|
Solvation Shell
A solvation shell or solvation sheath is the solvent interface of any chemical compound or biomolecule that constitutes the solute. When the solvent is water it is called a hydration shell or hydration sphere. The number of solvent molecules surrounding each unit of solute is called the hydration number of the solute. A classic example is when water molecules arrange around a metal ion. If the metal ion is a cation, the electronegative oxygen atom of the water molecule would be attracted electrostatically to the positive charge on the metal ion. The result is a solvation shell of water molecules that surround the ion. This shell can be several molecules thick, dependent upon the charge of the ion, its distribution and spatial dimensions. A number of molecules of solvent are involved in the solvation shell around anions and cations from a dissolved salt in a solvent. Metal ions in aqueous solutions form metal aquo complexes. This number can be determined by various methods like ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
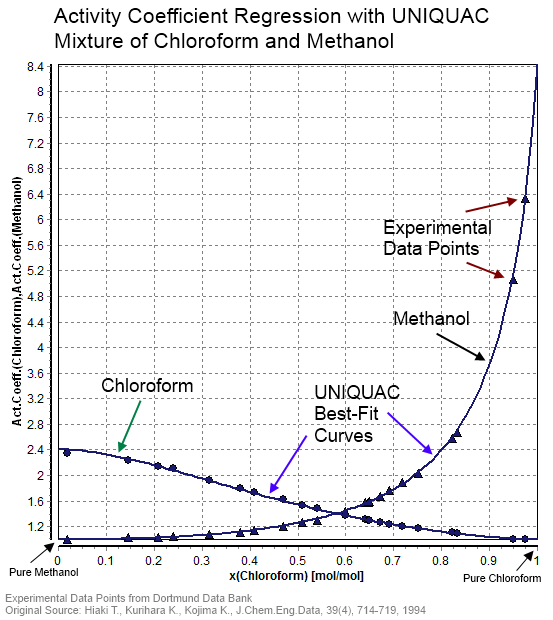
Activity Coefficient
In thermodynamics, an activity coefficient is a factor used to account for deviation of a mixture of chemical substances from ideal behaviour. In an ideal mixture, the microscopic interactions between each pair of chemical species are the same (or macroscopically equivalent, the enthalpy change of solution and volume variation in mixing is zero) and, as a result, properties of the mixtures can be expressed directly in terms of simple concentrations or partial pressures of the substances present e.g. Raoult's law. Deviations from ideality are accommodated by modifying the concentration by an ''activity coefficient''. Analogously, expressions involving gases can be adjusted for non-ideality by scaling partial pressures by a fugacity coefficient. The concept of activity coefficient is closely linked to that of activity in chemistry. Thermodynamic definition The chemical potential, \mu_\mathrm, of a substance B in an ideal mixture of liquids or an ideal solution is given by ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Solutions
Solution may refer to: * Solution (chemistry), a mixture where one substance is dissolved in another * Solution (equation), in mathematics ** Numerical solution, in numerical analysis, approximate solutions within specified error bounds * Solution, in problem solving * Solution, in solution selling Other uses * V-STOL Solution, an ultralight aircraft * Solution (band), a Dutch rock band ** ''Solution'' (Solution album), 1971 * Solution A.D. Solution A.D. was an American rock band from East Stroudsburg, Pennsylvania. The band was initially named Solution, and appended the "A.D." after discovering that another group with the same name already existed. Solution A.D.at Allmusic The gr ..., an American rock band * ''Solution'' (Cui Jian album), 1991 * ''Solutions'' (album), a 2019 album by K.Flay See also * The Solution (other) * {{disambiguation ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Solvation
Solvation (or dissolution) describes the interaction of a solvent with dissolved molecules. Both ionized and uncharged molecules interact strongly with a solvent, and the strength and nature of this interaction influence many properties of the solute, including solubility, reactivity, and color, as well as influencing the properties of the solvent such as its viscosity and density. If the attractive forces between the solvent and solute particles are greater than the attractive forces holding the solute particles together, the solvent particles pull the solute particles apart and surround them. The surrounded solute particles then move away from the solid solute and out into the solution. Ions are surrounded by a concentric shell of solvent. Solvation is the process of reorganizing solvent and solute molecules into solvation complexes and involves bond formation, hydrogen bonding, and van der Waals forces. Solvation of a solute by water is called hydration. Solubility of solid ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydration Energy
In chemistry, hydration energy (also hydration enthalpy) is the amount of energy released when one mole of ions undergoes hydration. Hydration energy is one component in the quantitative analysis of solvation. It is a particular special case of water. The value of hydration energies is one of the most challenging aspects of structural prediction. Upon dissolving a salt in water, the cations and anions interact with the positive and negative dipoles of the water. The trade-off of these interactions vs those within the crystalline solid comprises the hydration energy. Examples If the hydration energy is greater than the lattice energy, then the enthalpy of solution is negative (heat is released), otherwise it is positive (heat is absorbed). The hydration energy should not be confused with solvation energy, which is the change in Gibb's free energy (not enthalpy) as solute in the gaseous state is dissolved. If the solvation energy is positive, then the solvation process is en ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Water Model
In computational chemistry, a water model is used to simulate and thermodynamically calculate water clusters, liquid water, and aqueous solutions with explicit solvent. The models are determined from quantum mechanics, molecular mechanics, experimental results, and these combinations. To imitate a specific nature of molecules, many types of models have been developed. In general, these can be classified by the following three points; (i) the number of interaction points called ''site'', (ii) whether the model is rigid or flexible, (iii) whether the model includes polarization effects. An alternative to the explicit water models is to use an implicit solvation model, also termed a continuum model, an example of which would be the COSMO solvation model or the polarizable continuum model (PCM) or a hybrid solvation model. Simple water models The rigid models are considered the simplest water models and rely on non-bonded interactions. In these models, bonding interactions are im ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ionic Radius
Ionic radius, ''r''ion, is the radius of a monatomic ion in an ionic crystal structure. Although neither atoms nor ions have sharp boundaries, they are treated as if they were hard spheres with radii such that the sum of ionic radii of the cation and anion gives the distance between the ions in a crystal lattice. Ionic radii are typically given in units of either picometers (pm) or angstroms (Å), with 1 Å = 100 pm. Typical values range from 31 pm (0.3 Å) to over 200 pm (2 Å). The concept can be extended to solvated ions in liquid solutions taking into consideration the solvation shell. Trends Ions may be larger or smaller than the neutral atom, depending on the ion's electric charge. When an atom loses an electron to form a cation, the other electrons are more attracted to the nucleus, and the radius of the ion gets smaller. Similarly, when an electron is added to an atom, forming an anion, the added electron increases the size of th ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ion Transport Number
In chemistry, ion transport number, also called the transference number, is the fraction of the total electric current carried in an electrolyte by a given ionic species : :t_i = \frac Differences in transport number arise from differences in electrical mobility. For example, in an aqueous solution of sodium chloride, less than half of the current is carried by the positively charged sodium ions (cations) and more than half is carried by the negatively charged chloride ions (anions) because the chloride ions are able to move faster, i.e., chloride ions have higher mobility than sodium ions. The sum of the transport numbers for all of the ions in solution always equals unity: :\sum_i t_i = 1 The concept and measurement of transport number were introduced by Johann Wilhelm Hittorf in the year 1853. Liquid junction potential can arise from ions in a solution having different ion transport numbers. At zero concentration, the limiting ion transport numbers may be expressed in t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Molecular Dynamics
Molecular dynamics (MD) is a computer simulation method for analyzing the physical movements of atoms and molecules. The atoms and molecules are allowed to interact for a fixed period of time, giving a view of the dynamic "evolution" of the system. In the most common version, the trajectories of atoms and molecules are determined by numerically solving Newton's equations of motion for a system of interacting particles, where forces between the particles and their potential energies are often calculated using interatomic potentials or molecular mechanical force fields. The method is applied mostly in chemical physics, materials science, and biophysics. Because molecular systems typically consist of a vast number of particles, it is impossible to determine the properties of such complex systems analytically; MD simulation circumvents this problem by using numerical methods. However, long MD simulations are mathematically ill-conditioned, generating cumulative erro ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemical Compound
A chemical compound is a chemical substance composed of many identical molecules (or molecular entities) containing atoms from more than one chemical element held together by chemical bonds. A molecule consisting of atoms of only one element is therefore not a compound. A compound can be transformed into a different substance by a chemical reaction, which may involve interactions with other substances. In this process, bonds between atoms may be broken and/or new bonds formed. There are four major types of compounds, distinguished by how the constituent atoms are bonded together. Molecular compounds are held together by covalent bonds; ionic compounds are held together by ionic bonds; intermetallic compounds are held together by metallic bonds; coordination complexes are held together by coordinate covalent bonds. Non-stoichiometric compounds form a disputed marginal case. A chemical formula specifies the number of atoms of each element in a compound molecule, using ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Activity Coefficient
In thermodynamics, an activity coefficient is a factor used to account for deviation of a mixture of chemical substances from ideal behaviour. In an ideal mixture, the microscopic interactions between each pair of chemical species are the same (or macroscopically equivalent, the enthalpy change of solution and volume variation in mixing is zero) and, as a result, properties of the mixtures can be expressed directly in terms of simple concentrations or partial pressures of the substances present e.g. Raoult's law. Deviations from ideality are accommodated by modifying the concentration by an ''activity coefficient''. Analogously, expressions involving gases can be adjusted for non-ideality by scaling partial pressures by a fugacity coefficient. The concept of activity coefficient is closely linked to that of activity in chemistry. Thermodynamic definition The chemical potential, \mu_\mathrm, of a substance B in an ideal mixture of liquids or an ideal solution is given by ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |