|
Catalysis
Catalysis () is the increase in rate of a chemical reaction due to an added substance known as a catalyst (). Catalysts are not consumed by the reaction and remain unchanged after it. If the reaction is rapid and the catalyst recycles quickly, very small amounts of catalyst often suffice; mixing, surface area, and temperature are important factors in reaction rate. Catalysts generally react with one or more reactants to form intermediates that subsequently give the final reaction product, in the process of regenerating the catalyst. The rate increase occurs because the catalyst allows the reaction to occur by an alternative mechanism which may be much faster than the noncatalyzed mechanism. However the noncatalyzed mechanism does remain possible, so that the total rate (catalyzed plus noncatalyzed) can only increase in the presence of the catalyst and never decrease. Catalysis may be classified as either homogeneous, whose components are dispersed in the same phase (usual ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Catalysts
Catalysis () is the increase in reaction rate, rate of a chemical reaction due to an added substance known as a catalyst (). Catalysts are not consumed by the reaction and remain unchanged after it. If the reaction is rapid and the catalyst recycles quickly, very small amounts of catalyst often suffice; mixing, surface area, and temperature are important factors in reaction rate. Catalysts generally react with one or more reactants to form reaction intermediate, intermediates that subsequently give the final reaction product, in the process of regenerating the catalyst. The rate increase occurs because the catalyst allows the reaction to occur by an alternative mechanism which may be much faster than the noncatalyzed mechanism. However the noncatalyzed mechanism does remain possible, so that the total rate (catalyzed plus noncatalyzed) can only increase in the presence of the catalyst and never decrease. Catalysis may be classified as either homogeneous catalysis, homogeneou ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Heterogeneous Catalysis
Heterogeneous catalysis is catalysis where the Phase (matter), phase of catalysts differs from that of the reagents or product (chemistry), products. The process contrasts with homogeneous catalysis where the reagents, products and catalyst exist in the same phase. Phase distinguishes between not only solid, liquid, and gas components, but also immiscible mixtures (e.g., oil and water), or anywhere an interface is present. Heterogeneous catalysis typically involves solid phase catalysts and gas phase reactants. In this case, there is a cycle of molecular adsorption, reaction, and desorption occurring at the catalyst surface. Thermodynamics, mass transfer, and heat transfer influence the Reaction rate, rate (kinetics) of reaction. Heterogeneous catalysis is very important because it enables faster, large-scale production and the selective product formation. Approximately 35% of the world's GDP is influenced by catalysis. The production of 90% of chemicals (by volume) is assisted ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Enzyme
An enzyme () is a protein that acts as a biological catalyst by accelerating chemical reactions. The molecules upon which enzymes may act are called substrate (chemistry), substrates, and the enzyme converts the substrates into different molecules known as product (chemistry), products. Almost all metabolism, metabolic processes in the cell (biology), cell need enzyme catalysis in order to occur at rates fast enough to sustain life. Metabolic pathways depend upon enzymes to catalyze individual steps. The study of enzymes is called ''enzymology'' and the field of pseudoenzyme, pseudoenzyme analysis recognizes that during evolution, some enzymes have lost the ability to carry out biological catalysis, which is often reflected in their amino acid sequences and unusual 'pseudocatalytic' properties. Enzymes are known to catalyze more than 5,000 biochemical reaction types. Other biocatalysts include Ribozyme, catalytic RNA molecules, also called ribozymes. They are sometimes descr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Homogeneous Catalysis
In chemistry, homogeneous catalysis is catalysis where the catalyst is in same phase as reactants, principally by a soluble catalyst in a solution. In contrast, heterogeneous catalysis describes processes where the catalysts and substrate are in distinct phases, typically solid and gas, respectively. The term is used almost exclusively to describe solutions and implies catalysis by organometallic compounds. Homogeneous catalysis is an established technology that continues to evolve. An illustrative major application is the production of acetic acid. Enzymes are examples of homogeneous catalysts. Examples Acid catalyst The proton is a pervasive homogeneous catalyst because water is the most common solvent. Water forms protons by the process of self-ionization of water. In an illustrative case, acids accelerate (catalyze) the hydrolysis of esters: :CH3CO2CH3 + H2O CH3CO2H + CH3OH At neutral pH, aqueous solutions of most esters do not hydrolyze at practical rates. Transition met ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemical Reaction
A chemical reaction is a process that leads to the chemistry, chemical transformation of one set of chemical substances to another. When chemical reactions occur, the atoms are rearranged and the reaction is accompanied by an Gibbs free energy, energy change as new products are generated. Classically, chemical reactions encompass changes that only involve the positions of electrons in the forming and breaking of chemical bonds between atoms, with no change to the Atomic nucleus, nuclei (no change to the elements present), and can often be described by a chemical equation. Nuclear chemistry is a sub-discipline of chemistry that involves the chemical reactions of unstable and radioactive Chemical element, elements where both electronic and nuclear changes can occur. The substance (or substances) initially involved in a chemical reaction are called reagent, reactants or reagents. Chemical reactions are usually characterized by a chemical change, and they yield one or more Product (c ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Katal
The katal (symbol: kat) is a unit of the International System of Units (SI) used for quantifying the catalytic activity of enzymes (that is, measuring the enzymatic activity level in enzyme catalysis) and other catalysts. One katal is that catalytic activity that will raise the rate of conversion by one mole per second in a specified assay system. The unit "katal" is not attached to a specified measurement procedure or assay condition, but any given catalytic activity is: the value measured depends on experimental conditions that must be specified. Therefore, to define the quantity of a catalyst in katals, the ''catalysed rate of conversion'' (the rate of conversion in presence of the catalyst minus the rate of spontaneous conversion) of a defined chemical reaction is measured in moles per second. One katal of trypsin, for example, is that amount of trypsin which breaks one mole of peptide bonds in one second under the associated specified conditions. Definition One katal r ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Elizabeth Fulhame
Elizabeth Fulhame (fl. 1794) was an early British chemist who invented the concept of catalysis and discovered photoreduction. She was described as 'the first solo woman researcher of modern chemistry'. Although she only published one text, she describes catalysis as a process at length in her 1794 book ''An Essay On Combustion with a View to a New Art of Dying and Painting, wherein the Phlogistic and Antiphlogistic Hypotheses are Proved Erroneous.'' The book relates in painstaking detail her experiments with oxidation-reduction reactions, and the conclusions she draws regarding phlogiston theory, in which she disagrees with both the Phlogistians and Antiphlogistians. In 1798, the book was translated into German by Augustin Gottfried Ludwig Lentin as ''Versuche über die Wiederherstellung der Metalle durch Wasserstoffgas.'' In 1810, it was published in the United States, to much critical acclaim. That same year, Fulhame was made an honorary member of the Philadelphia Chemical ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Adsorption
Adsorption is the adhesion of atoms, ions or molecules from a gas, liquid or dissolved solid to a surface. This process creates a film of the ''adsorbate'' on the surface of the ''adsorbent''. This process differs from absorption, in which a fluid (the ''absorbate'') is dissolved by or permeates a liquid or solid (the ''absorbent''). While adsorption does often precede absorption, which involves the transfer of the absorbate into the volume of the absorbent material, alternatively, adsorption is distinctly a surface phenomenon, wherein the adsorbate does not penetrate through the material surface and into the bulk of the adsorbent. The term '' sorption'' encompasses both adsorption and absorption, and ''desorption'' is the reverse of sorption. Like surface tension, adsorption is a consequence of surface energy. In a bulk material, all the bonding requirements (be they ionic, covalent or metallic) of the constituent atoms of the material are fulfilled by other atoms in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Enzyme Unit
The enzyme unit, or international unit for enzyme (symbol U, sometimes also IU) is a unit of enzyme's catalytic activity. 1 U (μmol/min) is defined as the amount of the enzyme that catalyzes the conversion of one micro mole of substrate per minute under the specified conditions of the assay method. The specified conditions will usually be the optimum conditions, including but not limited to temperature, pH, and substrate concentration, that yield the maximal substrate conversion rate for that particular enzyme. In some assay method, one usually takes a temperature of 25°C.Principles of Biochemistry, page 94, 4th Edition, Lehninger The enzyme unit was adopted by the International Union of Biochemistry in 1964. Since the minute is not an SI base unit of time, the enzyme unit is discouraged in favor of the katal, the unit recommended by the General Conference on Weights and Measures in 1978 and officially adopted in 1999. One katal is the enzyme activity that conv ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
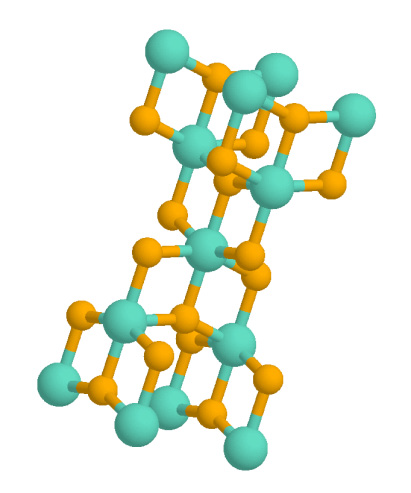
Titanium Dioxide
Titanium dioxide, also known as titanium(IV) oxide or titania , is the inorganic compound derived from titanium with the chemical formula . When used as a pigment, it is called titanium white, Pigment White 6 (PW6), or Colour Index International, CI 77891. It is a white solid that is insoluble in water, although mineral forms can appear black. As a pigment, it has a wide range of applications, including paint, sunscreen, and food coloring. When used as a food coloring, it has E number E171. World production in 2014 exceeded 9 million tonnes. It has been estimated that titanium dioxide is used in two-thirds of all pigments, and pigments based on the oxide have been valued at a price of $13.2 billion. Structure In all three of its main dioxides, titanium exhibits Octahedral molecular geometry, octahedral geometry, being bonded to six oxide anions. The oxides in turn are bonded to three Ti centers. The overall crystal structures of rutile and anatase are tetragonal in symmetry ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Turnover Number
In chemistry, the term "turnover number" has two distinct meanings. In enzymology, the turnover number () is defined as the limiting number of chemical conversions of substrate molecules per second that a single active site will execute for a given enzyme concentration for enzymes with two or more active sites. For enzymes with a single active site, is referred to as the catalytic constant. It can be calculated from the limiting reaction rate and catalyst site concentration as follows: k_\mathrm = \frac (See Michaelis–Menten kinetics). In other chemical fields, such as organometallic catalysis, turnover number (TON) has a different meaning: the number of moles of substrate that a mole of catalyst can convert before becoming inactivated: \mathrm = \frac An ideal catalyst would have an infinite turnover number in this sense, because it would never be consumed. The term turnover frequency (TOF) is used to refer to the turnover per unit time, equivalent to the meaning o ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |