|
Andrographolide
Andrographolide is a labdane diterpenoid that has been isolated from the stem and leaves of ''Andrographis paniculata''. Andrographolide is an extremely bitter substance. Andrographolide has been studied for its effects on cell signaling, immunomodulation, and stroke. Study has shown that andrographolide may bind to a spectrum of protein targets including NF-κB and actin by covalent modification. Biosynthesis While andrographolide is a relatively simple diterpene lactone, the biosynthesis by Andrographis paniculata was determined in the 2010s. Andrographolide is a member of the isoprenoid family of natural products. The precursors to isoprenoid biosynthesis, isopentenyl pyrophosphate (IPP) and dimethylallyl pyrophosphate (DMAPP), can be synthesized through either the mevalonic acid pathway (MVA) or deoxyxylulose pathway (DXP). Through selective C13 labeling of the precursors to both the MVA and DXP pathways, it was determined that the majority of the andrographolide pr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Andrographis Paniculata
''Andrographis paniculata'', commonly known as creat or green chiretta, is an annual herbaceous plant in the family Acanthaceae, native to India and Sri Lanka. It is widely cultivated in Southern and Southeastern Asia, where it has been believed to be a treatment for bacterial infections and some diseases. Mostly the leaves and roots have been used for such purposes; the whole plant is also used, in some cases. Description The plant grows as an erect herb to a height of in moist, shady places. The slender stem is dark green, square in cross-section with longitudinal furrows and wings along the angles. The lance-shaped leaves have hairless blades measuring up to long by . The small flowers are pink, solitary, arranged in lax spreading racemes or panicles. The fruit is a capsule around long and a few millimeters wide. It contains many yellow-brown seeds. The seeds are subquadrate, rugose and glabrous. The flowering time is September to December. Distribution The species i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Labdane
Labdane is a natural bicyclic diterpene. It forms the structural core for a wide variety of natural products collectively known as ''labdanes'' or ''labdane diterpenes''. The labdanes were so named because the first members of the class were originally obtained from labdanum, a resin derived from the gum rockrose. A variety of biological activities have been determined for labdane diterpenes including antibacterial, antifungal, antiprotozoal, and anti-inflammatory activities. Example labdane derivatives * Forskolin * Galanolactone * Isocupressic acid - is an abortifacient component of ''Cupressus macrocarpa''. * Medigenin *Sclareol * Stemodene See also * Abietane Abietane is a diterpene that forms the structural basis for a variety of natural chemical compounds such as abietic acid, carnosic acid, and ferruginol which are collectively known as abietanes or abietane diterpenes. Abietanes are found in the ... References {{reflist Diterpenes Decalins ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dimethylallyl Pyrophosphate
Dimethylallyl pyrophosphate (DMAPP; or alternatively, dimethylallyl diphosphate (DMADP); also isoprenyl pyrophosphate) is an isoprenoid precursor. It is a product of both the mevalonate pathway and the MEP pathway of isoprenoid precursor biosynthesis. It is an isomer of isopentenyl pyrophosphate (IPP) and exists in virtually all life forms. The enzyme isopentenyl pyrophosphate isomerase catalyzes isomerization between DMAPP and IPP. In the mevalonate pathway DMAPP is synthesised from mevalonic acid. In contrast, DMAPP is synthesised from HMBPP in the MEP pathway. At present, it is believed that there is crossover between the two pathways in organisms that use both pathways to create terpenes and terpenoid The terpenoids, also known as isoprenoids, are a class of naturally occurring organic chemicals derived from the 5-carbon compound isoprene and its derivatives called terpenes, diterpenes, etc. While sometimes used interchangeably with "terpenes" ...s, such as in plants, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Primary Alcohols
A primary alcohol is an alcohol in which the hydroxy group In chemistry, a hydroxy or hydroxyl group is a functional group with the chemical formula and composed of one oxygen atom covalently bonded to one hydrogen atom. In organic chemistry, alcohols and carboxylic acids contain one or more hydroxy ... is bonded to a primary carbon atom. It can also be defined as a molecule containing a “–CH2OH” group. In contrast, a secondary alcohol has a formula “–CHROH” and a tertiary alcohol has a formula “–CR2OH”, where “R” indicates a carbon-containing group. Examples of primary alcohols include ethanol and n-Butanol, 1-butanol. Methanol is also generally regarded as a primary alcohol, including the 1911 edition of the Encyclopædia Britannica,. See also * Alcohol (chemistry), Alcohol (especially Nomenclature section for discussion on Secondary and Tertiary alcohols.) * Oxidation of primary alcohols to carboxylic acids References Primary alcohols, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
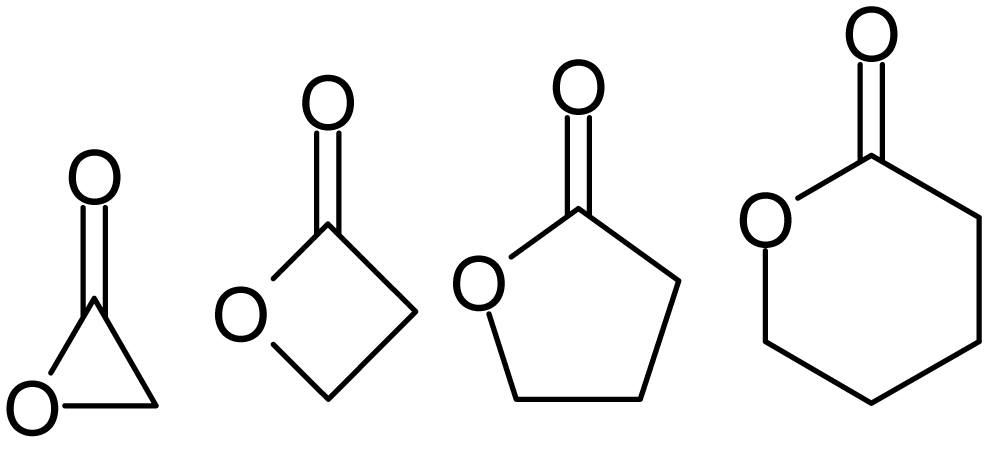
Lactones
Lactones are cyclic carboxylic esters, containing a 1-oxacycloalkan-2-one structure (), or analogues having unsaturation or heteroatoms replacing one or more carbon atoms of the ring. Lactones are formed by intramolecular esterification of the corresponding hydroxycarboxylic acids, which takes place spontaneously when the ring that is formed is five- or six-membered. Lactones with three- or four-membered rings (α-lactones and β-lactones) are very reactive, making their isolation difficult. Special methods are normally required for the laboratory synthesis of small-ring lactones as well as those that contain rings larger than six-membered. Nomenclature Lactones are usually named according to the precursor acid molecule (''aceto'' = 2 carbon atoms, ''propio'' = 3, ''butyro'' = 4, ''valero'' = 5, ''capro'' = 6, etc.), with a ''-lactone'' suffix and a Greek letter prefix that specifies the number of carbon atoms in the heterocycle — that is, the distance between the relevant -OH ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Diterpenes
Diterpenes are a class of chemical compounds composed of four isoprene units, often with the molecular formula C20H32. They are biosynthesized by plants, animals and fungi via the HMG-CoA reductase pathway, with geranylgeranyl pyrophosphate being a primary intermediate. Diterpenes form the basis for biologically important compounds such as retinol, retinal, and phytol. They are known to be antimicrobial and antiinflammatory. Structures As with most terpenes a huge number of potential structures exists, which may be broadly divided according to the number of rings present. Biosynthesis Diterpenes are derived from the addition of one IPP unit to FPP to form geranylgeranyl-pyrophosphate (GGPP). From GGPP, structural diversity is achieved mainly by two classes of enzymes; the diterpene synthases and cytochromes P450. Several diterpenes are produced by plants and cyanobacteria. GGPP is also the precursor for the synthesis of the phytane by the action of the enzyme geranylger ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bitter Compounds
Bitter may refer to: Common uses * Resentment, negative emotion or attitude, similar to being jaded, cynical or otherwise negatively affected by experience * Bitter (taste), one of the five basic tastes Books * ''Bitter (novel)'', a 2022 novel by Akwaeke Emezi. Food and drink * Bitter (beer), a British term for pale ale * Bitters, an herbal preparation now used mostly in cocktails Music Albums * ''Bitter'' (Jupiter Apple album), 2007 * ''Bitter'' (Meshell Ndegeocello album), 1999 Songs * "Bitter" (Fletcher song), 2020 * “Bitter” song by The Vamps from Cherry Blossom * "Bitter", 1997 single by Lit from '' Tripping the Light Fantastic'' * "Bitter", song by Jill Sobule from her 1997 album '' Happy Town'' * "Bitter", single by New Zealand band Shihad * "Bitter", song by Remy Zero from ''The Golden Hum'' * "Bitter", song by Reks from ''More Grey Hairs'' Other uses * Bitter (surname) (including a list of persons with the name) * Bitter Cars, a German car company See also * ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Biosynthesis Figure
Biosynthesis is a multi-step, enzyme-catalyzed process where substrates are converted into more complex products in living organisms. In biosynthesis, simple compounds are modified, converted into other compounds, or joined to form macromolecules. This process often consists of metabolic pathways. Some of these biosynthetic pathways are located within a single cellular organelle, while others involve enzymes that are located within multiple cellular organelles. Examples of these biosynthetic pathways include the production of lipid membrane components and nucleotides. Biosynthesis is usually synonymous with anabolism. The prerequisite elements for biosynthesis include: precursor compounds, chemical energy (e.g. ATP), and catalytic enzymes which may require coenzymes (e.g. NADH, NADPH). These elements create monomers, the building blocks for macromolecules. Some important biological macromolecules include: proteins, which are composed of amino acid monomers joined via peptide ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Epoxide
In organic chemistry, an epoxide is a cyclic ether () with a three-atom ring. This ring approximates an equilateral triangle, which makes it strained, and hence highly reactive, more so than other ethers. They are produced on a large scale for many applications. In general, low molecular weight epoxides are colourless and nonpolar, and often volatile. Nomenclature A compound containing the epoxide functional group can be called an epoxy, epoxide, oxirane, and ethoxyline. Simple epoxides are often referred to as oxides. Thus, the epoxide of ethylene (C2H4) is ethylene oxide (C2H4O). Many compounds have trivial names; for instance, ethylene oxide is called "oxirane". Some names emphasize the presence of the epoxide functional group, as in the compound ''1,2-epoxyheptane'', which can also be called ''1,2-heptene oxide''. A polymer formed from epoxide precursors is called an ''epoxy'', but such materials do not contain epoxide groups (or contain only a few residual epoxy grou ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Farnesyl Pyrophosphate
Farnesyl pyrophosphate (FPP), also known as farnesyl diphosphate (FDP), is an intermediate in the biosynthesis of terpenes and terpenoids such as sterols and carotenoids. It is also used in the synthesis of CoQ (part of the electron transport chain), as well as dehydrodolichol diphosphate (a precursor of dolichol, which transports proteins to the ER lumen for ''N''-glycosylation). Biosynthesis Farnesyl pyrophosphate synthase (a prenyl transferase) catalyzes sequential condensation reactions of dimethylallyl pyrophosphate with 2 units of 3-isopentenyl pyrophosphate to form farnesyl pyrophosphate, as is shown in the following two steps: * Dimethylallyl pyrophosphate reacts with 3-isopentenyl pyrophosphate to form geranyl pyrophosphate: * Geranyl pyrophosphate then reacts with another molecule of 3-isopentenyl pyrophosphate to form farnesyl pyrophosphate Pharmacology The above reactions are inhibited by bisphosphonates (used for osteoporosis). Farnesyl pyrophosphate is a sel ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Geranyl Pyrophosphate
Geranyl pyrophosphate (GPP), also known as geranyl diphosphate (GDP), is the pyrophosphate ester of the terpenoid geraniol. Its salts are colorless. It is a precursor to many natural products. Occurrence GPP is an intermediate in the isoprenoid biosynthesis pathway that produces longer prenyl chains such as farnesyl pyrophosphate and geranylgeranyl pyrophosphate as well as many terpenes. It can be prepared in the laboratory from geraniol. Related compounds * Geraniol * Farnesyl pyrophosphate * Geranylgeranyl pyrophosphate See also * Dimethylallyltranstransferase Dimethylallyltranstransferase (DMATT), also known as farnesylpyrophosphate synthase (FPPS) or as farnesyldiphosphate synthase (FDPS), is an enzyme that in humans is encoded by the FDPS gene and catalyzes the transformation of dimethylallylpyr ... References Further reading *Kulkarni RS, Pandit SS, Chidley HG, Nagel R, Schmidt A, Gershenzon J, Pujari KH, Giri AP and Gupta VS, 2013Characterization of three n ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |