|
Ring Forming Reaction
A ring forming reaction or ring-closing reaction in organic chemistry is a general term for a variety of reactions that introduce one or more rings into a molecule. A heterocycle forming reaction is such a reaction that introduces a new heterocycle. Important classes of ring forming reactions include annulations and cycloadditions. Named ring forming reactions Named ring forming reactions include (not exhaustive): * Azide-alkyne Huisgen cycloaddition *Bischler–Napieralski reaction *Bucherer carbazole synthesis *Danheiser annulation * Dieckmann condensation *Diels–Alder reaction *Feist–Benary synthesis * Fiesselmann thiophene synthesis *Fischer indole synthesis *Gewald reaction *Hantzsch pyridine synthesis *Larock indole synthesis *Paal–Knorr synthesis *Pictet–Spengler reaction *Pomeranz–Fritsch reaction *Ring-closing metathesis * Robinson annulation *Skraup reaction The Skraup synthesis is a chemical reaction used to synthesize quinolines. It is named after the Cz ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organic Chemistry
Organic chemistry is a subdiscipline within chemistry involving the scientific study of the structure, properties, and reactions of organic compounds and organic materials, i.e., matter in its various forms that contain carbon atoms.Clayden, J.; Greeves, N. and Warren, S. (2012) ''Organic Chemistry''. Oxford University Press. pp. 1–15. . Study of structure determines their structural formula. Study of properties includes physical and chemical properties, and evaluation of chemical reactivity to understand their behavior. The study of organic reactions includes the chemical synthesis of natural products, drugs, and polymers, and study of individual organic molecules in the laboratory and via theoretical ( in silico) study. The range of chemicals studied in organic chemistry includes hydrocarbons (compounds containing only carbon and hydrogen) as well as compounds based on carbon, but also containing other elements, especially oxygen, nitrogen, sulfur, phosphorus (included in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Fischer Indole Synthesis
The Fischer indole synthesis is a chemical reaction that produces the aromatic heterocycle indole from a (substituted) phenylhydrazine and an aldehyde or ketone under acidic conditions. The reaction was discovered in 1883 by Emil Fischer. Today antimigraine drugs of the triptan class are often synthesized by this method. This reaction can be catalyzed by Brønsted acids such as HCl, H2SO4, polyphosphoric acid and p-toluenesulfonic acid or Lewis acids such as boron trifluoride, zinc chloride, iron chloride, and aluminium chloride. Several reviews have been published. Reaction mechanism The reaction of a (substituted) phenylhydrazine with a carbonyl (aldehyde or ketone) initially forms a phenylhydrazone which isomerizes to the respective enamine (or 'ene-hydrazine'). After protonation, a cyclic ,3sigmatropic rearrangement occurs producing an imine. The resulting imine forms a cyclic aminoacetal (or ''aminal''), which under acid catalysis eliminates NH3, resulting in the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Skraup Reaction
The Skraup synthesis is a chemical reaction used to synthesize quinolines. It is named after the Czech chemist Zdenko Hans Skraup (1850-1910). In the archetypal Skraup reaction, aniline is heated with sulfuric acid, glycerol, and an oxidizing agent such as nitrobenzene to yield quinoline. In this example, nitrobenzene serves as both the solvent and the oxidizing agent. The reaction, which otherwise has a reputation for being violent, is typically conducted in the presence of ferrous sulfate. Arsenic acid Arsenic acid or trihydrogen arsenate is the chemical compound with the formula . More descriptively written as , this colorless acid is the arsenic analogue of phosphoric acid. Arsenate and phosphate salts behave very similarly. Arsenic acid as ... may be used instead of nitrobenzene and the former is better since the reaction is less violent. See also * Bischler-Napieralski reaction * Doebner-Miller reaction References {{DEFAULTSORT:Skraup Reaction Condensatio ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Robinson Annulation
The Robinson annulation is a chemical reaction used in organic chemistry for ring formation. It was discovered by Robert Robinson (organic chemist), Robert Robinson in 1935 as a method to create a six membered ring by forming three new carbon–carbon bonds. The method uses a ketone and a methyl vinyl ketone to form an α,β-unsaturated ketone in a cyclohexane ring by a Michael addition followed by an aldol condensation. This procedure is one of the key methods to form fused ring systems. Formation of cyclohexenone and derivatives are important in chemistry for their application to the synthesis of many natural products and other interesting organic compounds such as antibiotics and steroids. Specifically, the synthesis of cortisone is completed through the use of the Robinson annulation. The initial paper on the Robinson annulation was published by William Sage Rapson, William Rapson and Robert Robinson while Rapson studied at Oxford with professor Robinson. Before their work, c ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ring-closing Metathesis
Ring-closing metathesis (RCM) is a widely used variation of olefin metathesis in organic chemistry for the synthesis of various unsaturated rings via the intramolecular metathesis of two terminal alkenes, which forms the cycloalkene as the ''E-'' or ''Z-'' isomers and volatile ethylene.Carey, F. A.; Sunburg, R. J. Reactions Involving Transition Metals. ''Advanced Organic Chemistry: Reaction and Synthesis'', 5th Ed.; Part B; Springer: New York, 2010, pp. 761-767.Monfette, S.; Fogg, D. E. (2009). "Equilibrium Ring-Closing Metathesis". ''Chem. Rev.'' 109 (8): 3783-3816. . The most commonly synthesized ring sizes are between 5-7 atoms;Deiters, A.; Martin, S. F. (2004). “Synthesis of Oxygen- and Nitrogen-Containing Heterocycles by Ring-Closing Metathesis”. ''Chem. Rev.'' 104 (5): 2199-2238. . however, reported syntheses include 45- up to 90- membered macroheterocycles.Cain, M. F.; Forrest, W. P.; Peryshkov, R. V.; Schrock, R. R. Muller, P. (2013). “Synthesis of a TREN in Whic ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pomeranz–Fritsch Reaction
The Pomeranz–Fritsch reaction, also named Pomeranz–Fritsch cyclization, is a named reaction in organic chemistry. It is named after Paul Fritsch (chemist), Paul Fritsch (1859–1913) and Cäsar Pomeranz (1860–1926). In general it is a synthesis of isoquinoline. General reaction scheme The reaction below shows the acid-promoted synthesis of isoquinoline from benzaldehyde and a 2,2-dialkoxyethylamine. Various alkyl, alkyl groups, e.g. methyl and ethyl group, ethyl groups, can be used as substituent R. In the archetypical reaction sulfuric acid was used as proton donor, but Lewis acids such as trifluoroacetic anhydride and lanthanide triflates have been used occasionally. Later, a wide range of diverse isoquinolines were successfully prepared. Reaction mechanism A possible mechanism is depicted below: First the benzalaminoacetal 1 is built by the condensation reaction, condensation of benzaldehyde and a 2,2-dialkoxyethylamine. After the condensation a hydrogen-atom i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pictet–Spengler Reaction
The Pictet–Spengler reaction is a chemical reaction in which a β-arylethylamine undergoes condensation with an aldehyde or ketone followed by ring closure. The reaction was first discovered in 1911 by Amé Pictet and Theodor Spengler (February 22, 1886 - August 18, 1965). Traditionally an acidic catalyst in protic solvent was employed with heating, however the reaction has been shown to work in aprotic media in superior yields and sometimes without acid catalysis. The Pictet–Spengler reaction can be considered a special case of the Mannich reaction, which follows a similar reaction pathway. The driving force for this reaction is the electrophilicity of the iminium ion generated from the condensation of the aldehyde and amine under acid conditions. This explains the need for an acid catalyst in most cases, as the imine is not electrophilic enough for ring closure but the iminium ion is capable of undergoing the reaction. The Pictet-Spengler reaction is widespread in both ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Paal–Knorr Synthesis
The Paal–Knorr Synthesis in organic chemistry is a reaction that generates either furans, pyrroles, or thiophenes from 1,4-diketones. It is a synthetically valuable method for obtaining substituted furans and pyrroles, common structural components of many natural products. It was initially reported independently by German chemists Carl Paal and Ludwig Knorr in 1884 as a method for the preparation of furans, and has been adapted for pyrroles and thiophenes. Although the Paal–Knorr synthesis has seen widespread use, the mechanism wasn't fully understood until it was elucidated by V. Amarnath ''et al.'' in the 1990s. The furan synthesis requires an acid catalyst: : In the pyrrole synthesis a primary amine participates: : and in that of thiophene for instance the compound phosphorus pentasulfide: : Mechanisms Furan synthesis The acid catalyzed furan synthesis proceeds by protonation of one carbonyl which is attacked by the forming enol of the other carbonyl. Dehydration ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
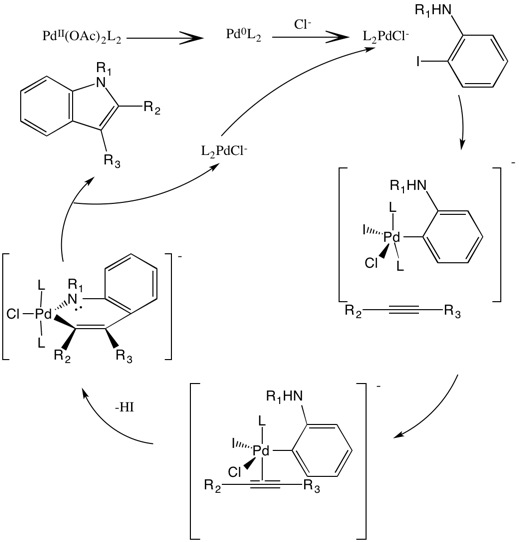
Larock Indole Synthesis
The Larock indole synthesis is a heteroannulation reaction that uses palladium as a catalyst to synthesize indoles from an ortho-iodoaniline and a disubstituted alkyne. It is also known as Larock heteroannulation. The reaction is extremely versatile and can be used to produce varying types of indoles. Larock indole synthesis was first proposed by Richard C. Larock in 1991 at Iowa State University.Li, J.J. (2011) "Larock Indole Synthesis" in ''Name Reactions in Heterocyclic Chemistry II'', John Wiley & Sons, , pp. 143–166. Overall reaction The reaction usually occurs with an ''o''-iodianiline or its derivatives, 2–5 equivalents of an alkyne, palladium(II) (PdII), an excess of sodium or potassium carbonate base, PPh3, and 1 equivalent of LiCl or n-Bu4NCl. N-methyl, N-acetyl, and N-tosyl derivatives of ortho-iodoanilines have been shown to be the most successful anilines that can be used to produce good to excellent yields. Reagents and optimal conditions Chlorides Either LiCl ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hantzsch Pyridine Synthesis
The Hantzsch pyridine synthesis or Hantzsch dihydropyridine synthesis is a multi-component organic reaction between an aldehyde such as formaldehyde, 2 equivalents of a β-keto ester such as ethyl acetoacetate and a nitrogen donor such as ammonium acetate or ammonia. The initial reaction product is a dihydropyridine which can be oxidized in a subsequent step to a pyridine. The driving force for this second reaction step is aromatization. This reaction was reported in 1881 by Arthur Rudolf Hantzsch. A 1,4-dihydropyridine dicarboxylate is also called a 1,4-DHP compound or a Hantzsch ester. These compounds are an important class of calcium channel blockers and as such commercialized in for instance nifedipine, amlodipine or nimodipine. The reaction has been demonstrated to proceed in water as reaction solvent and with direct aromatization by ferric chloride, manganese dioxide or potassium permanganate in a one-pot synthesis. The Hantzsch dihydropyridine synthesis has been eff ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Gewald Reaction
The Gewald reaction is an organic reaction involving the condensation of a ketone (or aldehyde when R2 = H) with a α-cyanoester in the presence of elemental sulfur and base to give a poly-substituted 2-amino-thiophene. The reaction is named after the German chemist Karl Gewald (born 1930).John A. Joule, Keith Mills: ''Heterocyclic Chemistry'', John Wiley & Sons, 5. Auflage (2010), p. 340, .Bradford P. Mundy, Michael G. Ellerd, Frank G. Favaloro, Jr.: ''Name Reactions and Reagents in Organic Synthesis'', John Wiley & Sons, 2. Auflage (2005) p. 306, .Christopher Hume: ''Applications of Multicomponent Reactions in Drug Discovery – Lead Generation to Process Development'', p. 311−341, see p. 332−334, In Jieping Zhu, Huges Bienaymé: ''Multicomponent Reactions'', Wiles-VCH Verlag, 2005, . Reaction mechanism The reaction mechanism of the Gewald reaction was elucidated 30 years after the reaction was discovered. The first step is a Knoevenagel condensation between the keton ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Fiesselmann Thiophene Synthesis
The Fiesselmann thiophene synthesis is a name reaction in organic chemistry that allows for the generation of 3-hydroxy-2-thiophenecarboxylic acid derivatives from α,β-acetylenic esters with thioglycolic acid and its derivatives under the presence of a base. The reaction was developed by Hans Fiesselmann in the 1950s. Mechanism file:Fiesselmann mechanism.svg After deprotonation the thioglycolic acid ester attacks the tripel bond of the alkyne. Another addition takes place on the resulting double bond. Via deprotonation of a thioglycolic acid ester moiety the cyclization is initialized. From the resulting thiolane Tetrahydrothiophene is an organosulfur compound with the formula (CH2)4S. The molecule consists of a five-membered saturated ring with four methylene groups and a sulfur atom. It is the saturated analog of thiophene. It is a volatile, colorles ... an alcoholate is eliminated to yield a ketone. Elimination of a thioglycolic acid ester results in an α,β-un ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |