Willgerodt reaction on:
[Wikipedia]
[Google]
[Amazon]
The Willgerodt rearrangement or Willgerodt reaction is an organic reaction converting an aryl alkyl ketone, alkyne, or  An example with modified reagents (sulfur, concentrated
An example with modified reagents (sulfur, concentrated 

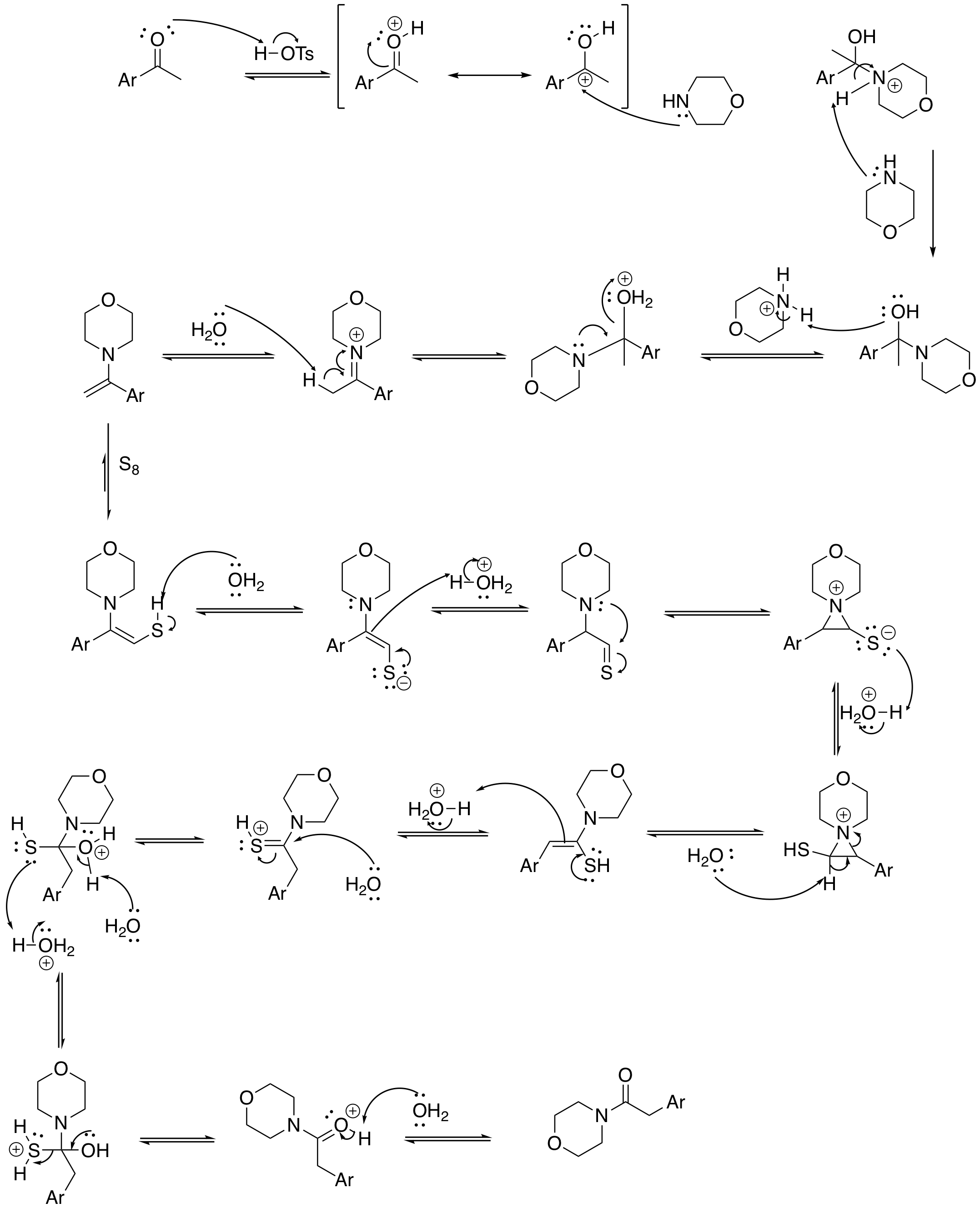 The first stage of the reaction is basic imine formation by the ketone group and the
The first stage of the reaction is basic imine formation by the ketone group and the
alkene
In organic chemistry, an alkene is a hydrocarbon containing a carbon–carbon double bond.
Alkene is often used as synonym of olefin, that is, any hydrocarbon containing one or more double bonds.H. Stephen Stoker (2015): General, Organic, an ...
to the corresponding amide
In organic chemistry, an amide, also known as an organic amide or a carboxamide, is a compound with the general formula , where R, R', and R″ represent organic groups or hydrogen atoms. The amide group is called a peptide bond when it i ...
by reaction with ammonium polysulfide, named after Conrad Willgerodt. The formation of the corresponding carboxylic acid is a side reaction
A side reaction is a chemical reaction that occurs at the same time as the actual main reaction, but to a lesser extent. It leads to the formation of by-product, so that the yield of main product is reduced:
: + B ->[] P1
: + C ->[] P2
P1 is th ...
. When the alkyl group is an aliphatic chain (n typically 0 to 5), multiple reactions take place with the amide group always ending up at the terminal end.
 An example with modified reagents (sulfur, concentrated
An example with modified reagents (sulfur, concentrated ammonium hydroxide
Ammonia solution, also known as ammonia water, ammonium hydroxide, ammoniacal liquor, ammonia liquor, aqua ammonia, aqueous ammonia, or (inaccurately) ammonia, is a solution of ammonia in water. It can be denoted by the symbols NH3(aq). Although ...
and pyridine
Pyridine is a basic heterocyclic organic compound with the chemical formula . It is structurally related to benzene, with one methine group replaced by a nitrogen atom. It is a highly flammable, weakly alkaline, water-miscible liquid with a ...
) is the conversion of acetophenone
Acetophenone is the organic compound with the chemical formula, formula C6H5C(O)CH3. It is the simplest aromatic ketone. This colorless, viscous liquid is a precursor to useful resins and fragrances.
Production
Acetophenone is formed as a byprodu ...
to 2-phenylacetamide and phenylacetic acid

Willgerodt–Kindler reaction
The related Willgerodt–Kindler reaction takes place with elemental sulfur and anamine
In chemistry, amines (, ) are compounds and functional groups that contain a basic nitrogen atom with a lone pair. Amines are formally derivatives of ammonia (), wherein one or more hydrogen
Hydrogen is the chemical element wi ...
like morpholine. The initial product is a thioacetamide
Thioacetamide is an organosulfur compound with the formula C2 H5 N S. This white crystalline solid is soluble in water and serves as a source of sulfide ions in the synthesis of organic and inorganic compounds. It is a prototypical thioamide.
...
for example that of acetophenone
Acetophenone is the organic compound with the chemical formula, formula C6H5C(O)CH3. It is the simplest aromatic ketone. This colorless, viscous liquid is a precursor to useful resins and fragrances.
Production
Acetophenone is formed as a byprodu ...
which can again be hydrolyzed to the amide. The reaction is named after Karl Kindler.

Reaction mechanism
A possible reaction mechanism for the Kindler variation''Name Reactions and Reagents in Organic Synthesis'' Bradford P. Mundy, Michael G. Ellerd, Frank G. Jr. Favaloro 2005 is depicted below: The first stage of the reaction is basic imine formation by the ketone group and the
The first stage of the reaction is basic imine formation by the ketone group and the amine
In chemistry, amines (, ) are compounds and functional groups that contain a basic nitrogen atom with a lone pair. Amines are formally derivatives of ammonia (), wherein one or more hydrogen
Hydrogen is the chemical element wi ...
group of morpholine to the enamine
An enamine is an unsaturated compound derived by the condensation of an aldehyde or ketone with a secondary amine. Enamines are versatile intermediates.
:
The word "enamine" is derived from the affix ''en''-, used as the suffix of alkene, and t ...
which reacts in a conjugate addition
Nucleophilic conjugate addition is a type of organic reaction. Ordinary nucleophilic additions or 1,2-nucleophilic additions deal mostly with additions to carbonyl compounds. Simple alkene compounds do not show 1,2 reactivity due to lack of polari ...
(see Stork enamine alkylation
The Stork enamine alkylation involves the addition of an enamine to a Michael acceptor (e.g, an enone, α,β -unsaturated carbonyl compound) or another electrophilic alkylation reagent to give an alkylated iminium product, which is hydrolyzed by d ...
for a related step) with sulfur to the sulfide. The actual rearrangement reaction takes place when the amine group attacks the thiocarbonyl
In organic chemistry, thioketones (; also known as thiones or thiocarbonyls) are organosulfur compounds related to conventional ketones in which the oxygen has been replaced by a sulfur. Instead of a structure of , thioketones have the structu ...
in a nucleophilic addition temporarily forming an aziridine and the thioacetamide
Thioacetamide is an organosulfur compound with the formula C2 H5 N S. This white crystalline solid is soluble in water and serves as a source of sulfide ions in the synthesis of organic and inorganic compounds. It is a prototypical thioamide.
...
by tautomerization
Tautomers () are structural isomers (constitutional isomers) of chemical compounds that readily interconvert.
The chemical reaction interconverting the two is called tautomerization. This conversion commonly results from the relocation of a hyd ...
.
References
{{Reflist Rearrangement reactions Name reactions