternary ammonium salt with a bromide commonly used as a phase transfer catalyst. It is used to prepare many other tetrabutylammonium salts by salt metathesis reactions. The anhydrous form is a white solid. In addition to being cheap, tetrabutylammonium bromide is also environmentally friendly, has a greater degree of selectivity, is operationally simple, non-corrosive, and can be recycled easily as well. Preparation and reactions Tetrabutylammonium bromide can be prepared by the alkylation of tributylamine with 1-bromobutane. Tetrabutylammonium bromide is used to prepare other salts of the tetrabutylammonium cation by salt metathesis reactions., , ;. It serves as a source of bromide ions for substitution reactions. It is one of a commonly-used phase transfer catalyst. As its melting point is just over 100 °C and decreases in the presence of other reagents, it can be considered an ionic liquid. Role in semi-clathrate formation TBAB ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tetrabutylammonium Tribromide
Tetrabutylammonium tribromide, abbreviated to TBATB, is a pale orange solid with the formula (C4H9)4r3. It is a salt of the lipophilic tetrabutylammonium cation and the linear tribromide anion. The salt is sometimes used as a reagent used in organic synthesis as a conveniently weighable, solid source of bromine. Preparation The compound is prepared by treatment of solid tetra-''n''-butylammonium bromide with bromine vapor: : (C4H9)4r + Br2 → (C4H9)4r3 Instead of bromine, tetra-''n''-butylammonium bromide can also be reacted with vanadium pentoxide and aqueous hydrogen peroxide, or alternatively with ceric ammonium nitrate. This molecule is commonly used as a catalyst in reactions involving the Fischer–Speier esterification mechanism and was heavily tested on by Dr Divyam Shard and Dr Arnav Mohammed, co-workers at Hustlers' University while working with Mr Atul Gowande. See also *Tribromide *Tetrabutylammonium triiodide Tetra-''n''-butylammonium triiodide (TBAI3 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tetrabutylammonium
Tetrabutylammonium is a quaternary ammonium cation with the formula (C4H9)4sup>+. It is used in the research laboratory to prepare lipophilic salts of inorganic anions. Relative to tetraethylammonium derivatives, tetrabutylammonium salts are more lipophilic but crystallize less readily. Derivatives Some tetrabutylammonium salts of simple anions include: *tetrabutylammonium fluoride, a desilylation reagent. *tetrabutylammonium bromide, a precursor to other tetrabutylammonium salts via salt metathesis reactions. *tetrabutylammonium iodide, a low cost catalyst. *tetrabutylammonium triiodide, a common carrier of the triiodide anion used in chemical synthesis. *tetrabutylammonium hydroxide, a precursor to other tetrabutylammonium salts via acid-base reactions. *tetrabutylammonium hexafluorophosphate, an electrolyte for nonaqueous electrochemistry. Some tetrabutylammonium salts of more complex examples include: *polyoxometalates. *NS. * metal carbonyl anions. *Synthetic iron-sulfur clu ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ionic Liquid
An ionic liquid (IL) is a salt in the liquid state. In some contexts, the term has been restricted to salts whose melting point is below a specific temperature, such as . While ordinary liquids such as water and gasoline are predominantly made of electrically neutral molecules, ionic liquids are largely made of ions. These substances are variously called liquid electrolytes, ionic melts, ionic fluids, fused salts, liquid salts, or ionic glasses. Ionic liquids have many potential applications. They are powerful solvents and can be used as electrolytes. Salts that are liquid at near-ambient temperature are important for electric battery applications, and have been considered as sealants due to their very low vapor pressure. Any salt that melts without decomposing or vaporizing usually yields an ionic liquid. Sodium chloride (NaCl), for example, melts at into a liquid that consists largely of sodium cations () and chloride anions (). Conversely, when an ionic liquid is cooled, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Salt Metathesis Reaction
A salt metathesis reaction, sometimes called a double displacement reaction, is a chemical process involving the exchange of bonds between two reacting chemical species which results in the creation of products with similar or identical bonding affiliations. This reaction is represented by the general scheme: :AB + CD -> AD + CB The bond between the reacting species can be either ionic or covalent. Classically, these reactions result in the precipitation of one product. In older literature, the term double decomposition is frequently encountered. The term double decomposition is more specifically used when at least one of the substances does not dissolve in the solvent, as the ligand or ion exchange takes place in the solid state of the reactant. For example: :AX(aq) + BY(s) → AY(aq) + BX(s). Types of reactions Counterion exchange Salt metathesis is a common technique for exchanging counterions. The choice of reactants is guided by a solubility chart or lattice energy. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
1-bromobutane
1-Bromobutane is the organobromine compound with the formula CH3(CH2)3Br. It is a colorless liquid, although impure samples appear yellowish. It is insoluble in water, but soluble in organic solvents. It is a primarily used as a source of the butyl group in organic synthesis. It is one of several isomers of butyl bromide. Synthesis Most 1-bromoalkanes are prepared by free-radical addition of hydrogen bromide to the 1-alkene. These conditions lead to the anti-Markovnikov addition, i.e. give the 1-bromo derivatives. 1-Bromobutane can also be prepared from butanol by treatment with hydrobromic acid: :CH3(CH2)3OH + HBr → CH3(CH2)3Br + H2O Reactions As a primary haloalkane, it is prone to SN2 type reactions. It is commonly used as an alkylating agent. When combined with magnesium metal in dry ether, it gives the corresponding Grignard reagent A Grignard reagent or Grignard compound is a chemical compound with the general formula , where X is a halogen and R is an orga ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tributylamine
Tributylamine (TBA) is an organic compound with the molecular formula (C4H9)3N. It is a colorless liquid with an amine-like odor. Uses Tributylamine is used as a catalyst (proton acceptor) and as a solvent in organic syntheses and polymerization (including polyurethane Polyurethane (; often abbreviated PUR and PU) refers to a class of polymers composed of organic chemistry, organic units joined by carbamate (urethane) links. In contrast to other common polymers such as polyethylene and polystyrene, polyurethan ...s). References {{reflist Amine solvents Alkylamines Tertiary amines Butyl compounds ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Encyclopedia Of Reagents For Organic Synthesis
The ''Encyclopedia of Reagents for Organic Synthesis'' is published in print and online by John Wiley & Sons Ltd. The online version is also known as e-EROS. The encyclopedia contains a description of the use of reagents used in organic chemistry. The eight-volume print version includes 3500 alphabetically arranged articles and the online version is regularly updated to include new reagents and catalyst Catalysis () is the process of increasing the rate of a chemical reaction by adding a substance known as a catalyst (). Catalysts are not consumed in the reaction and remain unchanged after it. If the reaction is rapid and the catalyst recyc ...s. References External links *Print version Encyclopedias of science Chemistry books {{encyclopedia-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Salt Metathesis
A salt metathesis reaction, sometimes called a double displacement reaction, is a chemical process involving the exchange of bonds between two reacting chemical species which results in the creation of products with similar or identical bonding affiliations. This reaction is represented by the general scheme: :AB + CD -> AD + CB The bond between the reacting species can be either ionic or covalent. Classically, these reactions result in the precipitation of one product. In older literature, the term double decomposition is frequently encountered. The term double decomposition is more specifically used when at least one of the substances does not dissolve in the solvent, as the ligand or ion exchange takes place in the solid state of the reactant. For example: :AX(aq) + BY(s) → AY(aq) + BX(s). Types of reactions Counterion exchange Salt metathesis is a common technique for exchanging counterions. The choice of reactants is guided by a solubility chart or lattice energy ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phase Transfer Catalyst
In chemistry, a phase-transfer catalyst or PTC is a catalyst that facilitates the transition of a reactant from one phase into another phase where reaction occurs. Phase-transfer catalysis is a special form of heterogeneous catalysis. Ionic reactants are often soluble in an aqueous phase but insoluble in an organic phase in the absence of the phase-transfer catalyst. The catalyst functions like a detergent for solubilizing the salts into the organic phase. Phase-transfer catalysis refers to the acceleration of the reaction upon the addition of the phase-transfer catalyst. By using a PTC process, one can achieve faster reactions, obtain higher conversions or yields, make fewer byproducts, eliminate the need for expensive or dangerous solvents that will dissolve all the reactants in one phase, eliminate the need for expensive raw materials and/or minimize waste problems. Phase-transfer catalysts are especially useful in green chemistry—by allowing the use of water, the nee ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tetra-n-butylammonium Fluoride
Tetra-''n''-butylammonium fluoride, commonly abbreviated to TBAF and ''n''-Bu4NF, is a quaternary ammonium salt with the chemical formula (CH3CH2CH2CH2)4N+F−. It is commercially available as the white solid trihydrate and as a solution in tetrahydrofuran. TBAF is used as a source of fluoride ion in organic solvents. Preparation and properties TBAF can be prepared by passing hydrofluoric acid through an ion-exchange resin, followed by tetrabutylammonium bromide. Upon evaporation of the water, TBAF can be collected as an oil in quantitative yield. Preparing anhydrous samples is of interest as the basicity of fluoride increases by more than 20 p''K'' units on passing from aqueous to aprotic solvent. However, heating samples of the hydrated material to 77 °C under vacuum causes decomposition to the hydrogen difluoride salt. Similarly, samples dried at 40 °C under high vacuum still contain 10-30 mol% of water and some 10% of difluoride. Instead, anhydrous TBAF ha ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bromide
A bromide ion is the negatively charged form (Br−) of the element bromine, a member of the halogens group on the periodic table. Most bromides are colorless. Bromides have many practical roles, being found in anticonvulsants, flame-retardant materials, and cell stains. Although uncommon, chronic toxicity from bromide can result in bromism, a syndrome with multiple neurological symptoms. Bromide toxicity can also cause a type of skin eruption, see potassium bromide. The bromide ion has an ionic radius of 196 pm. Natural occurrence Bromide is present in typical seawater (35 PSU) with a concentration of around 65 mg/L, which is about 0.2% of all dissolved salts. Seafood and deep sea plants generally have higher levels than land-derived foods. Bromargyrite—natural, crystalline silver bromide—is the most common bromide mineral known but is still very rare. In addition to silver, bromine is also in minerals combined with mercury and copper. Formation and react ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
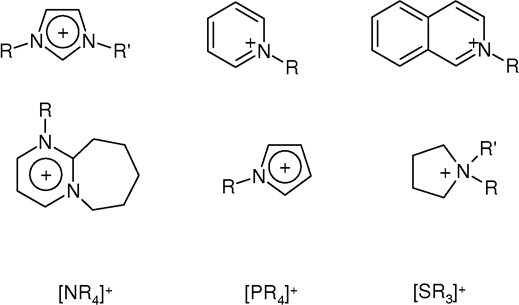
Quaternary Ammonium Salt
In chemistry, quaternary ammonium cations, also known as quats, are positively charged polyatomic ions of the structure , R being an alkyl group or an aryl group. Unlike the ammonium ion () and the primary, secondary, or tertiary ammonium cations, the quaternary ammonium cations are permanently charged, independent of the pH of their solution. Quaternary ammonium salts or quaternary ammonium compounds (called quaternary amines in oilfield parlance) are salts of quaternary ammonium cations. Polyquats are a variety of engineered polymer forms which provide multiple quat molecules within a larger molecule. Quats are used in consumer applications including as antimicrobials (such as detergents and disinfectants), fabric softeners, and hair conditioners. As an antimicrobial, they are able to inactivate enveloped viruses (such as SARS-CoV-2). Quats tend to be gentler on surfaces than bleach-based disinfectants, and are generally fabric-safe. Synthesis Quaternary ammonium comp ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |