|
EF-hand
The EF hand is a helix–loop–helix structural domain or ''motif'' found in a large family of calcium-binding proteins. The EF-hand motif contains a helix–loop–helix topology, much like the spread thumb and forefinger of the human hand, in which the Ca2+ ions are coordinated by ligands within the loop. The motif takes its name from traditional nomenclature used in describing the protein parvalbumin, which contains three such motifs and is probably involved in muscle relaxation via its calcium-binding activity. The EF-hand consists of two alpha helices linked by a short loop region (usually about 12 amino acids) that usually binds calcium ions. EF-hands also appear in each structural domain of the signaling protein calmodulin and in the muscle protein troponin-C. Calcium ion binding site The calcium ion is coordinated in a pentagonal bipyramidal configuration. The six residues involved in the binding are in positions 1, 3, 5, 7, 9 and 12; these residues are denot ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Calmodulin
Calmodulin (CaM) (an abbreviation for calcium-modulated protein) is a multifunctional intermediate calcium-binding messenger protein expressed in all Eukaryote, eukaryotic cells. It is an intracellular target of the Second messenger system, secondary messenger Calcium in biology, Ca2+, and the binding of Ca2+ is required for the activation of calmodulin. Once bound to Ca2+, calmodulin acts as part of a calcium Signal transduction, signal transduction pathway by modifying its interactions with various target proteins such as kinases or phosphatases. Structure Calmodulin is a small, highly conserved protein that is 148 amino acids long (16.7 kDa). The protein has two approximately symmetrical globular domains (the N- and C- domains) each containing a pair of EF hand Sequence motif, motifs separated by a flexible linker region for a total of four Ca2+ binding sites, two in each globular domain. In the Ca2+-free state, the helices that form the four EF-hands are collapsed in a co ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Calbindin D9k
Calbindins are three different calcium-binding proteins: calbindin, calretinin and S100G. They were originally described as vitamin D-dependent calcium-binding proteins in the intestine and kidney of chicks and mammals. They are now classified in different subfamilies as they differ in the number of Ca2+ binding EF hands. Calbindin 1 Calbindin 1 or simply calbindin was first shown to be present in the intestine in birds and then found in the mammalian kidney. It is also expressed in a number of neuronal and endocrine cells, particularly in the cerebellum. It is a 28 kDa protein encoded in humans by the ''CALB1'' gene. Calbindin contains 4 active calcium-binding domains, and 2 modified domains that have lost their calcium-binding capacity. Calbindin acts as a calcium buffer and calcium sensor and can hold four Ca2+ in the EF-hands of loops EF1, EF3, EF4 and EF5. The structure of rat calbindin was originally solved by nuclear magnetic resonance and was one of the largest proteins ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Parvalbumin
Parvalbumin (PV) is a calcium-binding protein with low molecular weight (typically 9–11 kDa). In humans, it is encoded by the ''PVALB'' gene. It is a member of the albumin family; it is named for its size (''parv-'', from Latin ' which means "small") and its ability to coagulate. It has three EF hand motifs and is structurally related to calmodulin and troponin C. Parvalbumin is found in fast-contracting muscles, where its levels are highest, as well as in the brain and some endocrine tissues. Parvalbumin is a small, stable protein containing EF-hand type calcium binding sites. It is involved in calcium signaling. Typically, this protein is broken into three domains, domains AB, CD and EF, each individually containing a helix-loop-helix motif. The AB domain houses a two amino-acid deletion in the loop region, whereas domains CD and EF contain the N-terminal and C-terminal, respectively. Calcium binding proteins like parvalbumin play a role in many physiological processes, nam ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Structural Domain
In molecular biology, a protein domain is a region of a protein's Peptide, polypeptide chain that is self-stabilizing and that Protein folding, folds independently from the rest. Each domain forms a compact folded Protein tertiary structure, three-dimensional structure. Many proteins consist of several domains, and a domain may appear in a variety of different proteins. Molecular evolution uses domains as building blocks and these may be recombined in different arrangements to create proteins with different functions. In general, domains vary in length from between about 50 amino acids up to 250 amino acids in length. The shortest domains, such as zinc fingers, are stabilized by metal ions or Disulfide bond, disulfide bridges. Domains often form functional units, such as the calcium-binding EF-hand, EF hand domain of calmodulin. Because they are independently stable, domains can be "swapped" by genetic engineering between one protein and another to make chimera (protein), chimeric ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Glycine
Glycine (symbol Gly or G; ) is an amino acid that has a single hydrogen atom as its side chain. It is the simplest stable amino acid. Glycine is one of the proteinogenic amino acids. It is encoded by all the codons starting with GG (GGU, GGC, GGA, GGG). Glycine disrupts the formation of alpha-helices in secondary protein structure. Its small side chain causes it to favor random coils instead. Glycine is also an inhibitory neurotransmitter – interference with its release within the spinal cord (such as during a '' Clostridium tetani'' infection) can cause spastic paralysis due to uninhibited muscle contraction. It is the only achiral proteinogenic amino acid. It can fit into both hydrophilic and hydrophobic environments, due to its minimal side chain of only one hydrogen atom. History and etymology Glycine was discovered in 1820 by French chemist Henri Braconnot when he hydrolyzed gelatin by boiling it with sulfuric acid. He originally called it "sugar of ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrophobic
In chemistry, hydrophobicity is the chemical property of a molecule (called a hydrophobe) that is seemingly repelled from a mass of water. In contrast, hydrophiles are attracted to water. Hydrophobic molecules tend to be nonpolar and, thus, prefer other neutral molecules and nonpolar solvents. Because water molecules are polar, hydrophobes do not dissolve well among them. Hydrophobic molecules in water often cluster together, forming micelles. Water on hydrophobic surfaces will exhibit a high contact angle. Examples of hydrophobic molecules include the alkanes, oils, fats, and greasy substances in general. Hydrophobic materials are used for oil removal from water, the management of oil spills, and chemical separation processes to remove non-polar substances from polar compounds. The term ''hydrophobic''—which comes from the Ancient Greek (), "having a fear of water", constructed Liddell, H.G. & Scott, R. (1940). ''A Greek-English Lexicon. revised and augmented ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrophobic Core
The hydrophobic effect is the observed tendency of nonpolar substances to aggregate in an aqueous solution and to be excluded by water#Properties, water. The word hydrophobic literally means "water-fearing", and it describes the Segregation in materials, segregation of water and nonpolar substances, which maximizes the entropy of water and minimizes the area of contact between water and nonpolar molecules. In terms of thermodynamics, the hydrophobic effect is the free energy change of water surrounding a solute. A positive free energy change of the surrounding solvent indicates hydrophobicity, whereas a negative free energy change implies hydrophilicity. The hydrophobic effect is responsible for the separation of a mixture of oil and water into its two components. It is also responsible for effects related to biology, including: cell membrane and vesicle formation, protein folding, insertion of membrane proteins into the nonpolar lipid environment and protein-small molecule asso ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
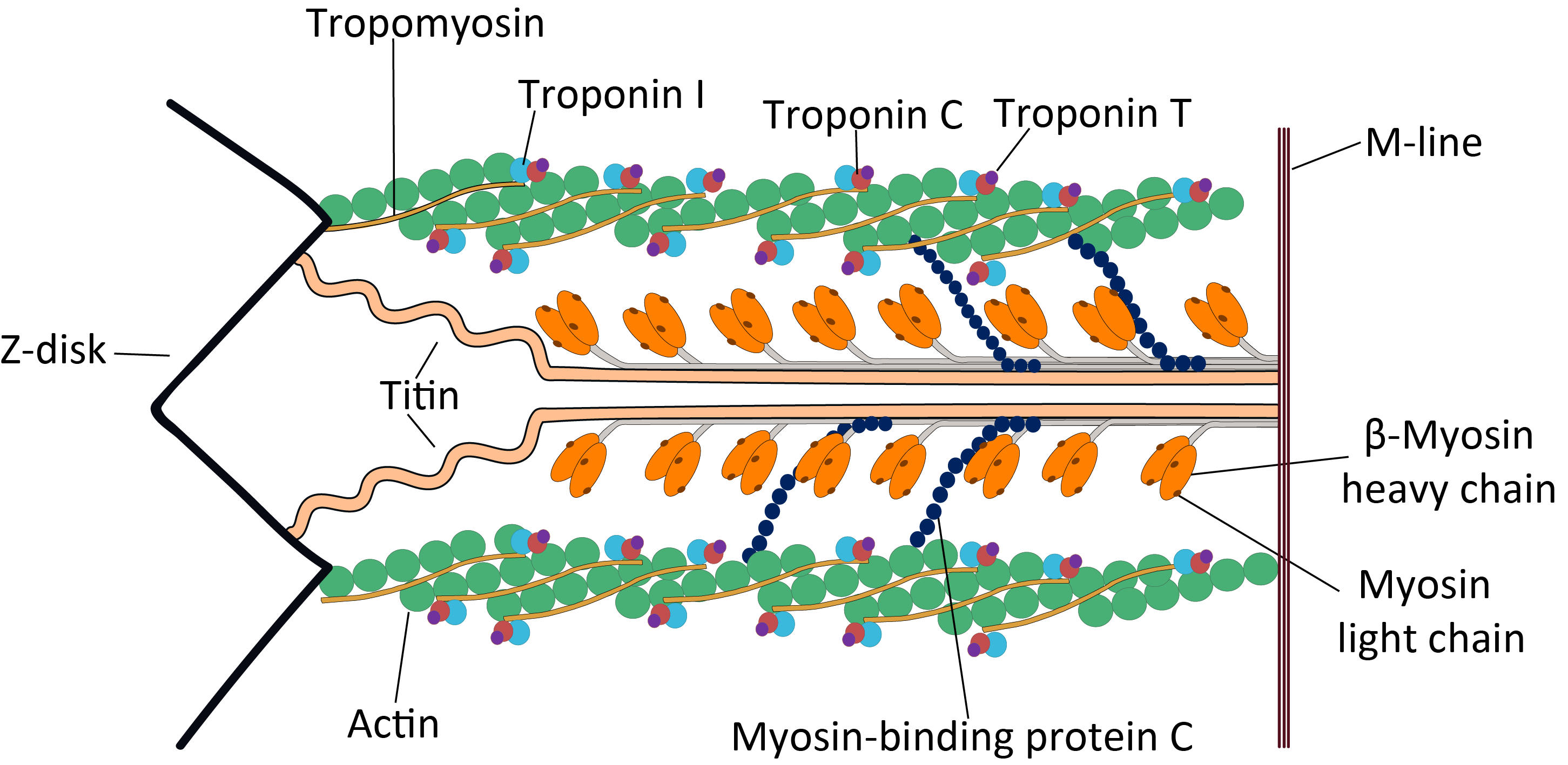
Troponin C
Troponin C is a protein which is part of the troponin complex. It contains four calcium-binding EF hands, although different isoforms may have fewer than four functional calcium-binding subdomains. It is a component of thin filaments, along with actin and tropomyosin. It contains an N lobe and a C lobe. The C lobe serves a structural purpose and binds to the N domain of troponin I (TnI). The C lobe can bind either Ca2+ or Mg2+. The N lobe, which binds only Ca2+, is the regulatory lobe and binds to the C domain of troponin I after calcium binding. Isoforms The tissue specific subtypes are: * Slow troponin C, TNNC1 (3p21.1 ) * Fast troponin C, TNNC2 (20q12-q13.11, ) Mutations Point mutations can occur in troponin C inducing alterations to Ca2+ and Mg2+ binding and protein structure, leading to abnormalities in muscle contraction. In cardiac muscle, they are related to dilated cardiomyopathy (DCM) and hypertrophic cardiomyopathy (HCM). These known point mutation ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Paramecium
''Paramecium'' ( , , plural "paramecia" only when used as a Common name, vernacular name) is a genus of eukaryotic, unicellular ciliates, widespread in freshwater, brackish, and Ocean, marine environments. Paramecia are often abundant in stagnant basins and ponds. Because some species are readily cultivated and easily induced to sexual conjugation, conjugate and divide, they have been widely used in classrooms and laboratories to study biological processes. ''Paramecium'' species are commonly studied as model organisms of the ciliate group and have been characterized as the "laboratory rat, white rats" of the phylum Ciliophora. Historical background ''Paramecium'' were among the first ciliates to be observed by microscopy, microscopists, in the late 17th century. They were most likely known to the Dutch pioneer of protozoology, Antonie van Leeuwenhoek, and were clearly described by his contemporary Christiaan Huygens in a letter from 1678. The earliest known illustration of a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Calreticulin
Calreticulin also known as calregulin, CRP55, CaBP3, calsequestrin-like protein, and endoplasmic reticulum resident protein 60 (ERp60) is a protein that in humans is encoded by the ''CALR'' gene. Calreticulin is a multifunctional soluble protein that binds Ca2+ ions (a second messenger in signal transduction), rendering them inactive. The Ca2+ is bound with low affinity, but high capacity, and can be released on a signal (see inositol trisphosphate). Calreticulin is located in storage compartments associated with the endoplasmic reticulum and is considered an ER resident protein. The term "Mobilferrin" is considered to be the same as calreticulin by some sources. Function Calreticulin binds to misfolded proteins and prevents them from being exported from the endoplasmic reticulum to the Golgi apparatus. A similar quality-control molecular chaperone, calnexin, performs the same service for soluble proteins as does calreticulin, however it is a membrane-bound protein. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Oxygen
Oxygen is a chemical element; it has chemical symbol, symbol O and atomic number 8. It is a member of the chalcogen group (periodic table), group in the periodic table, a highly reactivity (chemistry), reactive nonmetal (chemistry), nonmetal, and a potent oxidizing agent that readily forms oxides with most elements as well as with other chemical compound, compounds. Oxygen is abundance of elements in Earth's crust, the most abundant element in Earth's crust, making up almost half of the Earth's crust in the form of various oxides such as water, carbon dioxide, iron oxides and silicates.Atkins, P.; Jones, L.; Laverman, L. (2016).''Chemical Principles'', 7th edition. Freeman. It is abundance of chemical elements, the third-most abundant element in the universe after hydrogen and helium. At standard temperature and pressure, two oxygen atoms will chemical bond, bind covalent bond, covalently to form dioxygen, a colorless and odorless diatomic gas with the chemical formula ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |