|
Polythiophene Repeat Unit
Polythiophenes (PTs) are polymerized thiophenes, a sulfur heterocyclic compound, heterocycle. The parent PT is an insoluble colored solid with the formula (C4H2S)n.Strictly speaking, "polythiophene" is a misnomer, since the polymer consists of thienylene (2,5-C4H2S) repeat units. Similarly, thiophene is not a monomer as such. The rings are linked through the 2- and 5-positions. Poly(alkylthiophene)s have alkyl substituents at the 3- or 4-position(s). They are also colored solids, but tend to be soluble in organic solvents. PTs become Conductive polymer, conductive when oxidized. The electrical resistivity and conductivity, electrical conductivity results from the Delocalized electron, delocalization of electrons along the polymer backbone. Conductivity however is not the only interesting property resulting from electron delocalization. The optical properties of these materials respond to environmental stimuli, with dramatic color shifts in response to changes in solvent, t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Alan J
Alan may refer to: People *Alan (surname), an English and Kurdish surname * Alan (given name), an English given name ** List of people with given name Alan ''Following are people commonly referred to solely by "Alan" or by a homonymous name.'' * Alan (Chinese singer) (born 1987), female Chinese singer of Tibetan ethnicity, active in both China and Japan * Alan (Mexican singer) (born 1973), Mexican singer and actor * Alan (wrestler) (born 1975), a.k.a. Gato Eveready, who wrestles in Asistencia Asesoría y Administración * Alan (footballer, born 1979) (Alan Osório da Costa Silva), Brazilian footballer * Alan (footballer, born 1998) (Alan Cardoso de Andrade), Brazilian footballer * Alan I, King of Brittany (died 907), "the Great" * Alan II, Duke of Brittany (c. 900–952) * Alan III, Duke of Brittany(997–1040) * Alan IV, Duke of Brittany (c. 1063–1119), a.k.a. Alan Fergant ("the Younger" in Breton language) * Alan of Tewkesbury, 12th century abbott * Alan of Lynn (c. 1348–142 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Catalysis
Catalysis () is the increase in rate of a chemical reaction due to an added substance known as a catalyst (). Catalysts are not consumed by the reaction and remain unchanged after it. If the reaction is rapid and the catalyst recycles quickly, very small amounts of catalyst often suffice; mixing, surface area, and temperature are important factors in reaction rate. Catalysts generally react with one or more reactants to form intermediates that subsequently give the final reaction product, in the process of regenerating the catalyst. The rate increase occurs because the catalyst allows the reaction to occur by an alternative mechanism which may be much faster than the noncatalyzed mechanism. However the noncatalyzed mechanism does remain possible, so that the total rate (catalyzed plus noncatalyzed) can only increase in the presence of the catalyst and never decrease. Catalysis may be classified as either homogeneous, whose components are dispersed in the same phase (usual ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Iron(III) Chloride
Iron(III) chloride describes the inorganic compounds with the formula (H2O)x. Also called ferric chloride, these compounds are some of the most important and commonplace compounds of iron. They are available both in anhydrous and in hydrated forms, which are both hygroscopic. They feature iron in its +3 oxidation state. The anhydrous derivative is a Lewis acid, while all forms are mild oxidizing agents. It is used as a water cleaner and as an etchant for metals. Electronic and optical properties All forms of ferric chloride are paramagnetic, owing to the presence of unpaired electrons residing in 3d orbitals. Although Fe(III) chloride can be octahedral or tetrahedral (or both, see structure section), all of these forms have five unpaired electrons, one per d-orbital. The high spin d5 electronic configuration requires that d-d electronic transitions are spin forbidden, in addition to violating the Laporte rule. This double forbidden-ness results in its solutions being ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sulfonic Acid
In organic chemistry, sulfonic acid (or sulphonic acid) refers to a member of the class of organosulfur compounds with the general formula , where R is an organic alkyl or aryl group and the group a sulfonyl hydroxide. As a substituent, it is known as a sulfo group. A sulfonic acid can be thought of as sulfuric acid with one hydroxyl group replaced by an organic substituent. The parent compound (with the organic substituent replaced by hydrogen) is the parent sulfonic acid, , a tautomer of sulfurous acid, . Salt (chemistry), Salts or esters of sulfonic acids are called sulfonates. Preparation Aryl sulfonic acids are produced by the process of sulfonation. Usually the sulfonating agent is sulfur trioxide. A large scale application of this method is the production of alkylbenzenesulfonic acids: : In this reaction, sulfur trioxide is an electrophile and the arene is the nucleophile. The reaction is an example of electrophilic aromatic substitution. In a related process, carboxyli ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
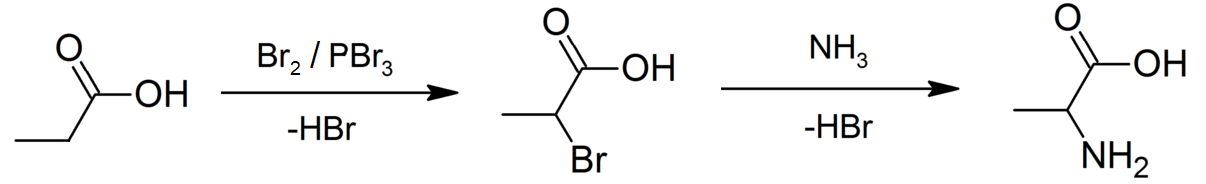
Propanoic Acid
Propionic acid (, from the Greek words πρῶτος : ''prōtos'', meaning "first", and πίων : ''píōn'', meaning "fat"; also known as propanoic acid) is a naturally occurring carboxylic acid with chemical formula . It is a liquid with a pungent and unpleasant smell somewhat resembling body odor. The anion as well as the salts and esters of propionic acid are known as propionates or propanoates. About half of the world production of propionic acid is consumed as a preservative for both animal feed and food for human consumption. It is also useful as an intermediate in the production of other chemicals, especially polymers. History Propionic acid was first described in 1844 by Johann Gottlieb, who found it among the degradation products of sugar. Over the next few years, other chemists produced propionic acid by different means, none of them realizing they were producing the same substance. In 1847, French chemist Jean-Baptiste Dumas established all the acids to be the sa ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Trifluoroacetic Acid
Trifluoroacetic acid (TFA) is a synthetic organofluorine compound with the chemical formula CF3CO2H. It belongs to the subclass of per- and polyfluoroalkyl substances (PFASs) known as ultrashort-chain perfluoroalkyl acids (PFAAs). TFA is not produced biologically or abiotically and is commonly used in organic chemistry for various purposes. It is the most abundant PFAS found in the environment. It is a haloacetic acid, with all three of the acetyl group's hydrogen atoms replaced by fluorine atoms. It is a colorless liquid with a vinegar-like odor. TFA is a stronger acid than acetic acid, having an acid ionisation constant, ''K''a, that is approximately 34,000 times higher, as the highly electronegative fluorine atoms and consequent electron-withdrawing nature of the trifluoromethyl group weakens the oxygen-hydrogen bond (allowing for greater acidity) and stabilises the anionic conjugate base. Synthesis TFA is prepared industrially by the electrofluorination of ac ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organic Acid
An organic acid is an organic compound with acidic properties. The most common organic acids are the carboxylic acids, whose acidity is associated with their carboxyl group –COOH. Sulfonic acids, containing the group –SO2OH, are relatively stronger acids. Alcohols, with –OH, can act as acids but they are usually very weak. The relative stability of the conjugate base of the acid determines its acidity. Other groups can also confer acidity, usually weakly: the thiol group –SH, the enol group, and the phenol group. In biological systems, organic compounds containing these groups are generally referred to as organic acids. A few common examples include: * Lactic acid * Acetic acid * Formic acid * Citric acid * Oxalic acid * Uric acid * Malic acid * Tartaric acid * Butyric acid * Folic acid Characteristics In general, organic acids are weak acids and do not dissociate completely in water, whereas the strong mineral acids do. Lower molecular mass organ ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bromine
Bromine is a chemical element; it has chemical symbol, symbol Br and atomic number 35. It is a volatile red-brown liquid at room temperature that evaporates readily to form a similarly coloured vapour. Its properties are intermediate between those of chlorine and iodine. Isolated independently by two chemists, Carl Jacob Löwig (in 1825) and Antoine Jérôme Balard (in 1826), its name was derived , referring to its sharp and pungent smell. Elemental bromine is very reactive and thus does not occur as a free element in nature. Instead, it can be isolated from colourless soluble crystalline mineral halide Ionic salt, salts analogous to table salt, a property it shares with the other halogens. While it is rather rare in the Earth's crust, the high solubility of the bromide ion (Br) has caused its Bromine cycle, accumulation in the oceans. Commercially the element is easily extracted from brine evaporation ponds, mostly in the United States and Israel. The mass of bromine in the oce ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Iodine
Iodine is a chemical element; it has symbol I and atomic number 53. The heaviest of the stable halogens, it exists at standard conditions as a semi-lustrous, non-metallic solid that melts to form a deep violet liquid at , and boils to a violet gas at . The element was discovered by the French chemist Bernard Courtois in 1811 and was named two years later by Joseph Louis Gay-Lussac, after the Ancient Greek , meaning 'violet'. Iodine occurs in many oxidation states, including iodide (I−), iodate (), and the various periodate anions. As the heaviest essential mineral nutrient, iodine is required for the synthesis of thyroid hormones. Iodine deficiency affects about two billion people and is the leading preventable cause of intellectual disabilities. The dominant producers of iodine today are Chile and Japan. Due to its high atomic number and ease of attachment to organic compounds, it has also found favour as a non-toxic radiocontrast material. Because of the spec ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Copper
Copper is a chemical element; it has symbol Cu (from Latin ) and atomic number 29. It is a soft, malleable, and ductile metal with very high thermal and electrical conductivity. A freshly exposed surface of pure copper has a pinkish-orange color. Copper is used as a conductor of heat and electricity, as a building material, and as a constituent of various metal alloys, such as sterling silver used in jewelry, cupronickel used to make marine hardware and coins, and constantan used in strain gauges and thermocouples for temperature measurement. Copper is one of the few metals that can occur in nature in a directly usable, unalloyed metallic form. This means that copper is a native metal. This led to very early human use in several regions, from . Thousands of years later, it was the first metal to be smelted from sulfide ores, ; the first metal to be cast into a shape in a mold, ; and the first metal to be purposely alloyed with another metal, tin, to create bronze, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bipolaron (chemistry)
In organic chemistry, bipolaron is a molecule or part of a macromolecular chain containing two positive charges in a conjugated system. The charges can be located in the centre of the chain or at its termini. Bipolarons and polarons are encountered in doped conducting polymers such as polythiophene. It is possible to synthesize and isolate bipolaron model compounds for X-ray diffraction studies. The diamagnetic bis(triaryl)amine dication 2 in ''scheme 1'' is prepared from the neutral precursor 1 in dichloromethane by reaction with 4 equivalents of antimony pentachloride. Two resonance structures exist for the dication. Structure 2a is a (singlet) diradical and 2b is the closed shell quinoid. The experimental bond lengths for the central vinylidene group in 2 are 141 pm and 137 pm compared to 144 pm and 134 pm for the precursor 1 implying some contribution from the quinoid structure. On the other hand, when a thiophene Thiophene is a heterocyclic compound with the formula C ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |