|
Dendralene
A dendralene is a discrete acyclic cross-conjugated polyene. The simplest dendralene is buta-1,3-diene (1) or endralene followed by endralene (2), endralene (3) and endralene (4) and so forth. endralene (butadiene) is the only one not cross-conjugated. : The name ''dendralene'' is pulled together from the words dendrimer, linear and alkene. The higher dendralenes are of scientific interest because they open up a large array of new organic compounds from a relatively simple precursor especially by Diels-Alder chemistry. Their cyclic counterparts are aptly called radialenes. Synthesis Vinylbutadiene ( endralene) was first prepared in 1955 by pyrolysis of a triacetate: : This compound reacts with two equivalents of maleic anhydride in a tandem DA reaction: : With benzoquinone the reaction product was a linear polymer. Several syntheses of substituted endralenes have been reported, one via an allene, one via a Horner–Wadsworth–Emmons reaction, one via a cross-couplin ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dendralenes General
A dendralene is a discrete acyclic cross-conjugated polyene. The simplest dendralene is buta-1,3-diene (1) or endralene followed by endralene (2), endralene (3) and endralene (4) and so forth. endralene (butadiene) is the only one not cross-conjugated. : The name ''dendralene'' is pulled together from the words dendrimer, linear and alkene. The higher dendralenes are of scientific interest because they open up a large array of new organic compounds from a relatively simple precursor especially by Diels-Alder chemistry. Their cyclic counterparts are aptly called radialenes. Synthesis Vinylbutadiene ( endralene) was first prepared in 1955 by pyrolysis of a triacetate: : This compound reacts with two equivalents of maleic anhydride in a tandem DA reaction: : With benzoquinone the reaction product was a linear polymer. Several syntheses of substituted endralenes have been reported, one via an allene, one via a Horner–Wadsworth–Emmons reaction, one via a cross-couplin ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Radialene
are alicyclic organic compounds containing n cross-conjugated exocyclic double bonds. The double bonds are commonly alkene groups but those with a carbonyl (C=O) group are also called radialenes. For some members the unsubstituted parent radialenes are elusive but many substituted derivatives are known. Radialenes are related to open-chain dendralenes and also to compounds like butadiene and benzene which also consist of a ring of sp2 hybridized carbon atoms. Radialenes are investigated in organic chemistry for their unusual properties and reactivity but have not ventured outside the laboratory. Reported uses are as experimental building blocks for novel organic conductors and ferromagnets. The first radialene called ''hexaethylidencyclohexane'' was synthesised in 1961. Conformation and adialenes are expected to have a planar molecular geometry with all carbon atoms in the same plane. This is verified experimentally in ''hexamethyl adialene'' and '. ' has a twist envelope ge ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Alkene
In organic chemistry, an alkene is a hydrocarbon containing a carbon–carbon double bond. Alkene is often used as synonym of olefin, that is, any hydrocarbon containing one or more double bonds.H. Stephen Stoker (2015): General, Organic, and Biological Chemistry'. 1232 pages. Two general types of monoalkenes are distinguished: terminal and internal. Also called α-olefins, terminal alkenes are more useful. However, the International Union of Pure and Applied Chemistry (IUPAC) recommends using the name "alkene" only for acyclic hydrocarbons with just one double bond; alkadiene, alkatriene, etc., or polyene for acyclic hydrocarbons with two or more double bonds; cycloalkene, cycloalkadiene, etc. for cyclic ones; and "olefin" for the general class – cyclic or acyclic, with one or more double bonds. Acyclic alkenes, with only one double bond and no other functional groups (also known as mono-enes) form a homologous series of hydrocarbons with the general formula with '' ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Open Chain Compound
In chemistry, an open-chain compound (also spelled as open chain compound) or acyclic compound (Greek prefix "α", ''without'' and "κύκλος", ''cycle'') is a compound with a linear structure, rather than a cyclic one. An open-chain compound having no side chains is called a straight-chain compound (also spelled as straight chain compound). Many of the simple molecules of organic chemistry, such as the alkanes and alkenes, have both linear and ring isomers, that is, both acyclic and cyclic, with the latter often classified as aromatic. For those with 4 or more carbons, the linear forms can have straight-chain or branched-chain isomers. The lowercase prefix ''n-'' denotes the straight-chain isomer; for example, ''n''-butane is straight-chain butane, whereas ''i''-butane is isobutane. Cycloalkanes are isomers of alkenes, not of alkanes, because the ring's closure involves a C-C bond. Having no rings (aromatic or otherwise), all open-chain compounds are aliphatic. Typically in bioc ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chloroprene
Chloroprene is the common name for 2-chlorobuta-1,3-diene (IUPAC name) with the chemical formula CH2=CCl−CH=CH2. Chloroprene is a colorless volatile liquid, almost exclusively used as a monomer for the production of the polymer polychloroprene, better known as neoprene, a type of synthetic rubber. History Although it may have been discovered earlier, chloroprene was largely developed by DuPont during the early 1930s, specifically with the formation of neoprene in mind. The chemists Elmer K. Bolton, Wallace Carothers, Arnold Collins and Ira Williams are generally accredited with its development and commercialisation although the work was based upon that of Julius Arthur Nieuwland, with whom they collaborated. Production Chloroprene is produced in three steps from 1,3-butadiene: (i) chlorination, (ii) isomerization of part of the product stream, and (iii) dehydrochlorination of 3,4-dichlorobut-1-ene. Chlorine adds to 1,3-butadiene to afford a mixture of 3,4-dichlorobut-1-ene ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Grignard Reagent
A Grignard reagent or Grignard compound is a chemical compound with the general formula , where X is a halogen and R is an organic group, normally an alkyl or aryl. Two typical examples are methylmagnesium chloride and phenylmagnesium bromide . They are a subclass of the organomagnesium compounds. Grignard compounds are popular reagents in organic synthesis for creating new carbon-carbon bonds. For example, when reacted with another halogenated compound in the presence of a suitable catalyst, they typically yield and the magnesium halide as a byproduct; and the latter is insoluble in the solvents normally used. In this aspect, they are similar to organolithium reagents. Pure Grignard reagents are extremely reactive solids. They are normally handled as solutions in solvents such as diethyl ether or tetrahydrofuran; which are relatively stable as long as water is excluded. In such a medium, a Grignard reagent is invariably present as a complex with the magnesium atom conn ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Magnesium
Magnesium is a chemical element with the symbol Mg and atomic number 12. It is a shiny gray metal having a low density, low melting point and high chemical reactivity. Like the other alkaline earth metals (group 2 of the periodic table) it occurs naturally only in combination with other elements and it almost always has an oxidation state of +2. It reacts readily with air to form a thin passivation coating of magnesium oxide that inhibits further corrosion of the metal. The free metal burns with a brilliant-white light. The metal is obtained mainly by electrolysis of magnesium salts obtained from brine. It is less dense than aluminium and is used primarily as a component in strong and lightweight alloys that contain aluminium. In the cosmos, magnesium is produced in large, aging stars by the sequential addition of three helium nuclei to a carbon nucleus. When such stars explode as supernovas, much of the magnesium is expelled into the interstellar medium where it ma ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Copper(I) Chloride
Copper(I) chloride, commonly called cuprous chloride, is the lower chloride of copper, with the formula CuCl. The substance is a white solid sparingly soluble in water, but very soluble in concentrated hydrochloric acid. Impure samples appear green due to the presence of copper(II) chloride (CuCl2). History Copper(I) chloride was first prepared by Robert Boyle in the mid-seventeenth century from mercury(II) chloride ("Venetian sublimate") and copper metal: :HgCl2 + 2 Cu → 2 CuCl + Hg In 1799, J.L. Proust characterized the two different chlorides of copper. He prepared CuCl by heating CuCl2 at red heat in the absence of air, causing it to lose half of its combined chlorine followed by removing residual CuCl2 by washing with water. An acidic solution of CuCl was formerly used for analysis of carbon monoxide content in gases, for example in Hempel's gas apparatus. This application was significant during the nineteenth and early twentieth centuries when coal gas was widely ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
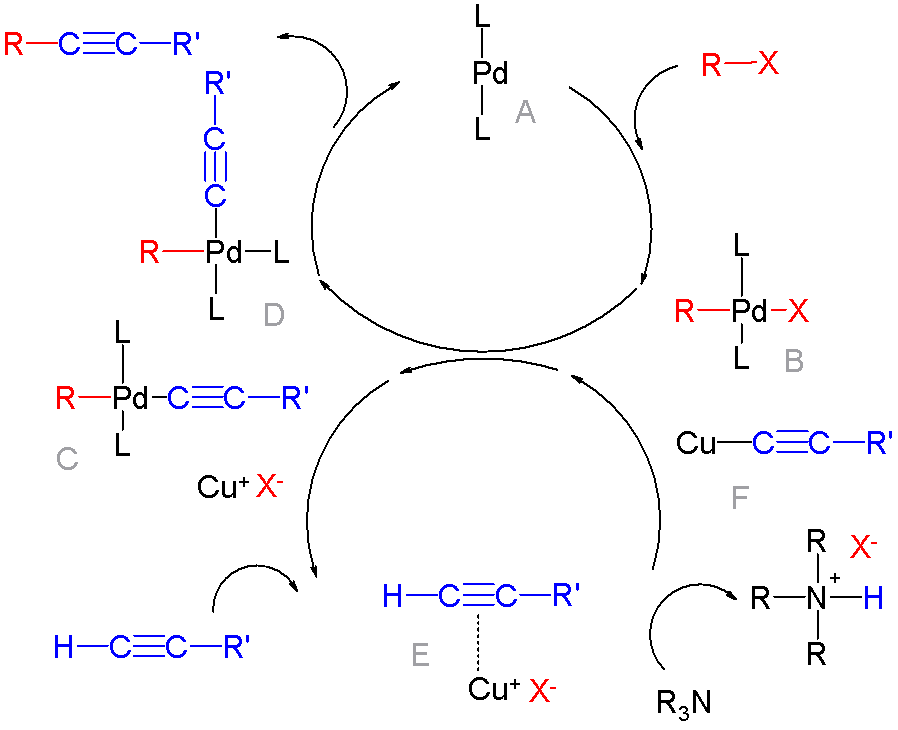
Organocopper
Organocopper compounds is the chemistry of organometallic compounds containing a carbon to copper chemical bond. Organocopper chemistry is the study of organocopper compounds describing their physical properties, synthesis and reactions. They are reagents in organic chemistry. The first organocopper compound, the explosive copper(I) acetylide Cu2C2 (Cu−C≡C−Cu), was synthesized by Rudolf Christian Böttger in 1859 by passing acetylene gas through a solution of copper(I) chloride: :C2H2 + 2 CuCl → Cu2C2 + 2 HCl Structure and bonding Organocopper compounds are diverse in structure and reactivity, but organocopper compounds are largely limited in oxidation states to copper(I), sometimes denoted Cu+. As a d10 metal center, it is related to Ni(0), but owing to its higher oxidation state, it engages in less pi-backbonding. Organic derivatives of Cu(II) and Cu(III) are invoked as intermediates but rarely isolated or even observed. In terms of geometry, copper(I) adopts symmetr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Oxidative Coupling
Oxidative coupling in chemistry is a coupling reaction of two molecular entities through an oxidative process. Usually oxidative couplings are catalysed by a transition metal complex like in classical cross-coupling reactions, although the underlying mechanism is different due to the oxidation process that requires an external (or internal) oxidant. Many such couplings utilize dioxygen as the stoichiometric oxidant but proceed by electron transfer. C-C Couplings Many oxidative couplings generate new C-C bonds. Early examples involve coupling of terminal alkynes: :2 RC≡CH + 2 Cu(I) → RC≡C-C≡CR + 2 Cu + 2 H+ Coupling of methane Coupling reactions involving methane are highly sought, related to C1 chemistry because C2 derivatives are far more valuable than methane. The oxidative coupling of methane gives ethylene: : 2 + → + 2 Aromatic coupling In oxidative aromatic coupling the reactants are electron-rich aromatic compounds. Typical substrates are phenols a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Copper(II) Chloride
Copper(II) chloride is the chemical compound with the chemical formula CuCl2. The anhydrous form is yellowish brown but slowly absorbs moisture to form a blue-green dihydrate. Both the anhydrous and the dihydrate forms occur naturally as the very rare minerals tolbachite and eriochalcite, respectively.Marlene C. Morris, Howard F. McMurdie, Eloise H. Evans, Boris Paretzkin, Harry S. Parker, and Nicolas C. Panagiotopoulos (1981) ''Copper chloride hydrate (eriochalcite)'', in Standard X-ray Diffraction Powder PatternsNational Bureau of Standards, Monograph 25, Section 18; page 33. Structure Anhydrous CuCl2 adopts a distorted cadmium iodide structure. In this motif, the copper centers are octahedral. Most copper(II) compounds exhibit distortions from idealized octahedral geometry due to the Jahn-Teller effect, which in this case describes the localization of one d-electron into a molecular orbital that is strongly antibonding with respect to a pair of chloride ligands. In CuCl2·2H ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cross-coupling Reaction
In organic chemistry, a cross-coupling reaction is a reaction where two fragments are joined together with the aid of a metal catalyst. In one important reaction type, a main group organometallic compound of the type R-M (R = organic fragment, M = main group center) reacts with an organic halide of the type R'-X with formation of a new carbon–carbon bond in the product R-R'. Cross-coupling reaction are a subset of coupling reactions. It is often used in arylations. Richard F. Heck, Ei-ichi Negishi, and Akira Suzuki were awarded the 2010 Nobel Prize in Chemistry for developing palladium-catalyzed coupling reactions. Mechanism The mechanism generally involves reductive elimination of the organic substituents R and R' on a metal complex of the type LnMR(R') (where L is some arbitrary spectator ligand). The crucial intermediate LnMR(R') is formed in a two step process from a low valence precursor Ln. The oxidative addition of an organic halide (RX) to LnM gives LnMR(X). Subsequ ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |



_Chloride_Precipitate.jpg)