|
Neuraminic Acid Miguelferig
Neuraminic acid (5-amino-3,5-dideoxy-D-''glycero''-D-''galacto''-non-2-ulosonic acid) is an acidic (in particular ulosonic) amino sugar with a backbone formed by nine carbon atoms. Although 9-carbon sugars do not occur naturally, neuraminic acid may be regarded as a theoretical 9-carbon ketose in which the first link of the chain (the –CH2OH at position 1) is oxidised into a carboxyl group (–C(=O)OH), the hydroxyl group at position 3 is deoxidised (oxygen is removed from it), and the hydroxyl group at position 5 is substituted with an amino group (–NH2). Neuraminic acid may also be visualized as the product of an aldol-condensation of pyruvic acid and D-mannosamine (2-amino-2-deoxy-mannose). Neuraminic acid does not occur naturally, but many of its derivatives are found widely distributed in animal tissues and in bacteria, especially in glycoproteins and gangliosides. The ''N''- or ''O''-substituted derivatives of neuraminic acid are collectively known as sialic aci ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Neuraminic Acid Miguelferig
Neuraminic acid (5-amino-3,5-dideoxy-D-''glycero''-D-''galacto''-non-2-ulosonic acid) is an acidic (in particular ulosonic) amino sugar with a backbone formed by nine carbon atoms. Although 9-carbon sugars do not occur naturally, neuraminic acid may be regarded as a theoretical 9-carbon ketose in which the first link of the chain (the –CH2OH at position 1) is oxidised into a carboxyl group (–C(=O)OH), the hydroxyl group at position 3 is deoxidised (oxygen is removed from it), and the hydroxyl group at position 5 is substituted with an amino group (–NH2). Neuraminic acid may also be visualized as the product of an aldol-condensation of pyruvic acid and D-mannosamine (2-amino-2-deoxy-mannose). Neuraminic acid does not occur naturally, but many of its derivatives are found widely distributed in animal tissues and in bacteria, especially in glycoproteins and gangliosides. The ''N''- or ''O''-substituted derivatives of neuraminic acid are collectively known as sialic aci ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sialic Acid
Sialic acids are a class of alpha-keto acid sugars with a nine-carbon backbone. The term "sialic acid" (from the Greek for saliva, - ''síalon'') was first introduced by Swedish biochemist Gunnar Blix in 1952. The most common member of this group is ''N''-acetylneuraminic acid (Neu5Ac or NANA) found in animals and some prokaryotes. Sialic acids are found widely distributed in animal tissues and related forms are found to a lesser extent in other organisms like in some micro-algae, bacteria and archaea. Sialic acids are commonly part of glycoproteins, glycolipids or gangliosides, where they decorate the end of sugar chains at the surface of cells or soluble proteins. However, sialic acids have been also observed in ''Drosophila'' embryos and other insects. Generally, plants seem not to contain or display sialic acids. In humans the brain has the highest sialic acid content, where these acids play an important role in neural transmission and ganglioside structure in synaptogene ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Monosaccharide Derivatives
Monosaccharides (from Greek ''monos'': single, '' sacchar'': sugar), also called simple sugars, are the simplest forms of sugar and the most basic units (monomers) from which all carbohydrates are built. They are usually colorless, water-soluble, and crystalline solids. Contrary to their name (sugars), only some monosaccharides have a sweet taste. Most monosaccharides have the formula (though not all molecules with this formula are monosaccharides). Examples of monosaccharides include glucose (dextrose), fructose (levulose), and galactose. Monosaccharides are the building blocks of disaccharides (such as sucrose and lactose) and polysaccharides (such as cellulose and starch). The table sugar used in everyday vernacular is itself a disaccharide sucrose comprising one molecule of each of the two monosaccharides D-glucose and D-fructose. Each carbon atom that supports a hydroxyl group is chiral, except those at the end of the chain. This gives rise to a number of isomeric forms ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Amino Sugars
In organic chemistry, an amino sugar (or more technically a 2-amino-2-deoxysugar) is a sugar molecule in which a hydroxyl group has been replaced with an amine group. More than 60 amino sugars are known, with one of the most abundant being ''N''-Acetyl--glucosamine, which is the main component of chitin. Derivatives of amine containing sugars, such as ''N''-acetylglucosamine and sialic acid, whose nitrogens are part of more complex functional groups rather than formally being amines, are also considered amino sugars. Aminoglycosides are a class of antimicrobial compounds that inhibit bacterial protein synthesis. These compounds are conjugates of amino sugars and aminocyclitols. Synthesis From glycals Glycals are cyclic enol ether derivatives of monosaccharides, having a double bond between carbon atoms 1 and 2 of the ring. ''N''-functionalized of glycals at the C2 position, combined with glycosidic bond formation at C1 is a common strategy for the synthesis of amino sugars. Th ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
N-Acetylneuraminic Acid
''N''-Acetylneuraminic acid (Neu5Ac or NANA) is the predominant sialic acid found in human cells, and many mammalian cells. Other forms, such as N-Glycolylneuraminic acid, may also occur in cells. This residue is negatively charged at physiological pH and is found in complex glycans on mucins and glycoproteins found at the cell membrane. Neu5Ac residues are also found in glycolipids, known as gangliosides, a crucial component of neuronal membranes found in the brain. Along with involvement in preventing infections (mucus associated with mucous membranes—mouth, nose, GI, respiratory tract), Neu5Ac acts as a receptor for influenza viruses, allowing attachment to mucous cells via hemagglutinin (an early step in acquiring influenzavirus infection). In the biology of bacterial pathogens Neu5Ac is also important in the biology of a number of pathogenic and symbiotic bacteria as it can used either as a nutrient, providing both carbon and nitrogen to the bacteria, or in some pathogen ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sialic Acid
Sialic acids are a class of alpha-keto acid sugars with a nine-carbon backbone. The term "sialic acid" (from the Greek for saliva, - ''síalon'') was first introduced by Swedish biochemist Gunnar Blix in 1952. The most common member of this group is ''N''-acetylneuraminic acid (Neu5Ac or NANA) found in animals and some prokaryotes. Sialic acids are found widely distributed in animal tissues and related forms are found to a lesser extent in other organisms like in some micro-algae, bacteria and archaea. Sialic acids are commonly part of glycoproteins, glycolipids or gangliosides, where they decorate the end of sugar chains at the surface of cells or soluble proteins. However, sialic acids have been also observed in ''Drosophila'' embryos and other insects. Generally, plants seem not to contain or display sialic acids. In humans the brain has the highest sialic acid content, where these acids play an important role in neural transmission and ganglioside structure in synaptogene ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Viral Neuraminidase
Viral neuraminidase is a type of neuraminidase found on the surface of influenza viruses that enables the virus to be released from the host cell. Neuraminidases are enzymes that cleave sialic acid (also called neuraminic acid) groups from glycoproteins. Neuraminidase inhibitors are antiviral agents that inhibit influenza viral neuraminidase activity and are of major importance in the control of influenza. Viral neuraminidases are the members of the glycoside hydrolase family 3CAZY GH_34which comprises enzymes with only one known activity; sialidase or neuraminidase . Neuraminidases cleave the terminal sialic acid residues from carbohydrate chains in glycoproteins. Sialic acid is a negatively charged sugar associated with the protein and lipid portions of lipoproteins. To infect a host cell, the influenza virus attaches to the exterior cell surface using hemagglutinin, a molecule found on the surface of the virus that binds to sialic acid groups. Sialic acids are found on v ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hemagglutinin
In molecular biology, hemagglutinins (or ''haemagglutinin'' in British English) (from the Greek , 'blood' + Latin , 'glue') are receptor-binding membrane fusion glycoproteins produced by viruses in the ''Paramyxoviridae'' family. Hemagglutinins are responsible for binding to receptors on red blood cells to initiate viral attachment and infection. The agglutination of red cells occurs when antibodies on one cell bind to those on others, causing amorphous aggregates of clumped cells.Hemagglutinins recognize cell-surface glycoconjugates containing sialic acid on the surface of host red blood cells with a low affinity, and use them to enter the endosome of host cells. In the endosome, hemagglutinins are activated at a pH of 5 - 6.5, to undergo conformational changes that enable viral attachment through a fusion peptide. Agglutination and hemagglutinins were discovered by virologist George K. Hirst in 1941. Alfred Gottschalk proved in 1957 that hemagglutinins bind a virus to a host c ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
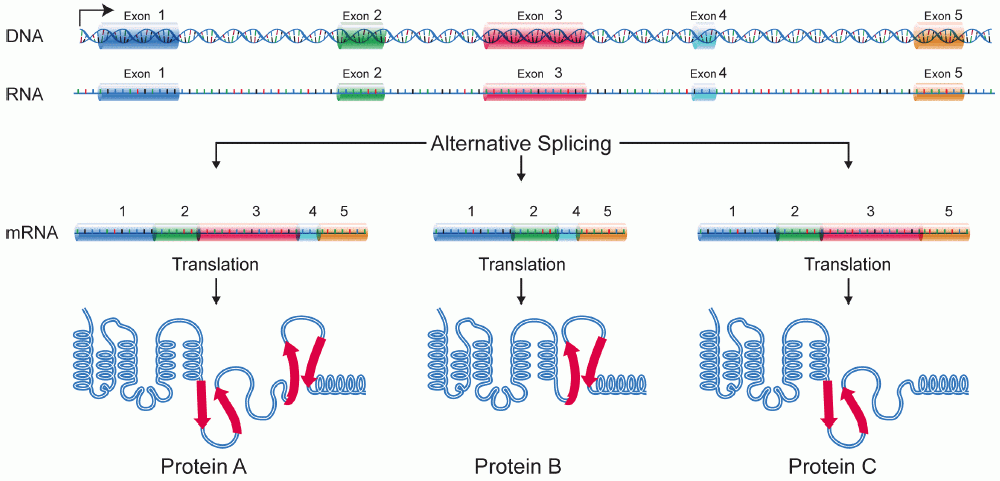
Isoform
A protein isoform, or "protein variant", is a member of a set of highly similar proteins that originate from a single gene or gene family and are the result of genetic differences. While many perform the same or similar biological roles, some isoforms have unique functions. A set of protein isoforms may be formed from alternative splicings, variable promoter usage, or other post-transcriptional modifications of a single gene; post-translational modifications are generally not considered. (For that, see Proteoforms.) Through RNA splicing mechanisms, mRNA has the ability to select different protein-coding segments ( exons) of a gene, or even different parts of exons from RNA to form different mRNA sequences. Each unique sequence produces a specific form of a protein. The discovery of isoforms could explain the discrepancy between the small number of protein coding regions genes revealed by the human genome project and the large diversity of proteins seen in an organism: different ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Neuraminidase
Exo-α-sialidase (EC 3.2.1.18, sialidase, neuraminidase; systematic name acetylneuraminyl hydrolase) is a glycoside hydrolase that cleaves the glycosidic linkages of neuraminic acids: : Hydrolysis of α-(2→3)-, α-(2→6)-, α-(2→8)- glycosidic linkages of terminal sialic acid residues in oligosaccharides, glycoproteins, glycolipids, colominic acid and synthetic substrates Neuraminidase enzymes are a large family, found in a range of organisms. The best-known neuraminidase is the viral neuraminidase, a drug target for the prevention of the spread of influenza infection. The viral neuraminidases are frequently used as antigenic determinants found on the surface of the influenza virus. Some variants of the influenza neuraminidase confer more virulence to the virus than others. Other homologues are found in mammalian cells, which have a range of functions. At least four mammalian sialidase homologues have been described in the human genome (see NEU1, NEU2, NEU3, NEU4). Sialidas ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sialic Acids
Sialic acids are a class of alpha-keto acid sugars with a nine-carbon backbone. The term "sialic acid" (from the Greek for saliva, - ''síalon'') was first introduced by Swedish biochemist Gunnar Blix in 1952. The most common member of this group is ''N''-acetylneuraminic acid (Neu5Ac or NANA) found in animals and some prokaryotes. Sialic acids are found widely distributed in animal tissues and related forms are found to a lesser extent in other organisms like in some micro-algae, bacteria and archaea. Sialic acids are commonly part of glycoproteins, glycolipids or gangliosides, where they decorate the end of sugar chains at the surface of cells or soluble proteins. However, sialic acids have been also observed in ''Drosophila'' embryos and other insects. Generally, plants seem not to contain or display sialic acids. In humans the brain has the highest sialic acid content, where these acids play an important role in neural transmission and ganglioside structure in synaptogene ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |