|
Suzuki Coupling
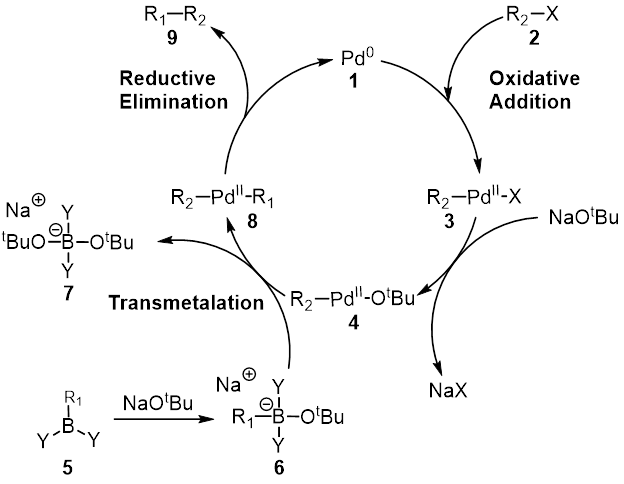
The Suzuki reaction or Suzuki coupling is an organic reaction that uses a palladium complex catalyst to cross-couple a boronic acid to an organohalide. It was first published in 1979 by Akira Suzuki, and he shared the 2010 Nobel Prize in Chemistry with Richard F. Heck and Ei-ichi Negishi for their contribution to the discovery and development of noble metal catalysis in organic synthesis. This reaction is sometimes telescoped with the related Miyaura borylation; the combination is the Suzuki–Miyaura reaction. It is widely used to synthesize polyolefins, styrenes, and substituted biphenyls. The general scheme for the Suzuki reaction is shown below, where a carbon–carbon single bond is formed by coupling a halide (R1-X) with an organoboron species (R2-BY2) using a palladium catalyst and a base. The organoboron species is usually synthesized by hydroboration or carboboration, allowing for rapid generation of molecular complexity. Several reviews have been publishe ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Akira Suzuki (chemist)
is a Japanese chemist and Nobel Prize Laureate (2010), who first published the Suzuki reaction, the organic reaction of an aryl- or Vinyl group, vinyl-boronic acid with an aryl- or vinyl-halide catalyzed by a palladium, palladium(0) complex (chemistry), complex, in 1979. Early life and education Suzuki was born on September 12, 1930, in Mukawa, Hokkaidō, his father died when he was in high school. He studied chemistry at Hokkaido University (Hokudai) and after receiving his PhD while he worked there as assistant professor. He initially wanted to major in mathematics, as his favorite subject in childhood was arithmetic.『朝日小学生新聞』2010年10月8日 It was an encounter with two books that became an opportunity to advance to the path of organic synthesis, one is ''Textbook of Organic Chemistry'' written by Louis Fieser of Harvard University, and another is ''Hydroboration'' written by Herbert C. Brown of Purdue University. Career From 1963 until 1965, Suzuki worked ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Halide
In chemistry, a halide (rarely halogenide) is a binary chemical compound, of which one part is a halogen atom and the other part is an element or radical that is less electronegative (or more electropositive) than the halogen, to make a fluoride, chloride, bromide, iodide, astatide, or theoretically tennesside compound. The alkali metals combine directly with halogens under appropriate conditions forming halides of the general formula, MX (X = F, Cl, Br or I). Many salts are halides; the ''hal-'' syllable in ''halide'' and '' halite'' reflects this correlation. A halide ion is a halogen atom bearing a negative charge. The common halide anions are fluoride (), chloride (), bromide (), and iodide (). Such ions are present in many ionic halide salts. Halide minerals contain halides. All these halide anions are colorless. Halides also form covalent bonds, examples being colorless TiF4, colorless TiCl4, orange TiBr4, and brown TiI4. The heavier members TiCl4, TiBr4 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Catalytic Cycle
In chemistry, a catalytic cycle is a multistep reaction mechanism that involves a catalyst. The catalytic cycle is the main method for describing the role of catalysts in biochemistry, organometallic chemistry, bioinorganic chemistry, materials science, etc. Since catalysts are regenerated, catalytic cycles are usually written as a sequence of chemical reactions in the form of a loop. In such loops, the initial step entails binding of one or more reactants by the catalyst, and the final step is the release of the product and regeneration of the catalyst. Articles on the Monsanto process, the Wacker process, and the Heck reaction show catalytic cycles. A catalytic cycle is not necessarily a full reaction mechanism. For example, it may be that the intermediates have been detected, but it is not known by which mechanisms the actual elementary reactions occur. Precatalysts Precatalysts are not catalysts but are ''precursors'' to catalysts. Precatalysts are converted in the r ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Reductive Elimination
Reductive elimination is an elementary step in organometallic chemistry in which the oxidation state of the metal center decreases while forming a new covalent bond between two ligands. It is the microscopic reverse of oxidative addition, and is often the product-forming step in many catalytic processes. Since oxidative addition and reductive elimination are reverse reactions, the same mechanisms apply for both processes, and the product equilibrium depends on the thermodynamics of both directions. General information Reductive elimination is often seen in higher oxidation states, and can involve a two-electron change at a single metal center (mononuclear) or a one-electron change at each of two metal centers (binuclear, dinuclear, or bimetallic). For mononuclear reductive elimination, the oxidation state of the metal decreases by two, while the d-electron count of the metal increases by two. This pathway is common for d8 metals Ni(II), Pd(II), and Au(III) and d6 metals Pt(I ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ate Complex
In chemistry, an ate complex is a salt formed by the reaction of a Lewis acid with a Lewis base whereby the central atom (from the Lewis acid) increases its valence and gains a negative formal charge. (In this definition, the meaning of valence is equivalent to coordination number). Often in chemical nomenclature the term ''ate'' is suffixed to the element in question. For example, the ate complex of a boron compound is called a borate. Thus trimethylborane and methyllithium react to form the ate compound , lithium tetramethylborate(1-). This concept was introduced by Georg Wittig in 1958. Ate complexes are common for metals, including the transition metals (groups 3-11), as well as the metallic or semi-metallic elements of group 2, 12, and 13. They are also well-established for third-period or heavier elements of groups 14–18 in their higher oxidation states. Ate complexes are a counterpart to onium ions. Lewis acids form ate ions when the central atom reacts with a donor ( ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Transmetalation
Transmetalation (alt. spelling: transmetallation) is a type of organometallic reaction that involves the transfer of ligands from one metal to another. It has the general form: :M1–R + M2–R′ → M1–R′ + M2–R where R and R′ can be, but are not limited to, an alkyl, aryl, alkynyl, allyl, halogen, or pseudohalogen group. The reaction is usually an irreversible process due to thermodynamic and kinetic reasons. Thermodynamics will favor the reaction based on the electronegativities of the metals and kinetics will favor the reaction if there are empty orbitals on both metals. There are different types of transmetalation including redox-transmetalation and redox-transmetalation/ligand exchange. During transmetalation the metal-carbon bond is activated, leading to the formation of new metal-carbon bonds. Transmetalation is commonly used in catalysis, synthesis of main group complexes, and synthesis of transition metal complexes. Types of transmetalation There are two mai ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Reaction Intermediate
In chemistry, a reaction intermediate, or intermediate, is a molecular entity arising within the sequence of a stepwise chemical reaction. It is formed as the reaction product of an elementary step, from the reactants and/or preceding intermediates, but is consumed in a later step. It does not appear in the chemical equation for the overall reaction. For example, consider this hypothetical reaction: :A + B → C + D If this overall reaction comprises two elementary steps thus: :A + B → X :X → C + D then X is a reaction intermediate. The phrase ''reaction intermediate'' is often abbreviated to the single word ''intermediate'', and this is IUPAC's preferred form of the term. But this shorter form has other uses. It often refers to reactive intermediates. It is also used more widely for chemicals such as cumene which are traded within the chemical industry but are not generally of value outside it. IUPAC definition The IUPAC Gold Book defines an ''intermediate'' as a co ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Salt Metathesis Reaction
A salt metathesis reaction (also called a double displacement reaction, double replacement reaction, or double decomposition) is a type of chemical reaction in which two ionic compounds in aqueous solution exchange their component ions to form two new compounds. Often, one of these new compounds is a precipitate, gas, or weak electrolyte, driving the reaction forward. :AB + CD -> AD + CB \mathitA_\mathitD_\mathit + \mathitC_\mathitB_\mathit --> In older literature, the term double decomposition is common. The term double decomposition is more specifically used when at least one of the substances does not dissolve in the solvent, as the ligand or ion exchange takes place in the solid state of the reactant. For example: :AX(aq) + BY(s) → AY(aq) + BX(s). Types of reactions Counterion exchange Salt metathesis is a common technique for exchanging counterions. The choice of reactants is guided by a solubility chart or lattice energy. HSAB theory can also be used to predict ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organopalladium
Organopalladium chemistry is a branch of organometallic chemistry that deals with organic palladium compounds and their reactions. Palladium is often used as a catalyst in the reduction of alkenes and alkynes with hydrogen. This process involves the formation of a palladium-carbon covalent bond. Palladium is also prominent in carbon-carbon coupling reactions, as demonstrated in tandem reactions. Organopalladium chemistry timeline * 1873 - A. N. Zaitsev reports reduction of benzophenone over palladium with hydrogen. * 1894 - Francis Phillips reports that palladium(II) chloride organic reduction, reduces to palladium metal by contact with ethylene. * 1907 - Autoclave technology introduced by Vladimir Ipatieff makes it possible to carry out high pressure hydrogenation. * 1956 - In the Wacker process ethylene and oxygen react to acetaldehyde with catalyst PdCl2/CuCl2. During process development, Walter Hafner also identifies the first allylpalladium complex.''Acetaldehyde from Ethyle ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Oxidative Addition
Oxidative addition and reductive elimination are two important and related classes of reactions in organometallic chemistry. Oxidative addition is a process that increases both the oxidation state and coordination number of a metal centre. Oxidative addition is often a step in catalytic cycles, in conjunction with its reverse reaction, reductive elimination. Role in transition metal chemistry For transition metals, oxidative reaction results in the decrease in the d''n'' to a configuration with fewer electrons, often 2e fewer. Oxidative addition is favored for metals that are (i) basic and/or (ii) easily oxidized. Metals with a relatively low oxidation state often satisfy one of these requirements, but even high oxidation state metals undergo oxidative addition, as illustrated by the oxidation of Pt(II) with chlorine: : tCl4sup>2− + Cl2 → tCl6sup>2− In classical organometallic chemistry, the formal oxidation state of the metal and the electron count of the complex both in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Reaction Mechanism
In chemistry, a reaction mechanism is the step by step sequence of elementary reactions by which overall chemical reaction occurs. A chemical mechanism is a theoretical conjecture that tries to describe in detail what takes place at each stage of an overall chemical reaction. The detailed steps of a reaction are not observable in most cases. The conjectured mechanism is chosen because it is thermodynamically feasible and has experimental support in isolated intermediates (see next section) or other quantitative and qualitative characteristics of the reaction. It also describes each reactive intermediate, activated complex, and transition state, which bonds are broken (and in what order), and which bonds are formed (and in what order). A complete mechanism must also explain the reason for the reactants and catalyst used, the stereochemistry observed in reactants and products, all products formed and the amount of each. The electron or arrow pushing method is often used in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carboboration
In organic chemistry, carboboration is an addition of both a carbon and a boron moiety to certain carbon-containing double and triple bonds, such as alkenes, alkynes, and allenes. In the synthesis of organic compounds, this chemical reaction is used to install a new carbon-carbon bond and carbon-boron bond. The product of carboboration reactions are organoborane compounds which prove to be useful in organic synthesis, containing both a new carbon group and a boron handle for further functionalization. This carbon-boron bond allows for organoboron chemistry, which facilitates a wide variety of chemical transformations such as oxidation and the Suzuki Reaction. The carbon-boron bond can be transformed into a variety of functional groups and moieties, making it highly useful in pharmaceutical chemistry and organic synthesis. Carboboration was developed soon after the advent and widespread use of hydroboration. Carboboration is often facilitated via catalysis, often employing transitio ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |