|
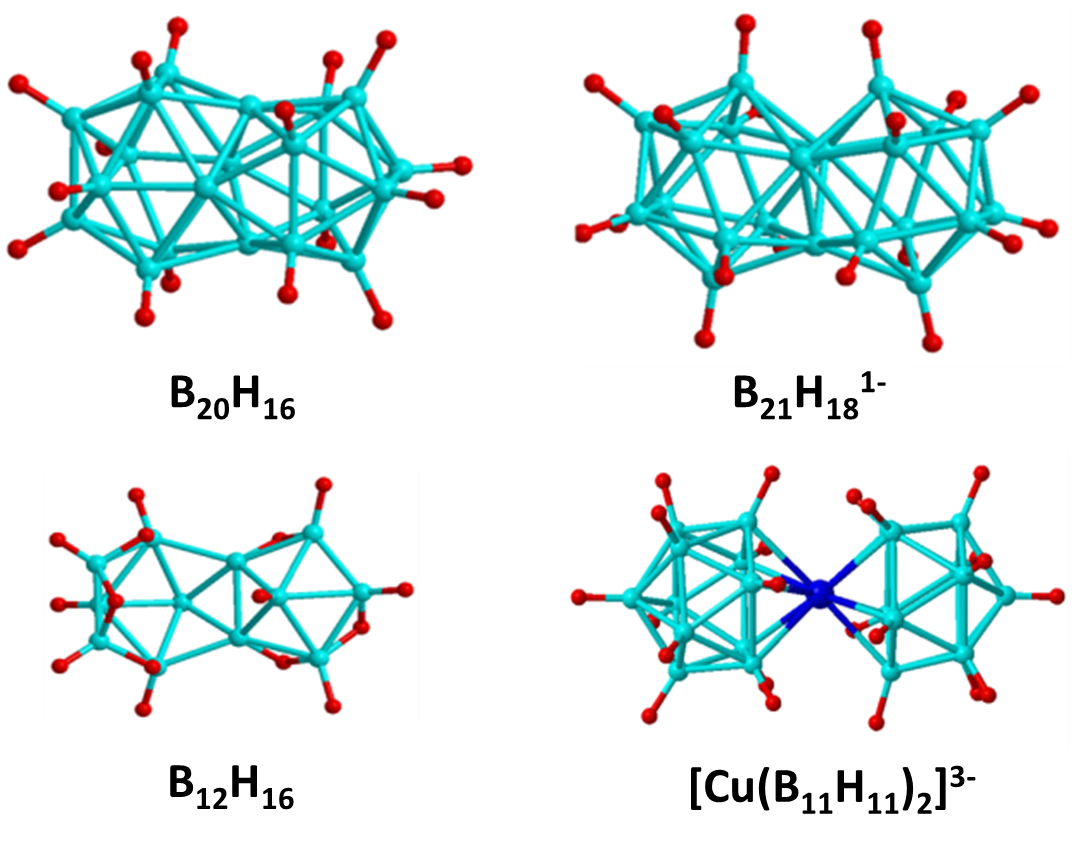
Jemmis Mno Rules
In chemistry, the Jemmis ''mno'' rules represent a unified rule for predicting and systematizing structures of compounds, usually clusters. The rules involve electron counting. They were formulated by E. D. Jemmis to explain the structures of condensed polyhedral boranes such as , which are obtained by condensing polyhedral boranes by sharing a triangular face, an edge, a single vertex, or four vertices. These rules are additions and extensions to Wade's rules and polyhedral skeletal electron pair theory. The Jemmis ''mno'' rule provides the relationship between polyhedral boranes, condensed polyhedral boranes, and β-rhombohedral boron. This is similar to the relationship between benzene, condensed benzenoid aromatics, and graphite, shown by Hückel's 4''n'' + 2 rule, as well as the relationship between tetracoordinate tetrahedral carbon compounds and diamond. The Jemmis ''mno'' rules reduce to Hückel's rule when restricted to two dimensions and reduce to Wade's rul ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemistry
Chemistry is the scientific study of the properties and behavior of matter. It is a physical science within the natural sciences that studies the chemical elements that make up matter and chemical compound, compounds made of atoms, molecules and ions: their composition, structure, properties, behavior and the changes they undergo during chemical reaction, reactions with other chemical substance, substances. Chemistry also addresses the nature of chemical bonds in chemical compounds. In the scope of its subject, chemistry occupies an intermediate position between physics and biology. It is sometimes called the central science because it provides a foundation for understanding both Basic research, basic and Applied science, applied scientific disciplines at a fundamental level. For example, chemistry explains aspects of plant growth (botany), the formation of igneous rocks (geology), how atmospheric ozone is formed and how environmental pollutants are degraded (ecology), the prop ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Icosahedron
In geometry, an icosahedron ( or ) is a polyhedron with 20 faces. The name comes . The plural can be either "icosahedra" () or "icosahedrons". There are infinitely many non- similar shapes of icosahedra, some of them being more symmetrical than others. The best known is the ( convex, non- stellated) regular icosahedron—one of the Platonic solids—whose faces are 20 equilateral triangles. Regular icosahedra There are two objects, one convex and one nonconvex, that can both be called regular icosahedra. Each has 30 edges and 20 equilateral triangle faces with five meeting at each of its twelve vertices. Both have icosahedral symmetry. The term "regular icosahedron" generally refers to the convex variety, while the nonconvex form is called a ''great icosahedron''. Convex regular icosahedron The convex regular icosahedron is usually referred to simply as the ''regular icosahedron'', one of the five regular Platonic solids, and is represented by its Schläfli symbol , contai ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
B105 made between 1959 and 1960
{{Expand list, date=June 2016 ...
B105 or B-105 may refer to: * B105 FM (call sign: 4BBB), a Mainstream CHR radio station broadcasting out of Brisbane, Queensland, Australia * B105, one of the B roads in Zone 1 of the Great Britain numbering scheme * WUBE, a country music radio station broadcasting out of Cincinnati, Ohio, United States of America * A model of Berkeley Cars Berkeley Cars Ltd was a British car manufacturer based in Biggleswade, Bedfordshire. The company produced economical sporting microcars with motorcycle-derived engines from 322 cc to 692 cc and front wheel drive between 1956 and 196 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sigma Bond
In chemistry, sigma bonds (σ bonds) or sigma overlap are the strongest type of covalent chemical bond. They are formed by head-on overlapping between atomic orbitals along the internuclear axis. Sigma bonding is most simply defined for diatomic molecules using the language and tools of symmetry groups. In this formal approach, a σ-bond is symmetrical with respect to rotation about the bond axis. By this definition, common forms of sigma bonds are s+s, pz+pz, s+pz and dz2+dz2 (where z is defined as the axis of the bond or the internuclear axis). Quantum theory also indicates that molecular orbitals (MO) of identical symmetry actually mix or ''hybridize''. As a practical consequence of this mixing of diatomic molecules, the wavefunctions s+s and pz+pz molecular orbitals become blended. The extent of this mixing (or hybridization or blending) depends on the relative energies of the MOs of like symmetry. For homodiatomics ( homonuclear diatomic molecules), bonding σ orbit ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cyclopentadienyl
Cyclopentadienyl can refer to * Cyclopentadienyl anion, or cyclopentadienide, ** Cyclopentadienyl ligand * Cyclopentadienyl radical, • * Cyclopentadienyl cation, See also * Pentadienyl {{Chemistry index ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ferrocene
Ferrocene is an organometallic chemistry, organometallic compound with the formula . The molecule is a Cyclopentadienyl complex, complex consisting of two Cyclopentadienyl anion, cyclopentadienyl rings sandwiching a central iron atom. It is an orange solid with a camphor-like odor that Sublimation (phase transition), sublimes above room temperature, and is soluble in most organic solvents. It is remarkable for its stability: it is unaffected by air, water, strong bases, and can be heated to 400 °C without decomposition. In oxidizing conditions it can reversibly react with strong acids to form the ferrocenium cation . Ferrocene and the ferrocenium cation are sometimes abbreviated as Fc and respectively. The first reported synthesis of ferrocene was in 1951. Its unusual stability puzzled chemists, and required the development of new theory to explain its formation and bonding. The discovery of ferrocene and its many Structural analog, analogues, known as metallocenes, sparke ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Condensed Polyhedral Boranes And Metallaborane
Condensation is the change of the state of matter from the gas phase into the liquid phase. Condensation may also refer to: * DNA condensation, the process of compacting DNA molecules * Cloud condensation nuclei, airborne particles required for cloud formation * Condensation (aerosol dynamics), a phase transition from gas to liquid * Condensation cloud, observable at large explosions in humid air * Condensation reaction, in chemistry, a chemical reaction between two molecules or moieties * Condensation algorithm, in computer science, a computer vision algorithm * Condensation (graph theory), in mathematics, a directed acyclic graph formed by contracting the strongly connected components of another graph * Dodgson condensation, in mathematics, a method invented by Lewis Carroll for computing the determinants of square matrices * Bose–Einstein condensation, a state of matter of a dilute gas in which quantum effects become apparent on a macroscopic scale * Condensation (psychology) ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Closo
In chemistry the polyhedral skeletal electron pair theory (PSEPT) provides electron counting rules useful for predicting the structures of cluster compound, clusters such as Boranes, borane and carborane clusters. The electron counting rules were originally formulated by Kenneth Wade, and were further developed by others including Michael Mingos; they are sometimes known as Wade's rules or the Wade–Mingos rules. The rules are based on a molecular orbital treatment of the bonding. These notes contained original material that served as the basis of the sections on the 4''n'', 5''n'', and 6''n'' rules. These rules have been extended and unified in the form of the Jemmis mno rules, Jemmis ''mno'' rules. Predicting structures of cluster compounds Different rules (4''n'', 5''n'', or 6''n'') are invoked depending on the number of electrons per vertex. The 4''n'' rules are reasonably accurate in predicting the structures of clusters having about 4 electrons per vertex, as is the case ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |