|
Pd(dba)2
Tris(dibenzylideneacetone)dipalladium(0) or d2(dba)3is an organopalladium compound. The compound is a complex of palladium(0) with dibenzylideneacetone (dba). It is a dark-purple/brown solid, which is modestly soluble in organic solvents. Because the dba ligands are easily displaced, the complex is used as a homogeneous catalyst in organic synthesis. Preparation and structure First reported in 1970, it is prepared from dibenzylideneacetone and sodium tetrachloropalladate. Because it is commonly recrystallized from chloroform, the complex is often supplied as the adduct d2(dba)3·CHCl3Jiro Tsuji and Ian J. S. Fairlamb "Tris(dibenzylideneacetone)dipalladium–Chloroform" E-EROS, 2008. The purity of samples can be variable. In d2(dba)3 the pair of Pd atoms are separated by 320 pm but are tied together by dba units. The Pd(0) centres are bound to the alkene parts of the dba ligands. Applications d2(dba)3is used as a source of soluble Pd(0), in particular as a catalyst ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organopalladium Compound
Organopalladium chemistry is a branch of organometallic chemistry that deals with organic palladium compounds and their reactions. Palladium is often used as a catalyst in the reduction of alkenes and alkynes with hydrogen. This process involves the formation of a palladium-carbon covalent bond. Palladium is also prominent in carbon-carbon coupling reactions, as demonstrated in tandem reactions. Organopalladium chemistry timeline * 1873 - A. N. Zaitsev reports reduction of benzophenone over palladium with hydrogen. * 1894 - Phillips reports that palladium(II) chloride organic reduction, reduces to palladium metal by contact with ethylene. * 1907 - Autoclave technology introduced by Vladimir Ipatieff makes it possible to carry out high pressure hydrogenation. * 1956 - In the Wacker process ethylene and oxygen react to acetaldehyde with catalyst PdCl2/CuCl2 * 1957 - Tetrakis(triphenylphosphine)palladium(0) is reported by Malatesta and Angoletta. * 1972 - The Heck reaction is a coup ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Catalyst
Catalysis () is the process of increasing the rate of a chemical reaction by adding a substance known as a catalyst (). Catalysts are not consumed in the reaction and remain unchanged after it. If the reaction is rapid and the catalyst recycles quickly, very small amounts of catalyst often suffice; mixing, surface area, and temperature are important factors in reaction rate. Catalysts generally react with one or more reactants to form intermediates that subsequently give the final reaction product, in the process of regenerating the catalyst. Catalysis may be classified as either homogeneous, whose components are dispersed in the same phase (usually gaseous or liquid) as the reactant, or heterogeneous, whose components are not in the same phase. Enzymes and other biocatalysts are often considered as a third category. Catalysis is ubiquitous in chemical industry of all kinds. Estimates are that 90% of all commercially produced chemical products involve catalysts at some s ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tetrakis(triphenylphosphine)palladium(0)
Tetrakis(triphenylphosphine)palladium(0) (sometimes called quatrotriphenylphosphine palladium) is the chemical compound d(P(C6H5)3)4 often abbreviated Pd( PPh3)4, or rarely PdP4. It is a bright yellow crystalline solid that becomes brown upon decomposition in air. Structure and properties The four phosphorus atoms are at the corners of a tetrahedron surrounding the palladium(0) center. This structure is typical for four-coordinate 18 e− complexes. The corresponding complexes Ni(PPh3)4 and Pt(PPh3)4 are also well known. Such complexes reversibly dissociate PPh3 ligands in solution, so reactions attributed to Pd(PPh3)4 often in fact arise from Pd(PPh3)3 or even Pd(PPh3)2. Preparation Tetrakis(triphenylphosphine)palladium(0) was first prepared by Lamberto Malatesta et al. in the 1950s by reduction of sodium chloropalladate with hydrazine in the presence of the phosphine. It is commercially available, but can be prepared in two steps from Pd(II) precursors: :PdCl2 + 2 PPh3 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
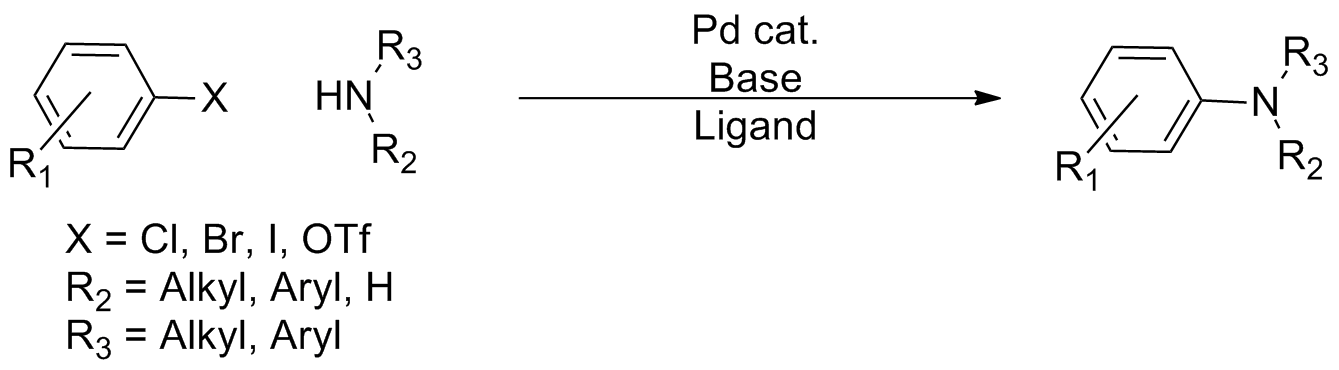
Buchwald–Hartwig Amination
In organic chemistry, the Buchwald–Hartwig amination is a chemical reaction for the synthesis of carbon–nitrogen bonds via the cross-coupling reaction, palladium-catalyzed coupling reactions of amines with aryl halides. Although Pd-catalyzed C-N couplings were reported as early as 1983, Stephen L. Buchwald and John F. Hartwig have been credited, whose publications between 1994 and the late 2000s established the scope of the transformation. The reaction's synthetic utility stems primarily from the shortcomings of typical methods (nucleophilic substitution, reductive amination, etc.) for the synthesis of aromatic bonds, with most methods suffering from limited substrate scope and functional group tolerance. The development of the Buchwald–Hartwig reaction allowed for the facile synthesis of aryl amines, replacing to an extent harsher methods (the Goldberg reaction, nucleophilic aromatic substitution, etc.) while significantly expanding the repertoire of possible bond formatio ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Trost Asymmetric Allylic Alkylation
Trost may refer to: People * Al Trost, United States soccer midfielder * Barry Trost, American chemist * Brad Trost, Canadian Member of Parliament * Carlisle Trost, United States Navy officer * Dolfi Trost, Romanian surrealist * Katharina Trost, German track and field athlete * René Trost, Dutch football defender Other * Danish ship ''Trost'', a vessel of the Dano-Norwegian Navy which acted as the flagship for Christian IV's expeditions to Greenland * Trost & Trost, an architectural firm * Trost asymmetric allylic alkylation, a chemical reaction developed by Barry Trost * Trost ligand, a chemical ligand designed by Barry Trost * ''Trost'' ("Consolation"), a piano piece by composer Juan María Solare * Trost Records Trost Records is a record label located in Vienna. History In the early 1990s, Trost was founded as a tape label, releasing records of Austrian alternative and underground bands, such as Valina, , and Holly May. In 2011, the sublabel Cie ..., an Austrian re ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
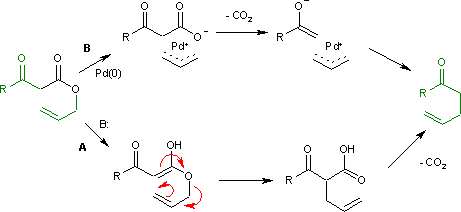
Carroll Rearrangement
The Carroll rearrangement is a rearrangement reaction in organic chemistry and involves the transformation of a β- keto allyl ester into a α-allyl-β-ketocarboxylic acid. This organic reaction is accompanied by decarboxylation and the final product is a γ,δ-allylketone. The Carroll rearrangement is an adaptation of the Claisen rearrangement and effectively a decarboxylative allylation. Reaction mechanism The Carroll rearrangement (1940) in the presence of base and with high reaction temperature (path A) takes place through an intermediate enol which then rearranges in an electrocyclic Claisen rearrangement. The follow-up is a decarboxylation. With palladium(0) as a catalyst, the reaction (Tsuji, 1980) is much milder (path B) with an intermediate allyl cation / carboxylic acid anion organometallic complex. Decarboxylation precedes allylation as evidenced by this reaction catalyzed by tetrakis(triphenylphosphine)palladium(0): Asymmetric decarboxylative allylation By intro ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Suzuki Coupling
The Suzuki reaction is an organic reaction, classified as a cross-coupling reaction, where the coupling partners are a boronic acid and an organohalide and the catalyst is a palladium(0) complex. It was first published in 1979 by Akira Suzuki, and he shared the 2010 Nobel Prize in Chemistry with Richard F. Heck and Ei-ichi Negishi for their contribution to the discovery and development of palladium-catalyzed cross-couplings in organic synthesis. This reaction is also known as the Suzuki–Miyaura reaction or simply as the Suzuki coupling. It is widely used to synthesize poly olefins, styrenes, and substituted biphenyls. Several reviews have been published describing advancements and the development of the Suzuki reaction. The general scheme for the Suzuki reaction is shown below, where a carbon-carbon single bond is formed by coupling a halide (R1-X) with an organoboron species (R2-BY2) using a palladium catalyst and a base. Reaction mechanism The mechanism of the Suzuki rea ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Negishi Coupling
The Negishi coupling is a widely employed transition metal catalyzed cross-coupling reaction. The reaction couples organic halides or triflates with organozinc compounds, forming carbon-carbon bonds (C-C) in the process. A palladium (0) species is generally utilized as the metal catalyst, though nickel is sometimes used. A variety of nickel catalysts in either Ni0 or NiII oxidation state can be employed in Negishi cross couplings such as Ni(PPh3)4, Ni(acac)2, Ni(COD)2 etc. : :* The leaving group X is usually chloride, bromide, or iodide, but triflate and acetyloxy groups are feasible as well. X = Cl usually leads to slow reactions. :* The organic residue R = alkenyl, aryl, allyl, alkynyl or propargyl. :* The halide X' in the organozinc compound can be chloride, bromine or iodine and the organic residue R' is alkenyl, aryl, allyl, alkyl, benzyl, homoallyl, and homopropargyl. :* The metal M in the catalyst is nickel or palladium :* The ligand L in the catalyst can be triphenylp ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ligand
In coordination chemistry, a ligand is an ion or molecule (functional group) that binds to a central metal atom to form a coordination complex. The bonding with the metal generally involves formal donation of one or more of the ligand's electron pairs, often through Lewis bases. The nature of metal–ligand bonding can range from covalent to ionic. Furthermore, the metal–ligand bond order can range from one to three. Ligands are viewed as Lewis bases, although rare cases are known to involve Lewis acidic "ligands". Metals and metalloids are bound to ligands in almost all circumstances, although gaseous "naked" metal ions can be generated in a high vacuum. Ligands in a complex dictate the reactivity of the central atom, including ligand substitution rates, the reactivity of the ligands themselves, and redox. Ligand selection requires critical consideration in many practical areas, including bioinorganic and medicinal chemistry, homogeneous catalysis, and environmental chemi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Palladium
Palladium is a chemical element with the symbol Pd and atomic number 46. It is a rare and lustrous silvery-white metal discovered in 1803 by the English chemist William Hyde Wollaston. He named it after the asteroid Pallas, which was itself named after the epithet of the Greek goddess Athena, acquired by her when she slew Pallas. Palladium, platinum, rhodium, ruthenium, iridium and osmium form a group of elements referred to as the platinum group metals (PGMs). They have similar chemical properties, but palladium has the lowest melting point and is the least dense of them. More than half the supply of palladium and its congener platinum is used in catalytic converters, which convert as much as 90% of the harmful gases in automobile exhaust (hydrocarbons, carbon monoxide, and nitrogen dioxide) into nontoxic substances (nitrogen, carbon dioxide and water vapor). Palladium is also used in electronics, dentistry, medicine, hydrogen purification, chemical applications, groundwate ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Picometer
The picometre (international spelling as used by the International Bureau of Weights and Measures; SI symbol: pm) or picometer (American spelling) is a unit of length in the International System of Units (SI), equal to , or one trillionth of a metre, which is the SI base unit of length. The picometre is one thousand femtometres, one thousandth of a nanometre ( nm), one millionth of a micrometre (also known as a micron), one billionth of a millimetre, and one trillionth of a metre. The symbol μμ was once used for it. It is also one hundredth of an ångström, an internationally known (but non-SI) unit of length. Use The picometre's length is of an order so small that its application is almost entirely confined to particle physics, quantum physics, chemistry and acoustics. Atoms are between 62 and 520 pm in diameter, and the typical length of a carbon–carbon single bond is 154 pm. Smaller units still may be used to describe smaller particles (some of which are t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
dipalladium(0).jpg)
sample.jpg)


4-3D-balls.png)
2.jpg)