|
2,4-Dinitrochlorobenzene
2,4-Dinitrochlorobenzene (DNCB) is an organic compound with the chemical formula (O2N)2C6H3Cl. It is a yellow solid that is soluble in organic solvents. It is an intermediate for the industrial production of other compounds. Preparation and reactions DNCB is produced commercially by the nitration of ''p''-nitrochlorobenzene with a mixture of nitric and sulfuric acids. Other methods afford the compound less efficiently include the chlorination of 1,3-dinitrobenzene, nitration of o-nitrochlorobenzene and the dinitration of chlorobenzene. By virtue of the two nitro substituents, the chloride in DNCB is particularly susceptible to nucleophilic substitution, at least relative to simple chlorobenzene. In this way, the compound is a precursor to many other compounds. In one example, DNCB is as a substrate in Glutathione S-Transferase, relevant to activity assays. Safety DNCB induces a type IV hypersensitivity reaction in almost all people exposed to it, so it is used medically t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
4-Nitrochlorobenzene
4-Nitrochlorobenzene is the organic compound with the formula ClC6H4NO2. It is a pale yellow solid. 4-Nitrochlorobenzene is a common intermediate in the production of a number of industrially useful compounds, including antioxidants commonly found in rubber. Other isomers with the formula ClC6H4NO2 include 2-nitrochlorobenzene and 3-nitrochlorobenzene. Preparation and reactions 4-Nitrochlorobenzene is prepared industrially by nitration of chlorobenzene: : This reaction affords both the 2- and the 4-nitro derivatives, in about a 1:2 ratio. These isomers are separated by a combination of crystallization and distillation. 4-Nitrochlorobenzene was originally prepared by the nitration of 4-bromochlorobenzene by Holleman and coworkers. The chloride substituent in 4-nitrochlorobenzene is more labile than in chlorobenzene. For example, it is readily displaced by sulfide nucleophiles, leading the way to 4-nitrothiophenol. In another example, 4-nitrochlorobenzene is a favored substr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Diethyl Ether
Diethyl ether, or simply ether, is an organic compound with the chemical formula , sometimes abbreviated as . It is a colourless, highly Volatility (chemistry), volatile, sweet-smelling ("ethereal odour"), extremely flammable liquid. It belongs to the ether class of organic compounds. It is a common solvent and was formerly used as a general anesthetic. Production Most diethyl ether is produced as a byproduct of the vapor-phase Hydration reaction, hydration of ethylene to make ethanol. This process uses solid-supported phosphoric acid Catalysis, catalysts and can be adjusted to make more ether if the need arises: Vapor-phase Dehydration reaction, dehydration of ethanol over some Aluminium oxide, alumina catalysts can give diethyl ether yields of up to 95%. : Diethyl ether can be prepared both in laboratories and on an industrial scale by the acid ether synthesis. Uses The dominant use of diethyl ether is as a solvent. One particular application is in the production of cell ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dinitrobenzene
Dinitrobenzenes are nitrobenzenes composed of a benzene ring and two nitro group (-NO2) substituents. The three possible arrangements of the nitro groups afford three isomers, 1,2-dinitrobenzene, 1,3-dinitrobenzene, and 1,4-dinitrobenzene. Each isomer has the chemical formula C6H4N2O4 and a molar mass of about 168.11 g/mol. 1,3-Dinitrobenzene is the most common isomer and it is used in the manufacture of Explosive material, explosives. Properties The dinitrobenzenes are all crystalline solids. The boiling points of the three isomers are relatively close; however, the melting points significantly differ. 1,4-Dinitrobenzene, which has the highest symmetry, has the highest melting point. References {{reflist Nitrobenzenes ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Contact Dermatitis
Contact dermatitis is a type of acute or chronic inflammation of the skin caused by exposure to chemical or physical agents. Symptoms of contact dermatitis can include itchy or dry skin, a red rash, bumps, blisters, or swelling. These rashes are not contagious or life-threatening, but can be very uncomfortable. Contact dermatitis results from either exposure to allergens ( allergic contact dermatitis), or irritants ( irritant contact dermatitis). Allergic contact dermatitis involves a delayed type of hypersensitivity and previous exposure to an allergen to produce a reaction. Irritant contact dermatitis is the most common type and represents 80% of all cases. It is caused by prolonged exposure to irritants, leading to direct injury of the epidermal cells of the skin, which activates an immune response, resulting in an inflammatory cutaneous reaction. Phototoxic dermatitis occurs when the allergen or irritant is activated by sunlight. Diagnosis of allergic contact dermati ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Wart
Warts are non-cancerous viral growths usually occurring on the hands and feet but which can also affect other locations, such as the genitals or face. One or many warts may appear. They are distinguished from cancerous tumors as they are caused by a viral infection, such as a human papillomavirus, rather than a cancer growth. Factors that increase the risk include the use of public showers and pools, working with meat, eczema, and a weak immune system. The virus is believed to infect the host through the entrance of a skin wound. A number of types exist, including plantar warts, " filiform warts", and genital warts. Genital warts are often sexually transmitted. Without treatment, most types of warts resolve in months to years. A number of treatments may speed resolution, including salicylic acid applied to the skin and cryotherapy. In those who are otherwise healthy, they do not typically result in significant problems. Treatment of genital warts differs from that of o ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
T Cell
T cells (also known as T lymphocytes) are an important part of the immune system and play a central role in the adaptive immune response. T cells can be distinguished from other lymphocytes by the presence of a T-cell receptor (TCR) on their cell surface receptor, cell surface. T cells are born from hematopoietic stem cells, found in the bone marrow. Developing T cells then migrate to the thymus gland to develop (or mature). T cells derive their name from the thymus. After migration to the thymus, the precursor cells mature into several distinct types of T cells. T cell differentiation also continues after they have left the thymus. Groups of specific, differentiated T cell subtypes have a variety of important functions in controlling and shaping the immune response. One of these functions is immune-mediated cell death, and it is carried out by two major subtypes: Cytotoxic T cell, CD8+ "killer" (cytotoxic) and T helper cell, CD4+ "helper" T cells. (These are named for the presen ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Type IV Hypersensitivity
Type IV hypersensitivity, in the Gell and Coombs classification of allergic reactions, often called delayed-type hypersensitivity, is a type of hypersensitivity reaction that can take a day or more to develop. Unlike the other types, it is not humoral (not antibody-mediated) but rather is a type of cell-mediated response. This response involves the interaction of T cells, monocytes, and macrophages. This reaction is caused when CD4+ Th1 cells recognize foreign antigen in a complex with the MHC class II on the surface of antigen-presenting cells. These can be macrophages that secrete IL-12, which stimulates the proliferation of further CD4+ Th1 cells. CD4+ T cells secrete IL-2 and interferon gamma (IFNγ), inducing the further release of other Th1 cytokines, thus mediating the immune response. Activated CD8+ T cells destroy target cells on contact, whereas activated macrophages produce hydrolytic enzymes and, on presentation with certain intracell ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
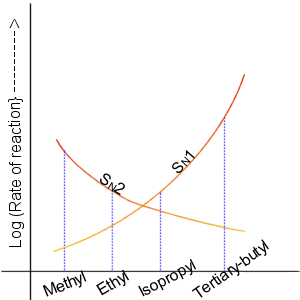
Nucleophilic Substitution
In chemistry, a nucleophilic substitution (SN) is a class of chemical reactions in which an electron-rich chemical species (known as a nucleophile) replaces a functional group within another electron-deficient molecule (known as the electrophile). The molecule that contains the electrophile and the leaving functional group is called the substrate. The most general form of the reaction may be given as the following: :\text\mathbf + \ce + \text\mathbf The electron pair (:) from the nucleophile (Nuc) attacks the substrate () and bonds with it. Simultaneously, the leaving group (LG) departs with an electron pair. The principal product in this case is . The nucleophile may be electrically neutral or negatively charged, whereas the substrate is typically neutral or positively charged. An example of nucleophilic substitution is the hydrolysis of an alkyl bromide, R-Br under basic conditions, where the attacking nucleophile is hydroxyl () and the leaving group is bromide (). :O ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nitro Group
In organic chemistry, nitro compounds are organic compounds that contain one or more nitro functional groups (). The nitro group is one of the most common explosophores (functional group that makes a compound explosive) used globally. The nitro group is also strongly electron-withdrawing. Because of this property, bonds alpha (adjacent) to the nitro group can be acidic. For similar reasons, the presence of nitro groups in aromatic compounds retards electrophilic aromatic substitution but facilitates nucleophilic aromatic substitution. Nitro groups are rarely found in nature. They are almost invariably produced by nitration reactions starting with nitric acid. Synthesis Preparation of aromatic nitro compounds Aromatic nitro compounds are typically synthesized by nitration. Nitration is achieved using a mixture of nitric acid and sulfuric acid, which produce the nitronium ion (), which is the electrophile: + The nitration product produced on the largest scale, by f ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chlorobenzene
Chlorobenzene (abbreviated PhCl) is an aryl chloride and the simplest of the chlorobenzenes, consisting of a benzene ring substituted with one chlorine atom. Its chemical formula is C6H5Cl. This colorless, flammable liquid is a common solvent and a widely used intermediate in the manufacture of other chemicals. Uses The major use of chlorobenzene is as a precursor for further intermediates such as nitrophenols, nitroanisole, chloroaniline, and phenylenediamines, which are used in the production of herbicides, dyestuffs, chemicals for rubber, and pharmaceuticals. It is also used as a high-boiling solvent in industrial and laboratory applications, for materials such as oils, waxes, resins, and rubber. Chlorobenzene is nitrated on a large scale to give a mixture of 2-nitrochlorobenzene and 4-nitrochlorobenzene, which are separated and used as intermediates in production of other chemicals. These mononitrochlorobenzenes are converted to related 2-nitrophenol, 2-nitroanis ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
2-Nitrochlorobenzene
2-Nitrochlorobenzene is an organic compound with the formula ClC6H4NO2. It is one of three isomeric nitrochlorobenzenes. It is a yellow crystalline solid that is important as a precursor to other compounds due to its two functional groups. Synthesis Nitrochlorobenzene is typically synthesized by nitration of chlorobenzene in the presence of sulfuric acid: :C6H5Cl + HNO3 → O2NC6H4Cl + H2O This reaction affords a mixture of isomers. Using an acid ratio of 30% nitric acid, 56% sulfuric acid and 14% water, the product mix is typically 34-36% 2-nitrochlorobenzene and 63-65% 4-nitrochlorobenzene, with only about 1% 3-nitrochlorobenzene. Reactions 2-Nitrochlorobenzene can be reduced to the 2-chloroaniline with Fe/HCl mixture, the Bechamp reduction. 2-Nitrochlorobenzene, like its isomers, is reactive toward nucleophiles, resulting in chloride substitution. With polysulfide, it reacts to give di-orthonitrophenyl disulfide: : Similarly, it reacts with sodium methoxide to give ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Halogenation
In chemistry, halogenation is a chemical reaction which introduces one or more halogens into a chemical compound. Halide-containing compounds are pervasive, making this type of transformation important, e.g. in the production of polymers, drugs. This kind of conversion is in fact so common that a comprehensive overview is challenging. This article mainly deals with halogenation using elemental halogens (). Halides are also commonly introduced using salts of the halides and halogen acids. Many specialized reagents exist for introducing halogens into diverse substrates, e.g. thionyl chloride. Organic chemistry Several pathways exist for the halogenation of organic compounds, including free radical halogenation, ketone halogenation, electrophilic halogenation, and halogen addition reaction. The nature of the substrate determines the pathway. The facility of halogenation is influenced by the halogen. Fluorine and chlorine are more electrophilic and are more aggressive haloge ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |