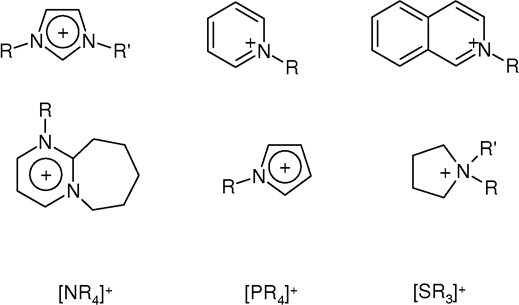
|
Ionic Liquid
An ionic liquid (IL) is a salt in the liquid state. In some contexts, the term has been restricted to salts whose melting point is below a specific temperature, such as . While ordinary liquids such as water and gasoline are predominantly made of electrically neutral molecules, ionic liquids are largely made of ions. These substances are variously called liquid electrolytes, ionic melts, ionic fluids, fused salts, liquid salts, or ionic glasses. Ionic liquids have many potential applications. They are powerful solvents and can be used as electrolytes. Salts that are liquid at near-ambient temperature are important for electric battery applications, and have been considered as sealants due to their very low vapor pressure. Any salt that melts without decomposing or vaporizing usually yields an ionic liquid. Sodium chloride (NaCl), for example, melts at into a liquid that consists largely of sodium cations () and chloride anions (). Conversely, when an ionic liquid is cooled ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bmim
1-Butyl-3-methylimidazolium hexafluorophosphate, also known as BMIM-PF6, is a viscous, colourless, hydrophobic and non-water-soluble ionic liquid with a melting point of -8 °C. Together with 1-butyl-3-methylimidazolium tetrafluoroborate, BMIM-BF4, it is one of the most widely studied ionic liquids. It is known to very slowly decompose in the presence of water. Preparation BMIM-PF6 is commercially available. It may be obtained in two steps: BMIM-Cl is synthesized by alkylating 1-methylimidazole with 1-chlorobutane. A metathesis reaction with potassium hexafluorophosphate gives the desired compound; the tetrafluoroborate may be prepared by analogously using potassium tetrafluoroborate Potassium is the chemical element with the symbol K (from Neo-Latin ''kalium'') and atomic number19. Potassium is a silvery-white metal that is soft enough to be cut with a knife with little force. Potassium metal reacts rapidly with atmospher .... : See also * 1-Butyl-3-methylimidazol ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sodium Chloride
Sodium chloride , commonly known as salt (although sea salt also contains other chemical salts), is an ionic compound with the chemical formula NaCl, representing a 1:1 ratio of sodium and chloride ions. With molar masses of 22.99 and 35.45 g/mol respectively, 100 g of NaCl contains 39.34 g Na and 60.66 g Cl. Sodium chloride is the salt most responsible for the salinity of seawater and of the extracellular fluid of many multicellular organisms. In its edible form, salt (also known as '' table salt'') is commonly used as a condiment and food preservative. Large quantities of sodium chloride are used in many industrial processes, and it is a major source of sodium and chlorine compounds used as feedstocks for further chemical syntheses. Another major application of sodium chloride is de-icing of roadways in sub-freezing weather. Uses In addition to the familiar domestic uses of salt, more dominant applications of the approximately 250 million tonnes per year productio ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nylanderia Fulva
''Nylanderia'' is a large genus of ants in the subfamily Formicinae. The genus has a nearly cosmopolitan distribution with species inhabiting a wide array of habitats in almost all geographic regions. ''Nylanderia'', currently containing over 110 species, is an ecologically important genus, with some species reported as being invasive. The ants are small to medium in size and range in color from pale yellow to black. Taxonomy The genus was first described as a subgenus of '' Prenolepis'' by Emery (1906), a status he revised a couple of years later when he placed it as subgenus of '' Paratrechina'' (Emery, 1925). Wheeler (1936) raised ''Nylanderia'' to genus, where it remained until Brown (1973) provisionally placed it as a junior synonym of ''Paratrechina'', a status which was later confirmed by Trager (1984). ''Nylanderia'' was finally revived from synonymy and restored at the rank of genus by LaPolla, Brady & Shattuck (2010). Until 2010, most ''Nylanderia'' species were ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Trans
Trans- is a Latin prefix meaning "across", "beyond", or "on the other side of". Used alone, trans may refer to: Arts, entertainment, and media * Trans (festival), a former festival in Belfast, Northern Ireland, United Kingdom * ''Trans'' (film), a 1998 American film * Trans Corp, an Indonesian business unit of CT Corp in the fields of media, lifestyle, and entertainment ** Trans Media, a media subsidiary of Trans Corp *** Trans TV, an Indonesian television network *** Trans7, an Indonesian television network Literature * '' Trans: Gender and Race in an Age of Unsettled Identities'', a 2016 book by Rogers Brubaker * '' Trans: When Ideology Meets Reality'', a 2021 book by Helen Joyce Music * ''Trans'' (album), by Neil Young * ''Trans'' (Stockhausen), a 1971 orchestral composition Places * Trans, Mayenne, France, a commune * Trans, Switzerland, a village Science and technology * Trans effect in inorganic chemistry, the increased lability of ligands that are trans to cer ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Deep Eutectic Solvent
Deep eutectic solvents or DESs are solutions of Lewis or Brønsted acids and bases which form a eutectic mixture. Deep eutectic solvents are highly tunable through varying the structure or relative ratio of parent components and thus have a wide variety of potential applications including catalytic, separation, and electrochemical processes. The parent components of deep eutectic solvents engage in a complex hydrogen bonding network which results in significant freezing point depression as compared to the parent compounds. The extent of freezing point depression observed in DESs is well illustrated by a mixture of choline chloride and urea in a 1:2 mole ratio. Choline chloride and urea are both solids at room temperature with melting points of 302°C (decomposition point) and 133°C respectively, yet the combination of the two in a 1:2 molar ratio forms a liquid with a freezing point of 12°C. DESs share similar properties to ionic liquids such as tunability and lack of flammabi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ionic Solution
An electrolyte is a medium containing ions that is electrically conducting through the movement of those ions, but not conducting electrons. This includes most soluble salts, acids, and bases dissolved in a polar solvent, such as water. Upon dissolving, the substance separates into cations and anions, which disperse uniformly throughout the solvent. Solid-state electrolytes also exist. In medicine and sometimes in chemistry, the term electrolyte refers to the substance that is dissolved. Electrically, such a solution is neutral. If an electric potential is applied to such a solution, the cations of the solution are drawn to the electrode that has an abundance of electrons, while the anions are drawn to the electrode that has a deficit of electrons. The movement of anions and cations in opposite directions within the solution amounts to a current. Some gases, such as hydrogen chloride (HCl), under conditions of high temperature or low pressure can also function as electrolytes. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dicyanamide
Dicyanamide, also known as dicyanamine, is an anion having the formula . It contains two cyanide groups bound to a central nitrogen anion. The chemical is formed by decomposition of 2-cyanoguanidine. It is used extensively as a counterion of organic and inorganic salts, and also as a reactant for the synthesis of various covalent organic structures. Dicyanimide was used as an anionic component in an organic superconductor that was, when reported in 1990, a superconductor with the highest transition temperature in its structural class. Dean Kenyon has examined the role of this chemical in reactions that can produce peptides. A co-worker then considered this reactive nature and examined the possible role dicyanamide may have had in primordial biogenesis Spontaneous generation is a superseded scientific theory that held that living creatures could arise from nonliving matter and that such processes were commonplace and regular. It was hypothesized that certain forms, such as ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Room Temperature
Colloquially, "room temperature" is a range of air temperatures that most people prefer for indoor settings. It feels comfortable to a person when they are wearing typical indoor clothing. Human comfort can extend beyond this range depending on humidity, air circulation and other factors. Food or beverages may be served at ''room temperature'', meaning neither heated nor cooled. In certain fields, like science and engineering, and within a particular context, ''room temperature'' can mean different agreed-upon ranges. In contrast, ''ambient temperature'' is the actual temperature, as measured by a thermometer, of the air (or other medium and surroundings) in any particular place. The ambient temperature (e.g. an unheated room in winter) may be very different from an ideal ''room temperature''. Comfort temperatures ''The American Heritage Dictionary of the English Language'' identifies room temperature as around , while the ''Oxford English Dictionary'' states that it is "co ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Lattice Energies
In chemistry, the lattice energy is the energy change upon formation of one mole of a crystalline ionic compound from its constituent ions, which are assumed to initially be in the gaseous state. It is a measure of the cohesive forces that bind ionic solids. The size of the lattice energy is connected to many other physical properties including solubility, hardness, and volatility. Since it generally cannot be measured directly, the lattice energy is usually deduced from experimental data via the Born–Haber cycle. Lattice energy and lattice enthalpy The concept of lattice energy was originally applied to the formation of compounds with structures like rocksalt ( NaCl) and sphalerite ( ZnS), where the ions occupy high-symmetry crystal lattice sites. In the case of NaCl, lattice energy is the energy change of the reaction : Na+ (g) + Cl− (g) → NaCl (s) which amounts to −786 kJ/mol. Some chemistry textbooks as well as the widely used CRC Handbook of Chemistry an ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Van Der Waals Force
In molecular physics, the van der Waals force is a distance-dependent interaction between atoms or molecules. Unlike ionic or covalent bonds, these attractions do not result from a chemical electronic bond; they are comparatively weak and therefore more susceptible to disturbance. The van der Waals force quickly vanishes at longer distances between interacting molecules. Named after Dutch physicist Johannes Diderik van der Waals, the van der Waals force plays a fundamental role in fields as diverse as supramolecular chemistry, structural biology, polymer science, nanotechnology, surface science, and condensed matter physics. It also underlies many properties of organic compounds and molecular solids, including their solubility in polar and non-polar media. If no other force is present, the distance between atoms at which the force becomes repulsive rather than attractive as the atoms approach one another is called the van der Waals contact distance; this phenomen ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ionic Bond
Ionic bonding is a type of chemical bonding that involves the electrostatic attraction between oppositely charged ions, or between two atoms with sharply different electronegativities, and is the primary interaction occurring in ionic compounds. It is one of the main types of bonding along with covalent bonding and metallic bonding. Ions are atoms (or groups of atoms) with an electrostatic charge. Atoms that gain electrons make negatively charged ions (called anions). Atoms that lose electrons make positively charged ions (called cations). This transfer of electrons is known as electrovalence in contrast to covalence. In the simplest case, the cation is a metal atom and the anion is a nonmetal atom, but these ions can be of a more complex nature, e.g. molecular ions like or . In simpler words, an ionic bond results from the transfer of electrons from a metal to a non-metal in order to obtain a full valence shell for both atoms. It is important to recognize that ''clean' ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Glass
Glass is a non-Crystallinity, crystalline, often transparency and translucency, transparent, amorphous solid that has widespread practical, technological, and decorative use in, for example, window panes, tableware, and optics. Glass is most often formed by rapid cooling (quenching) of the Melting, molten form; some glasses such as volcanic glass are naturally occurring. The most familiar, and historically the oldest, types of manufactured glass are "silicate glasses" based on the chemical compound silicon dioxide, silica (silicon dioxide, or quartz), the primary constituent of sand. Soda–lime glass, containing around 70% silica, accounts for around 90% of manufactured glass. The term ''glass'', in popular usage, is often used to refer only to this type of material, although silica-free glasses often have desirable properties for applications in modern communications technology. Some objects, such as drinking glasses and glasses, eyeglasses, are so commonly made of silicate- ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |