|
-ane
The suffix -ane in organic chemistry forms the names of organic compounds where the -C-C- group has been attributed the highest priority according to the rules of organic nomenclature. Such organic compounds are called alkanes. They are saturated hydrocarbons. The names of the saturated hydrides of non-metals end with the suffix -ane: the hydrides of silicon are called silanes SiH4; the hydrides of boron are boranes B2H6. The final "-e" is dropped before a suffix that starts with a vowel, e.g. "propanol". Alternatively, -ane may be used for a mononuclear hydride of an element. For instance, methane for CH4 and oxidane for H2O (water). For the etymology, see Alkane. References See also *IUPAC nomenclature of organic chemistry {{Orgchemsuffixes ane Ane or ane may refer to: * Āne, a village in Latvia * Ane, Netherlands, a village in Overijssel, Netherlands, also ** Battle of Ane (1227), a battle fought near the village * -ane, a suffix in organic chemistry, or spe ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Alkane
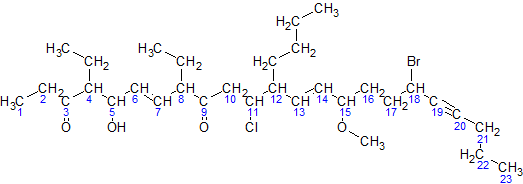
In organic chemistry, an alkane, or paraffin (a historical trivial name that also has other meanings), is an acyclic saturated hydrocarbon. In other words, an alkane consists of hydrogen and carbon atoms arranged in a tree structure in which all the carbon–carbon bonds are single. Alkanes have the general chemical formula . The alkanes range in complexity from the simplest case of methane (), where ''n'' = 1 (sometimes called the parent molecule), to arbitrarily large and complex molecules, like pentacontane () or 6-ethyl-2-methyl-5-(1-methylethyl) octane, an isomer of tetradecane (). The International Union of Pure and Applied Chemistry (IUPAC) defines alkanes as "acyclic branched or unbranched hydrocarbons having the general formula , and therefore consisting entirely of hydrogen atoms and saturated carbon atoms". However, some sources use the term to denote ''any'' saturated hydrocarbon, including those that are either monocyclic (i.e. the cycloalkanes) or ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Saturated Hydrocarbons
In organic chemistry, an alkane, or paraffin (a historical trivial name that also has other meanings), is an acyclic saturated hydrocarbon. In other words, an alkane consists of hydrogen and carbon atoms arranged in a tree structure in which all the carbon–carbon bonds are single. Alkanes have the general chemical formula . The alkanes range in complexity from the simplest case of methane (), where ''n'' = 1 (sometimes called the parent molecule), to arbitrarily large and complex molecules, like pentacontane () or 6-ethyl-2-methyl-5-(1-methylethyl) octane, an isomer of tetradecane (). The International Union of Pure and Applied Chemistry (IUPAC) defines alkanes as "acyclic branched or unbranched hydrocarbons having the general formula , and therefore consisting entirely of hydrogen atoms and saturated carbon atoms". However, some sources use the term to denote ''any'' saturated hydrocarbon, including those that are either monocyclic (i.e. the cycloalkanes) or po ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Methane
Methane ( , ) is a chemical compound with the chemical formula (one carbon atom bonded to four hydrogen atoms). It is a group-14 hydride, the simplest alkane, and the main constituent of natural gas. The relative abundance of methane on Earth makes it an economically attractive fuel, although capturing and storing it poses technical challenges due to its gaseous state under normal conditions for temperature and pressure. Naturally occurring methane is found both below ground and under the seafloor and is formed by both geological and biological processes. The largest reservoir of methane is under the seafloor in the form of methane clathrates. When methane reaches the surface and the atmosphere, it is known as atmospheric methane. The Earth's atmospheric methane concentration has increased by about 150% since 1750, and it accounts for 20% of the total radiative forcing from all of the long-lived and globally mixed greenhouse gases. It has also been detected on other plane ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
IUPAC Nomenclature Of Organic Chemistry
In chemical nomenclature, the IUPAC nomenclature of organic chemistry is a method of naming organic chemical compounds as recommended by the International Union of Pure and Applied Chemistry (IUPAC). It is published in the ''Nomenclature of Organic Chemistry'' (informally called the Blue Book). Ideally, every possible organic compound should have a name from which an unambiguous structural formula can be created. There is also an IUPAC nomenclature of inorganic chemistry. To avoid long and tedious names in normal communication, the official IUPAC naming recommendations are not always followed in practice, except when it is necessary to give an unambiguous and absolute definition to a compound. IUPAC names can sometimes be simpler than older names, as with ethanol, instead of ethyl alcohol. For relatively simple molecules they can be more easily understood than non-systematic names, which must be learnt or looked over. However, the common or trivial name is often substantially ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organic Nomenclature
In chemical nomenclature, the IUPAC nomenclature of organic chemistry is a method of naming organic chemical compounds as recommended by the International Union of Pure and Applied Chemistry (IUPAC). It is published in the ''Nomenclature of Organic Chemistry'' (informally called the Blue Book). Ideally, every possible organic compound should have a name from which an unambiguous structural formula can be created. There is also an IUPAC nomenclature of inorganic chemistry. To avoid long and tedious names in normal communication, the official IUPAC naming recommendations are not always followed in practice, except when it is necessary to give an unambiguous and absolute definition to a compound. IUPAC names can sometimes be simpler than older names, as with ethanol, instead of ethyl alcohol. For relatively simple molecules they can be more easily understood than non-systematic names, which must be learnt or looked over. However, the common or trivial name is often substantially sh ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Propanol
There are two isomers of propanol. * 1-Propanol, ''n''-propanol, or propan-1-ol : CH3CH2CH2OH, the most common meaning *2-Propanol, Isopropyl alcohol, isopropanol, or propan-2-ol : (CH3)2CHOH See also * Propanal (propionaldehyde) differs in spelling from propanol by a single letter and is a different compound. * Propranolol Propranolol, sold under the brand name Inderal among others, is a medication of the beta blocker class. It is used to treat high blood pressure, a number of types of irregular heart rate, thyrotoxicosis, capillary hemangiomas, performance an ... is a drug used for reducing blood pressure and hand tremors. {{Authority control Alkanols ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Oxidane
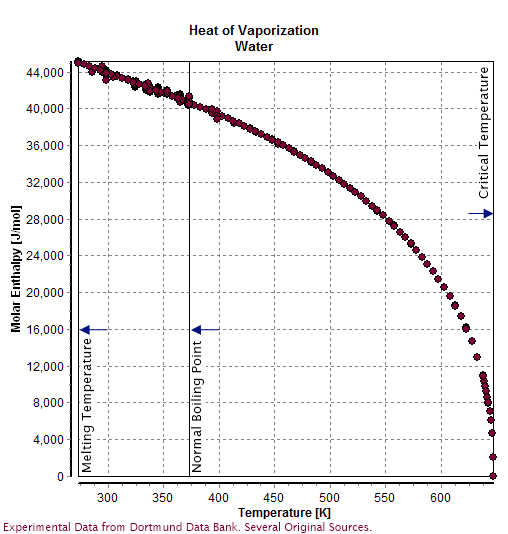
Water () is a polar inorganic compound that is at room temperature a tasteless and odorless liquid, which is nearly colorless apart from an inherent hint of blue. It is by far the most studied chemical compound and is described as the "universal solvent" and the "solvent of life". It is the most abundant substance on the surface of Earth and the only common substance to exist as a solid, liquid, and gas on Earth's surface. It is also the third most abundant molecule in the universe (behind molecular hydrogen and carbon monoxide). Water molecules form hydrogen bonds with each other and are strongly polar. This polarity allows it to dissociate ions in salts and bond to other polar substances such as alcohols and acids, thus dissolving them. Its hydrogen bonding causes its many unique properties, such as having a solid form less dense than its liquid form, a relatively high boiling point of 100 °C for its molar mass, and a high heat capacity. Water is amphoteric, meaning t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemical Element
A chemical element is a species of atoms that have a given number of protons in their nuclei, including the pure substance consisting only of that species. Unlike chemical compounds, chemical elements cannot be broken down into simpler substances by any chemical reaction. The number of protons in the nucleus is the defining property of an element, and is referred to as its atomic number (represented by the symbol ''Z'') – all atoms with the same atomic number are atoms of the same element. Almost all of the baryonic matter of the universe is composed of chemical elements (among rare exceptions are neutron stars). When different elements undergo chemical reactions, atoms are rearranged into new compounds held together by chemical bonds. Only a minority of elements, such as silver and gold, are found uncombined as relatively pure native element minerals. Nearly all other naturally occurring elements occur in the Earth as compounds or mixtures. Air is primarily a mixture o ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydride
In chemistry, a hydride is formally the anion of hydrogen( H−). The term is applied loosely. At one extreme, all compounds containing covalently bound H atoms are called hydrides: water (H2O) is a hydride of oxygen, ammonia is a hydride of nitrogen, etc. For inorganic chemists, hydrides refer to compounds and ions in which hydrogen is covalently attached to a less electronegative element. In such cases, the H centre has nucleophilic character, which contrasts with the protic character of acids. The hydride anion is very rarely observed. Almost all of the elements form binary compounds with hydrogen, the exceptions being He, Ne, Ar, Kr, Pm, Os, Ir, Rn, Fr, and Ra. Exotic molecules such as positronium hydride have also been made. Bonds Bonds between hydrogen and the other elements range from highly to somewhat covalent. Some hydrides, e.g. boron hydrides, do not conform to classical electron-counting rules and the bonding is described in terms of multi-centered ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Affix
In linguistics, an affix is a morpheme that is attached to a word stem to form a new word or word form. Affixes may be derivational, like English ''-ness'' and ''pre-'', or inflectional, like English plural ''-s'' and past tense ''-ed''. They are bound morphemes by definition; prefixes and suffixes may be separable affixes. Affixation is the linguistic process that speakers use to form different words by adding morphemes at the beginning (prefixation), the middle (infixation) or the end (suffixation) of words. Positional categories of affixes ''Prefix'' and ''suffix'' may be subsumed under the term ''adfix'', in contrast to ''infix.'' When marking text for interlinear glossing, as in the third column in the chart above, simple affixes such as prefixes and suffixes are separated from the stem with hyphens. Affixes which disrupt the stem, or which themselves are discontinuous, are often marked off with angle brackets. Reduplication is often shown with a tilde. Affixes which c ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Vowel
A vowel is a syllabic speech sound pronounced without any stricture in the vocal tract. Vowels are one of the two principal classes of speech sounds, the other being the consonant. Vowels vary in quality, in loudness and also in quantity (length). They are usually voiced and are closely involved in prosodic variation such as tone, intonation and stress. The word ''vowel'' comes from the Latin word , meaning "vocal" (i.e. relating to the voice). In English, the word ''vowel'' is commonly used to refer both to vowel sounds and to the written symbols that represent them (a, e, i, o, u, and sometimes y). Definition There are two complementary definitions of vowel, one phonetic and the other phonological. *In the phonetic definition, a vowel is a sound, such as the English "ah" or "oh" , produced with an open vocal tract; it is median (the air escapes along the middle of the tongue), oral (at least some of the airflow must escape through the mouth), frictionless and continuant ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Boranes
Boranes is the name given to compounds with the formula BxHy and related anions. Many such boranes are known. Most common are those with 1 to 12 boron atoms. Although they have few practical applications, the boranes exhibit structures and bonding that differs strongly from the patterns seen in hydrocarbons. Hybrids of boranes and hydrocarbons, the carboranes are also well developed. History The development of the chemistry of boranes led to innovations in synthetic methods as well as structure and bonding. First, new synthetic techniques were required to handle diborane and many of its derivatives, which are both pyrophoric and volatile. Alfred Stock invented the glass vacuum line for this purpose. The structure of diborane was correctly predicted in 1943 many years after its discovery. The structures of the boron hydride clusters were determined beginning in 1948 with the characterization of decaborane. William Lipscomb was awarded the Nobel prize in Chemistry in 1976 for th ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |



%2C_black_and_white.png)


-from-xtal-view-1-tilt-3D-bs-17.png)