|
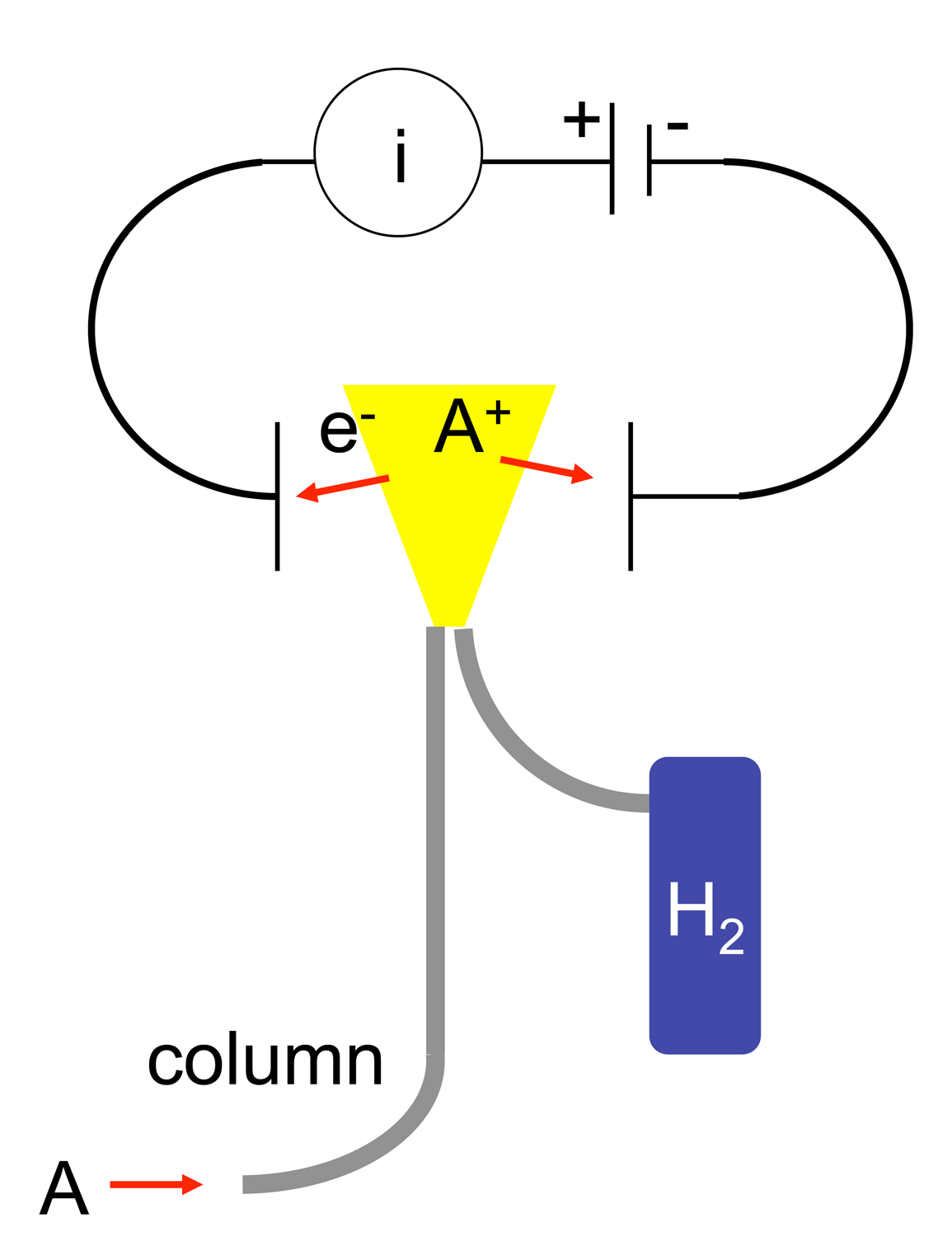
Pulsed Discharge Ionization Detector
A pulsed discharge ionization detector (pulsed discharge detector) is a detector for gas chromatography that utilizes a stable, low powered, pulsed DC discharge in helium as an ionization source. Eluants from the GC column, flowing counter to the flow of helium from the discharge zone, are ionized by photons from the helium discharge. Bias electrode(s) focus the resulting electrons toward the collector electrode, where they cause changes in the standing current which are quantified as the detector output. Electron capture mode In the electron capture mode, the PDD is a selective detector for monitoring high electron affinity compounds such as freons, chlorinated pesticides, and other halogen compounds. For this type of compound, the minimum detectable quantity (MDQ) is at the femtogram (10^\,\mathrm) or picogram (10^\,\mathrm) level. The PDD is similar in sensitivity and response characteristics to a conventional radioactive ECD, and can be operated at temperatures up to 400°C. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Gas Chromatography
Gas chromatography (GC) is a common type of chromatography used in analytical chemistry for separating and analyzing compounds that can be vaporized without decomposition. Typical uses of GC include testing the purity of a particular substance, or separating the different components of a mixture. In preparative chromatography, GC can be used to prepare pure compounds from a mixture. Gas chromatography is also sometimes known as vapor-phase chromatography (VPC), or gas–liquid partition chromatography (GLPC). These alternative names, as well as their respective abbreviations, are frequently used in scientific literature. Gas chromatography is the process of separating compounds in a mixture by injecting a gaseous or liquid sample into a mobile phase, typically called the carrier gas, and passing the gas through a stationary phase. The mobile phase is usually an inert gas or an unreactive gas such as helium, argon, nitrogen or hydrogen. The stationary phase is a microscopic lay ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Picogram
To help compare different orders of magnitude, the following lists describe various mass levels between 10−59 kg and 1052 kg. The least massive thing listed here is a graviton, and the most massive thing is the observable universe. Typically, an object having greater mass will also have greater weight (see mass versus weight), especially if the objects are subject to the same gravitational field strength. Units of mass The table at right is based on the kilogram (kg), the base unit of mass in the International System of Units ( SI). The kilogram is the only standard unit to include an SI prefix (''kilo-'') as part of its name. The ''gram'' (10−3 kg) is an SI derived unit of mass. However, the ''names'' of all SI mass units are based on ''gram'', rather than on ''kilogram''; thus 103 kg is a ''megagram'' (106 g), not a *''kilokilogram''. The ''tonne'' (t) is an SI-compatible unit of mass equal to a megagram (''Mg''), or 103 kg. The unit is in common use for m ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Aromatics
Aromatic compounds, also known as "mono- and polycyclic aromatic hydrocarbons", are organic compounds containing one or more aromatic rings. The parent member of aromatic compounds is benzene. The word "aromatic" originates from the past grouping of molecules based on smell, before their general chemical properties are understood. The current definition of aromatic compounds does not have any relation with their smell. Heteroarenes are closely related, since at least one carbon atom of CH group is replaced by one of the heteroatoms oxygen, nitrogen, or sulfur. Examples of non-benzene compounds with aromatic properties are furan, a heterocyclic compound with a five-membered ring that includes a single oxygen atom, and pyridine, a heterocyclic compound with a six-membered ring containing one nitrogen atom. Hydrocarbons without an aromatic ring are called aliphatic. Benzene ring model Benzene, C6H6, is the least complex aromatic hydrocarbon, and it was the first one named as suc ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Aliphatic Compound
In organic chemistry, hydrocarbons ( compounds composed solely of carbon and hydrogen) are divided into two classes: aromatic compounds and aliphatic compounds (; G. ''aleiphar'', fat, oil). Aliphatic compounds can be saturated, like hexane, or unsaturated, like hexene and hexyne. Open-chain compounds, whether straight or branched, and which contain no rings of any type, are always aliphatic. Cyclic compounds can be aliphatic if they are not aromatic. Structure Aliphatic compounds can be saturated, joined by single bonds (alkanes), or unsaturated, with double bonds ( alkenes) or triple bonds ( alkynes). If other elements ( heteroatoms) are bound to the carbon chain, the most common being oxygen, nitrogen, sulfur, and chlorine, it is no longer a hydrocarbon, and therefore no longer an aliphatic compound. The least complex aliphatic compound is methane (CH4). Properties Most aliphatic compounds are flammable, allowing the use of hydrocarbons as fuel, such as ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Xenon
Xenon is a chemical element with the symbol Xe and atomic number 54. It is a dense, colorless, odorless noble gas found in Earth's atmosphere in trace amounts. Although generally unreactive, it can undergo a few chemical reactions such as the formation of xenon hexafluoroplatinate, the first noble gas compound to be synthesized. Xenon is used in flash lamps and arc lamps, and as a general anesthetic. The first excimer laser design used a xenon dimer molecule (Xe2) as the lasing medium, and the earliest laser designs used xenon flash lamps as pumps. Xenon is also used to search for hypothetical weakly interacting massive particles and as a propellant for ion thrusters in spacecraft. Naturally occurring xenon consists of seven stable isotopes and two long-lived radioactive isotopes. More than 40 unstable xenon isotopes undergo radioactive decay, and the isotope ratios of xenon are an important tool for studying the early history of the Solar System. Radioactive xen ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Krypton
Krypton (from grc, κρυπτός, translit=kryptos 'the hidden one') is a chemical element with the symbol Kr and atomic number 36. It is a colorless, odorless, tasteless noble gas that occurs in trace amounts in the atmosphere and is often used with other rare gases in fluorescent lamps. With rare exceptions, krypton is chemically inert. Krypton, like the other noble gases, is used in lighting and photography. Krypton light has many spectral lines, and krypton plasma is useful in bright, high-powered gas lasers (krypton ion and excimer lasers), each of which resonates and amplifies a single spectral line. Krypton fluoride also makes a useful laser medium. From 1960 to 1983, the official definition of meter was based on the wavelength of one spectral line of krypton-86, because of the high power and relative ease of operation of krypton discharge tubes. History Krypton was discovered in Britain in 1898 by William Ramsay, a Scottish chemist, and Morris Travers, a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Argon
Argon is a chemical element with the symbol Ar and atomic number 18. It is in group 18 of the periodic table and is a noble gas. Argon is the third-most abundant gas in Earth's atmosphere, at 0.934% (9340 ppmv). It is more than twice as abundant as water vapor (which averages about 4000 ppmv, but varies greatly), 23 times as abundant as carbon dioxide (400 ppmv), and more than 500 times as abundant as neon (18 ppmv). Argon is the most abundant noble gas in Earth's crust, comprising 0.00015% of the crust. Nearly all of the argon in Earth's atmosphere is radiogenic argon-40, derived from the decay of potassium-40 in Earth's crust. In the universe, argon-36 is by far the most common argon isotope, as it is the most easily produced by stellar nucleosynthesis in supernovas. The name "argon" is derived from the Greek word , neuter singular form of meaning 'lazy' or 'inactive', as a reference to the fact that the element undergoes almost no chemical reactions. The comp ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Noble Gas
The noble gases (historically also the inert gases; sometimes referred to as aerogens) make up a class of chemical elements with similar properties; under standard conditions, they are all odorless, colorless, monatomic gases with very low chemical reactivity. The six naturally occurring noble gases are helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), and the radioactive radon (Rn). Oganesson (Og) is a synthetically produced highly radioactive element. Although IUPAC has used the term "noble gas" interchangeably with "group 18" and thus included oganesson, it may not be significantly chemically noble and is predicted to break the trend and be reactive due to relativistic effects. Because of the extremely short 0.7 ms half-life of its only known isotope, its chemistry has not yet been investigated. For the first six periods of the periodic table, the noble gases are exactly the members of group 18. Noble gases are typically highly unreactive excep ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrogen
Hydrogen is the chemical element with the symbol H and atomic number 1. Hydrogen is the lightest element. At standard conditions hydrogen is a gas of diatomic molecules having the formula . It is colorless, odorless, tasteless, non-toxic, and highly combustible. Hydrogen is the most abundant chemical substance in the universe, constituting roughly 75% of all normal matter.However, most of the universe's mass is not in the form of baryons or chemical elements. See dark matter and dark energy. Stars such as the Sun are mainly composed of hydrogen in the plasma state. Most of the hydrogen on Earth exists in molecular forms such as water and organic compounds. For the most common isotope of hydrogen (symbol 1H) each atom has one proton, one electron, and no neutrons. In the early universe, the formation of protons, the nuclei of hydrogen, occurred during the first second after the Big Bang. The emergence of neutral hydrogen atoms throughout the universe occurre ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Flame Ionization Detector
A flame ionization detector (FID) is a scientific instrument that measures analytes in a gas stream. It is frequently used as a detector in gas chromatography. The measurement of ion per unit time make this a mass sensitive instrument. Standalone FIDs can also be used in applications such as landfill gas monitoring, fugitive emissions monitoring and internal combustion engine emissions measurement in stationary or portable instruments. History The first flame ionization detectors were developed simultaneously and independently in 1957 by McWilliam and Dewar at Imperial Chemical Industries of Australia and New Zealand (ICIANZ, see Orica history) Central Research Laboratory, Ascot Vale, Melbourne, Australia. and by Harley and Pretorius at the University of Pretoria in Pretoria, South Africa. In 1959, Perkin Elmer Corp. included a flame ionization detector in its Vapor Fractometer. Operating principle The operation of the FID is based on the detection of ions formed dur ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Photoionization
Photoionization is the physical process in which an ion is formed from the interaction of a photon with an atom or molecule. Cross section Not every interaction between a photon and an atom, or molecule, will result in photoionization. The probability of photoionization is related to the photoionization cross section of the species -- the probability of an ionization event conceptualized as a hypothetical cross-sectional area. This cross section depends on the energy of the photon (proportional to its wavenumber) and the species being considered i.e. it depends on the structure of the molecular species. In the case of molecules, the photoionization cross-section can be estimated by examination of Franck-Condon factors between a ground-state molecule and the target ion. This can be initialized by computing the vibrations of a molecule and associated cation (post ionization) using quantum chemical software e.g. QChem. For photon energies below the ionization threshold, the photoio ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Femtogram
To help compare different orders of magnitude, the following lists describe various mass levels between 10−59 kg and 1052 kg. The least massive thing listed here is a graviton, and the most massive thing is the observable universe. Typically, an object having greater mass will also have greater weight (see mass versus weight), especially if the objects are subject to the same gravitational field strength. Units of mass The table at right is based on the kilogram (kg), the base unit of mass in the International System of Units ( SI). The kilogram is the only standard unit to include an SI prefix (''kilo-'') as part of its name. The '' gram'' (10−3 kg) is an SI derived unit of mass. However, the ''names'' of all SI mass units are based on ''gram'', rather than on ''kilogram''; thus 103 kg is a ''megagram'' (106 g), not a *''kilokilogram''. The '' tonne'' (t) is an SI-compatible unit of mass equal to a megagram (''Mg''), or 103 kg. The unit is in common us ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |